Abstract
Background:
The chemokine receptor CXCR4 and the transcription factor JUNB, expressed on a variety of tumor cells, seem to play an important role in the metastatic process. Since disseminated tumor cells (DTCs) in the bone marrow (BM) have been associated with worse outcomes, we evaluated the expression of CXCR4 and JUNB in DTCs of primary, nonmetastatic breast cancer (BC) patients before the onset of any systemic treatment.
Methods:
Bilateral BM (10 ml) aspirations of 39 hormone receptor (HR)-positive, HER2-negative BC patients were assessed for the presence of DTCs using the following combination of antibodies: pan-cytokeratin (A45-B/B3)/CXCR4/JUNB. An expression pattern of the examined proteins was created using confocal laser scanning microscopy, Image J software and BC cell lines.
Results:
CXCR4 was overexpressed in cancer cells and DTCs, with the following hierarchy of expression: SKBR3 > MCF7 > DTCs > MDA-MB231. Accordingly, the expression pattern of JUNB was: DTCs > MDA-MB231 > SKBR3 > MCF7. The mean intensity of CXCR4 (6411 ± 334) and JUNB (27725.64 ± 470) in DTCs was statistically higher compared with BM hematopoietic cells (2009 ± 456, p = 0.001; and 11112.89 ± 545, p = 0.001, respectively). The (CXCR4+JUNB+CK+) phenotype was the most frequently detected [90% (35/39)], followed by the (CXCR4–JUNB+CK+) phenotype [36% (14/39)]. However, (CXCR4+JUNB–CK+) tumor cells were found in only 5% (3/39) of patients. Those patients harboring DTCs with the (CXCR4+JUNB+CK+) phenotype revealed lower overall survival (Cox regression: p = 0.023).
Conclusions:
(CXCR4+JUNB+CK+)-expressing DTCs, detected frequently in the BM of BC patients, seem to identify a subgroup of patients at higher risk for relapse that may be considered for close follow up.
Introduction
Breast cancer (BC) is the leading cause of cancer-related deaths among women in Europe and the USA. 1 BC-related mortality is associated with the development of metastatic potential of primary tumor lesions, due mostly to early dissemination of tumor cells that enter the lymph and blood systems as circulating tumor cells (CTCs) to move into secondary organs, preferentially the bone marrow (BM), to survive as disseminated tumor cells (DTCs) in the foreign microenvironment to form metastasis, often 10–20 years after first diagnosis.2,3 A variety of studies has already proven that DTCs are present in up to 40% of primary BC patients, and that their presence, as well as persistence, is associated with a reduced progression-free survival (PFS) and overall survival (OS).4–8 The lack of therapeutic approaches to eliminate these cells constitutes a major obstacle to the successful treatment of this disease. Due to the nature and rarity of DTCs, only a few studies have already unraveled their characteristics. In this regard, it has been demonstrated that most existing DTCs are nonproliferating, which explains why chemotherapy is not able to eliminate them. Furthermore, discordant expression of so-called predictive markers on the primary tumor (hormonal and growth factor receptors) and corresponding DTCs has been described.5,9,10 Whereas therapy decisions are based solely on the expression of these markers on the primary tumor, targets in DTCs might be useful to eliminate minimal residual disease at primary diagnosis. In addition, some DTCs have been identified as cancer stem cells (CSCs) that might have the ability to renew themselves.11,12
However, not all patients with detectable DTCs have a higher risk of relapse. This implies that not all DTCs have metastatic potential, but, unfortunately, it is currently not predictable which of these cells will evolve to metastasis. Recently, three models of metastatic potential have been proposed. The first postulates that patients who will not develop metastasis harbor DTCs that are exclusively tumor bulk cells, containing no, or only a very few, CSCs. Secondly, patients facing metastasis a few years after first diagnosis seem to have CSCs with full metastatic potential among the bulk cells. Finally, it is suggested that especially hormone-receptor-positive (HR+) BCs, which recur more than 5 years after first diagnosis, pass through a period of dormancy because existing CSCs have the potential for self-renewal and immortality but lack other mechanisms to metastasize. 13 Consequently, to treat patients appropriately, and avoid probable overtreatment, we have to identify those patients who harbor DTCs that are associated with a higher risk of relapse.
To approach this goal, we recently reported that chemokine receptor type 4 (CXCR4) and the transcription factor JUNB are overexpressed in CTCs. 14 CXCR4 has been found to be a prognostic marker in various types of cancer, including BC. 15 It is upregulated in tumor tissue as compared with normal tissues, and, together with its ligand CXCL12, seems to play a role in the metastasis of these tumor entities.15–17 Interestingly, in early stage BC, tumors of node-positive patients had superior CTC-seeding and metastatic potential compared with node-negative patients, which was attributed to the expression of vimentin, urokinase receptor, and CXCR4, all known to be involved in epithelial-mesenchymal transition (EMT), and, thus, associated with a particularly malignant phenotype. 18
JUNB has been analyzed primarily in the context of cell cycle regulation and differentiation; however, several recent studies have reported a close relationship between JUNB and invasion/metastasis in solid tumors, including BC.19–21 JUNB also represents an important target in diseases associated with EMT, including cancer and fibrosis,22,23 and has been implicated in the earliest events in the development of resistance to kinase inhibitors in BC. 24
The expression of these markers in DTCs has not been evaluated so far. The aim of our study was to identify CXCR4 and JUNB expression in DTCs derived from primary, nonmetastatic, HR+ BC patients, and to explore their frequency and potential role in follow up of the disease. For this purpose, we retrospectively stained archived BM slides with a known number of cytokeratin-positive (CK+) cells for the DTC phenotypes (CXCR4+JUNB+CK+), (CXCR4–JUNB+CK+) and (CXCR4+JUNB–CK+), respectively.
Materials and methods
Cell cultures
Three different BC cell lines (MCF7, SKBR3, and MDA-MB231) were used as controls in this study. All cell lines were obtained from ATCC (American Type Culture Collection, Manassas, VA, USA). MCF7 adenocarcinoma HR+ cells were cultured in 1:1 Dulbecco’s Modified Eagle Medium (DMEM, GlutaMAX) (Gibco-BRL, Gaithersburg, MD, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco-BRL), 16 ng/ml insulin, and 50 mg/ml penicillin/streptomycin (Gibco-BRL). SKBR3 (HER2-positive) BC cells were cultured in RPMI 1640 supplemented with 10% FBS. MDA-MB231 (ER–PR–HER– triple-negative) cells were cultured in DMEM, supplemented with 10% FBS and 50 mg/ml penicillin/streptomycin. Cells were maintained in a humidified atmosphere of 5% CO2 in air. Subcultivation was performed with 0.25% trypsin and 5 mM EDTA (Gibco-BRL). All cell lines were spiked into normal donors’ peripheral blood mononuclear cells (PBMCs) fraction (100/106). All experiments were performed during the logarithmic growth phase.
Patient population and patient characteristics
A total of 39 nonmetastatic BC patients who presented with first diagnosis of BC at the Department of Gynecology and Obstetrics in Essen, Germany, were included in the study. The median age of the patients was 59 years (range 43–85 years). Of the 39 patients, 25 (64%) had T1 tumors, 28 (72%) were node-negative, most of the patients (32/39, 82%) had a ductal carcinoma, and a predominantly poor or moderately differentiated tumor. All patients were HR+ and HER2-negative. Patients’ characteristics at the time of diagnosis are shown in Table 1.
Clinical data of patients.
BM, bone marrow; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; PR, progesterone receptor.
Study design
We analyzed the expression of CXCR4 and JUNB in DTCs retrospectively in the BM of primary, nonmetastatic BC patients to determine their prognostic value in long-term follow up of BC. The median follow-up time for these patients was 92 months (range: 41–135 months).
Eligibility criteria
The eligibility criteria were histologically proven BC; BM aspiration at time of primary diagnosis, and before the administration of any systemic treatment; no severe uncontrolled comorbidities or medical conditions, no further malignancies currently or in history. Adjuvant treatment included antihormonal therapy (tamoxifen or an aromatase inhibitor) and chemotherapy/radiotherapy. In addition, based on the publication of Diel and colleagues, 16 all DTC-positive patients were recommended an additional therapy with oral bisphosphonate clodronate (2 × 520 mg per day for at least 2 years), which resulted in improved prognosis for our patients. 25
A total of 10 healthy blood donors (HDs) were also included in the study and analyzed for the same staining as study controls to evaluate the sensitivity and specificity of the method/antibodies. PBMCs from HDs were extracted following Ficoll-Hypaque density gradient centrifugation (Gibco-BRL).
Study approval
All specimens were taken in the Department of Gynecology and Obstetrics of the University Hospital in Essen, Germany. All patients gave informed written consent prior to inclusion in the study. The patients gave their written consent for taking blood and BM for diagnostic and research purposes, and also allowed clinical and follow-up data to be used for research purposes.
All protocols were approved by the institutional review board (05/2856): Institutional Ethic committee of the Medical Faculty of the University Hospital of Essen, Germany.
Selection and detection of DTCs
Samples (10 ml) of BM were aspirated from the anterior iliac crests of all patients at the beginning of surgery of the primary tumor, before starting any therapy, and processed within 24 h. BM tumor cell isolation and detection have been described elsewhere. 25 Briefly, BM cells were isolated from heparinized BM (5000 U/ml BM) by Ficoll-Hypaque density gradient centrifugation (density 1.077 g/mol; Pharmacia, Freiburg, Germany) at 400 × g for 30 min. Slides with 106 cells were analyzed for DTCs by immunocytochemistry using the pan-cytokeratin antibody (A45-B/B3). Microscopic evaluation of the slides was carried out using the ARIOL system (Genetix, New Milton, UK). The remaining cells were spun onto glass slides for further characterization, and stored at −80°C until further use.
Triple immunofluorescence and confocal laser scanning microscopy
Two cytospins from all patients, containing 106 MNCs, were used for triple immunofluoresence (CK/CXCR4/JUNB) stainings. Cells were fixed and permeabilized with a mixture of acetone/methanol (9:1) for 20 min at room temperature (RT). After blocking with phosphate-buffered saline (PBS) supplemented with 10% (v/v) FBS for 1 h, cells were incubated with JUNB anti-mouse (Santa Cruz Biotechnology, Santa Cruz, CA, USA) antibody for 1 h. Alexa 633 anti-mouse was used as a secondary antibody for 45 min. Consequently, the samples were stained with CXCR4 anti-rabbit (ABCAM, Cambridge, MA, USA) for 1 h, followed by the corresponding Alexa555 anti-rabbit antibody (Molecular Probes, Invitrogen, Carlsbad, CA, USA).
A45-B/B3 (detecting CK8, CK18, and CK19) antibody was used conjugated with Alexa 488, applying Zenon technology (Molecular Probes) according to the manufacturer’s instructions. Zenon antibodies were prepared within 30 min of use. Subsequently, cells were incubated with the antibody complex for 1 h. Finally, cells were stained with 4′,6-diamidino-2-phenylindole (DAPI) conjugated with antifade.
Positive and negative controls were used in each experiment, using BC cell lines’ cytospins, by omitting one of the first antibodies. Therefore, each experiment included three different negative controls and one positive control for all antibodies. Slides were then analyzed with confocal laser scanning microscopy and Image J to quantify expression of tumor markers.
Statistical analysis
Statistical tests were performed at the 5% level of significance. SPSS version 20 (SPSS, Chicago, IL, USA) software was used for the analysis. The statistical tests between the mean intensity variables in cell lines and DTCs were carried out with either Student’s t test for variables following the normal distribution or with Wilcoxon signed-rank nonparametric test for variables with binomial distribution. Furthermore, for nominal variables, chi-squared analysis was performed.
OS was defined as the time from treatment initiation until death from any cause. PFS was defined from the enrolment of the study until disease relapse or death, whichever occurred first. Kaplan–Meier curves and Cox regression analysis for PFS and OS were compared using the log-rank test to provide a univariate and multivariate assessment of the prognostic value of selected clinical risk factors.
Results
CXCR4 and JUNB expression in cell lines
Three cancer cell lines representative of different BC subtypes (Liminal: MCF7; HER-positive: SKBR3; basal like: MDA-MB231) were used to create an expression pattern of CXCR4 and JUNB in cancer cells. Both molecules were quantified in BM hematopoietic cells (Figure 1a, b).
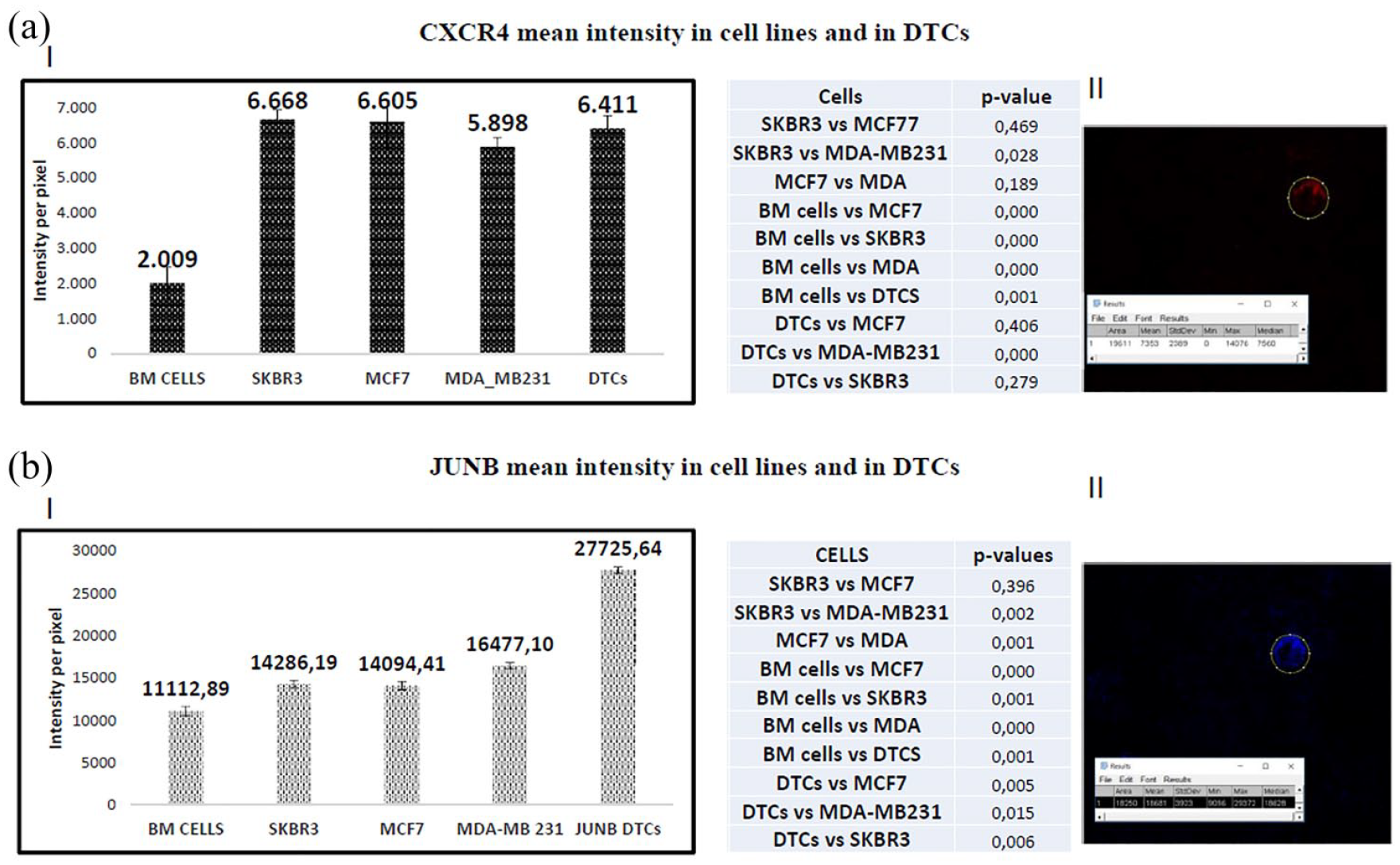
Quantification of CXCR4 (a) and JUNB (b) in BC cell lines and in patients’ DTCs. (I) Quantification of the mean intensity of CXCR4/JUNB in the examined BC cell lines, BM cells, and in patients’ DTCs. At least 50 cells were examined from each cell line (MCF7, SKBR3, and MDA-MB231) and BM cells. The total number of isolated DTCs is also included in the quantification analysis of CXCR4/JUNB. (II) Representative DTC analyzed for CXCR4/JUNB expression with Image J software.
CXCR4 was overexpressed in cancer cells (Figure 1a). The highest intensity was observed in SKBR3 (6668 ± 272) cells, and was statistically different from the intensity in BM hematopoietic cells (p = 0.00001). MCF7 and MDA-MB231 cells also revealed increased expression compared with BM cells (p = 0.00001). The hierarchy of expression among the cell lines was SKBR3 > MCF7 > MDA-MB231 > BM hematopoietic cells.
Quantification of JUNB among BC cell lines revealed that the highest expression was observed in MDA-MB231 (16477 ± 330) triple-negative cells, following the hierarchy: MDA-MB231 > SKBR3 > MCF7 > BM hematopoietic cells. The expression of JUNB in all BC cell lines was statistically increased compared with BM hematopoietic cells (MDA-MB231: p = 0.0001; SKBR3: p = 0.001; MCF7: p = 0.0001, respectively).
DTCs in BC patients
At primary diagnosis, we evaluated all 39 BM aspirations for the presence of CK+ DTCs, resulting in 37/39 positive patients. Thus, in 2/39 patients, no CK+ cells were detected on the slides that were used for the detection of DTCs in routine clinical analysis. However, residual slides of these two patients were also included in the study. Finally, slides of 39 patients were stained with CXCR4/JUNB/cytokeratin antibodies. PBMCs from HDs were also stained, and were negative for triple-positive cells (CK/CXCR4/JUNB); however, there were a few PBMCs positive for CXCR4 in four samples. All DTCs with expression of CXCR4 or JUNB higher than in BM hematopoietic cells were considered as positive for the corresponding molecules.
Quantification of CXCR4 and JUNB in DTCs isolated from BC patients
Quantification of CXCR4 expression in patients’ DTCs revealed that the receptor was overexpressed in these cells, and the expression level was similar to that seen in BC cell lines (MCF7 and SKBR3). CXCR4 expression was also increased statistically compared with MDA-MB231 (6.411 ± 334 versus 5898 ± 247, p = 0.0001) and BM hematopoietic cells (6.411 ± 334 versus 2009 ± 456, p = 0.001) (Figure 1a).
The expression of JUNB in DTCs was statistically significantly higher than in any BC cell line. In particular, JUNB in DTCs was more enhanced than in MCF7 cells (27725.64 ± 470 versus 14094.41 ± 486, p = 0.005), SKBR3 (14286.2 ± 428, p = 0.006), and MDA-MB231 cells (16477.10 ± 330, p = 0.015), respectively. JUNB expression was also increased significantly compared with BM hematopoietic cells (11112.89 ± 545, p = 0.001) (Figure 1b).
Among the whole cohort of patients, CXCR4-positive DTCs were detected in 92% (36/39), while JUNB-positive DTCs were found in 95% (37/39) of patients.
(CXCR4+JUNB+CK+) cells were the most abundant phenotype, observed in 90% (35/39) of patients, while (CXCR4–JUNB+CK+) cells were detected in 36% (14/39) of patients. A lower frequency was observed for the (CXCR4+JUNB–CK+) phenotype, (5%, 2/39 patients), as well as for the (CXCR4–JUNB–CK+) phenotype (31%, 12/39 patients) (Figure 2a).

Percentage of distinct phenotypes among early BC patients. (a) Percentage of patients harboring DTCs with distinct phenotypes. The majority of patients (90%) had at least one DTC expressing the (CXCR4+JUNB+CK+) phenotype, while the (CXCR4+JUNB–CK+) phenotype (5%) was less common. (b) The mean value of the percentage of DTCs per patient revealed that the most frequent phenotype (75%) among the whole number of isolated DTCs was (CXCR4+JUNB+CK+), while the phenotype observed least (1.4%) was (CXCR4+JUNB–CK+).
The percentage of DTCs with (CXCR4+JUNB+CK+) phenotype among the total number of examined DTCs was 75%, which was statistically significantly higher than any other phenotype (Figure 2b). Furthermore, the percentage of (CXCR4–JUNB+CK+)-expressing DTCs was statistically significantly increased (12%, p = 0.002) compared with (CXCR4+JUNB–CK+)-expressing DTCs. Finally, the percentage of DTCs representing the (CXCR4–JUNB–CK+) phenotype was higher compared with the (CXCR4+JUNB–CK+) phenotype [8.6% versus 1.4%, p = 0.002 (Figure 2b)].
As shown in Table 2, 21/39 patients (54%) harbored more than one phenotype in their DTCs. Exclusively (CXCR+JUNB+CK+)-expressing DTCs were observed in 16/39 patients (41%). Interestingly, none of the patients had exclusively (CXCR4–JUNB–CK+)-DTCs.
DTCs per patient and per phenotype.

DTC, disseminated tumor cell.
As shown in Figure 3(a, b), JUNB expression was observed in the cytoplasm and in the nucleus. CXCR4 was located mainly in the plasma membrane or in the cytoplasm. In one of the patients, both single DTCs and DTC clusters could be detected. Figure 3b shows two different phenotypes within the same patient [(CXCR4+JUNB+CK+) and (CXCR4–JUNB+CK+) cells]. DTC clusters in this patient were negative for CXCR4 and positive for JUNB, whereas single DTCs in the same aspiration expressed higher levels of CXCR4. Table 2 lists the absolute number of DTCs per patient and phenotype.
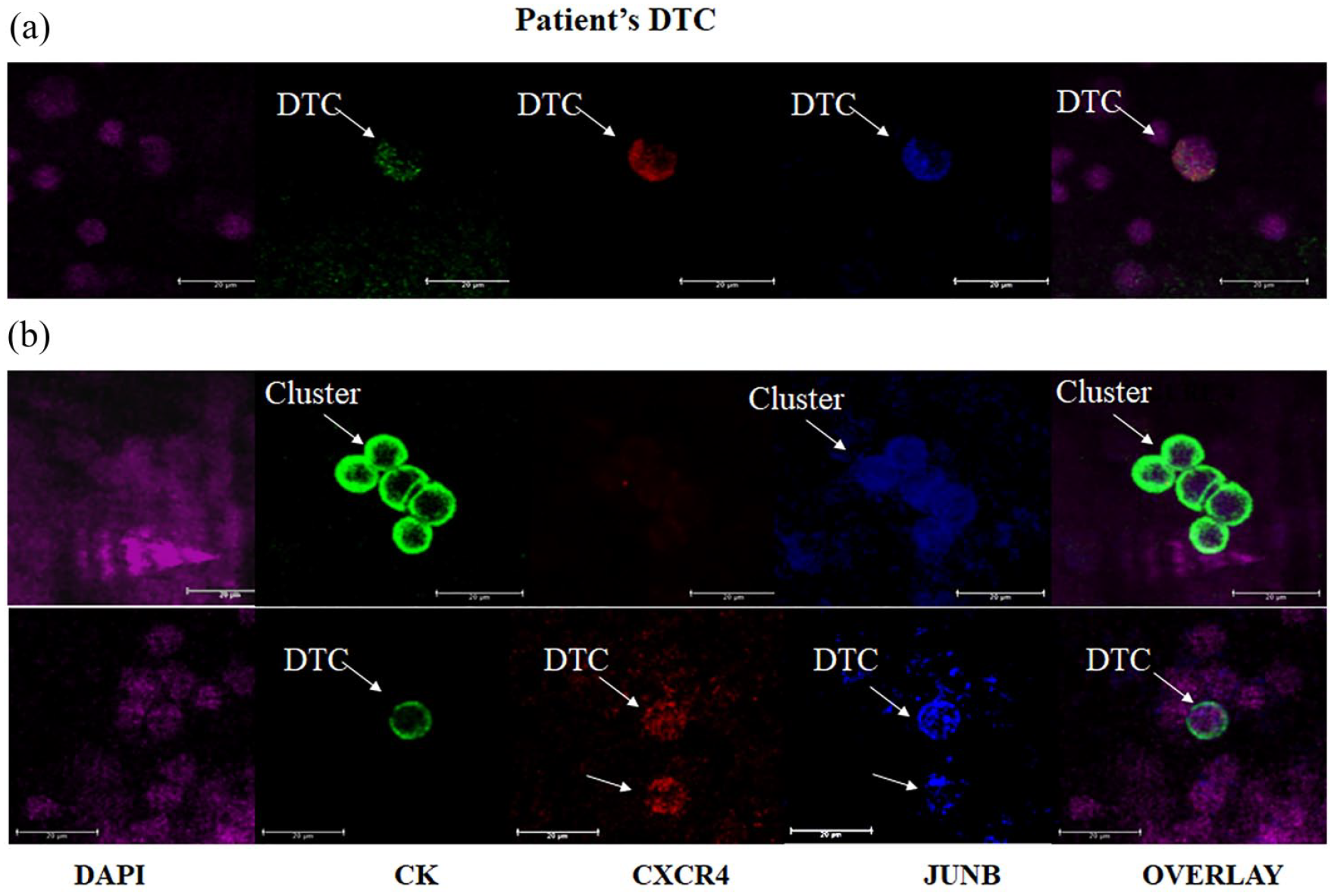
Expression of cytokeratin (green), CXCR4 (red), JUNB (blue), and DAPI (purple) in DTCs isolated from BC patients. (a) Representative confocal laser scanning images of a patient’s DTC expressing CK, JUNB, and CXCR4. Samples were triple stained with pan-cytokeratin A45-B/B3-zenon-conjugated antibody (green) along with CXCR4 anti-rabbit, JUNB anti-mouse antibodies, and DAPI. (b) Representative confocal laser scanning images of two different phenotypes observed in the same patient. The first panel shows a DTC cluster with the (CXCR4–JUNB+CK+) phenotype, while the second panel shows a single DTC expressing the (CK+CXCR4+JUNB+) phenotype.
The (CXCR4+JUNB+CK-)-phenotype could potentially represent tumor cells in EMT, based on their cytomorphological characteristics (Figure 3b, second panel); however, further investigation is needed to answer this question. Consequently, these cells were not included in the analysis.
Clinical relevance
Evaluation of patients’ clinicopathological characteristics revealed that patients with lobular histological subtype experienced lower OS [(Kaplan–Meier, p = 0.002, 69 months (range 43–111) versus 97.5 months (range 41–135), (supplementary Figure S1a)] and DFS [Kaplan–Meier, p = 0.013, 51 months (range 42–111) versus 93 months (range 10–135), (supplementary Figure 1b)].
(CXCR4–JUNB+CK+)-expressing DTCs were significantly statistically correlated (p = 0.044, Spearman’s rho analysis) with the lobular carcinoma subtype.
Finally, the absolute number of DTCs was related to lower OS [(Cox regression, p = 0.026, hazard ratio: 1.025)]. Examination all the different DTC-phenotypes showed that patients harboring the (CXCR4+JUNB+CK+)-phenotype had lower OS compared with patients without this phenotype [(Cox regression, p = 0.023, hazard ratio = 1.026) (supplementary Figure 1d)]. In addition, Kaplan–Meier analysis documented that patients harboring more than 15 (CXCR+JUNB+CK+) DTCs experienced shorter OS compared with patients with fewer, or no, double-positive cells [p = 0.003, 79 months (range 65–93) versus 118 months (range 107–128), supplementary Figure 1].
Discussion
The current study demonstrates for the first time that DTCs in the BM of primary, nonmetastatic BC patients frequently express (CXCR4+JUNB+CK+) cells, and that the presence of these DTCs is associated with unfavorable clinical outcomes. While this phenotype was observed exclusively in about 41% of patients, none of the patients exhibited exclusively (CXCR4–JUNB–CK+)-expressing DTCs. However, there was a phenotypic heterogeneity in DTCs, since most patients harbored both (CXCR4+JUNB+CK+) and (CXCR4–JUNB+CK+) cells. Furthermore, DTC clusters observed in one patient presented as negative for CXCR4 and positive for JUNB, whereas single DTCs in the same aspiration expressed higher levels of CXCR4. Finally, based on their cytomorphological characteristics (nuclear/cytoplasmic ratio, number of nuclei, etc.), (CXCR4+JUNB+CK–) cells detected in some cases could potentially represent tumor cells undergoing EMT.
The presence, as well as the persistence, of DTCs in primary and locally advanced BC is a poor prognostic factor, and has been associated with reduced PFS and OS.4–8 While some DTCs have been identified as CSCs, it is well known that DTCs are nonproliferating cells, and that they differ from the corresponding primary tumor with regard to HR and HER2 expression.5,9,10 These findings explain why chemotherapy, as well as antihormonal or HER2-targeted therapies, are not able to eliminate these cells. In order to find effective therapies, new targets on DTCs have to be identified so that patients can be treated accordingly, and allowing identification of patients at higher risk of relapse.
In this study, we examined CXCR4 and JUNB expression in DTCs. Both molecules are related to metastatic progression.15,20,21 Quantification of CXCR4 in DTCs revealed that the intensity of this receptor was statistically significantly higher than in BM cells, implying that CXCR4 could potentially help to distinguish tumor cells in the BM of BC patients. Interestingly, this observation is in line with previous studies by our group showing that CXCR4 is highly expressed in CTCs. 14 In particular, CXCR4-positive CTCs were detected in 92% of the patients in this study. These findings are strengthened those of Salgia and colleagues, 26 who demonstrated that a baseline CXCR4-positive CTC level of ⩾7% in extensive-stage disease small cell lung cancer was prognostic for shorter PFS, and CTC counts ⩾6 at baseline, and after 1 cycle of treatment, were prognostic for shorter PFS and OS, respectively. Interestingly, baseline CXCR4 expression in tumor tissue was not prognostic of survival or predictive of LY2510924 (an CXCR4 antagonist) treatment response. 26 In addition to these findings, it has been shown using quantitative polymerase chain reaction (qPCR) that CXCR4 expression in CD45-depleted patient blood was correlated with increased lymph node spread (>3LN), 18 and meta-analysis studies revealed that DFS and OS were negatively correlated with CXCR4 expression in primary tumors.27,28
For JUNB, a transcription factor upregulated during EMT,21,22,29 the current study documented a statistically significant upregulation in DTCs compared with all BC cell lines and hematopoietic cells (BM), implying that JUNB could also serve as a reliable marker to identify patients harboring DTCs. This assumption is further confirmed by the detection of CXCR4+JUNB+double-positive but CK– negative cells in 50% of the patients showing tumor cytomorphological characteristics, implicating an EMT phenotype. This suggestion is not only reinforced by bibliographic data, indicating an upregulation of these markers during EMT, but also by our and other studies demonstrating the presence of CTCs in this patient group.30–32 However, further studies are needed to address this question.
The triple-positive phenotype (CXCR+JUNB+CK+) was observed in 90% of patients. In addition, the percentage of triple-positive (CXCR4+JUNB+CK+) cells (72%) among the total number of examined DTCs was also significantly enhanced compared with the other phenotypes. Interestingly, none of the patients harbored exclusively (CXCR4–JUNB–CK+) DTCs.
The analysis of patients’ clinical characteristics determined that the presence of JUNB-positive DTCs (CXCR4–JUNB+CK+) was related to the lobular histological subtype (p = 0.044) of the primary tumor. The fact that this subtype was associated with poorer OS in this cohort of patients (p = 0.002) suggested that expression of JUNB could be related to more aggressive disease. The data reported in the current study are in line with previous studies showing the role of JUNB in cancer development.19–21 Moreover, it has been demonstrated that JUNB is induced by ALK-NPM, which participates in the mTOR pathway, 33 and is required for cell cycle re-entry after quiescence, cooperating with c-jun for the development of fibrosarcoma. 34 In addition, there are some recent data indicating that JUNB is implicated in the earliest events of development of resistance to kinase inhibitors in BC. 24 Combining all these data with our findings strongly supports the view that JUNB plays a critical role in cancer progression. Statistical analysis of patient survival in our cohort confirmed this assumption, showing that patients harboring DTCs belonging to the (CXCR+JUNB+CK+)-phenotype, experienced poorer OS (p = 0.023). Interestingly, it has been considered recently that both CXCR4 and JUNB could serve as therapeutic targets, especially CXCX4. 15 In this regard, blockade of CXCR4 with Plerixafor in the MMTV-PyMT model of BC selectively reduced M2 tumor-associated macrophages after chemotherapy, thereby reducing tumor burden. 35 It has further been reported that the CXCR4 antagonist AMD3465 inhibited BC growth and metastasis by acting on tumor cells, as well as on immune cells present in the tumor microenvironment. 36 In triple-negative BC (TNBC), although a therapeutic response of a fully human CXCR4 antibody correlating with the expression of CXCR4 has been demonstrated, 37 a very recently published study showed that CXCR4 inhibitors did not reduce tumor growth in TNBC, but even increased metastatic spread, although they efficiently impaired tumor growth and metastasis in HER2-positive BC. 38
For JUNB, knockout in metastatic cells significantly suppressed invasion and migration, and repressed the incidence of lung metastases, which resulted in prolonged survival in vivo. These data suggest that downregulation of JUNB might be an effective treatment strategy for patients with head and neck squamous cell carcinoma. 39
Conclusions and limitations of the study
CXCR4 and JUNB were frequently overexpressed in DTCs isolated from early stage BC patients, which could potentially be related to patient clinical outcomes. Considering the observation that all patients expressed at least one of these molecules in their DTCs, CXCR4 and JUNB might be promising markers to identify BC patients at higher risk for relapse, and patients who should be considered for closer follow up, or even for additional targeted therapy.
However, we would like to emphasize that this is a small ‘proof of principle’ study, which has to be confirmed in a larger, as well as in an independent, patient cohort. Due to the small number of patients, and the preliminary nature of this work, our results are exploratory only, also with regard to statistical analysis. Further studies should include other BC subtypes (HER2-positive, TNBC) to elucidate the relevance of these findings, and to confirm the clinical utility of these markers.
Supplemental Material
SUPL_FIGURE_1 – Supplemental material for CXCR4 and JUNB double-positive disseminated tumor cells are detected frequently in breast cancer patients at primary diagnosis
Supplemental material, SUPL_FIGURE_1 for CXCR4 and JUNB double-positive disseminated tumor cells are detected frequently in breast cancer patients at primary diagnosis by Galatea Kallergi, Oliver Hoffmann, Ann-Kathrin Bittner, Lina Papadimitriou, Spyridoula D. Katsarou, Nefeli Zacharopoulou, Michalis Zervakis, Stelios Sfakianakis, Christos Stournaras, Vassilis Georgoulias, Rainer Kimmig and Sabine Kasimir-Bauer in Therapeutic Advances in Medical Oncology
Footnotes
Author contributions
Development of methodology: G. Kallergi, Acquisition of data acquired and managed patients, provided facilities: G. Kallergi, S. Kasimir Bauer, C. Stournaras, G Georgoulias.
Analysis and interpretation of data (e.g. statistical analysis, biostatistics, computational analysis): G. Kallergi, M. Zervakis, S. Sfakianakis, Spyridoula D. Katsarou, N. Zacharopoulou, L. Papadimitriou, O. Hoffmann, AK Bittner, R. Kimmig.
Writing, review, and revision of the manuscript: G. Kallergi, C. Stournaras, V. Georgoulias, S. Kasimir Bauer.
Administrative, technical, or material support (i.e. reporting or organizing data, constructing databases): G. Kallergi, S. Kasimir Bauer.
Study supervision: G. Kallergi, S. Kasimir Bauer.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and publication of this article: This research has been cofinanced by the European Regional Development Fund of the European Union and Greek funds through the Operational Program Competiveness Entrepreneurship and Innovation, under the call RESEARCH–CREATE–INNOVATE (project code:T1EDK-04489 e-MASS). Partial supported was received from the Cretan Association for Biomedical Research (CABR) and the Hellenic Oncology Research Group (HORG).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
