Abstract
Aims:
To review the burden of allergic and infectious diseases and the evidence for a link to microbial exposure, the human microbiome and immune system, and to assess whether we could develop lifestyles which reconnect us with exposures which could reduce the risk of allergic disease while also protecting against infectious disease.
Methods:
Using methodology based on the Delphi technique, six experts in infectious and allergic disease were surveyed to allow for elicitation of group judgement and consensus view on issues pertinent to the aim.
Results:
Key themes emerged where evidence shows that interaction with microbes that inhabit the natural environment and human microbiome plays an essential role in immune regulation. Changes in lifestyle and environmental exposure, rapid urbanisation, altered diet and antibiotic use have had profound effects on the human microbiome, leading to failure of immunotolerance and increased risk of allergic disease. Although evidence supports the concept of immune regulation driven by microbe–host interactions, the term ‘hygiene hypothesis’ is a misleading misnomer. There is no good evidence that hygiene, as the public understands, is responsible for the clinically relevant changes to microbial exposures.
Conclusion:
Evidence suggests a combination of strategies, including natural childbirth, breast feeding, increased social exposure through sport, other outdoor activities, less time spent indoors, diet and appropriate antibiotic use, may help restore the microbiome and perhaps reduce risks of allergic disease. Preventive efforts must focus on early life. The term ‘hygiene hypothesis’ must be abandoned. Promotion of a risk assessment approach (targeted hygiene) provides a framework for maximising protection against pathogen exposure while allowing spread of essential microbes between family members. To build on these findings, we must change public, public health and professional perceptions about the microbiome and about hygiene. We need to restore public understanding of hygiene as a means to prevent infectious disease.
Introduction
Allergic diseases including asthma, hay fever, eczema and food allergies have dramatically increased over the last century, initially in high-income communities but now elsewhere. At the same time, threats of infectious disease pandemics, antibiotic resistance and numbers of immune-compromised people living in the community have increased. Taken together, these diseases are a significant burden on health and prosperity.
The idea that there might be a link between the rise in allergic disease and reduced microbial exposure as a result of measures introduced to protect against infection was first proposed in 1989.1,2 This so-called hygiene hypothesis, as outlined by Dr David Strachan, proposed that a lower incidence of infection in early childhood could be an explanation for the 20th century rise in atopic diseases. Although a simple idea in itself, it raised the thought that rising allergies may be an inevitable price to be paid for freedom from the burden of killer infectious diseases. Although evidence still supports the concept that immune regulation is driven by microbe–host interactions, the term ‘hygiene hypothesis’ is now being seen by many as a misleading misnomer for a concept with far-reaching consequences for public health and an issue which needs to be addressed.3,4
Humans are ecosystems, where the microbes that live on and within us (the human microbiome) constitute an organ at least as essential to health as our liver or kidneys. 5 The immune system is a learning device, and at birth it resembles a computer with hardware and software but few data. Additional data must be supplied during the first years of life, through contact with microorganisms from other humans and the natural environment. If these inputs are inadequate or inappropriate, the regulatory mechanisms of the immune system can fail. As a result, the system attacks not only harmful organisms which cause infections but also innocuous targets such as pollen, house dust and food allergens resulting in allergic diseases.
Despite this new understanding, the hygiene hypothesis concept – that we have become too clean – still persists in the minds of the public. As a result, the public has lost confidence in hygiene. This is happening at a time when infectious disease issues mean that hygiene is becoming more, rather than less, important.
The aim of this study is to review the burden of allergic and infectious diseases and the evidence for a link to microbial exposure, the human microbiome and immune system. Also, it is to assess whether and to what extent we could develop lifestyles which reconnect us with exposures and thereby reduce the risks of allergic disease while also protecting against infectious disease.
Methods
Using methodology based on the Delphi technique,6–9 six experts in infectious and allergic disease were surveyed to allow for elicitation of group judgement in order to arrive at a consensus view on issues pertinent to the aim of the study.
Key themes emerged: first, the extent of the health burden of allergic and hygiene-related diseases; second, the most recent evidence regarding the nature of the link between reduced microbial exposure and its impact on the human microbiome and the immune regulatory system; third, the question of relationship between lifestyles and protection against infectious diseases. The Delphi technique is a qualitative research method that relies on the judgement of individuals presumed to be knowledgeable and expert at what they do. When a sufficient degree of consensus is achieved, the Delphi process is curtailed and the resulting judgement is published. Six experts in infectious diseases and allergies were invited to participate, and the issues to be addressed were agreed via online communication. The authors participated in a conference in which each presented evidence related to their area of expertise. Following this, authors submitted a written contribution. These were analysed and key themes were integrated into a paper which was made available online to all authors for review. This included further questions soliciting the author’s views. After further rounds of questions and revision, a consensus position was obtained.
Results
Why hygiene is important in the 21st century
In the 1950s and 1960s, there was optimism that, with vaccination and antibiotics freely available, conquest of most infections would follow. During the last four decades, this opinion has been reversed. Infectious disease continues to exert a heavy burden on health and prosperity. The various infectious disease issues are most often considered in isolation, but when viewed together, they represent a powerful argument for renewed emphasis on hygiene, which alongside vaccination strategies remain key to containing infectious disease. 10
During the 1980s, there was a rapid increase in reported cases of food poisoning in the United Kingdom, particularly related to Salmonella and Campylobacter. 11 Although reported cases have somewhat declined, food, waterborne, and non-food-related infectious intestinal diseases (IIDs) remain at unacceptable levels. The latest study of IID (food and non-foodborne IID) reported that the true incidence in the community is 43% higher than in the mid-1990s: this study estimated 17 million cases a year in the United Kingdom. 12 The estimated cost of food-related IID is £1.5 billion a year, including resource and welfare losses. 12 Norovirus, mainly spread from person-to-person, is the most significant cause of intestinal infections in the developed world, including 3 million cases per year in the United Kingdom. 12
Evidence shows that respiratory hygiene involving hands and surfaces can limit spread of respiratory infections, particularly colds, and also influenza.13–15 Since respiratory and intestinal viral infections are not treatable by antibiotics, prevention through hygiene is key.
In developed countries, about 7% of inpatients acquire an infection in hospital. 16 Recent figures show a decline in health-care-associated infection (HCAI), in the United Kingdom, particularly of Clostridium difficile and MRSA (methicillin-resistant Staphylococcus aureus),17,18 while other causes of HCAI have emerged, including new epidemic strains of Escherichia coli, Pseudomonas spp. and viruses.
Governments, looking at prevention as a means to reduce health spending, have introduced shorter hospital stays and increased homecare. This requires new policies to prevent HCAIs in community settings 19 where there is no evidence of a decline. Until recently, most episodes of C. difficile infection were believed to result from acquisition in health-care settings. There is now increasing evidence of multiple other potential sources, including asymptomatic patients, and sources in the wider environment, such as water, farm animals or pets, and food. 20 The contribution of cases acquired from these sources to the overall burden of disease is unclear, particularly with concerns about increased community-associated C. difficile infection. 21
Societal changes mean that people with greater susceptibility to infectious disease make up an increasing proportion of the population, up to 20% or more. 10 The largest proportion comprises the elderly who have reduced immunity, often exacerbated by other illnesses. It also includes the very young and family members with invasive devices such as catheters and people whose immuno-competence is impaired as a result of chronic and degenerative illness (including HIV/AIDS) or drug therapies such as cancer chemotherapy.
Emerging pathogens and new strains are a significant concern. It is remarkable that norovirus, Campylobacter and Legionella were largely unknown as human pathogens before the 1970s, with others such as E. coli O157 and O104 emerging in subsequent decades. It is now thought likely that we shall identify many more, the latest being Zika virus. 22 Agencies worldwide recognise that for threats such as new influenza strains, SARS (severe acute respiratory syndrome) and Ebola, hygiene is a first line of defence during the early critical period before mass measures such as vaccination become available. 23 The low infectious dose observed for several of the emerging pathogens, such as E. coli O157:H7 and norovirus, is an additional concern that emphasises the role that hygiene can play in prevention.24,25
Antibiotic resistance is a global priority. 26 Hygiene addresses this problem by reducing the need for antibiotic prescribing and reducing ‘silent’ spread of antibiotic resistant strains in the community and hospitals. 27 As persistent nasal or bowel carriage of these strains spreads in the healthy population, this increases the risk of infection with resistant strains in both hospitals and the community. 27
Infections can act as co-factors in diseases, such as cancer and chronic degenerative diseases. Syndromes such as Guillain–Barré 28 and triggering of allergy by viral infections 29 add to the burden of hygiene-related infection.
The rise of allergies in the 20th century
While infectious disease and hygiene have been key public health issues for centuries, 30 allergic diseases have only relatively recently been regarded as a significant health burden. The marked increase in prevalence of allergic diseases, such as eczema, 31 allergic rhinitis 32 and food allergy, 32 has been a prominent trend over the past century in all regions of the world, but most characterised in Western countries. 33 While this is frequently presented as an ‘epidemic’, epidemiological data indicate the situation is more complex. As highlighted by Platts-Mills 34 (Figure 1), the ‘spikes’ in prevalence of allergic rhinitis, asthma and food allergy have occurred at different times in the past 120 years, and thus different atopic diseases may have different contributing factors. Indeed, there are emerging data that in some areas (mostly in ‘Western’ countries) these increases may have plateaued and even begun to subside. 34 A further issue is that at least for food allergy, prevalence may have been overestimated, depending on the methodology used. Venter et al. 35 assessed the rate of challenge-positive food allergy in three birth cohorts on the Isle of Wight (UK) between 1989 and 2002. A major finding of this study, confirmed in other reports, is that rates of parent-reported allergy were significantly higher (33%) than those confirmed by placebo-controlled food challenge (6%) (the accepted gold standard for diagnosis). For peanut allergy, the same study reported a rate of 1 in 200 children aged 3–4 years in 1989, increasing to 1 in 70 in the mid-1990s, but plateauing thereafter. A 2016 UK intervention study, in children breast-fed to at least 6 months of age, reported a rate of 1 in 40. 36 Of note, the development of an inappropriate immune response to foods (‘sensitisation’), which occurs before onset of clinical disease, is an early event often occurring in the first few months of life. 36
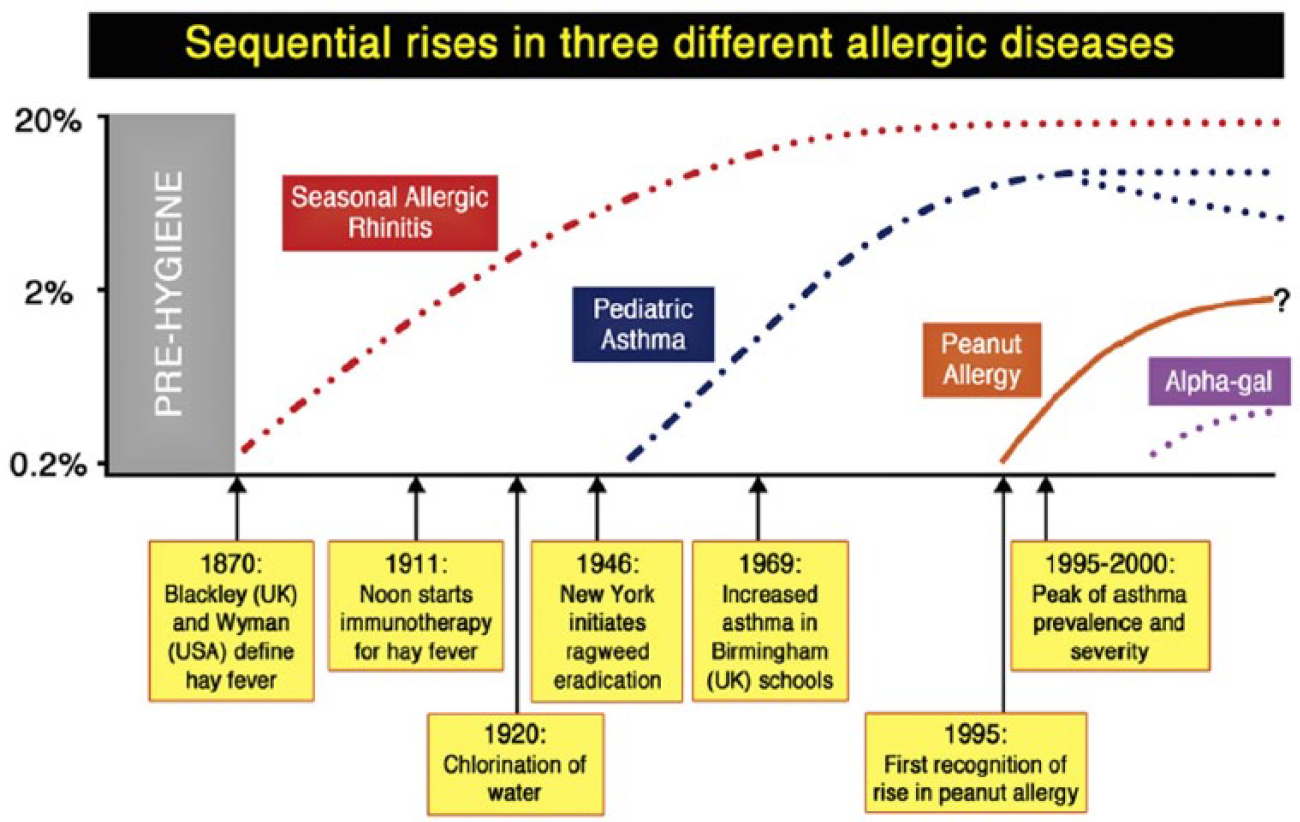
Trends in allergic disease
Perhaps the relatively late appearance of food allergy over the past few decades is a consequence of a progression from allergic airways disease (hay fever, asthma) in parents to a more severe clinical phenotype (food allergy) in their offspring. 37 However, a compelling alternative is the interaction between genetic predisposition and environmental influences, particularly for food allergy, where immune sensitisation to foods may originate with exposure to food allergens in the environment through the skin, a situation exacerbated by eczema and reduced skin barrier function. 36 At the same time, there have been changes in how foods are consumed (e.g. roasted peanut, as consumed in Europe and North America, is more allergenic than raw or other forms of processed peanut).
From Hygiene Hypothesis to Old Friends Mechanism
Building on the significant amount of research published since 1989, a number of refinements to the original hygiene hypothesis now seem to offer more plausible explanations. The Old Friends (OF) Mechanism was proposed by Rook in 2003 and argues that the vital microbial exposures are not colds, measles and other childhood infections (the crowd infections), but rather microbes already present during primate evolution and in hunter-gatherer times when the human immune system was evolving.38–40 OF microbes include environmental species which inhabit indoor and outdoor environments, and the largely non-harmful commensal microbes acquired from the skin, gut and respiratory tract of other humans. In evolving humans, before the advent of modern medicine, the OF also included organisms such as helminths, Helicobacter pylori, and hepatitis A virus that could persist for life in hunter-gatherer groups and that needed to be tolerated. They all therefore activated immunoregulatory mechanisms, 38 but few experts believe that they need to be replaced or even that there is any feasible way of doing so.
Whereas the hygiene hypothesis implicated childhood virus infections as the vital exposures, from an evolutionary point of view this was never likely. Crowd infections were not part of human evolutionary experience because they either kill or induce solid immunity, so could not persist in small hunter-gatherer groups. 41 Epidemiological studies carried out in Finland, Denmark and the United Kingdom now confirm that childhood infections do not protect against allergic disorders.42–44
Studies show how OF exposures are vital because they interact with the regulatory systems that keep the immune system in balance and prevent overreaction, which is an underlying cause of allergies. Diversity of microbial exposure is key. First, a large experience of harmless bacteria and archaea during infancy, when immunoregulatory systems are being established, increases the repertoire of organisms that can be tolerated. Second, since all life-forms are ultimately constructed from similar building blocks, exposed individuals acquire some memory lymphocytes that recognise novel pathogens or even novel viruses. 45
What are the likely causes of reduced or altered microbial exposures
In order to look for strategies which might restore the necessary microbial exposures, it is first necessary to understand the underlying causes of the loss of exposure. Since allergic diseases are largely conditions of the last 100 years, an obvious assumption is that the sanitary revolution is a root cause. The latter part of the 19th century saw radical improvements in sanitation, cleaner food and water, clean-up of cities, and rapid decline in infectious diseases. 46 However, it is likely that these changes also inadvertently reduced exposure to OF microbes which occupy the same habitats. Since the major changes in water, sanitation and hygiene had occurred by 1920, it is difficult to ascribe the massive changes in the asthma prevalence from 1960 onwards to these changes. 34
It is now clear that the most important times for OF exposure are early in development, during pregnancy, delivery, and the first few days or months of infancy.47,48 A 2008 review of epidemiological studies show that Caesarean section is linked to increased risk of allergy. 49 C-sections have become increasingly common since 1950 and now account for 25% of UK births. 50 Furthermore, transfer of microbiota occurs via the mother’s milk, which is not sterile. 51 Breast versus bottle feeding has a large influence on gut microbiome,52,53 but further studies are needed to confirm any association with allergic disease. In high-income settings, there is likely to be a trans-generational effect where each generation receives a more impoverished microbiota, and essential microbiota are lost from the community. 54
Continuing early-life exposure from the mother and siblings is also important.55,56 Studies show that children from large families are at lower risk of developing allergies.52,57 Exposure to pets protects against allergies,58,59 although domestic animals in the home have increased rather than decreased. 60 People seem to share their microbiota via dogs, 61 which greatly increase the microbial biodiversity of the home.62,63
There is good evidence that contact with microbial diversity from the natural environment is crucial. Numerous studies now show that exposure to farm environments during the first 2–3 years of life protects against allergic disorders64–66 and correlates with microbial biodiversity in the air 67 and the home. 68 Animal models show candidate organisms from these environments protect against allergic disorders. 69 Studies in Finland show that living close to green space and agriculture rather than close to a town increases biodiversity of the skin microbiota and correlates with reduced allergic sensitisation. 70 Urbanisation has accelerated loss of exposure to the natural environment. In the United Kingdom, 82% of the population now live in urban areas, 71 with up to 90% of our time spent indoors. 72
Although research has tended to focus on the gut microbiome, it seems likely that the microbiome of skin and airways is also involved.73–75 Much of the exposure obtained from outdoor environments is likely to be via the airways. The air contains bacteria, archaea, viruses, fungi, spores, pollen, plant biomass and dust. Depending on the environment and on degree of exertion, the number of bacteria/archaea breathed in could vary between about 106 and 1010 in 24 h. A proportion of these will be retained in the airways, and recent work reveals that exposure to bacterial components causes increased expression of a protein that inhibits inflammation.73,74 Gut exposure is also mediated via the airways where ciliary action brings about transfer to the gut. The likelihood that skin microbiota are OF microbes is indicated by studies showing that Acinetobacter species in skin protect against allergy. 75
Factors that maintain the gut microbiota
Once the microbiome has been acquired and evolved during childhood, 48 the critical question becomes what factors maintain optimum composition and biodiversity, because loss of biodiversity is strongly associated with disease states, inflammation and decline.76–78
Increasingly, the answer appears to be that the optimal composition of the microbiota is maintained by diet, 79 which needs to be diverse, and contain fibre (polysaccharides digested by the microbiota rather than the human host), 80 and polyphenols found in plant products.81–83 A diet deficient in fibre can lead to progressive extinctions of important groups of organisms, 54 which are cumulative and increasingly difficult to reverse in subsequent generations. 42 Polyphenols and also fish oils also appear to modulate the composition of the microbiota.84,85
Citizens of high-income countries have less diverse microbiota than do hunter-gatherers.77–79 Other studies show that the elderly living in the community with healthy diets 78 have higher gut microbiota diversity than those in long-stay residential care who have a less diverse diet. Studies in Sweden and Denmark show that reduced gut microbiota diversity in infants is associated with increased risk of allergic disease in childhood.86–88
Introduction of antibiotics in the 1950s and subsequent prescribing trends, show a compelling temporal fit with rising allergies since the 1970s. A 2014 review of evidence from over 50 epidemiological studies shows a reasonably consistent relationship between excessive antibiotic use, particularly in early childhood, and increased risk of allergic disease. 89 Evidence showing that exposure to antibiotics during pregnancy increases the risk of allergic disorders in infants90,91 has been further confirmed in recent studies.92,93 Antibiotics, particularly macrolides, have lasting effects on the microbiota of young children and increase risks of asthma. 92 This mirrors effects documented in animal models, where early disruption of gut microbiota causes long-term damage to metabolic regulation. 94
Disruptions of maternal microbiota diversity by antibiotics or inadequate diet are found to be transmitted to future generations. 54
Domestic and personal hygiene
Of all the trends that might explain declining OF exposure, one of the weakest is the popular notion of ‘being too clean in our own homes’. If this factor contributes, its role is likely to be small relative to other factors. An explosion of data, obtained using high-throughput RNA sequencing of samples from US homes, suggests that modern homes are ‘teeming with microbes’. It also suggests that the bacterial communities found in the home relate to the people and domestic animals living there and the food they eat, together with input from the local outdoor environment.63,95
Microbiological studies in westernised homes indicate that routine daily or weekly cleaning habits (even involving use of antibacterial cleaners) have no sustained effect on levels of microbes in our homes.96–98 The idea that we could create ‘sterile’ homes through excessive cleanliness is implausible; as fast as microbes are removed, they are replaced, via dust and air from the outdoor environment, and commensal microbes shed from the human body and our pets, and contaminated foods brought into the homes. Strachan’s 1 1989 proposition that ‘higher standards of personal cleanliness’ could also contribute to reduced exposure to essential microbes may be compatible with increased bathing/showering/shampooing since around 1950s, 46 but although bathing and so on removes large numbers of microbes from the skin, these are rapidly replaced.
Although data from westernised homes suggest that more diverse communities can be found on less-cleaned surfaces (TV screen, door trims, floors) than regularly cleaned surfaces (cutting board, kitchen surface, toilet seat),63,95 to date, there is no confirmed evidence of a link between personal or home cleanliness and increased risk of allergic disease. In a German birth cohort study of 399 families, personal cleanliness (e.g. handwashing and showering) was associated with lower levels of endotoxin and muramic acid (bacterial markers) in bedding and floor dust. In comparison, household cleanliness (e.g. cleaning floors and bathrooms, dusting, and changing towels) was associated with less dust but not with lower microbial marker levels. Endotoxin in infancy was associated with less allergic sensitisation and less asthma when these children reached school age, whereas muramic acid exposure at school age, but not infancy, was associated with less school-age asthma and eczema. 99 It might seem surprising that neither personal nor home cleanliness activities were directly associated with allergy outcomes, but Liu 100 suggests that this may reflect the importance of early-life timing of microbial exposures and not cleanliness behaviours, with the influence of endotoxin exposure being in infancy. A 2002 data analysis of UK children born in 1991/1992 found association between parent-reported frequency of hand and face washing, showering and bathing at 15 months and wheezing and atopic eczema at 30–42 months, but this association was not reported in other studies.101,102
The key point may be that the microbial content of modern urban homes has altered relative to earlier generations, not because of home and personal cleanliness but because, prior to the 1800s, people lived in predominantly rural surroundings. Also, although human gut and skin microbiota are constantly shed from family members, it is likely that exposure has altered reflecting the reduced diversity of the human microbiota due to factors described above. This means we now interact with an altogether different and less diverse mix of microbes.
Other factors also argue against the role of hygiene. Hygiene is irrelevant to microbiome disruption through altered diet and antibiotics. Also, if contact with the natural environment and microbial components of house dust occurs mostly via the airways, hygiene and cleanliness is unlikely to be responsible for reduced inputs from this key source.
Communicating Microbiome Science to Society – Prelude to Reversing Immunoallergic Disorders
Although evidence suggests that strategies such as promoting natural childbirth and breast feeding, increased social exposure through sport, other outdoor activities, less time spent indoors, diet and appropriate antibiotic use could help restore the microbiome and perhaps reduce risks of allergic disease, clinical and other evaluations are required to establish whether and to what extent this might occur and when intervention is most beneficial.
There is a window of time when the developing microbiome is critical for the education of the maturing immune system. Disruption or delay in acquisition of the microbiome in the first few years of life may predispose to later immune dysfunction. It follows that preventive efforts against immuno-allergic disorders must be focussed on early life events. Attempts to correct abnormal host–microbe interactions, once immunological events which lead to allergic disease are established, may be too late. These issues are further discussed by Shanahan and colleagues.103–105 Gaps in understanding host–microbe interactions will be addressed as research continues, and one can anticipate a time, when optimal conditions for colonisation of the newborn are understood and can be controlled by strategies ensuring neonates begin life with a robust and diverse microbiota. In the interim, there is much that can be achieved by education and behaviour change, based on current information.
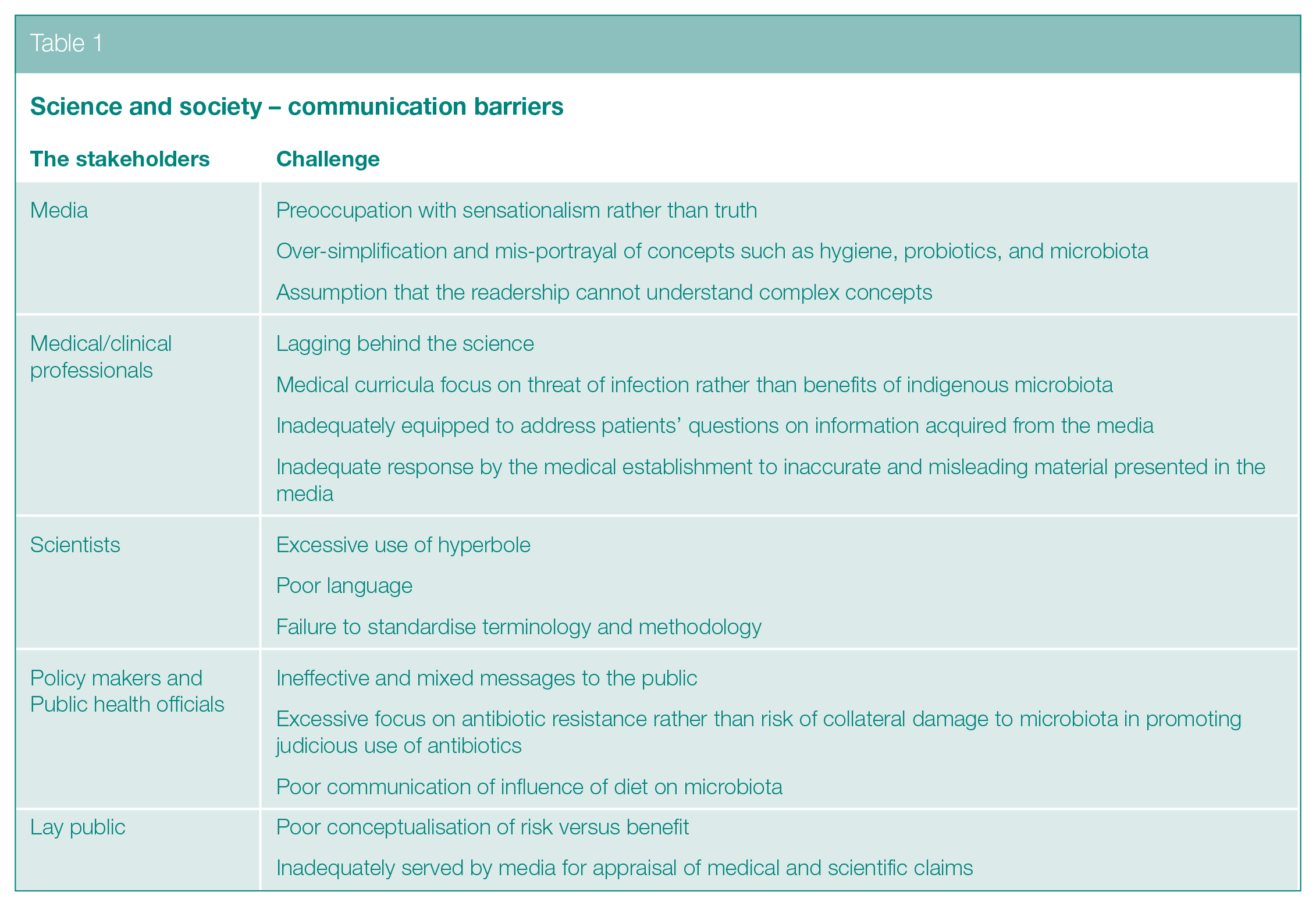
Several factors seem to conspire to limit effective communication of microbiome science to society (Table 1). Some elements within the popular media do disservice to their readership. Examples include mis-representation of the role of hygiene and cleanliness, failure to clarify that probiotics are not all the same, and failure to probe unsubstantiated health claims or address seemingly complex concepts in detail. Fault also lies elsewhere (Table 1). In contrast to policy makers and public health officials, clinicians deal with individual patients, not populations. Unless concerns about antibiotic usage are brought to an individual level, with emphasis on the consumer rather than the prescriber, reform initiatives will have limited impact. Patients are less likely to demand antibiotics if provided with information on the impact of such agents on the microbiota and the risk of immune disorders in later life. 106
Science and society – communication barriers
Promotion of breast feeding is lacking in precise rationale for modern women. Breast-feeding mothers need to know they are promoting a lifelong healthy microbiota for their offspring. Since the neonate acquires its microbiome primarily from its mother, greater attention needs to be paid to the mother’s diet, faecal and vaginal microbiome. Increasing awareness of the importance of the microbiome and the factors which sustain or disrupt it should be part of antenatal education.
Microbiome science already provides a glimpse of how the microbiota may be preserved or restored, including development of smart antibiotics, 107 non-antibiotic anti-microbials, microbial transplants, microbial consortia or single strains, and use of personalised biomarkers of disease risk prediction.108,109 Restoration of the microbiome by vaginal microbiota transplants in C-section infants has been demonstrated, 110 albeit of unproven long-term benefit and controversial. 111 In addition, the molecular basis by which bifidobacteria engage with the host immune system is emerging;112,113 this is important because such organisms are a predominant component of the microbiota in neonates.
Because of the multiplicity of factors involved, strategies to preserve or manipulate the microbiota will probably require a personalised approach tailored to individual genetics and lifestyle factors. 109
Developing and Promoting A Targeted Approach to Hygiene in Home and Everyday Life
Over the last 20 years or so, for reasons outlined above, there has not only been a revival of concern about infection and the role of hygiene10,114 but also a realisation that the ‘scrupulous cleanliness’ approach advocated by Florence Nightingale 115 is no longer appropriate. If, as this review suggests, allergic diseases are not the price we have to pay for protection against infection, this is good news for hygiene. However, if we are to maximise protection against infection while at the same time sustaining exposure to essential microbes, we need a revised approach to hygiene based on current scientific evidence.
The International Scientific Forum on Home Hygiene (IFH) (http://www.ifh-homehygiene.org) was established in 1997 with the aim of developing and promoting a more effective approach to hygiene, based on scientific principles and the growing database of evidence about pathogen transmission. 116 To achieve this, IFH adopted the principle of targeted hygiene. 117 Targeted Hygiene is based on a four-step risk assessment requiring identification of the sources and reservoirs of pathogens, the routes of transmission, the critical control points, and appropriate hygiene interventions.
Targeted hygiene is based on the chain of infection transmission (Figure 2) which shows that pathogenic organisms are continually shed into the environment from sources such as human occupants, pets and raw foods. 118
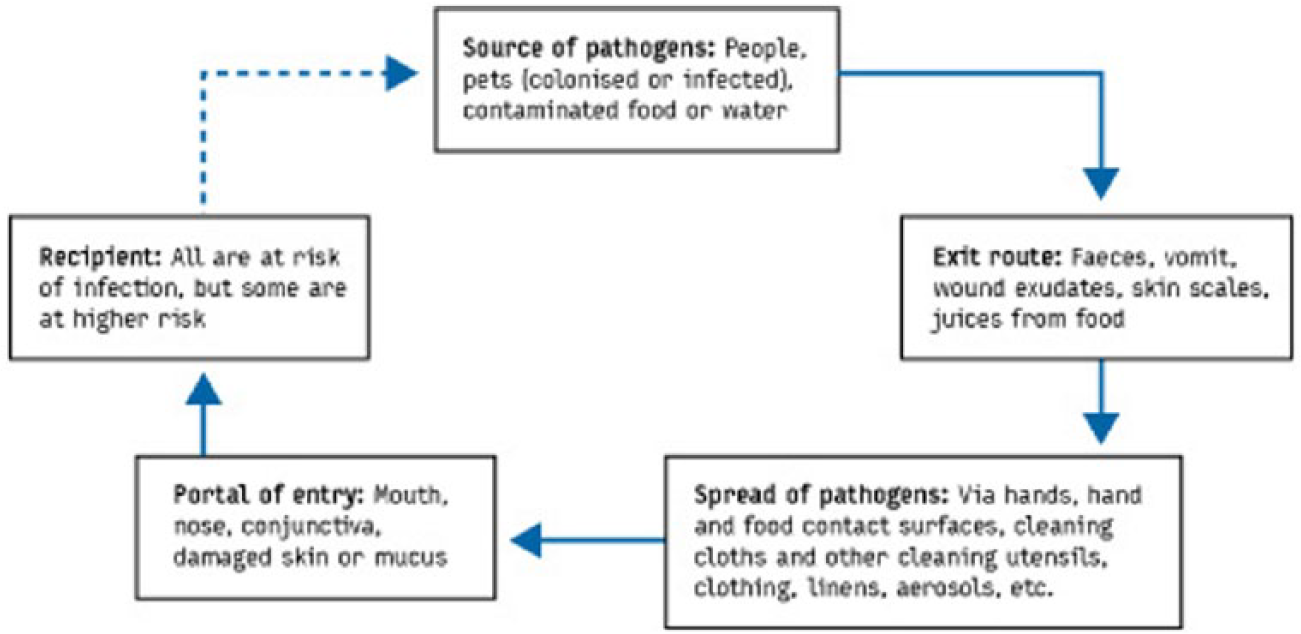
The chain of infection transmission in the home
To get from an infected source to another individual, pathogens use well defined routes. Sampling studies record the presence of non-pathogenic bacteria and bacteria and viruses of medical interest on environmental surfaces in home and community settings, and laboratory and field studies have evaluated the rates of transfer of viral and bacterial pathogens via hands and common touch surfaces. 116 These demonstrate that the critical control points for transmission of infection are the hands, hand contact surfaces, food contact surfaces, and cleaning utensils and that these present the highest risk of transmission (Figure 3).
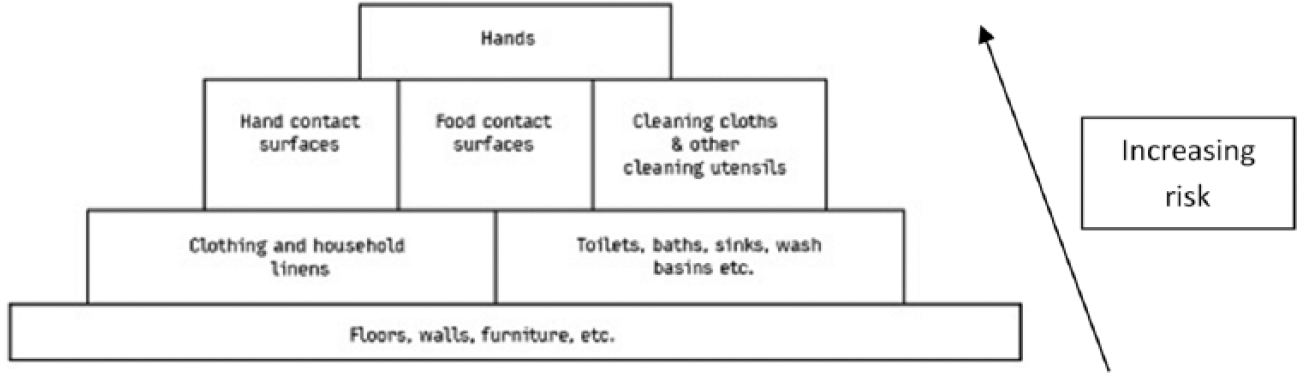
Ranking of sites and surfaces based on risk of transmission of infection
Equally important considerations are the interventions used to eliminate pathogens from critical control points before they spread further. This is important since inadequate procedures can increase transmission.119–123 Hygienic (as opposed to visible) cleaning of hands, surfaces, fabrics and so on can be achieved by the following:
Physical removal of pathogens from inanimate or skin surfaces using soap or detergent-based cleaning. To be effective as a hygiene measure, this should be accompanied with thorough rinsing under running water, such that pathogens are not further disseminated.
Using an antimicrobial product (disinfectants or alcohol hand sanitisers) or processes (heat) that inactivate pathogens in situ. Antimicrobials are required where adequate removal is not possible by wiping/cleaning and/or rinsing alone, or in situations of higher risk. 124
Combined action, for example, laundering, where physical removal is combined with inactivation by heat together with an oxygen bleach–based laundry product.
While it is difficult to quantify the impact, evidence suggests that targeted hygiene reduces spread of infection. A review of evidence published between 1980 and 2001 concluded that the strength of the association between hygiene in the community and infections, as measured by the relative reduction in risk of illness by one or more hygiene measures (including handwashing), was generally greater than 20%. 125 A meta-analysis of community studies showed that improvements in hand hygiene alone resulted in reductions in gastrointestinal and respiratory illness of 31% and 21%, respectively. 126
Changing hygiene behaviour, however, requires changing public perceptions about hygiene, most particularly that hygiene is different from cleanliness, that is, more than just absence of dirt. Hygiene is what we do in the places and at the times that matter (hand, food, toilet and respiratory hygiene, health care, etc.) to protect against infection.
Communication and social marketing campaigns are now being evaluated and used as a means to achieve behaviour change mainly (but not exclusively) in relation to food and respiratory hygiene. These campaigns, however, focus on changing behaviours rather than changing understanding and dispelling misconceptions.13,127–130 The e-bug project is a Europe-wide initiative aimed at ensuring all children leave school with an understanding of targeted hygiene. 131 An important feature of this teaching resource is that it is based on understanding infection and how it is transmitted.
Conclusion
The evidence reviewed in this study reflects the significant shift in thinking in the last 25 years. It shows that the interaction of the OF microbes which inhabit the natural environment and human microbiome with our immune system plays an essential role in immune regulation, promoting a tolerising milieu for the immune system which may impact against the development of allergic disease. Changes in lifestyle and environment, along with rapid urbanisation, have all contributed to changes in our exposure to essential microbes. 132 In addition, altered diet and excessive antibiotic use have also sustained detrimental effects on the content and diversity of the human microbiome. Together, these factors have had profound effects on the immune system, which are likely to have contributed to the onset of allergic disease.
By contrast, the public idea that obsessive hygiene and cleanliness is the root cause of the rise in allergies is no longer supported. Data show that relevant microbial exposures are almost entirely unrelated to hygiene as the public understands it. This is partly because sustaining the human microbiome through diet and avoiding excessive antibiotic usage are factors entirely unrelated to hygiene.
As far as understanding strategies which may reduce the risk of allergic disease, work is progressing fast, but there is still a long way to go. The multiple factors involved (including those not directly associated with microbiome interactions (allergen exposure, genetic, pollution, etc.)) make it impossible to assess the contribution of each factor. It is likely that success will only be achieved through combined effects of lifestyle changes, together with improved diet and reduced antibiotic prescribing. Nevertheless, data are now strong enough to encourage changes, such as encouraging natural childbirth, physical interaction between siblings and non-siblings, more sport and other outdoor activities (including babies in prams), and less time spent indoors, and reduced antibiotic consumption.
This review further supports the view that the term ‘hygiene hypothesis’ is a misleading and dangerous misnomer which needs to be abandoned in favour of a more appropriate term such as the OF Mechanism. However, in order to tackle both allergy and infection issues we also need to develop a smarter approach to hygiene. Although targeted hygiene was developed to optimise protection against infection, it provides a framework for maximising protection against pathogen exposure but, at the same time, minimising disturbance of the indoor microbiome and spread of essential microbes between family members.
As summarised in Table 1, if we want to take advantage of these new findings, we first have to change public, public health and professional perceptions about the microbiome and about hygiene. Unstructured and conflicting advice and vague health warnings in the consumer and professional media must be replaced with simple clear mechanistic explanations and consistent messages using consistent terminology which avoids the use of the term ‘hygiene hypothesis’ to define the concept of a link between microbial exposure and allergies. Recent media articles which promote unsubstantiated suggestions that reduced handwashing could be a means to build and sustain a diverse gut microbiome are in direct conflict with public health agency advice on handwashing which is identified as probably the most important ‘critical control point’ for preventing spread of infection in all settings.133,134
An underlying problem that needs addressing is that, both nationally and internationally there are no lead agencies which take ownership of hygiene promotion, looking at it from the point of view of the public at large and what they need to understand and know. Campaigns targeting food or respiratory, pet or health-care hygiene are developed by different agencies, often with conflicting messages. They also do little to address public misunderstandings about how infections are transmitted, the difference between hygiene, cleanliness and dirt, the widespread misuse of the term ‘germs’, and the hygiene hypothesis misnomer. 135
The imperative to understand and reverse the epidemiologic trends in allergic and immune-mediated disorders relates not solely to the personal suffering and health-care burden in the developed world. Without urgent effective intervention, such trends will be replicated around the globe as societies undergo socio-economic development. 105
Footnotes
Funding
P.J.T. holds a Clinician Scientist Award from the UK Medical Research Council (reference MR/K010468/1) and is supported through the National Institute for Health Research (NIHR)/Imperial Biomedical Research Centre. F.S. is a founder shareholder in Atlantia Food Clinical Trials, Tucana Health, and Alimentary Health Ltd. He is director of the APC Microbiome Institute, a research centre funded in part by Science Foundation Ireland (APC/SFI/12/RC/2273) and which has recently been in receipt of research grants from the following companies: Abbvie, Alimentary Health Ltd, Cremo, Danone, General Mills, Friesland Campina, Janssen, Kerry, MeadJohnson, Nutricia, 4D Pharma plc, Second Genome, and Sigmoid pharma. The authors received an honorarium from the International Scientific Forum on Home Hygiene for their time in preparation of this manuscript.
