Abstract
Background
This study investigated the effect of tranexamic acid (TXA) on postoperative outcomes in anatomic total shoulder (aTSA) or reverse total shoulder arthroplasty (rTSA) surgery.
Methods
We queried the TriNetX network for patients who underwent TSA from 2000 to 2025. Patients were 1:1 matched into aTSA or rTSA cohorts and subcategorized into TXA or non-TXA groups. Yearly trends in TXA utilization by TSA procedure were reported. Postoperative complications at 90 days and 2 years were assessed.
Results
TXA utilization increased 7% in aTSA and 11% in rTSA from 2020 to 2024. Within 90 days, TXA patients in both cohorts had a reduced risk of transfusions, acute post-hemorrhagic anemia, and hospital readmission. TXA patients in the aTSA cohort had a reduced risk of pulmonary embolism within 90 days. At 2 years, TXA patients in both cohorts had a reduced risk of transfusions, surgical site infection, pulmonary embolism, and hospital readmission. TXA patients in the rTSA cohort had a higher risk of periprosthetic loosening at both follow-ups.
Discussion
Patients receiving TXA had a significantly reduced risk of postoperative complications. TXA has a similar impact on outcomes in both TSA variations, and utilization should be considered as part of a comprehensive management strategy.
Keywords
Introduction
Total shoulder arthroplasty (TSA) is an orthopaedic procedure that has continued to steadily increase in prevalence in the United States over the past 3 decades.1–3 Within the two main variants of TSA, anatomic total shoulder arthroplasty (aTSA) has been mainly indicated for patients with primary osteoarthritis with a healthy rotator cuff, whereas reverse total shoulder arthroplasty (rTSA) has been reserved for patients with rotator cuff arthropathy.4,5 However, the indications for rTSA have broadened, as it is now also used to treat patients who would have traditionally been treated with an aTSA, leading to increased rTSA adoption.6–10 Therefore, there is a considerable need to evaluate how procedural variations influence postoperative outcomes.
Tranexamic acid (TXA) is a synthetic derivative of the amino acid lysine and acts as an anti-fibrinolytic agent by competitively inhibiting the conversion of plasminogen into plasmin.11–13 Due to the well-characterized complication of intraoperative blood loss during joint replacement procedures and subsequent need for transfusions, TXA has become a popular pharmacologic agent to minimize blood loss and the rate of postoperative transfusions in arthroplasty.14,15 Although TXA is well-studied in total hip arthroplasty (THA) and total knee arthroplasty (TKA), its role in TSA is less clearly defined.16–20 The risk of blood loss in TSA is less than in lower extremity arthroplasty, but transfusion rates in TSA have been reported to range from 4.5% to 43%, with patients undergoing rTSA having an even higher risk of transfusions and developing hematomas.6,21–26
Although prior research has established TXA as an effective pharmacological agent in TSA for reducing postoperative issues, the literature on the association between TXA and operative outcomes in TSA is still very limited. The scope of analysis in most of these studies was limited to hematological issues, including changes in blood loss, hemoglobin, and hematocrit, and none assessed the risks related to the prosthesis or surgery itself.17,18,27,28 As the use of TXA continues to grow in shoulder arthroplasty, it is imperative to further investigate its broader clinical impact, particularly across the aTSA and rTSA populations.29–32
The objective of this study was to investigate the utilization of TXA and the association of TXA administration with postoperative medical, surgical, and prosthetic complications following aTSA and rTSA. We hypothesized that TXA use would be associated with a lower risk of complications in both procedure types.
Methods
For this retrospective cohort study, we queried the TriNetX Research Network (Cambridge, Massachusetts, USA), a database that contains de-identified electronic health records and clinical data such as demographics, diagnoses (International Classification of Diseases, 10th Revision [ICD-10] codes), procedures (Current Procedural Terminology [CPT] codes), and medications of over 100 million patients across more than 50 healthcare organizations. The de-identification of the data is consistent with the Health Insurance Portability and Accountability Act Privacy Rule, and all data is reported as aggregate counts and statistical summaries to protect patients’ privacy and sensitive information. Due to the use of solely de-identified information in this study, it was exempt from Institutional Review Board approval.
The TriNetX database was queried on June 21, 2025 using CPT and ICD-10 codes for patients 18 years or older who underwent either an aTSA or rTSA from January 1, 2000 to January 1, 2025. Cohort 1 was defined as patients undergoing an aTSA, and Cohort 2 was defined as patients undergoing an rTSA. Both cohorts were then subcategorized into two groups as follows: (1) patients who were administered TXA within 24 h prior to their TSA procedure and (2) patients who were not administered TXA. Further information on cohort construction is available in Appendix 1.
Both cohorts were then matched 1:1 based on patient demographics (age, gender, ethnicity, race), existing comorbidities, and the use of antithrombotic medications using a propensity-score matching method, which is a statistical technique that aims to minimize the confounding bias by balancing the population in each cohort using logistic regression. Characteristics for propensity-matching were chosen based on their ability to influence outcomes of interest.2,25,33 Comorbidities were identified using the Elixhauser Comorbidity Index. 34 Statistical significance was defined as a two-tailed alpha value < 0.05.
This study analyzed the complications at the 90-day and 2-year follow-up points to investigate short- and long-term outcomes. Chi-square analysis was performed to determine the risk of surgical and prosthetic complications, including surgical site infections (SSI), wound dehiscence, prosthetic dislocation, prosthetic loosening, prosthetic fracture, and prosthetic joint infections (PJI), and medical complications including myocardial infarction (MI), pulmonary embolism (PE), deep vein thrombosis (DVT), hematoma, transfusions, shoulder stiffness, and hospital readmission. Additionally, rates of revision surgery were assessed at the 2-year mark. The index event, defined as the point at which analysis started for each patient, was the date of the aTSA or rTSA procedure. The outcomes between the two cohorts were reported through risk ratios (RR) and 95% confidence intervals (95% CI), using t tests and chi-squared tests with an alpha level of 0.05 for analysis. A Bonferroni correction was performed to account for multiple testing.
Before matching, our search results identified a total of 18,977 patients in the aTSA cohort, including 5636 patients in the TXA group (29.7%) and 13,341 patients in the non-TXA group (70.3%). For the rTSA cohort, our search identified 38,119 patients, including 13,963 patients in the TXA group (36.6%) and 24,156 patients in the non-TXA cohort (63.4%). Within both cohorts, the TXA group was significantly more likely to have used tobacco and have a diagnosis of diabetes mellitus, hypertensive diseases, or hypothyroidism (P < 0.001), and more likely to use apixaban, fondaparinux, heparin, or aspirin (P < 0.001) compared to the non-TXA group.
After matching, the aTSA cohort consisted of 5551 patients. There were no significant differences in terms of comorbidities or antithrombotic usage. The mean age was 66 years, and 50% were males. Across both the TXA and non-TXA groups, approximately 52% of patients had hypertensive diseases, 16% had diabetes mellitus, and 14% had hypothyroidism. The rTSA cohort consisted of 13,892 patients. There were also no significant differences in comorbidities or antithrombotic usage. The mean age was 71 years, and 57% were females. In both groups, 63% of patients had hypertensive disease, 23% had diabetes mellitus, and 19% had hypothyroidism. Comprehensive information on patient demographics, comorbidities, and medications before propensity matching is shown in Appendix 2, and characteristics after propensity matching are shown in Tables 1–4.
Anatomic total shoulder arthroplasty patient demographic characteristics (after match).
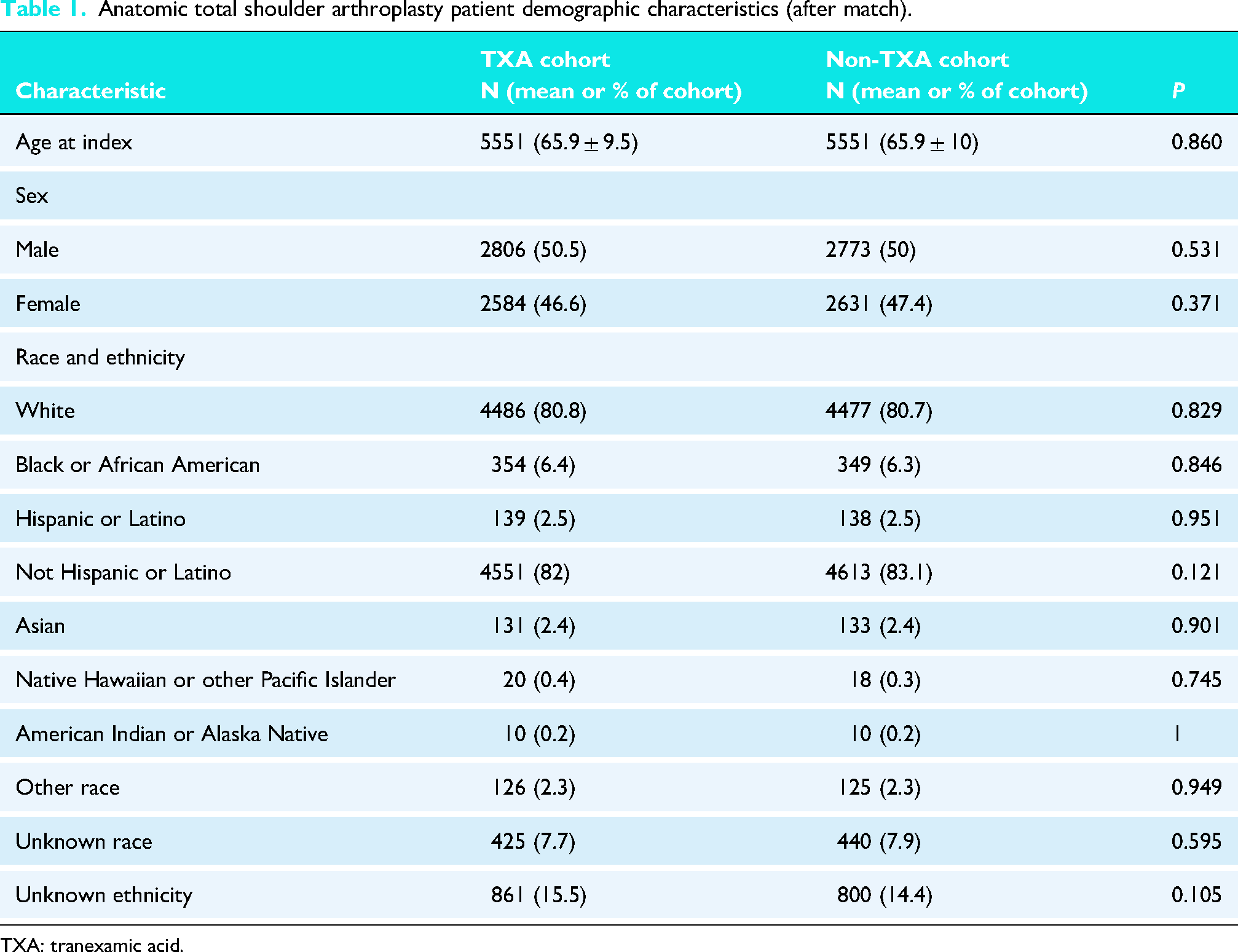
TXA: tranexamic acid.
Anatomic total shoulder arthroplasty patient comorbidities and medications (after match).
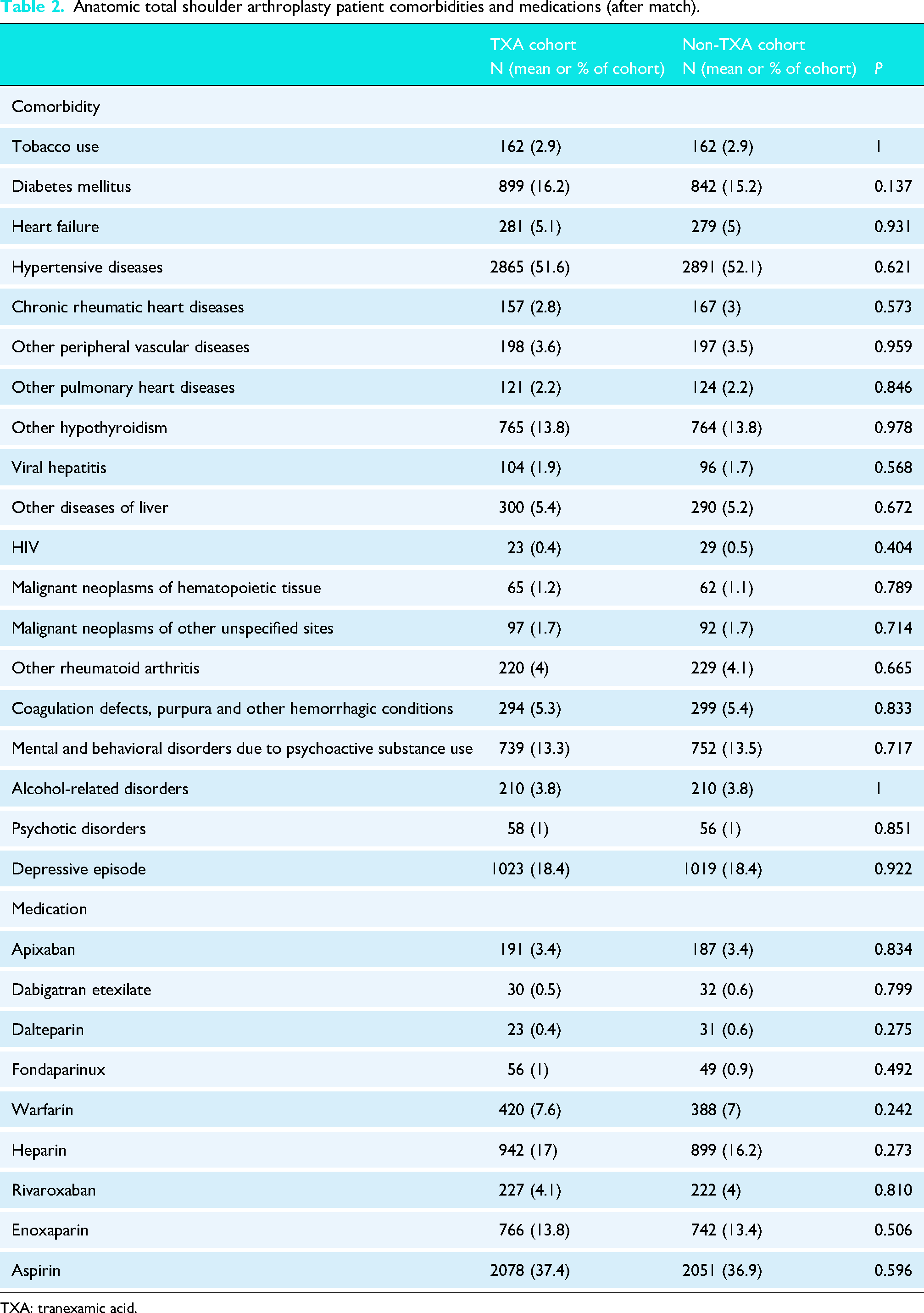
TXA: tranexamic acid.
Reverse total shoulder arthroplasty patient demographic characteristics (after match).
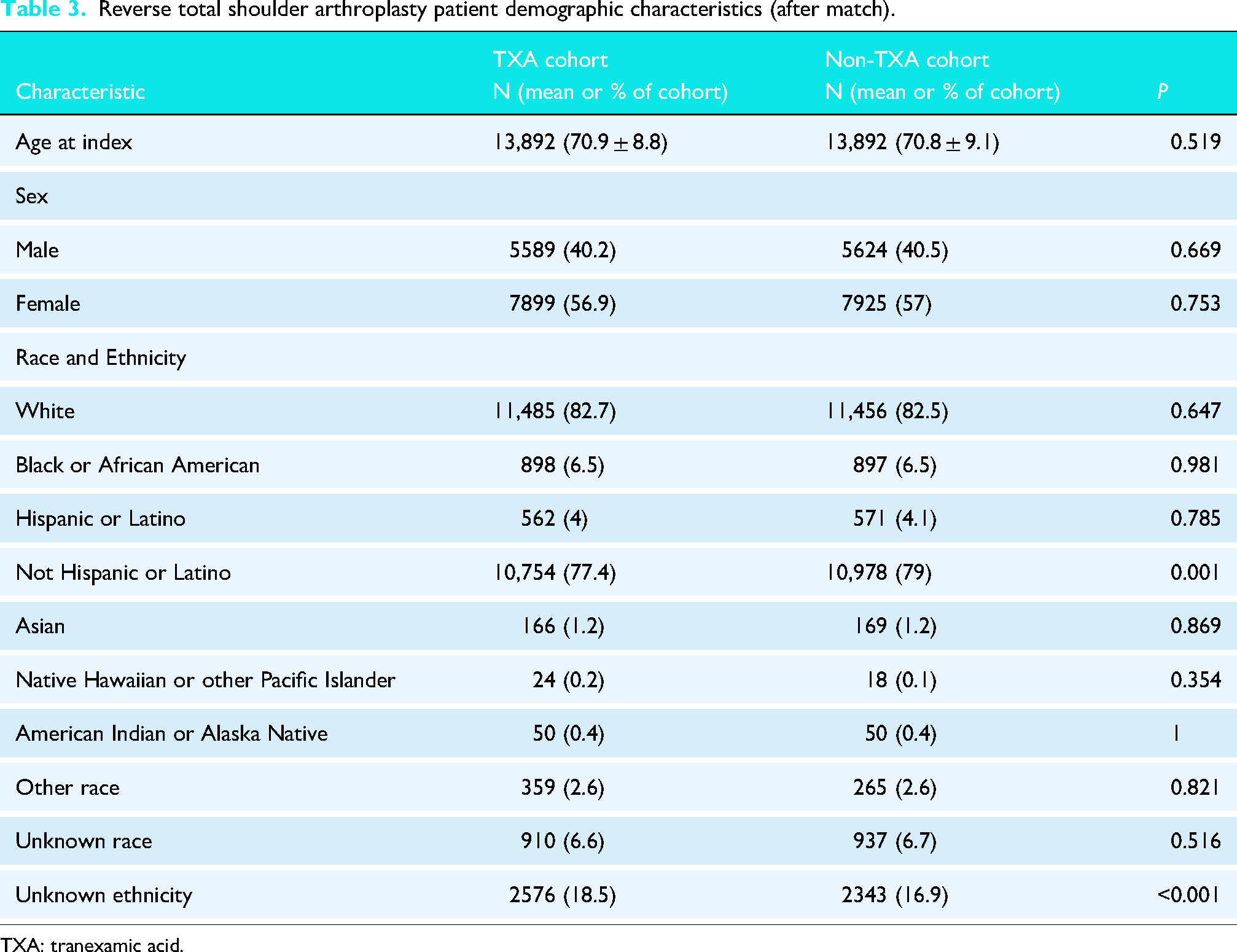
TXA: tranexamic acid.
Reverse total shoulder arthroplasty patient comorbidities and medications (after match).
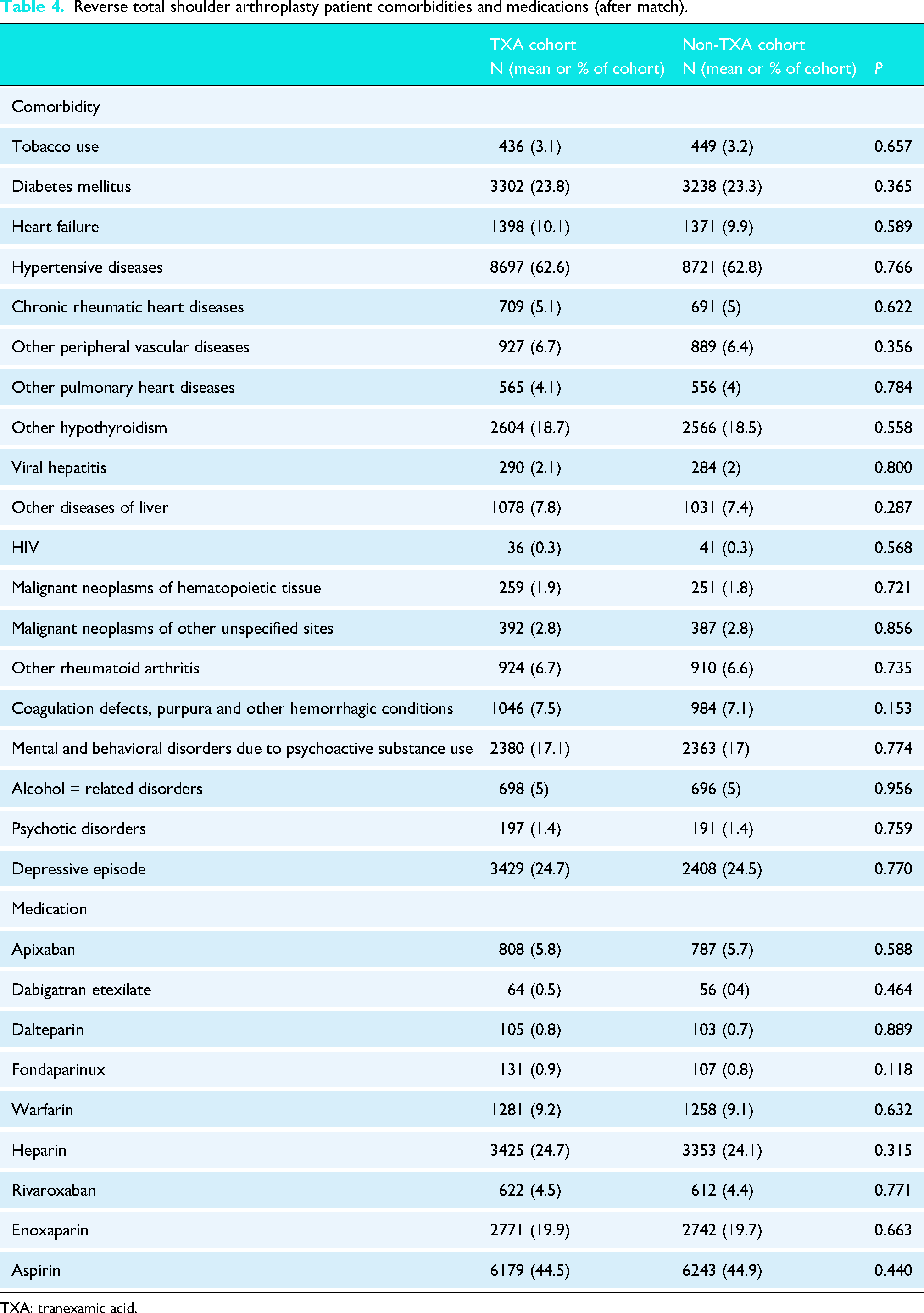
TXA: tranexamic acid.
Results
Anatomic TSA cohort
90-day postoperative complications
Within 90 days following the aTSA procedure, patients who received TXA were significantly less likely to have a transfusion (RR: 0.45; CI: 0.27, 0.75; P = 0.002), PE (RR: 0.61; CI: 0.38, 0.97; P = 0.036), acute post-hemorrhagic anemia (RR: 0.52; CI: 0.41, 0.65; P < 0.001), and hospital readmission (RR: 0.20; CI: 0.10, 0.39; P < 0.001). There were no significant differences between the TXA and non-TXA groups in terms of risk for SSI, prosthetic dislocation, prosthetic loosening, prosthetic fracture, PJI, shoulder stiffness, wound dehiscence, MI, DVT, or hematoma (Table 5).
Anatomic total shoulder arthroplasty complications at 90-day follow-up.
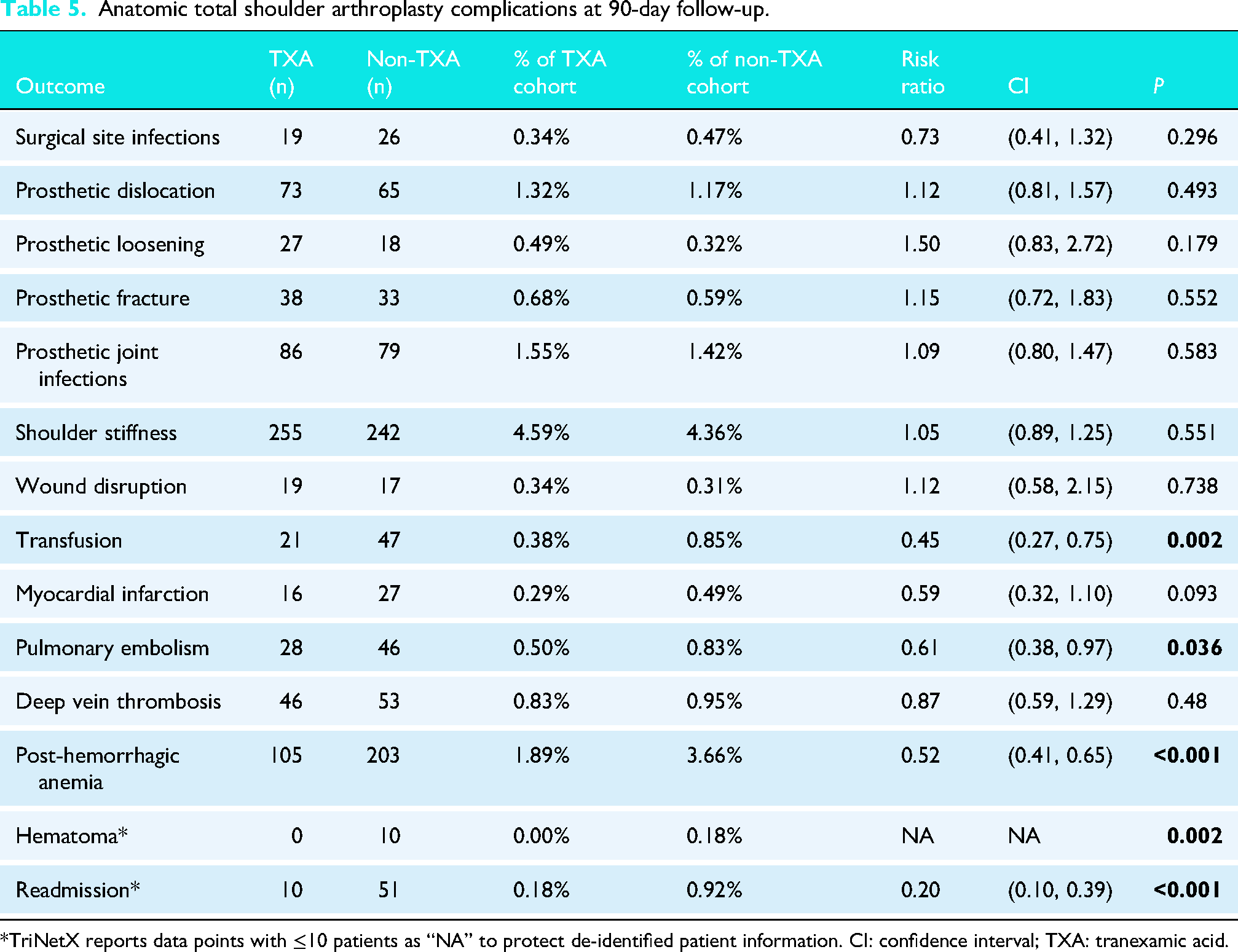
*TriNetX reports data points with ≤10 patients as “NA” to protect de-identified patient information. CI: confidence interval; TXA: tranexamic acid.
2-year postoperative complications
Within 2 years following the aTSA procedure, TXA-receiving patients were significantly less likely to have an SSI (RR: 0.65; CI: 0.46, 0.91; P = 0.011), transfusion (RR: 0.67; CI: 0.50, 0.91; P = 0.009), PE (RR: 0.59; CI: 0.44, 0.79; P < 0.001), and hospital readmission (RR: 0.19; CI: 0.12, 0.31; P < 0.001). There were no significant differences between the two groups in terms of risk for prosthetic dislocation, prosthetic loosening, prosthetic fracture, PJI, shoulder stiffness, MI, DVT, or hematoma. Additionally, the risk of revision was not significantly different between the TXA and non-TXA groups (Table 6).
Anatomic total shoulder arthroplasty complications at 2-year follow-up.
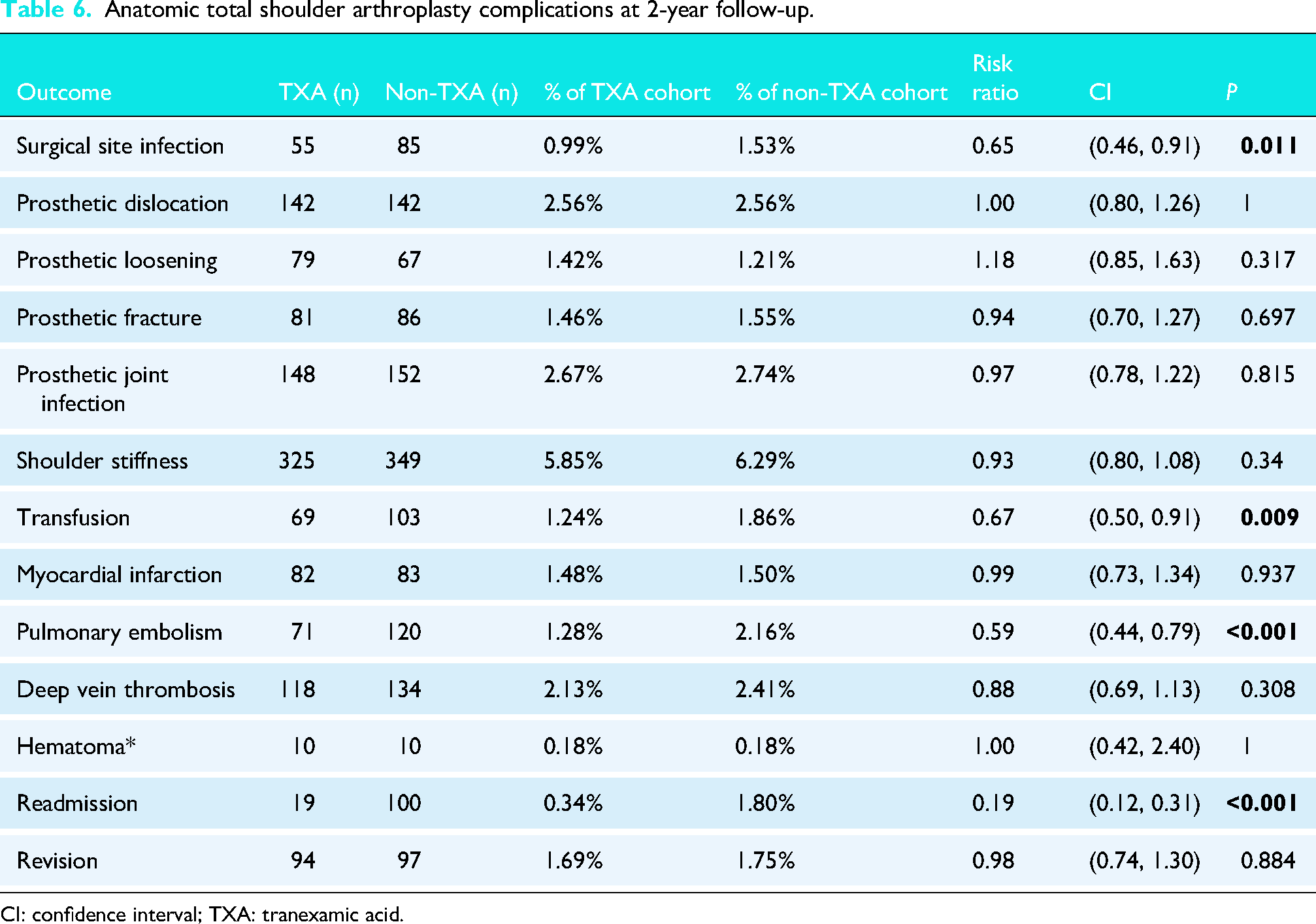
CI: confidence interval; TXA: tranexamic acid.
Reverse TSA cohort
90-day postoperative complications
Within 90 days following the rTSA procedure, patients receiving TXA were significantly less likely to have a transfusion (RR: 0.54; CI: 0.45, 0.64; P < 0.001), acute post-hemorrhagic anemia (RR: 0.61; CI: 0.54, 0.67; P < 0.001), and hospital readmission (RR: 0.11; CI: 0.07, 0.16; P < 0.001). However, the TXA group was at an increased risk for prosthetic loosening (RR: 1.36; CI: 1.01, 1.84; P = 0.045). There were no significant differences between the two groups in terms of risk for SSI, prosthetic dislocation, prosthetic fracture, PJI, shoulder stiffness, wound dehiscence, MI, PE, DVT, or hematoma (Table 7).
Reverse total arthroplasty complications at 90-day follow-up.
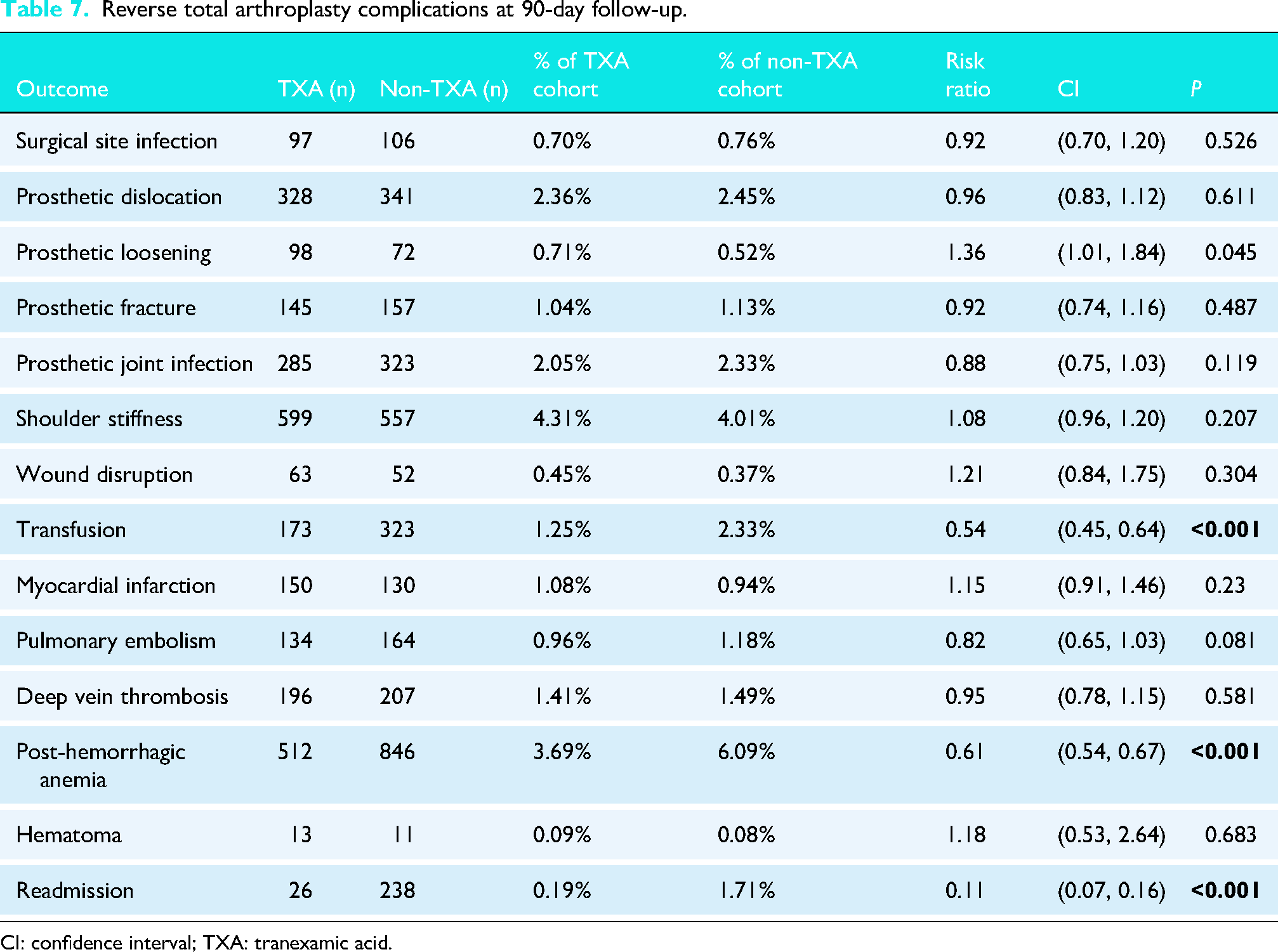
CI: confidence interval; TXA: tranexamic acid.
2-year postoperative complications
Within 2 years following the rTSA procedure, patients receiving TXA were significantly less likely to have an SSI (RR: 0.81; CI: 0.68, 0.96; P = 0.016), transfusion (RR: 0.61; CI: 0.53, 0.70; P < 0.001), PE (RR: 0.81; CI: 0.70, 0.95; P = 0.011), and hospital readmission (RR: 0.26; CI: 0.21, 0.33; P < 0.001). However, the TXA group was at increased risk of prosthetic loosening (RR: 1.25; CI: 1.04, 1.50; P = 0.017). There were no significant differences between the two groups in terms of risk for prosthetic dislocation, prosthetic fracture, PJI, shoulder stiffness, MI, DVT, hematoma, or revision surgery (Table 8).
Reverse total shoulder arthroplasty complications at 2-year follow-up.
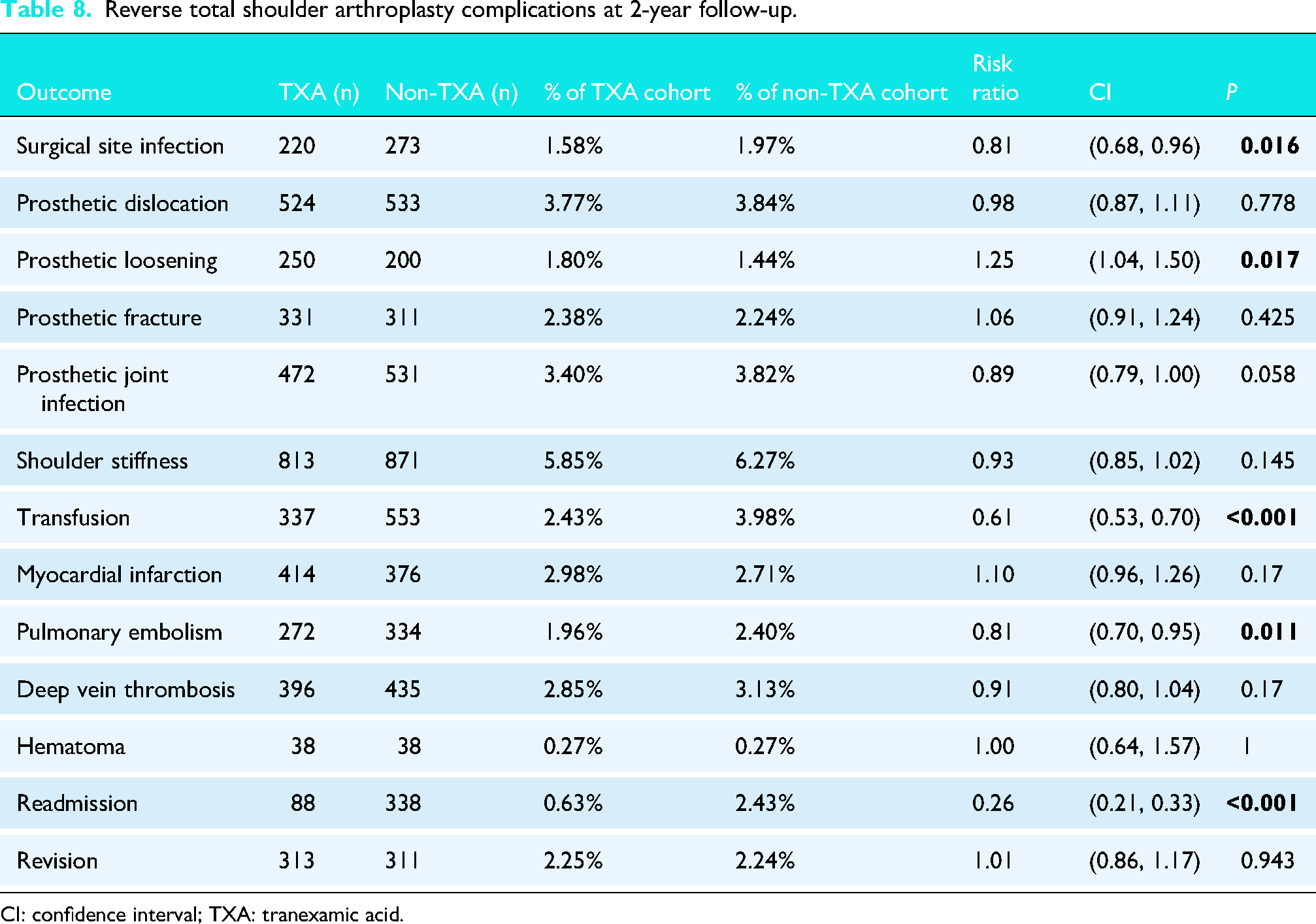
CI: confidence interval; TXA: tranexamic acid.
Trends in TXA usage
Our study uniquely tracked trends in TXA use by TSA technique from 2020–2024 (Figures 1 and 2). In this time period, TXA use increased from 43% to 54% in rTSA and from 44% to 51% in aTSA.
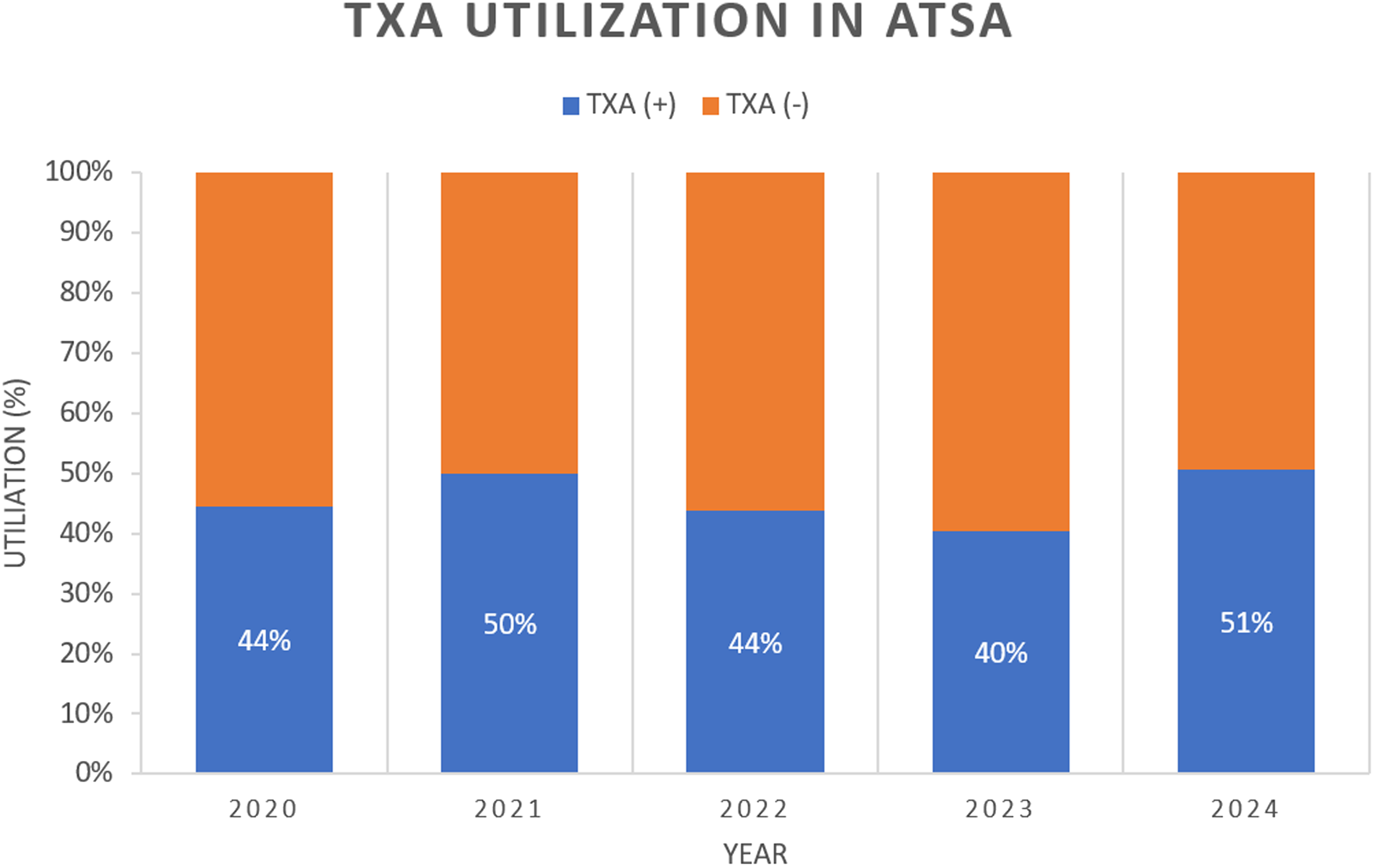
Rates of TXA utilization in aTSA procedures from 2020 to 2024.
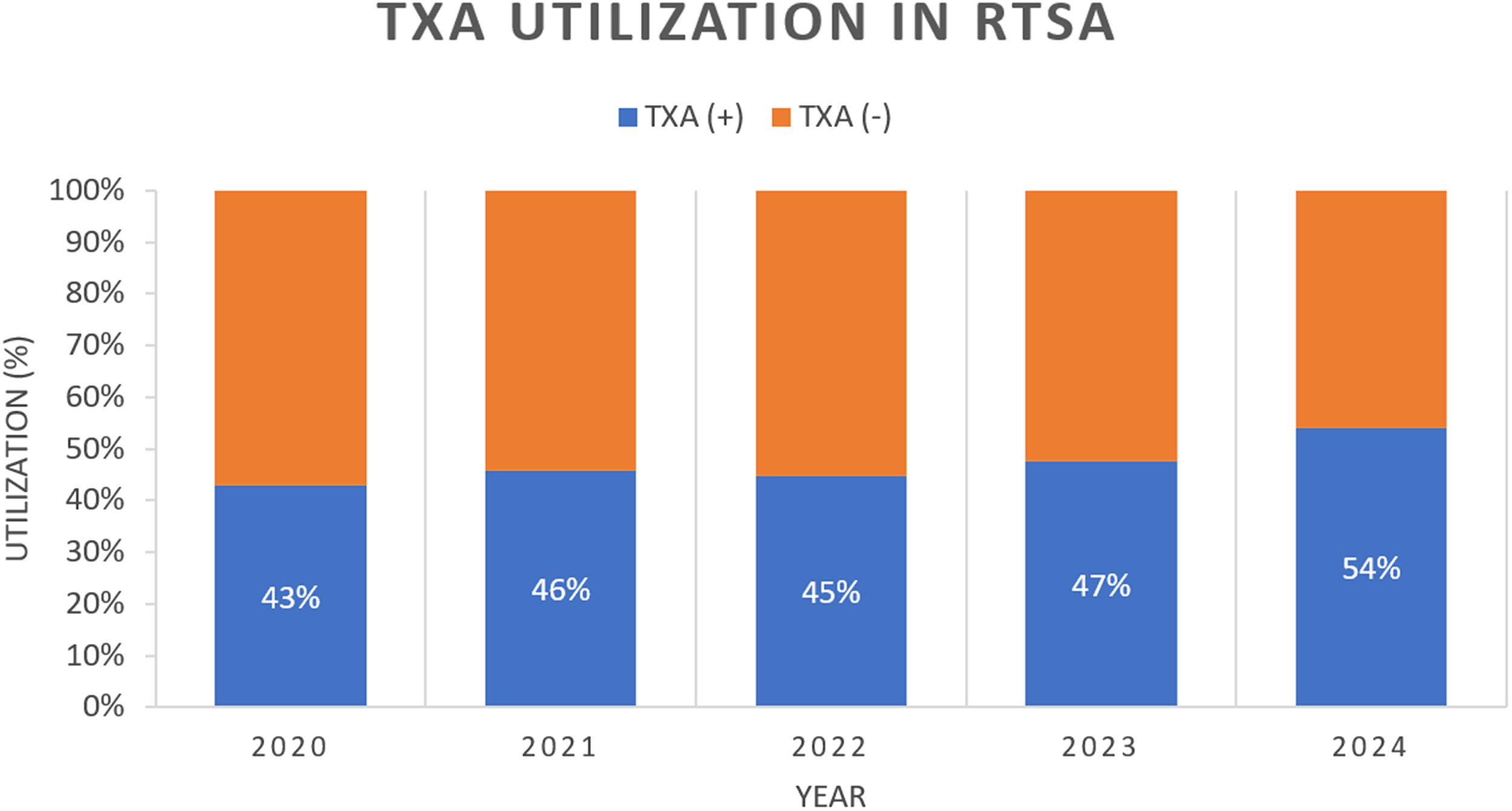
Rates of TXA utilization in rTSA procedures from 2020 to 2024.
Discussion
Due to the growing number of shoulder arthroplasties nationwide, expanding indications for TSA, and growing TXA use, there is a need to further characterize the risk profile of TXA in the context of shoulder replacements.2,35,36 To our knowledge, this study of 57,096 patients represents the largest analysis of the role of TXA in postoperative TSA complications stratified by procedure type. To date, only four studies have compared TXA outcomes in aTSA versus rTSA; however, their investigations were limited to hematological complications.17,18,27,28
Our study showed that at 90 days and 2 years postoperatively, TXA patients in both the aTSA and rTSA cohort had a lower risk of receiving a transfusion, having acute post-hemorrhagic anemia, and lower readmission rates compared to the non-TXA patients. At 2 years, patients receiving TXA in both cohorts were also at a lower risk for SSI and PE. These findings were consistent with our hypothesis and expand on the previous literature with the addition of surgical and prosthetic complications.
Regarding infection, TXA was associated with a significantly lower risk of SSI but an equivocal risk of PJI. Only one study has previously investigated the effects of TXA on SSIs in TSA and found no difference in infection rates between the TXA and non-TXA groups. 27 In contrast, TXA was associated with lower postoperative SSI risk after THA and TKA in a meta-analysis of 31 studies and a prospective study of 57 patients.37,38 The protective role of TXA against SSI in arthroplasty may be due to the decreased need for transfusions, lower rate of hematomas, or interaction with plasmin-mediated immune pathways.39–42 Additionally, TXA is established to protect against PJI in THA and TKA through possible antimicrobial properties and decreased blood loss, transfusion risk, wound drainage, and hematomas.36–38,43 However, this is not seen in TSA in our study or previous literature despite a decrease in transfusions and anemia.
The only significant prosthetic complication seen was increased risk of loosening in the TXA group following rTSA, a complication not described in previous THA studies.23,44 However, this is more likely attributable to older average age of rTSA patients who are already at a higher risk of postoperative prosthetic complications, or due to a higher survival rate in rTSA patients which allowed for the loosening.33,45 There were also similar levels of wound dehiscence in all groups. The effect of TXA on wound healing in arthroplasty is unclear, as some studies have reported a decrease in postoperative wound complications while others have found no difference.32,46,47
We found that the TXA patients in both TSA cohorts had significantly lower transfusion rates at all follow-ups. This is a finding consistent with the anti-fibrinolytic properties of TXA that allow it to mitigate blood loss during surgical procedures.48,49 These findings are notable, as the impact of TXA on transfusions in TSA was previously unclear. Three separate randomized control trials and a retrospective cohort study by Kissin et al. 28 found that the use of TXA in TSA was not associated with a decreased risk of postoperative transfusions.11,19,50 In contrast, three retrospective cohort studies reported that TXA usage was associated with a significant decrease in need for postoperative transfusion compared to non-TXA groups.35,51,52 Although all of these studies assessed the risk of transfusions, none stratified their results by the type of TSA procedure. Our study further contributes new evidence by demonstrating the risk associated with each subtype of TSA.
TXA was also associated with a decreased risk of PE and acute postoperative anemia in both cohorts. Our PE results are a novel finding, as no previous TSA study has demonstrated a PE benefit.28,35 Additionally, several meta-analyses have found TXA administration in TSA procedures to be associated with a decrease in total postoperative blood loss, need for transfusion, changes in hemoglobin and hematocrit, and length of hospital stay.29,53,54 Due to its efficacy in promoting hemostasis and decreasing blood loss, the use of TXA can play a crucial role in preventing postoperative anemia in both variations of TSA, as shown by our findings. There was no difference in the rates of MI or DVT across all cohorts. These findings are supported by prior studies reporting no effect of TXA on thromboembolic events in patients undergoing TSA.35,51
Our study found an increase in TXA utilization from 2020 to 2024 in both aTSA and rTSA procedures, aligning with trends of increased TXA popularity seen in other orthopaedic subspecialties such as lower extremity, spine, and orthopaedic trauma.14,22,43,46 Previous concern regarding the possible thromboembolic risks of TXA use, particularly in high-risk patients, has not been borne out in the literature, as multiple studies describe no increased risk of adverse outcomes after TXA use in high-risk patients after TSA.2,29,35 As the usage and efficacy of TXA continue to increase, we observe that this trend extends to both variations of TSA. Although further investigation is needed to fully understand the hazards in high-risk patient populations, this safety data may explain the steady increase in TXA use and offer reassurance to surgeons incorporating TXA into their practice protocols.
Overall, this study has strong clinical implications. Our results support the routine use of TXA in both rTSA and aTSA given its association with reduced transfusion rates, anemia, SSI, PE, and hospital readmissions without an observed increase in thromboembolic complications. As the population undergoing TSA continues to grow in size and medical complexity, TXA may offer a safe and effective option to improve perioperative outcomes and reduce healthcare utilization. These results may inform clinical protocols and preoperative decision-making, especially in our high-risk and elderly patients, where the benefits of meticulous hemostasis and reduced infection may be more pronounced.
The strengths of our study include our extensive outcome analysis, comprehensive statistical testing, and large sample size. Utilizing the propensity-matching technique and Elixhauser Comorbidity Index allowed our study to minimize the influence of external confounders and mitigate bias, while enhancing the internal validity of the results. However, limitations still exist. As with any other retrospective database analysis, the data used in this study comes from patient coding in electronic health records, which may be subject to error. Administrative patient coding for procedures, medications and comorbidities may be incorrectly entered or missed, which could impact the results of the study. While the Elixhauser Comorbidity Index helps adjust for comorbidities, unmeasured cofounders may still exist. In addition, TXA can be administered through a topical or intravenous method of delivery, and our study did not stratify results based on this, as TriNetX does not allow for that analysis. However, a retrospective cohort study by Budge 7 found both topical and intravenous TXA to be equivalently effective in controlling post-operative hemoglobin reductions in both rTSA and aTSA.
Further research on this topic is warranted. Prospective trials stratifying by topical versus intravenous TXA administration routes and dosing regimens are indicated to determine the most effective and safest approach. Further subgroup analysis in high-risk patients may also help refine specific risk profiles. Finally, incorporating patient-reported outcome scores and functional scores could offer further insight into whether the perioperative benefits of TXA affect short or long-term quality of life following TSA.
In this large, propensity-matched study, preoperative administration of TXA was associated with significantly lower rates of transfusion, anemia, SSI, PE, and hospital readmission following both anatomic and reverse TSA. These findings support the routine use of TXA as a safe and effective adjunct in TSA, with benefits that extend beyond hematologic outcomes to include reductions in infectious and thromboembolic complications.
As TSA volumes continue to rise and surgical indications broaden, TXA offers a cost-effective strategy to improve perioperative safety, enhance patient recovery, and reduce healthcare utilization. Future research should focus on patient outcome scores, the optimal route and dosing of TXA in TSA, and its safety in high-risk and medically complex patients. These efforts will be critical to inform evidence-based guidelines and ensure high-value care across all TSA populations.
Footnotes
Ethical approval
This study used anonymized data and was exempt from Institutional Review Board approval.
Informed consent
Not applicable, as this study used de-identified data from the TriNetX database.
Contributorship
JS, NRB, and JSS contributed to the conceptualization, formal analysis, investigation, methodology, writing of the original draft, and review and editing of the final draft. NRB and JSS additionally provided supervision. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
JSS
