Abstract
Surveillance for surgical site infections (SSIs) after intraocular surgery may enable the detection of outbreaks and reveal opportunities for quality improvement. The reported incidence of SSIs after intraocular surgery varies widely, and there are no benchmark studies for SSI surveillance based on surveillance case definitions.
We performed surveillance for SSIs after intraocular surgery at a regional ophthalmology surgical center in Winnipeg, Manitoba, Canada, from April 2014 to March 2023. Intraocular infection was defined according to the United States Centers for Disease Control and Prevention (CDC), National Healthcare Safety Network definition (eye infection, other than conjunctivitis). We defined an SSI as an intraocular infection occurring within 1 year after intraocular surgery, in contrast with the CDC definition of 30 or 90 days for other types of surgery.
There were 96,322 intraocular operations performed during the 9-year surveillance period. The incidence of SSI for all types of intraocular surgery was 0.03% (3.0 infections per 10,000 operations), with substantially greater incidence after glaucoma valve implant surgery. We found that the incidence of SSI, identified using surveillance definitions, is lower than reported previously. This report provides benchmark data that may be useful for ophthalmological SSI surveillance initiatives at other institutions.
Keywords
Background
Surgical site infection (SSI) surveillance after intraocular surgery is important because it may enable the detection of outbreaks and reveal opportunities for quality improvement (Apisarnthanarak et al., 2008; Mateos et al., 2006). The reported incidence of infection after intraocular surgery ranges from 0.05% to 0.8%, with variation in part due to differences in case definition (Chen et al., 2024; Creuzot-Garcher et al., 2016; Mateos et al., 2006). Postoperative endophthalmitis, which is an infection of the vitreous or aqueous fluid, is rare but may cause permanent loss of vision.
In Canada, the reported incidence of endophthalmitis after cataract surgery ranges from 0.04%–0.15% (Freeman et al., 2010; Hatch et al., 2009; Lloyd and Braga-Mele, 2009). However, these studies use varied methods to monitor postoperative infection such as billing data and clinical judgment that are unsuitable for use in surveillance. A literature search showed no benchmark studies for SSI surveillance after intraocular surgery that are based on surveillance case definitions.
Intraocular surgery is performed at high volume in a regional referral center in Winnipeg, Manitoba. The purpose of this study was to determine the incidence over time of SSI after intraocular surgery that was performed at this referral center serving a large geographic region in central Canada.
Methods
Setting
This was a retrospective study of SSI surveillance data that were collected in real time for all intraocular operations from April 2014 to March 2023 at Misericordia Health Centre in Winnipeg, Manitoba, Canada. This regional eye center provides comprehensive ophthalmology services, including cataract, retina, and glaucoma surgery, to patients from Manitoba, northwestern Ontario, Northwest Territories, and Nunavut. There typically were more than 10,000 intraocular operations per year, except when there was a reduction of elective surgical procedures performed during the COVID-19 pandemic. As an infection prevention and control initiative, this study was exempted from review by the Health Research Ethics Board of the University of Manitoba.
Ophthalmology practice
Surgical preparation typically included iodine scrub, dilation, analgesia, and iodine drops to the eye. Postoperative care typically included a sterile gauze dressing with a protective plastic eye shield held in place with clean medical tape. There was variation between surgeons in the use of pre- and postoperative ocular antibiotics (most commonly, moxifloxacin ophthalmic 0.5%) and anti-inflammatory drugs (prednisolone acetate ophthalmic 1%). Postoperative care included eye pressure measurement on the day of cataract surgery and a postoperative visit on the day after retinal surgery. Patients residing outside Winnipeg usually returned home after the postoperative visit. There were no changes in infection prevention and control practices or interventions to improve infection rates during the period of surveillance.
The timing of follow-up varied by ophthalmologist, but most patients had follow-up scheduled within a year. In addition, patients with ophthalmological complaints after surgery were referred to the regional eye surgery center for assessment. Clinical diagnosis of postoperative infection typically was made by the ophthalmologist or clinical resource team at the eye center, which included an on-site physician and clinical resource nurse. Patients residing outside Winnipeg who had symptoms or signs of postoperative infection typically were evaluated in their community by a family, urgent care, emergency room, or nursing station practitioner and referred to the clinical resource team at the eye center for emergency examination when deemed necessary. When an infection was identified on clinical examination, antibiotics were prescribed.
Surveillance
Intraocular infection, other than conjunctivitis, was defined according to the surveillance definition of the United States Centers for Disease Control and Prevention, National Healthcare Safety Network (eye infection, other than conjunctivitis), with the patient having at least 1 of 2 criteria: (1) a microorganism identified from the anterior chamber, posterior chamber, or vitreous fluid or (2) the presence of at least 2 of 3 signs or symptoms (eye pain, visual disturbance, or hypopyon) and antimicrobial therapy started by a physician within 2 days after symptom onset or worsening (Centers for Disease Control and Prevention, 2024a). We also defined an SSI as occurring within 1 year after intraocular surgery, in contrast with the United States definition of 30 or 90 days for other types of surgery, to ensure that infections presenting beyond 90 days would be included (Centers for Disease Control and Prevention, 2024b). All patients who had ophthalmological surgery at our center between April 2014 and March 2023 were followed for one year after surgery to determine the incidence of SSI.
From 2014 to 2019, the medical records of all patients who had intraocular surgery were reviewed manually by an infection control professional for the occurrence of an infection after intraocular surgery. In 2019, the Infection Prevention and Control Program changed the data collection method as it was identified that all diagnosed cases of infection only received antibiotics from the same facility where surgery had been performed. As physician initiation of antimicrobial therapy within 2 days is an element of the surveillance definition, pharmacy records were used to identify the patients who had surgery and received antibiotics. Medical records were subsequently reviewed to determine whether these patients met the SSI case definition. When this procedure change was initiated, duplicate reviews were performed over 6 months to ensure that the same data were identified with each of the methods before and after this procedural change. Subsequently, only pharmacy records were used to identify potential SSIs.
Surgical Site Infections after Intraocular Surgery at a Regional Outpatient Surgical Center in Winnipeg, Canada, January 2017–March 2023. a
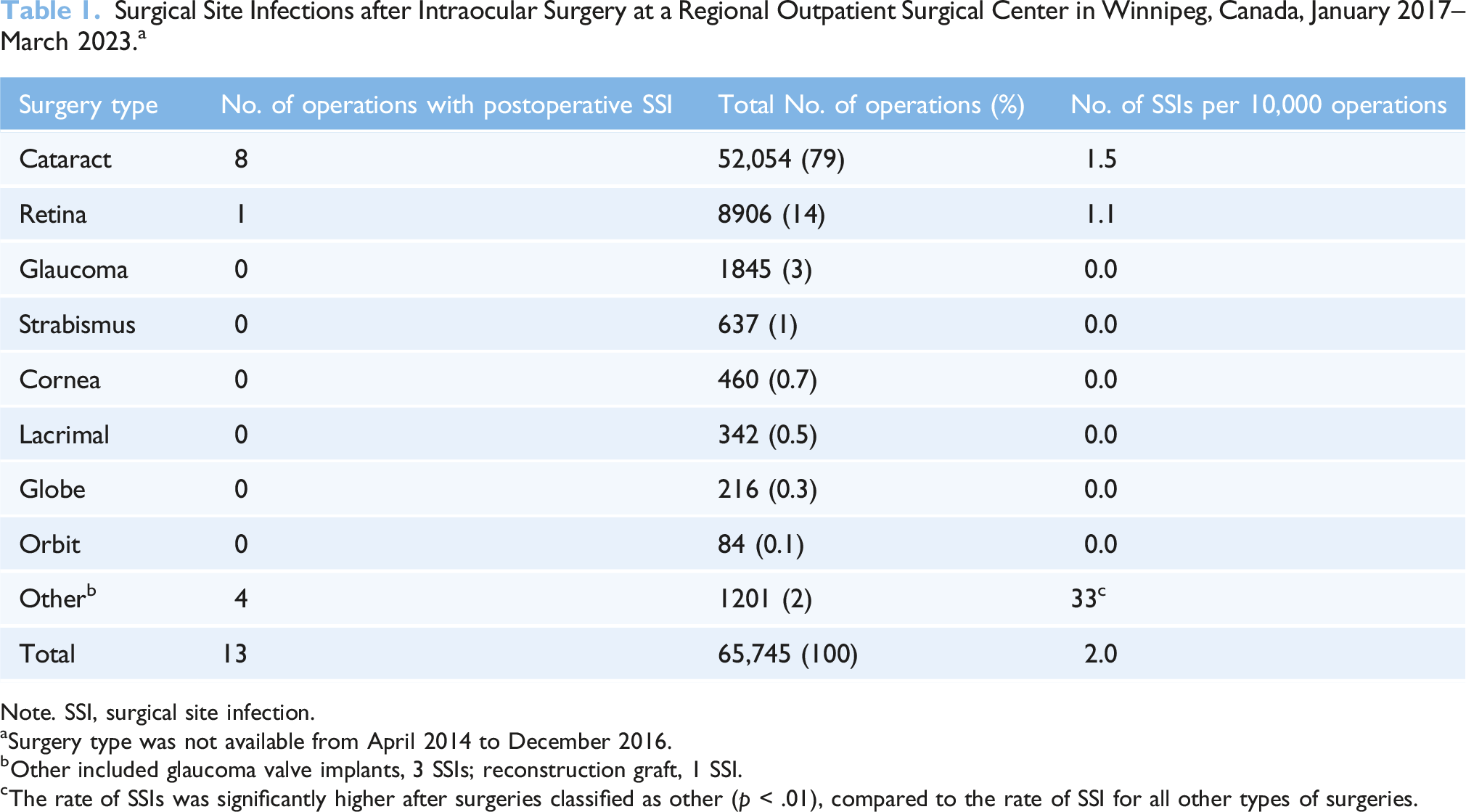
Note. SSI, surgical site infection.
aSurgery type was not available from April 2014 to December 2016.
bOther included glaucoma valve implants, 3 SSIs; reconstruction graft, 1 SSI.
cThe rate of SSIs was significantly higher after surgeries classified as other (p < .01), compared to the rate of SSI for all other types of surgeries.
Data analysis
Data analysis was performed with statistical software (Data Analysis ToolPak, Excel, Microsoft, Redmond, WA). Numeric data were reported including number of infections per 10,000 operations. Changes over time were evaluated with univariate linear regression (dependent variable, rate of infection per 10,000 operations; independent variable, time measured in quarter years). Statistical significance was defined by p ≤ .05.
Results
Surgical Site Infections after Intraocular Surgery at a Regional Outpatient Surgical Center in Winnipeg, Canada, April 2014–March 2023. a
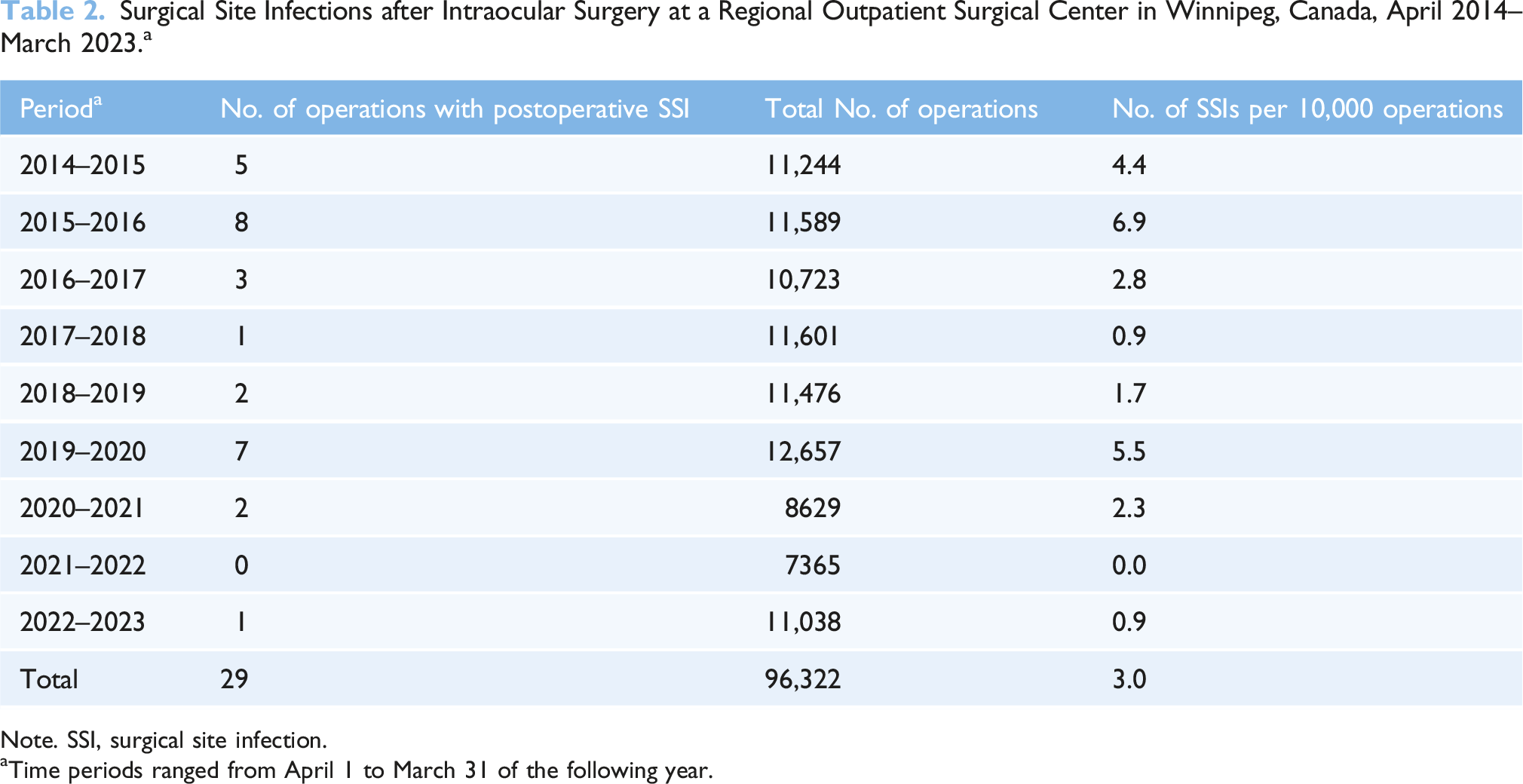
Note. SSI, surgical site infection.
aTime periods ranged from April 1 to March 31 of the following year.
There were no infections linked to any local outbreaks identified during the surveillance period. Linear regression showed a significant decrease in incidence of SSI over time (β = −0.12; R2 = 0.11; p < .05), and there was only 1 occurrence of 2 consecutive quarters having progressively increased SSI incidence over the previous quarter (quarters 10 and 11, July to December 2016).
Discussion
The incidence of SSI observed over the 9-year surveillance period (0.03%) was at the low end of the range reported previously (0.04%–0.15%). This may be due, in part, to our use of the United States Centers for Disease Control and Prevention, National Healthcare Safety Network, SSI surveillance definition, in contrast with other studies that define SSI from billing data (Freeman et al., 2010; Hatch et al., 2009) or clinical reporting by surgeons (Lloyd and Braga-Mele, 2009). Furthermore, the incidence of SSI decreased during the surveillance period, possibly because of changes in provider behavior associated with the implementation of the surveillance program (Chen et al., 2015).
Although most SSIs may occur within 90 days after surgery, which may justify the surveillance periods defined by the United States Centers for Disease Control and Prevention for surgery other than eye surgery, we observed 2 SSIs that were identified at more than 90 days after surgery. These two additional SSIs accounted for 7% of our SSIs, increasing our SSI rate from 2.8 to 3.0 per 10,000 operations. This is evidence in favor of the longer period of surveillance used for eye surgery and consistent with recommendations of varied durations of surveillance for different surgical procedures. Furthermore, there was no substantially increased workload related to extending the surveillance period from 90 days to 1 year.
Limitations of the present study include the potential for loss of follow-up, especially with patients living remotely from the surgical center. Data were not available about type of surgery during the initial 2.75 years of SSI surveillance (April 2014 to December 2016) or the number of operations associated with each type of implant. Although the surveillance program is ongoing, patients who underwent surgery after March 2023 were excluded because they were within the 1-year follow-up period for surveillance when the data were extracted. Some patients without infection may have died prior to the end of the surveillance period, potentially introducing a competing risk bias. While we do not have accurate data on how many patients died without infection during the surveillance period, we expect this number to be low and do not believe it is likely to affect our results meaningfully. No patients died as a result of SSI.
We found that using pharmacy records of prescriptions for antibiotics was an accurate method to identify SSI. Data collection overlapped by 2 quarters with new methods and older methods (double review) to ensure there was no differences between the two methods of surveillance. There were no differences found between the two methods. The incidence of SSI was similar prior to 2019 (3.3 infections per 10,000 operations) and from 2019 onwards (2.6 per 10,000 operations; p = .50).
In summary, the incidence of SSI after intraocular surgery was low and decreased over time. There were no identified changes to procedure that might account for this change over time. The present report provides benchmark data that may be useful for further ophthalmology SSI surveillance initiatives.
Footnotes
Author Contributions
Natalie Gibson and Molly Blake collected data for the study in their roles as infection control practitioners. Myrna Dyck analyzed the data and produced epidemiological reports in support of this project. Jeremy Li and Elly Trepman analyzed the data from the study and participated in writing and editing of the Manuscript. John Embil supervised the project as the medical director of infection control.
Declaration of conflicting interest
All authors report no conflicts of interest or commercial affiliations relevant to this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
