Abstract
Background:
Enfortumab vedotin (EV), an antibody-drug conjugate targeting Nectin-4, has demonstrated efficacy in advanced urothelial carcinoma (UC) following platinum-based chemotherapy and immune checkpoint inhibitor (ICI) therapy. However, real-world evidence on its effectiveness and safety remains limited.
Methods:
We conducted a multicenter retrospective study across Italian oncology centers to evaluate EV in patients with metastatic UC (mUC) who had progressed after prior platinum-based chemotherapy and ICI. The primary endpoints were progression-free survival (PFS) and overall survival (OS). Secondary endpoints included objective response rate (ORR), disease control rate (DCR), and safety. We also assessed prognostic factors, including a composite index (HERO score) based on baseline hemoglobin and neutrophil-to-lymphocyte ratio (NLR).
Results:
Fifty-three patients were included (median age 72 years; 41.5% ⩾75 years). The ORR was 34.0% (all partial responses), with a DCR of 58.5%. Median PFS and OS were 6.1 and 9.7 months, respectively. Multivariate analysis identified NLR ⩾ 4 and lung metastases as independent predictors of inferior PFS, while NLR ⩾ 4 remained independently associated with worse OS. Dose reductions and peripheral neuropathy were associated with improved outcomes. The HERO score significantly stratified patients by PFS and OS (p = 0.017 and p < 0.001, respectively). EV was generally well tolerated, with most adverse events being low-grade.
Conclusion:
In this real-world cohort, EV confirmed its efficacy and manageable safety profile in mUC. The HERO score may provide a simple tool for risk stratification in clinical practice, though prospective validation is needed.
Plain language summary
This study looked at how well a cancer drug called enfortumab vedotin (EV) works in real-world settings for people with advanced bladder cancer. These patients had already received both chemotherapy and immunotherapy but their cancer continued to grow. Researchers studied 53 patients treated at multiple hospitals in Italy. Most were older adults, with many having other health problems and advanced disease. The team tracked how long patients lived, how long their cancer stayed under control, and what side effects they experienced. Results showed that about one-third of patients had their tumors shrink with EV, and over half had some form of disease control. On average, the cancer stayed under control for just over 6 months, and patients lived nearly 10 months after starting treatment. Side effects like skin rash and nerve problems were common but usually mild. Interestingly, some patients who needed lower doses of the drug or developed nerve-related side effects actually lived longer. The researchers also tested a simple scoring system, called the HERO score, using two routine blood test results (hemoglobin and immune cell counts). Patients with a high HERO score tended to do much better than those with a low score. In conclusion, EV appears to be an effective and generally well-tolerated option for bladder cancer patients who have already tried other treatments. The HERO score may help doctors identify which patients are more likely to benefit from EV, although further studies are needed to confirm this.
Keywords
Introduction
Urothelial carcinoma (UC) is the most common histologic subtype of bladder cancer and represents a significant clinical challenge, particularly in its advanced or metastatic stages.1,2 Despite initial sensitivity to platinum-based chemotherapy, long-term outcomes remain poor due to high recurrence rates and eventual resistance to systemic therapy. In recent years, the advent of immune checkpoint inhibitors (ICIs) has improved survival in a subset of patients, yet many either do not respond or progress after an initial benefit. As such, there remains a substantial unmet need for effective treatment options in the post-chemotherapy and post-immunotherapy setting.3,4
Enfortumab vedotin (EV) is an antibody–drug conjugate (ADC) composed of a monoclonal antibody directed against Nectin-4, a protein highly expressed in UC, linked to the cytotoxic agent monomethyl auristatin E (MMAE). 5 By targeting Nectin-4, EV delivers MMAE directly into tumor cells, thereby disrupting microtubule dynamics and inducing apoptosis. 5 Clinical trials, including the pivotal EV-201 and EV-301 studies, have demonstrated significant antitumor activity and survival benefit in patients with previously treated locally advanced or metastatic UC, leading to its regulatory approval in multiple countries.6,7
Nevertheless, real-world evidence on the use of EV outside clinical trials remains limited. In daily clinical practice, patient populations often differ from those enrolled in trials, with greater comorbidity burden, poorer performance status, and more heterogeneous treatment histories. Evaluating EV in these settings is essential to better understand its effectiveness, safety, and prognostic implications across broader patient cohorts. Although the EV plus pembrolizumab combination has recently become a new standard of care for first-line treatment of advanced UC, EV monotherapy remains a clinically relevant option in several real-world scenarios. Many patients treated prior to combination approval, or those ineligible for pembrolizumab due to immune-related contraindications, comorbidities, or prior exposure, still receive single-agent EV. Therefore, real-world data on EV monotherapy continue to provide essential benchmarks for efficacy, tolerability, and prognostic stratification in current practice.
In this study, we retrospectively evaluated the clinical outcomes and toxicity profile of EV in a real-world cohort of patients with metastatic UC (mUC) treated in multiple Italian oncology centers. We aimed to assess its therapeutic efficacy, identify prognostic factors, and provide further insight into the management of this disease beyond the confines of controlled clinical trials.
Materials and methods
Patient selection and study design
This retrospective observational study included patients diagnosed with mUC who received at least one dose of EV following progression after prior platinum-based chemotherapy and ICI. Eligible patients were identified from electronic health records of four oncology centers affiliated with the GIOTTO network, a collaborative group of genitourinary oncology specialists in the Tuscany region of Italy.
Inclusion criteria comprised the availability of complete clinical and demographic data, including Eastern Cooperative Oncology Group (ECOG) performance status (PS), histologic subtype, prior systemic therapies, metastatic burden and sites, and laboratory parameters such as hemoglobin and neutrophil-to-lymphocyte ratio (NLR). Patients with incomplete follow-up or lacking documentation of response assessment were excluded.
The study was approved by the local ethics committee (Tuscany Area Vasta Centro, protocol number: 25351_OSS), and all patients provided written informed consent prior to inclusion. All procedures conformed to the ethical standards of the institutional and national research committees and to the 1964 Helsinki Declaration and its later amendments.
Treatment administration and clinical monitoring
EV was administered according to the approved European Medicines Agency prescribing information. Treatment adjustments, including dose reductions, omissions, or discontinuations, were made at the treating physician’s discretion based on clinical evaluation, patient tolerance, and toxicity management protocols.
Radiologic follow-up was performed using contrast-enhanced CT scans of the thorax, abdomen, and pelvis every 12 weeks or as clinically indicated. Treatment continued until radiographic or symptomatic disease progression or unacceptable toxicity. In selected cases, therapy continued beyond progression when clinical benefit was still considered possible, such as in the setting of oligoprogression or suspected pseudoprogression, following physician assessment.
Tumor responses were assessed using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 8 Adverse events (AEs) were graded in accordance with the Common Terminology Criteria for Adverse Events (CTCAE), version 5. 9
Efficacy endpoints and statistical analysis
The primary endpoints were progression-free survival (PFS), defined as the time from initiation of EV to radiologic progression or death from any cause, and overall survival (OS), defined as the time from treatment start to death. Secondary endpoints included objective response rate (ORR), calculated as the proportion of patients achieving complete (CR) or partial response (PR), and disease control rate (DCR), which included CR, PR, or stable disease (SD).
To further stratify outcomes and identify patients most likely to benefit from EV, we applied a composite prognostic index, the HERO score, which incorporates two easily accessible baseline laboratory parameters: hemoglobin ⩾11 g/dL and NLR < 4. Each variable contributes 1 point to the score, resulting in a total range from 0 to 2. The selected thresholds (hemoglobin ⩾ 11 g/dL and NLR < 4) were chosen based on prior studies demonstrating their independent prognostic value in metastatic UC and other solid tumors. In particular, hemoglobin < 11 g/dL and NLR ⩾ 4 have consistently been associated with inferior survival outcomes and have been incorporated into validated prognostic models such as the Bellmunt score and the ITACA score.10,11 These cut-offs were therefore adopted to ensure comparability and reproducibility across studies.
Comparisons between categorical variables were performed using Fisher’s exact test, while continuous variables were assessed using the Mann–Whitney U test. Survival outcomes were analyzed using the Kaplan–Meier method and compared via log-rank test. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated using Cox proportional hazards models. All statistical tests were two-sided, and significance was defined as p ⩽ 0.05. Given the limited sample size and to reduce the risk of overfitting, variables with a p-value ⩽ 0.05 in univariate analysis were entered into the multivariate model. Patients without documented progression or death at the time of analysis were censored at their last known follow-up.
Results
Patient characteristics
Of 61 patients screened across four participating centers, 8 were excluded (5 due to incomplete follow-up, 3 due to missing baseline data; Figure 1). A total of 53 patients met all eligibility criteria and were included in the final analysis. The median duration of follow-up was 9.8 months (range: 2.1–24.3). The median age was 72 years (range 47–84), with 41.5% of patients aged ⩾75 years. The majority were male (81.1%) and non-smokers (73.6%). The primary tumor site was the bladder in 84.9% of patients, and 86.8% had pure urothelial histology. Previous surgical intervention had been performed in 69.8% of cases, primarily as cystectomy (62.3%). Prior Bacillus Calmette–Guerin therapy was reported in 20.9%.
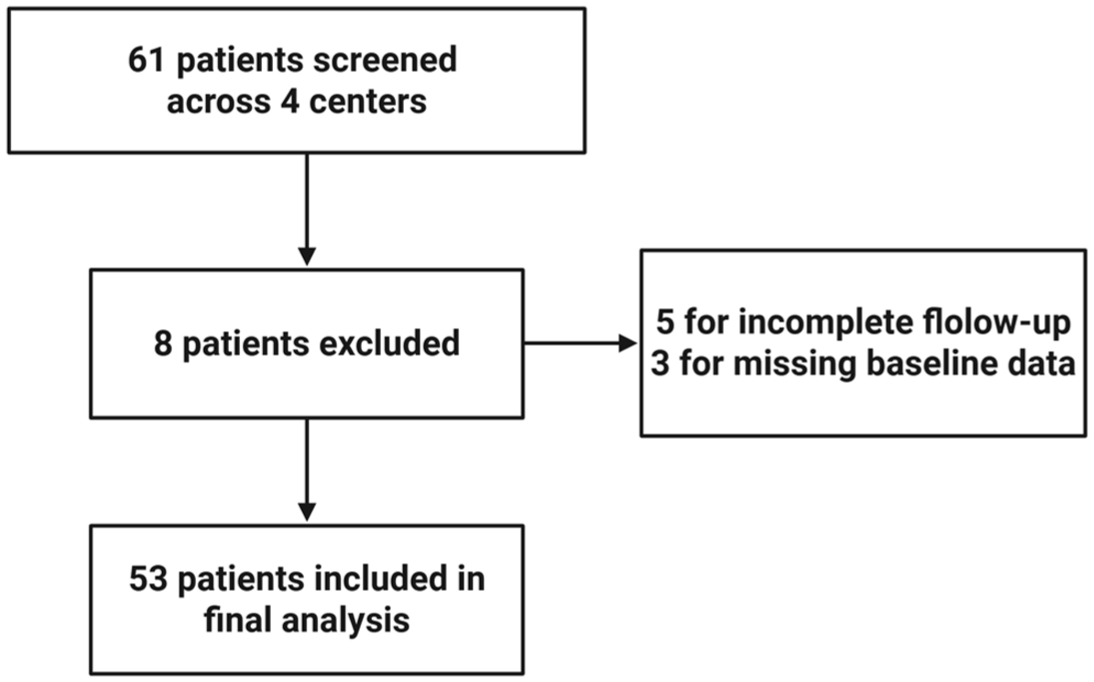
Patients selected flowchart.
ECOG PS was 0 in 41.5%, 1 in 47.2%, and 2 in 11.3%. The most frequent sites of metastases included lymph nodes (86.8%), bone (49.1%), lung (37.7%), and liver (32.1%). At diagnosis, 37.7% of patients were already metastatic. Baseline renal function was preserved in most patients, with a median estimated Glomerular Filtration Rate (eGFR) of 65 mL/min/1.73 m2 (range 38–92). Only nine patients (17%) had eGFR < 60 mL/min/1.73 m2, consistent with the age and comorbidity profile of this cohort. For broader international comparison, we evaluated patient eligibility according to the EVITA criteria, which define standard parameters for EV plus pembrolizumab therapy candidacy. 12 Approximately 78% of our cohort fulfilled EVITA eligibility (ECOG ⩽ 1, adequate and renal function), whereas the remaining 22% would have been considered ineligible mainly due to ECOG ⩾ 2. This distribution reflects real-world inclusiveness beyond trial-defined populations.
Chemotherapy had been administered in the neoadjuvant (11.3%) or adjuvant (15.1%) settings, with gemcitabine/cisplatin and gemcitabine/carboplatin as the most common regimens. Prior radiotherapy was noted in 45.3%. Response to prior chemotherapy included CR in 7.5%, PR in 30.2%, and SD or PD in 43.3%. Most patients had received prior immunotherapy, with avelumab (54.7%) and pembrolizumab (39.6%) being the most common agents. Patients’ characteristics are summarized in Table 1.
Patient baseline characteristics.

Data available for 43 patients.
BCG, Bacillus Calmette–Guerin; ECOG PS, European Committed Oncology Group Performance Status; EV, enfotumab vedotin.
Efficacy of EV
A median number of five cycles of EV (range 1–15) have been administered. Among the 53 patients treated with EV, the ORR was 34.0%, with no CR and 18 PR. SD was observed in 24.5% of patients, and 34.0% experienced disease progression as the best response. Forty-one PFS events and 32 OS events were recorded among the 53 patients included. The median PFS was 6.1 months (95% CI, 4.8–8.1), and the median OS was 9.7 months (95% CI, 6.6–15.4). Notably, patients who achieved a PR had a median OS of 13.4 months (95% CI, 9.7–NR), whereas patients who experience SD or PD had a median OS of 6.6 months (95% CI, 3.8–8.6). When stratified by broader categories (responders = PR + SD vs non-responders PD), the median OS was 13.4 months (95% CI, 8.6–17.7) versus 3.3 months (95% CI, 2.0–6.6), respectively (Figure 2).

Kaplan–Meier survival curve of overall survival according to treatment response.
Dose reductions were necessary in 41.5% of patients (one dose reduction) and 7.5% (two dose reductions). Treatment omission and discontinuation occurred in 20.7% and 5.7% of patients, respectively. Data are reported in Table 2.
Efficacy data of EV.

Data available for 49 patients (not evaluable in 4 patients).
CR, complete response; EV, enfotumab vedotin; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
Prognostic factors
In univariate Cox regression analysis for PFS, ECOG PS ⩾ 1 (HR 2.18, p = 0.023), lung metastases (HR 2.14, p = 0.029), bone metastases (HR 1.90, p = 0.047), proton pump inhibitor (PPI) use (HR 2.09, p = 0.026), NLR ⩾ 4 (HR 2.19, p = 0.023) were associated with worse outcomes, whereas cisplatin-based chemotherapy (HR 0.48, p = 0.050), presence of neuropathy (HR 0.28, p < 0.001), dose reductions (HR 0.42, p = 0.013), and Hb ⩾ 11 g/dL (HR 0.50, p = 0.040) correlated with improved PFS. In multivariate analysis, cisplatin-based chemotherapy (HR 0.33, p = 0.023), dose reduction (HR 0.30, p = 0.024), lung metastases (HR 4.34, p = 0.003), and NLR ⩾ 4 (HR 2.52, p = 0.044) remained significant (Table 3 and Figure 3).
Cox regression for PFS.
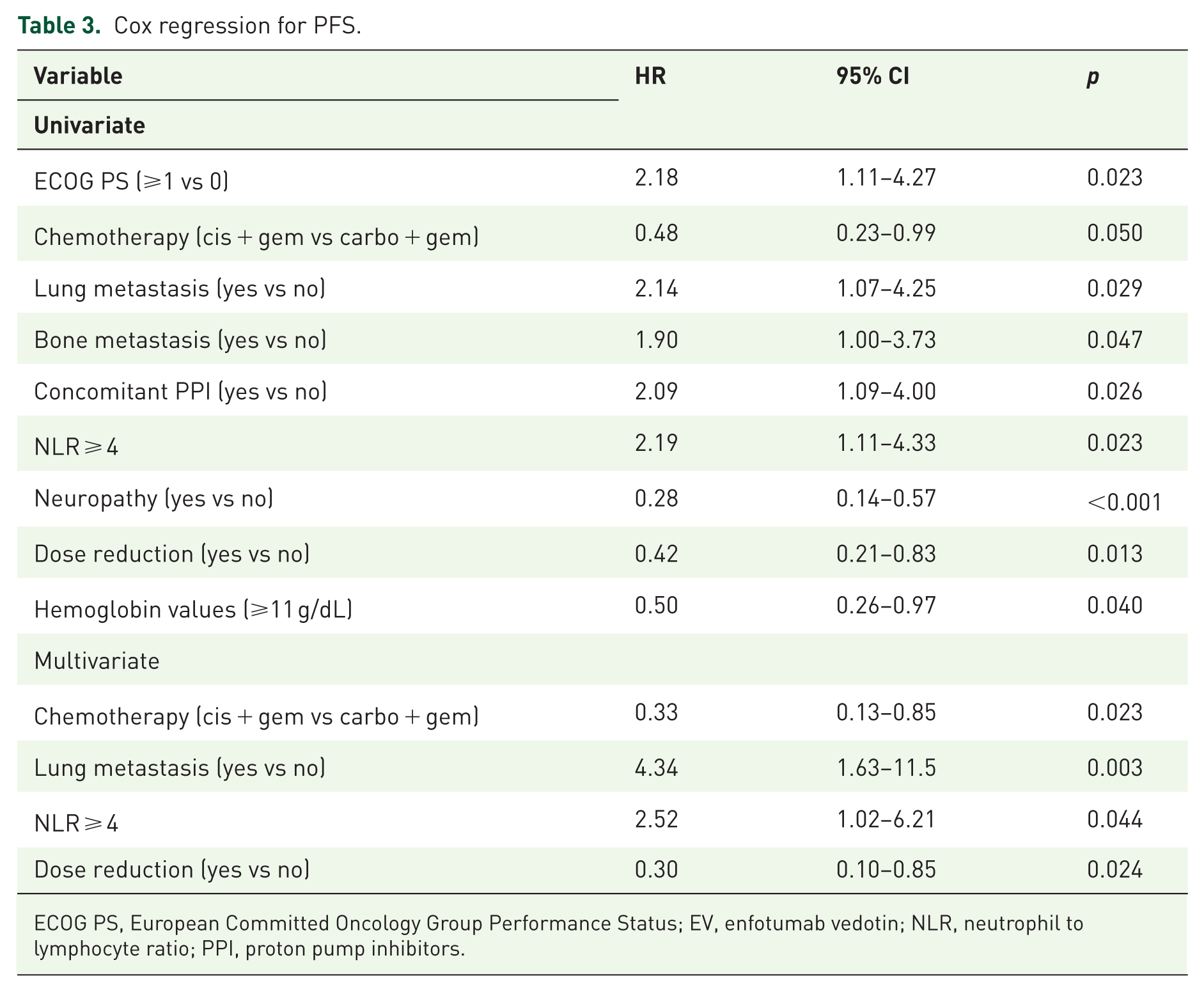
ECOG PS, European Committed Oncology Group Performance Status; EV, enfotumab vedotin; NLR, neutrophil to lymphocyte ratio; PPI, proton pump inhibitors.

Kaplan–Meier survival curve of progression-free survival according to (a) chemotherapy regimen, (b) lung metastases, (c) NLR, and (d) dose reduction.
Regarding OS, univariate predictors of worse survival included ECOG PS ⩾ 1 (HR 2.69, p = 0.013), neoadjuvant chemotherapy (HR 3.60, p = 0.014), bone metastases (HR 2.43, p = 0.023), PPI use (HR 2.10, p = 0.042), and NLR ⩾ 4 (HR 4.30, p = 0.001). Conversely, the presence of neuropathy (HR 0.31, p < 0.001), dose reduction (HR 0.39, p = 0.015), and Hb ⩾ 11 g/dL (HR 0.33, p = 0.004) were protective factors. In the multivariate model, only NLR ⩾ 4 remained a significant independent predictor of OS (HR 3.70, p = 0.011) (Table 4 and Figure 4).
Cox regression for OS.

ECOG PS, European Committed Oncology Group Performance Status; NLR, neutrophil to lymphocyte ratio; PPI, proton pump inhibitors.

Forest plot of HRs for factors associated with overall survival.
Clinical outcomes and HERO score prognostic stratification
Among the 53 patients in our cohort, 18 (34.0%) had a score of 0, 21 (39.6%) had a score of 1, and 14 (26.4%) had a score of 2. Although the HERO score appeared to reflect broader clinical characteristics, with lower scores more frequently associated with adverse prognostic factors such as ECOG PS ⩾ 1, variant histology, and absence of prior surgical intervention, and higher scores linked to a more favorable clinical profile, no significant differences were observed among the three groups (Table 5).
Patient baseline characteristics according to HERO score.

Data available for 43 patients.
BCG, Bacillus Calmette–Guerin; ECOG PS, Eastern Cooperative Oncology Group Performance Status.
Conversely, significant differences in clinical outcomes were observed across these groups. Patients with a HERO score of 2 achieved the highest ORR (60% PR), as well as superior median progression-free survival (8.1 months; 95% CI, 6.7–16.2) and overall survival (not reached; 95% CI, 9.8–NR). In contrast, those with a score of 0 had substantially worse outcomes, with a median PFS of 3.8 months (95% CI, 2.7–6.1) and OS of 5.3 months (95% CI, 3.3–8.6). These differences were statistically significant (PFS: p = 0.017; OS: p < 0.001; Figure 5 and Table 6).

Treatment response according to HERO score.
Efficacy data of enfotumab vedotin according to HERO score.
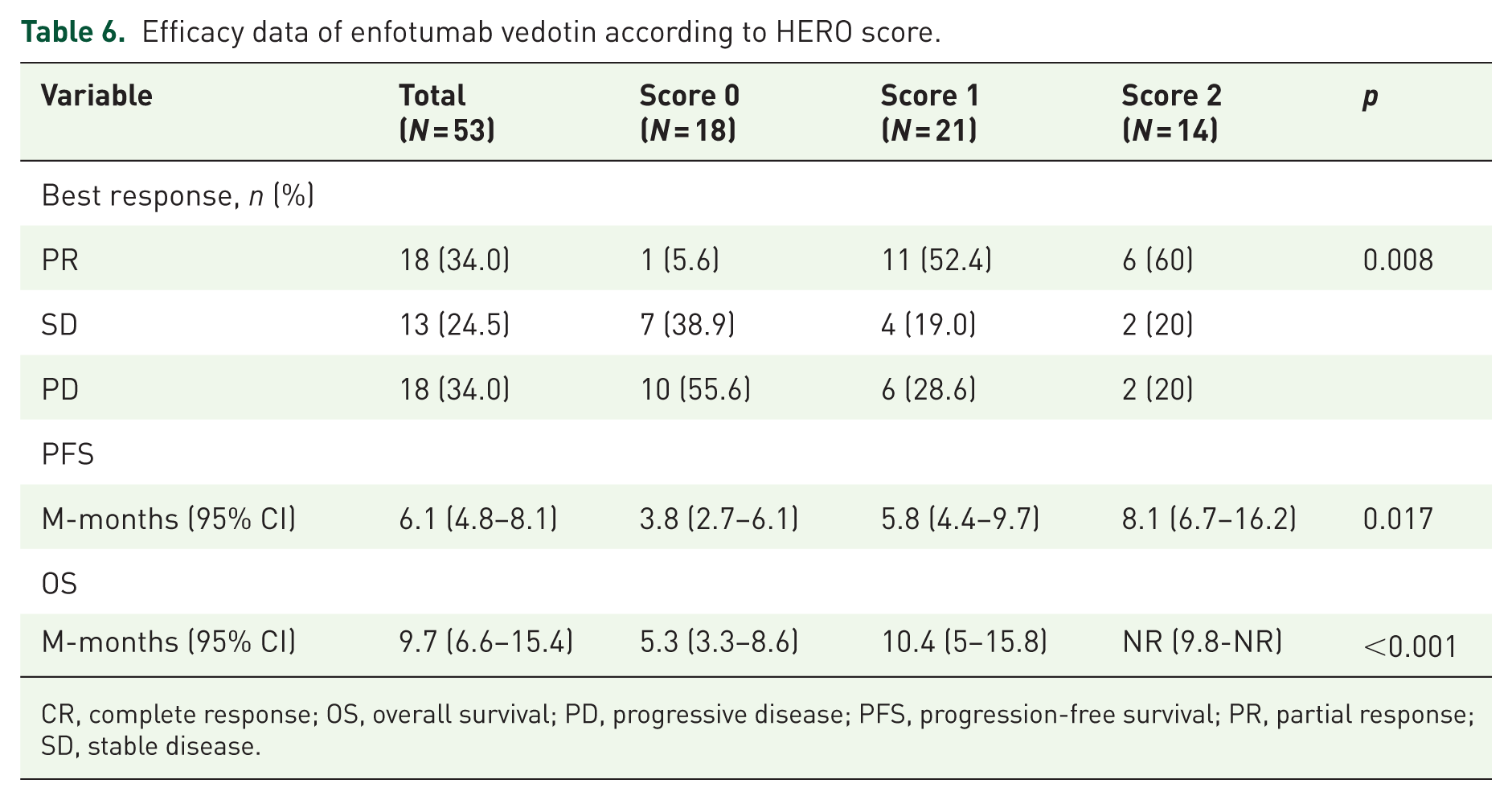
CR, complete response; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
Safety and tolerability
EV was generally well tolerated. Cutaneous toxicity was reported in 50.9% of patients, with most events being grade 1–2; only one case (1.9%) was grade 3–4. Peripheral neuropathy occurred in 45.3% of patients, with one case of grade 3–4 severity. Diarrhea and hyperglycemia (diabetes onset) were less frequent (17% and 5.7%, respectively), with no grade 3–4 events reported for either (Table S1 and Figure 6).
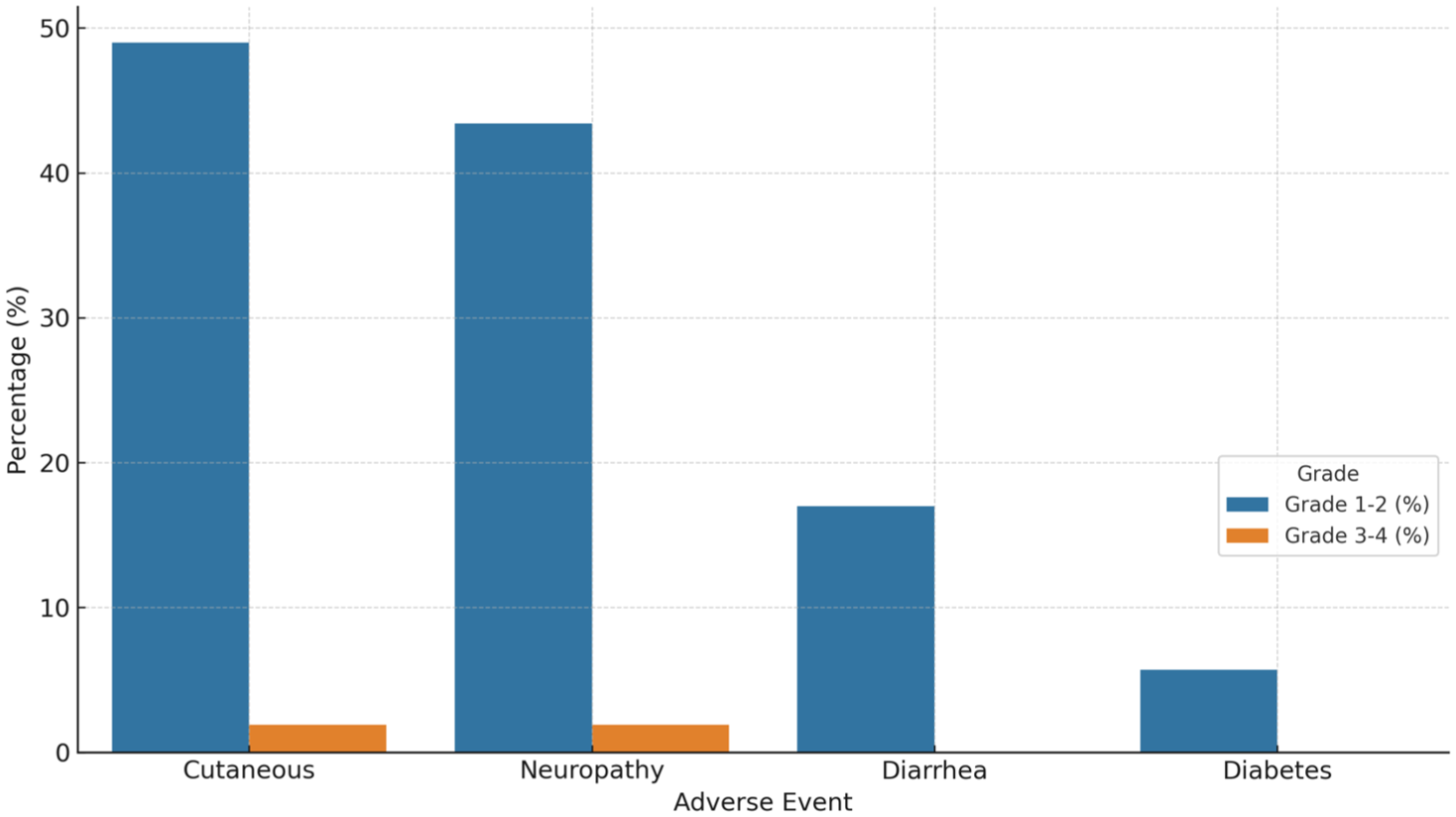
Incidence of adverse events.
Discussion
This real-world analysis confirms the clinical efficacy and manageable safety profile of EV in a cohort of patients with mUC who had previously received both platinum-based chemotherapy and ICI.
Compared to pivotal trials, median PFS in our real-world cohort was consistent with EV-201 (5.8) and EV-301 (5.6) while the median OS (9.7) and the ORR (34%) was slightly lower than that reported in EV-201 (11.7 and 44%–52%, respectively) and EV-301 (12.8% and 40.6%, respectively) reflecting the broader clinical characteristics and potential frailty of real-world patients.6,7 When contrasting our findings with pivotal trials, several differences must be acknowledged. Our cohort included patients treated in later lines of therapy, often after both platinum-based chemotherapy and maintenance or post-progression immunotherapy (avelumab or pembrolizumab), whereas trial populations were more homogeneous. Baseline risk factors such as a higher prevalence of visceral metastases, ECOG ⩾ 2, and advanced age may have contributed to slightly inferior outcomes. Follow-up duration in our series was shorter than in EV-301 (9.8 vs 12.9 months), which could also partly explain the differences in OS.
In our analysis, the ORR was slightly lower than those reported in other real-world studies, which ranged from 39% to 52%.13,14 The median PFS in our population was 6.1 months, consistent with findings from real-world studies ranged from 5.0 to 6.8 months.14–16 Conversely, although median OS in our study results within the expected range, it was shorter than those reported in the other real-world studies (range from 12.7 to 14.4 months).13–16 Compared with international cohorts, our Italian multicenter population exhibited distinctive features, including older median age (41.5% aged ⩾75 years; median age 72 vs 68 years in the US population), higher use of avelumab maintenance (54.7%), and a greater proportion of patients with ECOG ⩾ 2 (11.3%).
Moreover, in the ARON-2EV trial, patients were selected based on the ICI after platinum-based chemotherapy, usually patients with a better prognosis. 13 Despite these differences, efficacy outcomes were comparable to US and Asian datasets suggesting consistency of EV performance across diverse healthcare settings and patient profiles.
Similarly, the association between the emergence of peripheral neuropathy and improved survival, as identified in our analysis, has been reported across multiple series and may reflect an on-target effect of EV exposure.
Interestingly, our data also shed light on important prognostic factors influencing treatment outcomes. Lung and bone metastases, elevated neutrophil-to-lymphocyte ratio (NLR ⩾ 4), and concomitant use of PPIs were all associated with shorter PFS and OS.
In our study, dose reductions affected 49% of patients, in line with a recent real-world study conducted by Mamtani et al. 17 Interestingly, reductions were independently associated with improved outcomes, possibly reflecting a subgroup of patients who maintained prolonged treatment exposure despite toxicity, or a favorable biological profile enabling both tolerance and benefit from prolonged therapy.
A particularly noteworthy finding was the protective association of peripheral neuropathy with survival outcomes. While typically considered dose-limiting toxicity, the emergence of neuropathy may serve as a pharmacodynamic marker of adequate drug exposure and antitumor activity, as observed with other microtubule-targeting agents. Similar associations between treatment-emergent physical AEs and improved outcomes have been described with EV and other ADCs. In particular, the occurrence of skin rash has been linked to higher ORR and longer survival.15,17 Conversely, baseline anemia and other hematologic abnormalities have consistently predicted poorer prognosis across multiple datasets.10,14 These findings collectively support the prognostic interplay between systemic inflammation, hematologic status, and EV pharmacodynamics.
An additional finding of clinical relevance in our study was the prognostic utility of the HERO score, a simple composite index based on baseline hemoglobin levels (⩾11 g/dL) and NLR (<4). Stratifying patients into three risk groups (scores 0–2), we observed a clear and statistically significant gradient in treatment outcomes. Patients with a HERO score of 2 had markedly superior PFS (8.1 months) and OS (not reached), as well as a higher PR (60%), compared to those with a score of 0, whose median OS was limited to 5.3 months. These findings reinforce the importance of systemic inflammation and baseline hematologic status in modulating treatment response. Given the ease of obtaining these parameters in routine practice, the HERO score may offer a pragmatic tool to assist in early risk stratification and guide therapeutic expectations for patients receiving EV. Given the ease of obtaining these parameters in routine practice, the HERO score may offer a pragmatic tool to assist in early risk stratification and guide therapeutic expectations for patients receiving EV. Further validation in larger cohorts is warranted to confirm its prognostic robustness and potential use in clinical decision-making.
Compared to more established prognostic models such as the Bellmunt score, which includes ECOG performance status, hemoglobin levels, and the presence of liver metastases, or the Bajorin score, traditionally applied in the first-line setting and incorporating performance status and visceral metastases, the HERO score offers a streamlined approach based solely on two readily available laboratory parameters.10,18 In contrast to these historical models, which were developed mainly in pre-immunotherapy settings, the HERO score was intentionally designed as a pragmatic, laboratory-based tool applicable to contemporary, post-ICI populations. More recently, the ITACA score, developed in the immunotherapy setting, has combined clinical and laboratory factors to predict outcomes in patients receiving checkpoint inhibitors, but its role beyond the second-line remains to be clarified. 11 Although the limited sample size of our study warrants cautious interpretation, the HERO score demonstrated independent prognostic value in this real-world, post-immunotherapy population. While traditional scores remain valuable in broader clinical contexts, their applicability in later treatment lines, particularly after immunotherapy, is not fully established. In this regard, the HERO score may represent a pragmatic and accessible tool for routine use. Nonetheless, prospective validation in larger cohorts is necessary to confirm its predictive performance and to define how it might complement or integrate with existing models in guiding treatment strategies for advanced UC.
The modest toxicity profile observed in our study reported skin rash and peripheral neuropathy as the most common AEs. Generally, the cutaneous toxicities appear earliest than peripheral neuropathy that is cumulative dose toxicity. This pattern underscores the need for early dermatologic management and ongoing neurological monitoring during prolonged therapy. The absence of grade 3–4 events in most patients underscores the feasibility of EV administration in a real-world context, even among older patients or those with comorbidities.
Among the study limitations, there are, firstly, the retrospective nature that introduces potential bias in data collection and outcome assessment, and the relatively small sample size restricts the robustness of subgroup analyses. Secondly, no stratification was performed based on the specific ICI received in second-line therapy, limiting evaluation of its potential impact on subsequent EV efficacy. Moreover, treatment continuation beyond radiographic progression, although reflective of real-world management, may introduce some heterogeneity in the timing of progression events and should be considered when interpreting PFS results. Finally, it is to be considered that in the OS multivariable model, the variables ECOG performance status and prior neoadjuvant therapy did not meet the recommended events-per-variable threshold, which may limit the stability and interpretability of their estimates.
However, this real-world study offers valuable insights into the use of EV in mUC, based on data from multiple Italian centers with a consistent therapeutic approach. The homogeneity in treatment indication across institutions represents a key strength, ensuring comparability of clinical practices. Additionally, the analysis of prognostic factors, such as NLR and hemoglobin levels, adds depth to the clinical interpretation.
Conclusion
In this real-world cohort of pretreated metastatic UC patients, EV demonstrated meaningful clinical activity and a tolerable safety profile. Response rates and survival outcomes were comparable to those observed in pivotal clinical trials, reinforcing its role as an effective therapeutic option in the post-chemotherapy and post-immunotherapy setting. Dose reduction and the presence of neuropathy may serve as potential markers of treatment efficacy, while ECOG performance status, NLR, and the presence of visceral metastases remain critical prognostic indicators.
Importantly, our analysis also highlights the potential utility of the HERO score, a simple two-variable prognostic index combining hemoglobin and NLR. This score was significantly associated with both PFS and OS, identifying a subgroup of patients with particularly favorable outcomes following EV therapy. Given its simplicity and accessibility, the HERO score may serve as a practical tool to support treatment decisions and stratify patients, although further validation in larger cohorts is warranted.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251412927 – Supplemental material for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification
Supplemental material, sj-docx-1-tau-10.1177_17562872251412927 for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification by Martina Catalano, Sara Bartoli, Alessia Salfi, Mirko Bonalda, Lorenzo Della Seta, Alexandra Paulet, Francesco Bloise, Federico Paolieri, Luca Galli, Michele Sisani, Laura Doni, Armando Perrella, Lorenzo Antonuzzo and Giandomenico Roviello in Therapeutic Advances in Urology
Supplemental Material
sj-docx-2-tau-10.1177_17562872251412927 – Supplemental material for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification
Supplemental material, sj-docx-2-tau-10.1177_17562872251412927 for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification by Martina Catalano, Sara Bartoli, Alessia Salfi, Mirko Bonalda, Lorenzo Della Seta, Alexandra Paulet, Francesco Bloise, Federico Paolieri, Luca Galli, Michele Sisani, Laura Doni, Armando Perrella, Lorenzo Antonuzzo and Giandomenico Roviello in Therapeutic Advances in Urology
Supplemental Material
sj-pdf-3-tau-10.1177_17562872251412927 – Supplemental material for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification
Supplemental material, sj-pdf-3-tau-10.1177_17562872251412927 for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification by Martina Catalano, Sara Bartoli, Alessia Salfi, Mirko Bonalda, Lorenzo Della Seta, Alexandra Paulet, Francesco Bloise, Federico Paolieri, Luca Galli, Michele Sisani, Laura Doni, Armando Perrella, Lorenzo Antonuzzo and Giandomenico Roviello in Therapeutic Advances in Urology
Supplemental Material
sj-png-4-tau-10.1177_17562872251412927 – Supplemental material for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification
Supplemental material, sj-png-4-tau-10.1177_17562872251412927 for Real-world use of enfortumab vedotin in metastatic urothelial carcinoma: efficacy, safety, and risk stratification by Martina Catalano, Sara Bartoli, Alessia Salfi, Mirko Bonalda, Lorenzo Della Seta, Alexandra Paulet, Francesco Bloise, Federico Paolieri, Luca Galli, Michele Sisani, Laura Doni, Armando Perrella, Lorenzo Antonuzzo and Giandomenico Roviello in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
