Abstract
Retroperitoneal fibroids are a rare condition that is often misdiagnosed preoperatively. It typically occurs in women with a history of hysterectomy or myomectomy for benign uterine smooth muscle tumors. In this case report, we present the case of a 50-year-old woman who presented with discomfort in the right perineal area. Imaging showed a large retroperitoneal tumor. The patient underwent robotic-assisted laparoscopic surgery to remove the mass, which was confirmed by histopathological analysis to be a uterine smooth muscle tumor of undetermined malignant potential (STUMP). The patient had no recurrence during the 2-year follow-up. The overall prognosis of STUMP is favorable, but more rigorous and long-term follow-up is required to monitor potential recurrence. Our case highlights that retroperitoneal masses in women with prior uterine fibroid surgery should raise suspicion for retroperitoneal fibroids. Robot-assisted laparoscopic surgery may be a safe and feasible minimally invasive option.
Introduction
Retroperitoneal tumors are rare, accounting for only 0.2% of all tumors; however, approximately 70%–80% are malignant. 1 Retroperitoneal uterine leiomyoma (LRP) is a rare retroperitoneal tumor with an estimated incidence of 1.2% of primary retroperitoneal tumors. 2 The origin of LRP is currently unknown, but it often occurs in patients following surgery for utero-ovarian leiomyomas. 3 Although imaging can provide detailed information about the tumor and nearby neurovascular landmarks, preoperative misdiagnosis is common, and histopathology remains the gold standard for definitive diagnosis. 4 Surgery is the preferred option for the treatment of LRP but carries risks such as bleeding and hemodynamic instability. 5 In this report, we present a case of a patient with a history of uterine fibroid surgery who presented with a giant retroperitoneal tumor. The tumor was successfully resected by robotic surgery. Pathology revealed a uterine smooth muscle tumor of undetermined malignant potential (STUMP). While the overall prognosis for STUMP is favorable, there remains a certain risk of recurrence necessitating long-term follow-up. The patient in our case had no evidence of tumor recurrence at the 2-year postoperative follow-up.
Case summary
A 50-year-old female patient was admitted to our hospital with complaints of discomfort in the right perineal area for the past 2 months, accompanied by discomfort in the right inguinal area without abdominal pain. The patient also exhibited bloating, hematuria, and weight loss. She underwent laparoscopic myomectomy 12 years ago and open abdominal myomectomy, along with abdominal wall parasitic leiomyomectomy 7 years ago, respectively. In addition, she had been taking ovulation-inducing drugs for 2 years. Menarche was attained at the age of 12 years, and the patient is currently in the perimenopausal period with irregular menstrual cycles at every 30–40 days with bleeding lasting for 3–7 days. She had married but has no children. Specialist examination shows the abdomen was slightly distended without tenderness or rebound tenderness. A tough, well-defined oval mass could be palpated in the right mid-abdomen, with a maximum transverse diameter of about 15 cm.
A contrast-enhanced CT scan of the entire abdomen performed at an external hospital revealed a large soft tissue mass in the retroperitoneal hepatorenal space, measuring approximately 14*11*14 cm. The mass exhibits heterogeneous density and uneven arterial phase enhancement, suggesting a mesenchymal-derived tumor with low malignant potential. Positron Emission Tomography - Computed Tomography (PET-CT) showed a huge soft tissue mass in the right liver and kidney space, with a clear boundary and a size of about 14.9*9.7*13.2 cm. A mild abnormal concentration of PET in the corresponding part, with SUVmax of 3.0 and SUVave of 1.8, which was mostly considered a neurogenic tumor (Figure 1(a)–(d)). The lesion pushed against the adjacent liver, gallbladder, descending duodenum, and right kidney, with thickening of the adjacent right lateral retroperitoneum, and no enlarged lymph nodes or abnormally thick shadows in the retroperitoneal area. Routine laboratory tests and tumor markers (including alpha-fetoprotein, CA 19-9, and CA-125) were within normal limits. Following a multidisciplinary consultation involving urology, radiology, gynecology, and oncology, the tumor was characterized as a solid mass exhibiting features of rich vascular supply, heterogeneous density, uneven arterial phase enhancement, and mildly increased metabolic activity. Compression signs were observed, and invasion into surrounding tissues could not be ruled out. The preliminary diagnosis is a low-grade mesenchymal tumor with malignant potential, with the possibility of sarcoma remaining undetermined.

(a–d) The PET-CT scan reveals a large soft tissue mass in the right hepatorenal space, measuring approximately 14.9*9.7*13.2 cm, with a clear boundary. The mass is compressing the adjacent liver, gallbladder, descending part of the duodenum, and right kidney.
Based on these observations, robot-assisted laparoscopic right retroperitoneal giant mass resection was performed. The patient was positioned in a left lateral decubitus position at 70° (Supplemental Figure 1(A)). A five-point puncture technique was used to insert the trocar. First, we established port 2 at the junction of the right rectus abdominis lateral border and 3 cm below the umbilicus, which will be used to create pneumoperitoneum and insert the laparoscopic camera. Under direct laparoscopic visualization, 8 mm trocar sleeves were inserted at the following locations: 8 cm directly above and below the vertical line of port 2, and at the junction of the right costal margin and right midclavicular line. These three sites served as ports 1, 3, and 4, respectively, connected to a bipolar grasping forceps, a monopolar electrosurgical scissors, and a non-damaging grasping forceps. A 12-mm trocar was bluntly inserted 8 cm to the left of the midpoint between ports 2 and 3, functioning as an auxiliary port (Supplemental Figure 1(B)). We carefully dissected laterally to the paracolic gutter, retracting the colon toward the midline. A giant mass was seen at the liver–kidney hiatus, with a size of about 15*10*14 cm, compressing the adjacent liver and the right kidney. The lumbaris major muscle side and the suprarenal pole side of the mass were firmly adhered to the surrounding tissues. With adequate exposure and hemostasis, the tumor was completely dissected and placed into a specimen retrieval bag made from a large-sized endoscopic sleeve. The specimen was then extracted by enlarging the two and three surgical ports to 12 cm (Figure 2(a) and (b)). The surgery lasted nearly 3 h, with an estimated blood loss of approximately 100 ml.
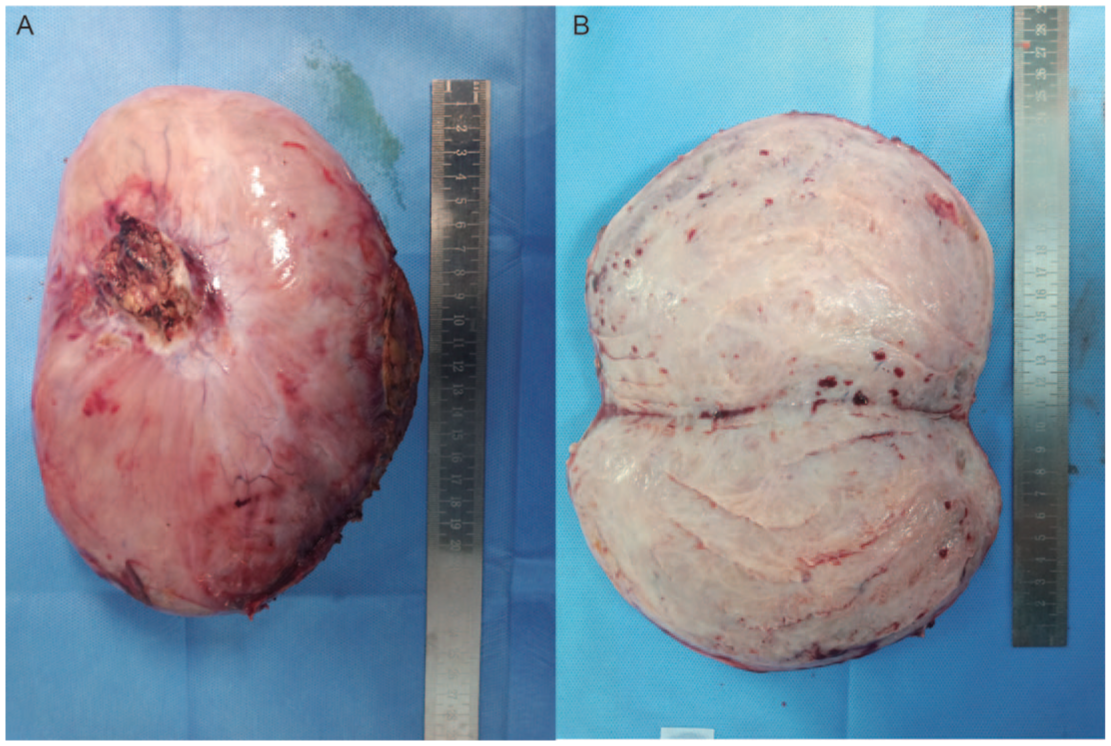
(a) The gross appearance of the tumor showed a complete fibrous envelope measuring 23*16.0*16.0 cm. (b) Maximum faceted sections are greyish-greyish red, firm and soft, locally cystic in sections.
Pathological diagnosis is leiomyoma with undetermined malignant potential. Cellular heterogeneity is not obvious with local mucous degeneration. There is no obvious necrosis, but nuclear division is clearly visible (>5/50HPF) (Figure 3(a) and (b)). The patient’s previous history of two uterine smooth muscle tumor surgeries suggests that the mass may have been the source of the uterine leiomyomas. The patient undergoes follow-up examinations every 6 months postoperatively, including clinical assessments and thoracic and abdominal imaging studies. Currently in the second year post-surgery, no tumor recurrence has been observed. Rigorous and long-term follow-up will continue. The reporting of this study conforms to the Consensus-based Clinical Case Reporting (CARE) guideline development 6 (Supplemental Figure 2).

(a, b) Hematoxylin and eosin staining showing smooth muscle cells with no obvious cell dysplasia, localized mucinous degeneration, no obvious necrosis, and easy nuclear division (>5/50 HPF). (a) (10×) and (b) (20×).
Discussion
Leiomyomas are most commonly found in the uterus and gastrointestinal tract, but they can occur anywhere smooth muscle cells are present. 7 Leiomyomas outside of the uterus can present as benign metastasizing leiomyoma, disseminated peritoneal leiomyomatosis, intravenous leiomyomatosis, parasitic leiomyomata, and retroperitoneal growth. 8 Benign metastasizing leiomyoma is a rare disease that can metastasize to distant sites such as the lungs, skin, bone, mediastinum, lymph nodes, muscle tissue, heart, and retroperitoneum. 9 LRP is most commonly found in the pelvis (73%). 3 The origin of LRP is currently unclear, although it has been suggested that they may originate from hormone-sensitive smooth muscle elements 10 or embryonic remnants of the Mullerian or Wolffian duct. 11 Zaitoon proposed a parasitic theory for the growth of uterine leiomyoma, suggesting that they adhere to surrounding structures and acquire an auxiliary blood supply, resulting in “parasitic” tumors. 12 Kang et al. suggested a primary multifocal origin for LRPs by analyzing a case reporting multiple, isolated, large smooth muscle tumors in the retroperitoneum without uterine fibroids. 13 Kho and Nezhat proposed that LRPs have a medical origin based on their analysis of a series of cases involving ectopic leiomyomas, which revealed that 83% of the patients underwent abdominal surgery and 67% underwent myomectomy, with most of the latter being laparoscopic fragmentation. In addition, more than 40% of LRP cases were associated with uterine leiomyoma or recent hysterectomy for uterine leiomyoma. 14 The possibility of the medical origin hypothesis is also supported by the fact that our case underwent two surgeries for uterine fibroids. Patients with LRP may experience a wide range of symptoms, including abdominal fullness, urinary symptoms, low back pain, pelvic pain, fatigue, weakness, and weight loss. 3 During the acute phase of spontaneous bleeding, patients may also experience a sudden onset of pain or hypovolemia. 15 The most common clinical feature is a palpable pelvic mass, which is present in nearly 90% of patients. 3 Retroperitoneal masses can be diagnosed using ultrasound and CT; however, these diagnostic modalities are not highly sensitive or specific for LRP, leading to frequent misdiagnoses. Magnetic resonance imaging can help differentiating leiomyoma from leiomyosarcoma. In patients with large leiomyoma, tumor markers such as CA-125 and carcinoembryonic antigen may be elevated. 16 The differential diagnosis involves other spindle cell tumors such as leiomyosarcoma and mesenchymal stromal tumors. Other rare diagnoses have been reported, such as malignant peripheral nerve sheath tumors, inflammatory myofibroblastic tumors, and pleomorphic sarcomas. 17 Surgical resection is the primary treatment for LRPs; however, the surgical approach is largely empirical due to the lack of published guidelines. Open surgery is the most commonly chosen option. 3 Research has demonstrated that robotic surgery in the retroperitoneal or pelvic region offers advantages such as reduced intraoperative bleeding, shorter hospital stays, and faster postoperative recovery when compared to conventional laparoscopic surgery.18–20 According to a large retrospective cohort study conducted by Crippa et al, 21 robotic surgery was found to be the most protective factor in terms of complication rates when compared to laparoscopic surgery, with lower transfusion requirements. Other treatment options include close monitoring and hormone deprivation therapy. Because estrogen is thought to contribute to the development of the disease, several treatments for hormone deprivation therapy have been suggested, including hysterectomy and Bilateral salpingo-oophorectomy, progestins, aromatase inhibitors, and luteinizing hormone-releasing hormone analogues. 22 Adjuvant therapy has not yet proven effective, so its use should be approached with caution.
In our case, the final pathology report indicated uterine STUMP. The STUMP classification requires three key histopathological criteria: diffuse moderate to severe atypia, at least 10 mitotic figures (MF)/10 high-power fields (HPFs), and tumor cell necrosis. The Stanford criteria require at least two of the following criteria for the histologic diagnosis of leiomyosarcoma. 23 When a tumor exhibits an abnormal combination of the three features mentioned above but does not meet the Stanford criteria for leiomyosarcoma, it can be diagnosed as STUMP. 24 The overall prognosis for STUMP is favorable, with a 5-year survival rate ranging from 60% to 90%. 25 Reported recurrence rates for STUMP range from 8.7% to 11%, with recurrence occurring as early as 50 months and as late as 9–10 years. 26 Recurrence may be presented as STUMP or leiomyosarcoma. Consequently, patients require rigorous follow-up, typically involving clinical and imaging examinations every 6 months for the first 5 years post-surgery, followed by annual monitoring through the 10th year. 27
Conclusion
Retroperitoneal uterine fibroids are rare tumors. For women with a history of uterine fibroid surgery, retroperitoneal masses should raise suspicion for retroperitoneal fibroids. Surgery is the primary treatment for retroperitoneal uterine fibroids, and minimally invasive techniques such as robot-assisted laparoscopic surgery are a safe and feasible option. For uterine STUMP, despite a favorable long-term prognosis, regular monitoring is necessary to exclude the possibility of recurrence.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251414923 – Supplemental material for A case of retroperitoneal giant leiomyoma and literature review
Supplemental material, sj-docx-1-tau-10.1177_17562872251414923 for A case of retroperitoneal giant leiomyoma and literature review by Zhuocheng Lai, Xiaojun Shi, Qiong Wang, Samir Lalani, Yiwei Wang, Yang Lian, Zaosong Zheng, Weiyu Huang, Shiyu Pang and Wanlong Tan in Therapeutic Advances in Urology
Supplemental Material
sj-pdf-2-tau-10.1177_17562872251414923 – Supplemental material for A case of retroperitoneal giant leiomyoma and literature review
Supplemental material, sj-pdf-2-tau-10.1177_17562872251414923 for A case of retroperitoneal giant leiomyoma and literature review by Zhuocheng Lai, Xiaojun Shi, Qiong Wang, Samir Lalani, Yiwei Wang, Yang Lian, Zaosong Zheng, Weiyu Huang, Shiyu Pang and Wanlong Tan in Therapeutic Advances in Urology
Supplemental Material
sj-pdf-3-tau-10.1177_17562872251414923 – Supplemental material for A case of retroperitoneal giant leiomyoma and literature review
Supplemental material, sj-pdf-3-tau-10.1177_17562872251414923 for A case of retroperitoneal giant leiomyoma and literature review by Zhuocheng Lai, Xiaojun Shi, Qiong Wang, Samir Lalani, Yiwei Wang, Yang Lian, Zaosong Zheng, Weiyu Huang, Shiyu Pang and Wanlong Tan in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
