Abstract
The use of mobile phones in the clinical environment by healthcare workers has become widespread. Despite evidence that these devices can harbour pathogenic micro-organisms there is little guidance on how to reduce contamination. Recently touchscreen phones with a single flat surface have been introduced. We hypothesise that bacterial contamination of phones used in hospitals will be lower on touchscreen devices compared to keypad devices. Sixty seven mobile phones belonging to health care workers were sampled. The median colony count for touchscreen phones and keypad devices was 0·09 colony forming units (cfu)/cm2 (interquartile range (IQR) 0.05–0·14) and 0·77 cfu/cm2 (IQR range 0·45–3.52) respectively. Colony counts were significantly higher on the keypad phones (Fisher’s exact test p<0.001). Multivariate analysis showed the type of phone (keypad vs. touch screen) was associated with increased colony counts (F-statistic 14.13: p<0.001). Overall, nine (13%) phones grew either meticillin resistant Staphylococcus aureus or vancomycin resistant enterococci. Eight (24%) keypad phones were contaminated with these organisms compared with one touch screen phone (3%). Our data indicate that touchscreen mobile phones are less contaminated than their keypad counterparts, and they are less likely to harbour pathogenic bacteria in the clinical setting.
Keywords
Introduction
The mobile phone has become an essential means of communication between doctors in hospitals and offers fast and efficient connection while reducing miscommunication and medical error. Previous anxieties about electro-magnetic interference have been allayed and in recent years the Medicines and Healthcare products Regulatory Agency (2011) and the UK Department of Health (2011) have issued guidance permitting the use of mobile phones in hospitals. While it is recommended that individual hospitals develop guidelines for the use of mobile phones, the result is likely to be a gradual phase out of pagers in all but the most sensitive of electro-magnetic interference areas.
Between 9% and 25% of mobile phones used by healthcare workers are contaminated with micro-organisms (Brady et al, 2009). Drug-resistant pathogens such as methicillin resistant Staphylococcus aureus (MRSA) and vancomycin resistant enterococci (VRE) have been recovered from as many as 10% of mobile phones (Goldblatt et al, 2007; Jeske et al, 2007; Brady et al, 2009). Contaminated hand-held devices have the potential to be reservoirs for cross contamination of patients and other staff. The majority of healthcare professionals use the same mobile phone inside and outside the workplace, and this risks contamination to other departments, hospitals and the community. There has been extensive guidance on how to reduce bacterial contaminants on hands and clothes and standards are regularly monitored in UK hospitals. There is relatively little guidance, however, on how to reduce contamination on mobile phones.
In 2011 market research by the independent regulator and competition authority for the UK communications industries (Ofcom, 2011), indicated that more than a quarter of adults use a smartphone, with increasing numbers of users among younger age groups. Many of these devices have a touch screen with a solitary smooth surface as opposed to a key pad with separate buttons and numerous crevices. We postulated that bacterial contamination of phones used in the healthcare setting will be lower on touchscreen devices compared with keypad devices.
Methods
During a six month period in 2011 healthcare professionals carrying mobile phones within a clinical environment in our hospital were approached randomly. To be included in the study, mobile phones had to be used to communicate clinical information; ‘on-call’ baton hospital phones that were passed from clinician to clinician were included. Healthcare workers had to be approached within the hospital. A single clinician could only have one phone sampled; and a single phone could not be sampled more than once. Consecutive clinicians were approached in the hospital. A similar number of keypad and touchscreen phones were sampled.
Conventional agar contact methodology was used to detect the presence of bacteria on the mobile phones. Direct contact methods are more sensitive than swabbing techniques,(Obee et al, 2007) and both sides of a numbered nutrient agar dipslide (Dimanco Ltd, Henlow, Bedfordshire, UK) were pressed firmly on the front surface of the phone. Using the dipslide uniform pressure was applied to the most handled parts of the phone (keypad, virtual keyboard, menu button, earpiece). All phones were sampled by one of three trained investigators. Each mobile phone was photographed and the clinicians were asked to fill out anonymous corresponding numbered questionnaires to record their grade, specialty, concomitant use of a pager and predominant working environment within the hospital. The microbiologist was blinded to the type of phone sampled.
All slides were incubated aerobically at 37°C for 48 hours. The incorporation of 2,3,5-triphenyltetrazolium (TTC ‘red spot dye’) within the agar aided the visualisation and enumeration of bacterial colonies. The aerobic colony count (ACC) as colony forming units (cfu)/cm2 was calculated by dividing the number of colonies isolated from each mobile phone by the area sampled (22 cm2). Confirmatory tests were conducted on all presumptive pathogens. Presumptive enterococci were Gram-stained and tested for aesculin hydrolysis activity (bile-esculin test). Once confirmed, the disc diffusion method was used to determine the susceptibility of each isolate to vancomycin (5 µg/disc; Oxoid Ltd, Basingstoke, UK). Presumptive S. aureus colonies were tested for DNase activity and resistance to cefoxitin (10 µg/disc; a surrogate marker of meticillin resistance). In both cases, zone sizes were interpreted according to British Society for Antimicrobial Chemotherapy guidelines (Andrews, 2009). Samples were considered unsuitable for analysis if there was an error in sampling technique or if slides were contaminated with a bacteria displaying swarming motility (e.g. Proteus spp) making the enumeration of colonies difficult.
Although not clearly linked to infection rates, a common threshold for the standard of hospital cleanliness at a hand touch site is an aerobic colony count less than 2.5 cfu/cm2 (Dancer, 2004). Accordingly, an aerobic colony count > 2.5 cfu/cm2 or the presence of potential pathogens were considered hygiene failures.
Statistical methods
The primary endpoint of the study was to investigate if there was a relationship between the number of bacteria recovered (cfu) and the type of mobile phone interface screen. This was performed using Fisher’s exact test. Multivariate analysis was performed to identify significant independent factors. Other factors taken into consideration included date the sample was taken, job description of phone holder (training doctor vs. consultant (attending) doctor, department of phone holder (medicine, surgery and anaesthetics). The multivariate analysis also considered if the phone holder carried an additional pager.
A secondary analysis was to determine the type of organism isolated and whether the type of phone was associated with the growth of antibiotic resistant organisms. A two sample test of proportion was used to assess these results.
Results
Between January and May 2011, 71 mobile phones were sampled. Thirty six phones were keypad and 35 phones were touch-screen, 17 of which were iPhones. All of these phones were used in the clinical environment on a daily basis. Four samples were deemed unsuitable for analysis, giving 67 results (three were smeared samples and one had an overgrowth of Proteus). They originated from the department of medicine (n=17), department of surgery (n=39) and department of anaesthetics (n=11). These included 41 from senior clinicians and 26 from training doctors and nurses. Overall, 23 of these clinicians also carried pagers at work – 44 did not carry a pager. Ten of the phones sampled were ‘on-call’ baton hospital phones that were passed from clinician to clinician.
The overall median cfu for the 67 phones was 0.23 cfu/cm2 (interquartile range (IQR) 0–2.14). The median cfu count for touchscreen phones and keypad phones was 0·09 cfu/cm2 (IQR 0.05–0·14) and 0·77 cfu/cm2 (IQR range 0·45–3.52) respectively. Colony counts were significantly higher on the keypad phones (Fisher’s exact test p<0.001) (Figure 1). Multivariate analysis showed only the type of phone (keypad vs. touch screen) was associated with increased cfu growth (above median) (F-statistic 14.13: p<0.001). Overall, nine (13%) phones grew either MRSA or VRE. Eight (24%) keypad phones were contaminated with these organisms compared with one touch screen phone (3%) (Two sample test of proportion p=0.01). None of the 17 iPhones sampled were contaminated at levels >1 cfu/cm2 and none were contaminated with potential pathogens. Five of these iPhones were enclosed in a protective case; 12 had their oleophobic screens exposed directly to fingertips.
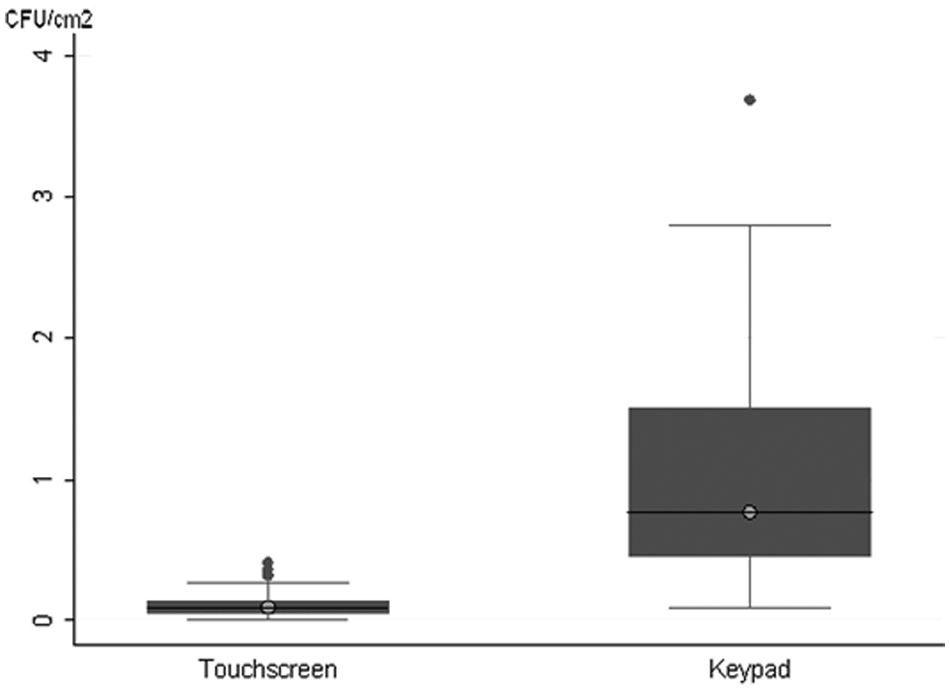
Box plot comparing colony forming units (cfu) values in touch screen and keypad phones
All the ‘on-call’ baton phones were keypad and had a median cfu count of 1.27 cfu/cm2 (IQR range 0.32–1.29); four (40%) of these phones were contaminated with MRSA or VRE.
In order to confirm our findings we repeated the exercise in another hospital. This hospital had a lower baseline MRSA rate in comparison to our institution (patients admitted with MRSA colonisation over the observed period – 3.9% in primary institution, 1.5% in parallel institution.)
In the parallel institution the median cfu/cm2 for 126 touchscreen vs. 47 keypad phones sampled was 0.23 cfu/cm2 (IQR 0.09–0.63) vs. 0.86 cfu/cm2 (IQR 0.77–1.35). 0.4 cfu/cm2. Five (4%) of the touchscreen phones were contaminated with MRSA; none of the keypad phones were contaminated with drug-resistant pathogens. Colony counts were significantly higher on the keypad phones (Fisher’s exact test p<0.001).
Discussion
Hospital acquired infection remains an important problem and there has been much work on minimising vectors that carry pathogenic bacteria within the setting. Hand washing significantly reduces the spread of infection (Mortimer et al, 1966) and hospital ties have been shown to carry the same bacteria that colonise wound infections (Steinlechner et al, 2002). These aspects are components of the ‘bare-below the elbows’ policy that has become healthcare standard. Equally, many hospital and healthcare authorities use fixed communication devices that can be sanitised, e.g. computer keyboards. Conversely, the use of mobile communication devices is relatively unregulated in respect to their potential for cross-infection.
Our study has shown that touchscreen mobile phones have lower bacterial colonisation when compared with keypad mobile phones. Keypad mobile phones were more likely to be contaminated at higher counts of bacteria and the majority of drug resistant bacteria were isolated from keypad phones. We propose that a keypad contact surface which is irregular and uneven can harbour bacteria and that the smooth surface of the touch screen has less potential for this. A comparative analogy with the computer keyboard has shown reduced colonisation rates where a smooth 2-D flat surface is used compared with a traditional 3-D keyboard (Wilson et al., 2008). There is evidence that cleaning mobile phones with an alcohol wipe can reduce contamination rates (Sumritivanicha et al, 2011) and it is conceivable that a touchscreen phone with a flat surface will be easier to decontaminate using simple recommended measures. However repeated use of alcohol may damage plastic.
Worryingly the keypad is the area in contact with the fingertips and intermittent handling of mobile phones during and between consultations is a means for co-transmission and does conceivably reduce the effectiveness of hand washing; a recent study genotyping bacteria confirms that organisms isolated from mobile phones are identical to isolates on the user’s hand (Khivsara et al, 2006).
The use of mobile phones in the clinical setting is inevitable. Indeed they are fast becoming a necessity to maintain accurate and efficient communication and have improved patient care (Soto et al, 2006). Healthcare professionals and patients welcome the use of mobile phones in the clinical setting (Brady et al, 2006) and the availability of low cost devices and instant communication means that the pager is being replaced; in our hospital, on-call pagers have been replaced by on-call mobile phones and 68% of the sampled clinicians did not carry a pager but used the mobile phone as their primary mode of communication.
Conclusion
Hospital acquired infections account for a significant burden of morbidity and mortality and reducing infections represents a significant saving both to the individual patient and financially to the whole healthcare system.
Mobile phones are likely to remain a part of the communications arsenal of modern healthcare practice. They can, however, act as a mobile reservoir for infection. We need to minimise the risk posed by these devices. Our study shows that when compared with keypad mobile phones, touchscreen mobile phones are considerably less contaminated, and have lower prevalence of multi-drug resistant bacteria.
Investing in touch screen phones for use in the hospital setting may be a cost-effective and safe way of reducing the infection risk associated with this now essential workplace tool.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interests
None declared.
