Abstract
VIPoma is a rare neuroendocrine tumor (NET) with a high potential to develop hepatic metastases and poor prognosis. The primitive tumor is nonsymptomatic and usually localized within the pancreas. Liver metastasis drives the prognosis and induces profuse watery diarrhea or renal failure. We herein present severe renal failure or diarrhea in two patients hospitalized in intensive care justifying emergency treatment of liver metastasis. The two patients experienced severe diarrhea due to a hypersecretion of vasoactive intestinal peptide (VIP) from liver metastasis released into the blood circulation. Therapeutic management was discussed and liver transarterial chemoembolization (TACE) was performed with chemotherapy-loaded embospheres, which cause necrosis of tumor lesions. TACE controlled the hormonal syndrome and made patients eligible for curative surgery. Tumor necrosis occurred and VIP levels collapsed. Surgery was performed in one of the two cases after TACE and the patient was considered in remission. Both patients were still alive after 3 years of follow up. Thus, TACE is feasible and appears to be an effective emergency treatment in patients with a VIP-hormonal syndrome due to liver metastases. Despite the biological disorder due to the hormonal secretion, an aggressive approach is warranted in VIP liver metastasis.
Keywords
Introduction
Vasoactive intestinal polypeptide-secreting tumor (VIPoma) is a rare neuroendocrine tumor described first by Priest and Alexander in 1957 [Priest and Alexander, 1957]. Most cases arise within the pancreas and appear as a large single tumor measuring >3 cm [Perry and Vinik, 1995]. VIPoma is a functional tumor, with potential malignancy characterized by slow and insidious tumor growth. Its prognosis depends on its differentiation (Ki-67 and mitotic index), tumor growth speed and metastatic extension [Strosberg et al. 2009; Halfdanarson et al. 2008]. Advanced stage VIPoma with hepatic metastases is mainly revealed by profuse diarrhea, also called ‘pancreatic cholera’, inducing a dehydration, severe hypokalemia and functional renal insufficiency [Verner and Morrison, 1958; Said and Faloona, 1975]. In those emergency situations, a multidisciplinary approach to therapy is warranted. Surgery appeared to be the only curative option but somatostatin analogs, by inhibiting hormone hypersecretion and preventing tumor growth, are currently used [Sundin et al. 2009]. In nonresectable liver metastases, a debulking surgery has been discussed for hormonal syndrome control [Leavey et al. 1995]. Liver transarterial chemoembolization (TACE) has been identified as a new therapeutic option in a nonemergency approach and leads to the resolution of VIPoma-induced symptoms [Sundin et al. 2009]. We herein report on two patients with life-threatening VIPoma due to hypokalemia induced by functional liver metastases.
Case presentation
Case 1
A 60-year old man attended a tertiary hospital for chronic diarrhea. He had a past history of gastroesophageal reflux and grade I esophagitis. Abdominal computerized tomography (CT) scan reported three hypervascularized liver lesions up to 100 mm size (Figure 1) and a 23 mm hypodense and poorly vascularized lesion in the tail of the pancreas. Upper gastrointestinal ultrasonography (EUS) suggested a metastatic pancreatic neuroendocrine tumor. Plasma levels were 578 ng/ml (N < 86 ng/ml), 260 ng/l (N < 65 ng/l), 664 pg/ml (N < 18 pg/ml) for chromogranin A, VIP, and serum calcitonin, respectively. In-octreotide scintigraphy (Octreoscan®) showed hyperfixation in the cephalic pancreas and in the liver. Liver biopsy confirmed the diagnosis of a grade II well-differentiated functional pancreatic neuroendocrine tumor. The mitotic index, Ki67, was 12%. The patient received a somatostatin analog and systemic chemotherapy with streptozotocin (500 mg/m2 J1–J5) and epirubicin (65 mg/m2 J1–J22) as first-line treatment. At month 2, no objective response to chemotherapy was identified and the patient experienced profuse watery diarrhea associated with vomiting, significant weight loss resulting in a severe dehydration with a functional renal failure (creatinine clearance = 12 ml/min), metabolic acidosis and hypokalemia (K+ = 2.6 mM). Considering the lack of efficacy of the conventional treatment and the metabolic disorders, a hepatic TACE with streptozotocin-loaded embospheres was performed without any complications. The patient showed normalization of renal function, and improved digestive symptoms within a week. The VIP level decreased to 82 ng/l. At 1 month, an abdominal CT scan revealed central necrosis and tumor response (Figure 2) allowing, at month 4, a splenopancreatectomy and a liver metastatic resection with curative intent. The pathological examination identified a complete response of the liver metastases. After 4 years, the patient remains free of tumor.
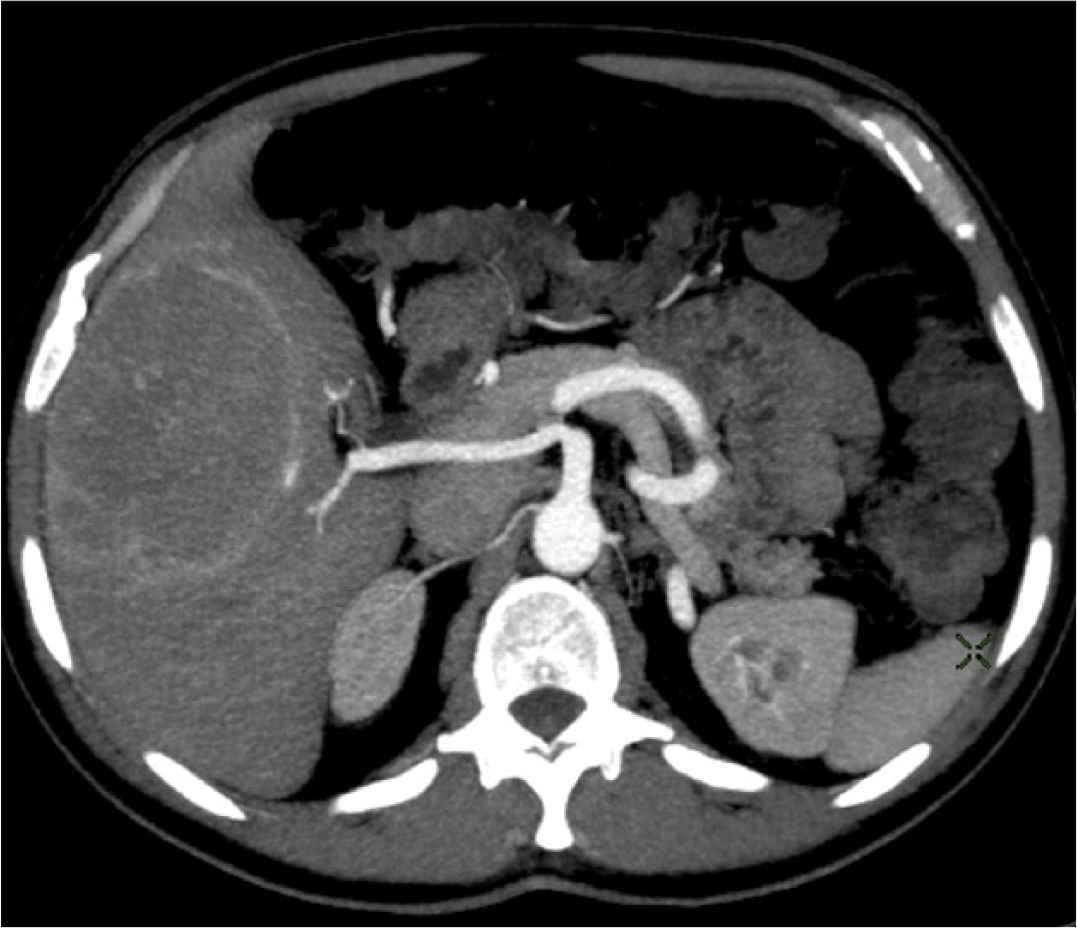
Computed tomography with intravenous contrast identified liver metastasis prior transarterial chemoembolization. The right liver metastases vascularized by an accessory right hepatic artery.

One month after TACE, computed tomography identified a necrosis of metastasis (with intravenous contrast).
Case 2
A 64-year old man was hospitalized in intensive care for hypovolemic shock, induced by watery diarrhea with acute renal failure and hypokalemia. A CT scan showed a 6 cm large lesion located in the tail of the pancreas and no hepatic lesions. Biopsy performed by EUS identified a grade II well-differentiated pancreatic neuroendocrine tumor. Octreotide scintigraphy and positron emission tomography identified an isolated pancreatic fixation. Blood tests found a calcitonin, VIP and chromogranin A hypersecretion with levels of 482 pg/ml (N < 18 pg/ml), 180 ng/l (N < 65 ng/l) and 257 ng/ml (N < 86 ng/ml), respectively. A curative distal pancreatectomy was realized and the diagnosis of VIPoma was confirmed. At year 1, the patient experienced diarrhea, dehydration and weight loss. Liver recurrence was identified on hepatic magnetic resonance imaging and Octreoscan® and no hepatic surgery remained possible. A selective hepatic TACE with streptozotocin-loaded embospheres was performed. At 1 month, the procedure was a therapeutic success with necrosis and decreased hepatic tumor volume. Post-operative VIP level was normal (46 ng/l). No recurrence was observed and the patient has remained symptom free. He did not experience tumor progression at 1 year.
Discussion
We report on two cases of symptomatic VIPoma treated with hepatic TACE. VIPoma is a rare subtype of pancreatic islet cell tumors with an incidence of 1 in 10 million people per year [Ghaferi et al. 2008]. Diagnosis is generally made in the fourth decade, mainly in females. VIPomas are predominantly localized in the pancreatic tail and are diagnosed at an advanced stage [Burns and Edil, 2012]. The management of VIPoma remains challenging [Song et al. 2009]. No recommendations exist for the therapeutic strategy in symptomatic patients. Considering that hepatic lesions from a VIPoma are responsible for digestive symptoms such as diarrhea, TACE appeared as an option to control the VIP hypersecretion. The two cases presented in this review reinforce this hypothesis and both patients have avoided systemic chemotherapy (i.e. no patient experienced disease progression at year 1). On the other hand, systemic cytotoxic chemotherapy and somatostatin analogs have been proposed when surgery is not an option [Moertel et al. 1992; Harris et al. 1995]. Those treatments are associated with a high rate of toxicity and morbidity; thus, making TACE a contender for first-line therapy.
Symptomatic VIPoma may induce massive diarrhea and metabolic disorders. VIPoma produces an important quantity of VIP released into the bloodstream. Clinical and biological manifestations are due to an excessive and unregulated VIP secretion. Plasma levels of VIP higher than 50 pmol/l have been identified to induce functional disorder [Perry and Vinik, 1995]. In our cases, we observed an elevation of calcitonin, serotonin, and VIP which decreased after TACE. High levels of VIP resulted in electrolyte over-secretion and water excretion [Bloom et al. 1973]. The decreased VIP level below 50 ng/l resulted in a symptom-free tumor in both cases. Our treatment approach in both cases reinforced the usefulness of an ‘emergency’ therapeutic option to decrease the VIP level and control patient symptoms, as has been proposed previously [Madeira et al. 1998]. In addition, there has been a recent demand among physicians for a market of efficacy to be identified for any new therapeutic option in oncology. We propose that the rapid decrease of the VIP below 50 ng/l following TACE might be considered as a marker of efficacy.
In our cases, TACE was considered as a preferred option compared with surgery. The surgical procedure in metastatic VIPoma includes an aggressive surgical therapy and is recommended when all disease can be safely removed [Song et al. 2009]. Surgery is commonly accepted as the standard treatment for localized VIPoma and resectable metastases. It is still the only curative treatment and should be considered even in case of metastases either as a curative intent or as a debulking therapy. Patients with a total curative surgery observed a 5 year 80% survival rate [Frilling et al. 2009]. Similarly, cytoreductive surgery should be considered to treat symptomatic patients, especially those with suboptimal palliation via medical therapy alone [Pavel et al. 2012]. The cytoreductive option has been identified as an effective to control the hormonal syndrome [Frilling et al. 2009]. TACE appears to be an efficient and well tolerated option to control symptomatic NETs [Frilling et al. 2009] and is considered as an option in hypersecretion syndrome [Ellison and Edil, 2012]. Surgery was not considered in our cases because of the intensity of the diarrhea, the imputability of hepatic lesions and the morbidity of such options compared with TACE.
Surgery permitted an overall control of the hormonal syndrome and allowed a curative treatment. Tumor necrosis induced by hepatic TACE offered a resolution of symptoms in our cases of metastatic VIPoma. Case and colleagues reported successful disease control of a patient with a VIPoma with progressive liver metastases, severe diarrhea and treated by hepatic artery embolization [Case et al. 2002]. Hepatic artery embolization [Markowitz, 1952] was developed to reduce symptoms due to advanced liver tumors and particularly hepatic metastases. In our cases, TACE was performed to obtain tumor reduction and control the hormonal syndrome. In the first case, hepatic embolization helped to reduce the large tumor size by allowing resection of the liver metastases. The complete response of the metastasis identified on pathological examination in the first case strengthened the efficacy of TACE in first-line treatment. The complete response did not play for a complement surgery. In our second case, no surgery was performed and no recurrence was identified during follow up.
Medical treatments do not appear to be sufficiently effective in the ‘emergency’ situation [Perry and Vinik, 1995]. Somatostatin analogs produce a partial reduction of VIP levels, and reduce tumor growth [Harris et al. 1995]. Advanced VIPomas with massive liver invasion requires additional treatment. Guidelines consider systemic cytotoxic chemotherapy with a combination of streptozotocin and adriamycin as the standard treatment for progressive or symptomatic unresectable neuroendocrine tumors, including VIPomas [Moertel et al. 1992]. Conventional chemotherapy is an alternative to traditional hepatic-directed therapies including resection, ablation, and hepatic artery embolization but it fails to control hormonal syndrome [Ghaferi et al. 2008]. The efficacy of chemotherapy deserves at least one or two cycles and appears to be clinically relevant at the end of the first month. Our cases pave the way for TACE as an ‘emergency’ option to obtain a rapid hepatic tumor regression. This option may be considered as the first option in symptomatic patients with metastatic lesions from a VIPoma.
The initial poor prognosis of VIPomas and the aggressiveness of liver metastases appears to be counterbalanced by ‘emergency’ TACE. Physicians should be aware that TACE might be used as a downsizing option prior to surgery in neuroendocrine tumors.
Footnotes
Acknowledgements
JD had the original idea for the research, collection of data, writing, responsibility for the first draft of the manuscript and proofreading. CL, MEH, and HG participated in data collection and proofreading. PR had the original idea for the research and participated in proofreading. RC took the responsibility for the first draft of the manuscript and proofreading.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Coriat has acted as a paid consultant for Roche, Merck and Novartis. The other authors have no conflicts of interest to declare.
