Abstract
This study aimed to offer important diagnostic insights and a sensible treatment plan for such challenging situations by presenting an incredibly unusual example of Skene’s gland cyst. We described a 39-year-old woman who suffered dysuria for 20 years with an acute exacerbation. Her sexual life was normal, and she did not exhibit any other significant clinical symptoms. However, positron emission tomography/computed tomography (PET/CT) and imaging tests gave contradictory results, with the former creating a high suspicion of malignancy, while the latter indicated a benign tumor. After a multidisciplinary discussion, a cautious treatment approach was adopted. Finally, the patient underwent a diagnostic puncture, and bacteriology detection, along with histological analysis, ultimately confirmed the diagnosis of a Skene’s gland cyst. Following needle puncture biopsy and subsequent antibiotic therapy, follow-up assessments revealed that the mass disappeared. Importantly, the patient’s clinical symptoms completely resolved, and no complications were observed. This case highlighted that puncture biopsy coupled with drainage was a safe and efficacious method, especially when faced with the challenge of diagnosing and treating paraurethral cystic masses.
Introduction
The Skene’s glands, which were homologous to the male prostate, were located in the region surrounding the external urethral meatus. 1 These glands possessed ducts that opened near the urethral opening and secreted fluid that provided lubrication and protection to the urethral mucosa. 2 When the ducts became obstructed, cysts or abscesses might form, which clinically presented as pain, a palpable mass, dysuria, and other lower urinary tract symptoms. 3 Skene’s gland cysts were extremely rare, particularly in adult women, and as such, there were no established guidelines or consensus regarding their diagnosis and treatment. 4
Puncture biopsy was a direct, accurate, and minimally invasive method to obtain pathological evidence, thereby providing a reliable basis for diagnosis. 5 Given the complexity of differentiating periurethral and urethral masses, pathological evidence was crucial in guiding the selection of appropriate management strategies. These strategies often had significant downstream effects on the extent of tissue damage, associated risks, and even patient survival outcomes. 6 We undertook a novel approach utilizing ultrasound-guided puncture biopsy combined with drainage for a patient presenting with a complex periurethral lesion. The diagnosis of Skene’s gland cyst was eventually confirmed through this approach, which also made diagnosis and treatment easier.
Case presentation
A 39-year-old woman presented to the urology clinic with acute urinary retention. She had a 20-year history of dysuria, which recently got worse. The patient denied fever, weight loss, pain during urination, and any other symptoms. In addition, her menstrual cycles and sexual activity were reported as normal. Upon physical examination, a painless lump with a hard substance, distinct edges, and separation from the vagina was found around the urethra.
Cystic-solid hyperplasia encircling the urethra beneath the bladder was seen on contrast-enhanced ultrasonography, which raised the possibility of a malignant urethral tumor. Meanwhile, enhanced pelvic magnetic resonance imaging (MRI) and CT urography (CTU) revealed a mass measuring 58 × 67 mm, as shown in Figure 1(a). On sagittal plane imaging, the mass appeared strikingly similar to the male prostate (Figure 1(b)). However, the findings suggested that the mass likely originated from cystic lesions within the paraurethral glands. As per the patient’s request and in light of the wildly disparate results from earlier imaging, we believed positron emission tomography/computed tomography (PET/CT) was necessary to further elucidate the probability of malignancy. In addition, PET/CT verified that the lesion was surrounded by a cyst wall and demonstrated an elevated FDG uptake maximum standardized uptake value (SUVmax) of roughly 19.9, suggesting a primary malignant origin (Figure 2).
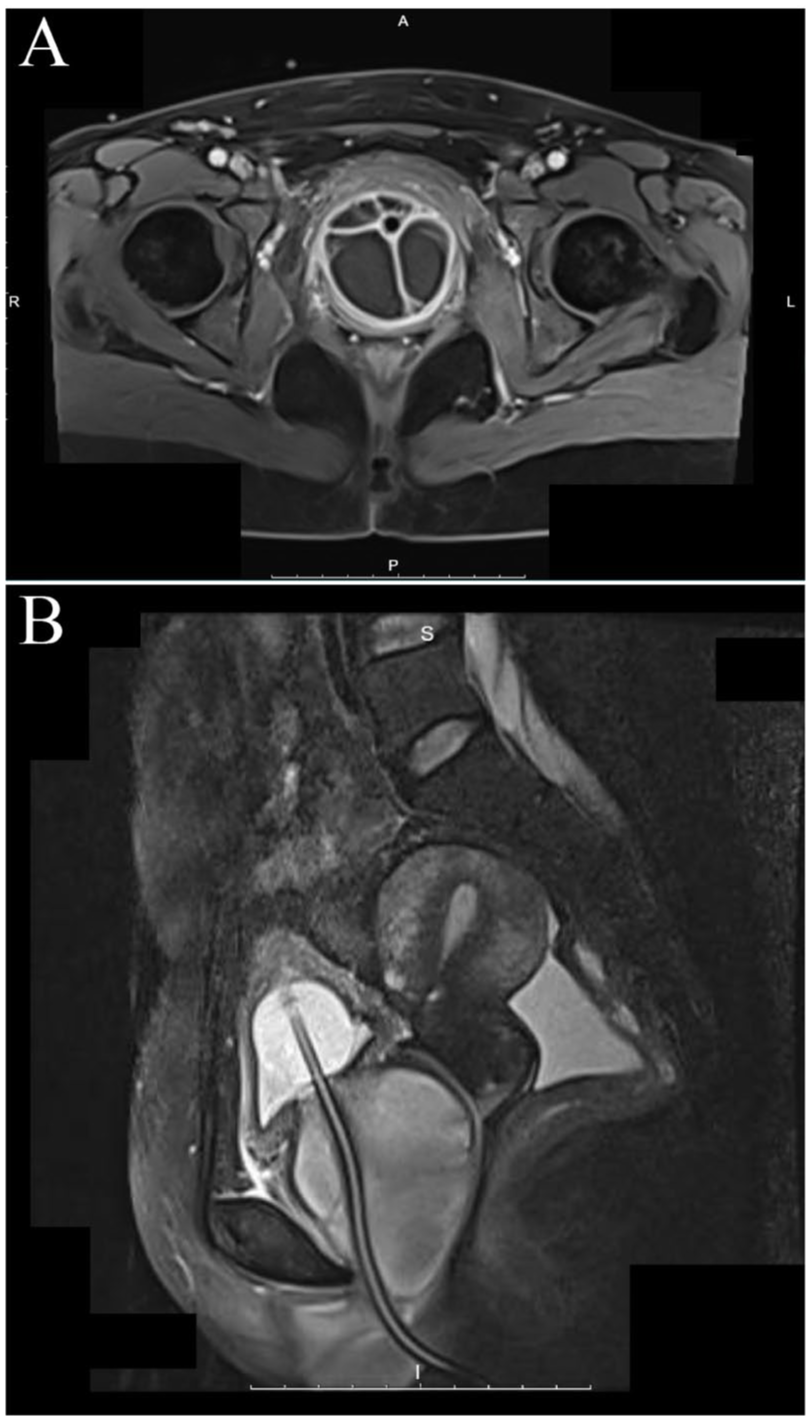
(a) High-resolution axial T1-weighted fat-suppressed dynamic contrast-enhanced VIBE sequence at the pubic-symphyseal level. The Skene’s gland cyst (para-urethral) was observed as a well-circumscribed, hyper-intense, multi-septated structure that embraced the urethra posterolaterally, with the outer ring of the cyst wall showing subtle enhancement. (b) Mid-sagittal T2-weighted image acquired while an indwelling Foley catheter was in place. The cyst lied caudal to the bladder neck and cranial to the external urethral meatus, elevating the ventral urethral wall without disrupting the peri-urethral sphincter complex.

18F-FDG PET/CT (six contiguous panels, craniocaudal from top left to bottom right; axial, anteroposterior, and lateral scout views). The Skene’s cyst was seen posterolateral to the urethra at the bladder neck and urethra, leveled with hypermetabolism (SUVmax 19.9).
Given the potential for malignancy, the treatment options for the patient would involve a decision between radical urethrectomy for carcinoma versus a paraurethral incision and drainage procedure. Both approaches might potentially carry the risk of damaging the urethral sphincter, which could result in urinary incontinence. In view of the patient’s relatively young age (39 years) and prolonged disease duration (20 years), coupled with her stated preferences, the therapeutic objectives were to achieve a definitive diagnosis while concurrently minimizing tissue damage and the risk of complications. In conclusion, the pathological diagnosis played a crucial role in guiding the subsequent treatment strategy.
Following a multidisciplinary team discussion (MDT), we decided to proceed with a needle puncture biopsy and aspiration of the fluid from the mass. Aspiration of the lesion yielded 70 ml of cloudy, purulent fluid without a significant odor. The pathological analysis that followed showed no signs of malignancy and just a small amount of fibrous tissue with a high infiltration of inflammatory cells. In addition, no bacterial or fungal growth was observed in the pus culture. On the third day after the puncture surgery, the catheter was removed after antimicrobial therapy, allowing the patient to resume regular, unhindered urination. Based on the imaging and pathological findings, the patient was diagnosed with a Skene’s gland cyst. A CT scan 1 month later (Figure 3) showed the bladder, uterus, and adnexa to be unremarkable, with only minimal residual ascites, and the patient was free of symptoms.

One-month post-treatment axial pelvic CT (1 mm) at the level of the pubic symphysis, which showed no definite abnormality with only minimal pelvic fluid.
Discussion
Skene’s cyst was an exceedingly rare condition, with only six case reports identified in the literature, encompassing a total of 15 patients.7–9 Notably, these reports did not include recent cases, and there was a lack of systematic reviews or comprehensive studies on this condition. 10 Our case report described an adult female patient presenting with a large paraurethral mass. Interestingly, imaging examinations and PET/CT results showed discrepancies, raising the possibility of a malignant lesion. After thorough multidisciplinary discussion and careful consideration, we ultimately opted for a needle biopsy as a diagnostic approach. The procedure confirmed the presence of a complex Skene’s cyst in the patient. This was the first minimally invasive drainage attempt in a patient with an extra-large periurethral mass exhibiting intense FDG avidity (SUVmax, 19.9) and discordant imaging findings. Moreover, treatment was successful, with a 1-month follow-up CT showing no appreciable abnormality and the patient remaining asymptomatic.
Periurethral and urethral masses in women typically originate from the urethra, vagina, or adjacent structures, posing significant challenges in diagnosis and differential identification. 11 Benign lesions to consider include urethral diverticulum, urethral caruncle, and Skene’s gland cyst, among others. However, the possibility of malignancy, such as primary urethral carcinoma or Skene’s gland malignancy, must also be thoroughly evaluated and ruled out. 4 Although primary urethral carcinoma was exceedingly rare, accounting for only 0.02% of female cancers, its highly aggressive nature and significant impact on patients’ quality of life make it a critical diagnostic consideration. Thus, it remained a key differential diagnosis in clinical practice. 12
The clinical symptoms of periurethral and urethral masses were often nonspecific and overlapping, primarily including common urinary tract symptoms such as dysuria, vaginal discharge, and recurrent urinary tract infections. 4 Although post-void dribbling was a classic sign of a urethral diverticulum, it was frequently hard to see or verify in clinical settings. Meanwhile, hematuria, vaginal bleeding, and menstrual bleeding required careful differentiation during clinical evaluation. 11 In addition, elevated PSA levels might be observed in cases of Skene’s gland malignancy. 13 In our clinical experience with differential diagnosis, ultrasound examination served as a fundamental screening modality, while contrast-enhanced ultrasound frequently supplied the earliest imaging clue to malignancy. Furthermore, CT urography excluded any urinary-tract origin, and pelvic MRI depicted a well-circumscribed, non-enhancing structure, which supported a benign Skene’s gland cyst. In addition, PET/CT quantifies intratumoral metabolic activity, enabling the distinction between benign and malignant lesions, as well as assessing tumor invasiveness and malignancy grade, and occasionally even suggesting tumor type. 14 However, given the rarity of both entities and the aggressive behavior of primary urethral carcinoma, we considered that PET/CT was deemed essential for definitive characterization. Notably, in our case, PET/CT indicated a potential malignancy, underscoring the necessity of further pathological confirmation. As a result, to reconcile the discordant imaging findings while sparing the patient unnecessary trauma, the needle puncture biopsy was therefore the optimal strategy for this special case, which promptly relieved obstruction and provided material for histopathology and microbial culture for a clear diagnosis.
Although PET/CT was widely used across oncologic and non-oncologic disorders, its well-documented pitfalls included false-positive hypermetabolism. Interpretation of PET/CT in urogenital malignancies was uniquely challenging because scattering artifacts, adjacent high-activity urine pools, and other factors frequently obscured or masqueraded as perilesional uptake. 15 Moreover, another PET capture following a furosemide injection could be useful in differentiating between actual pathologic uptake and urine pooling. 16 Besides, infectious and inflammatory lesions, which also exhibited increased glycolytic activity, commonly demonstrated avid FDG uptake. 17 These limitations mandated that clinicians, urologists in particular, interpreted PET/CT findings with circumspection.
However, the therapeutic approach differed significantly between benign and malignant lesions. For primary urethral carcinoma, which typically follows a poor natural course, current disease management recommendations are complete urethrectomy with surrounding tissue resection, along with lymph node dissection as indicated. 18 Equally, there was currently no clear consensus on the standard treatment protocol for Skene’s gland cysts. The clinicians might opt for conservative management with antibiotic therapy or surgical intervention. 19 In addition, it was noteworthy that both marsupialization and total excision were reported to carry the potential for serious complications, such as stress urinary incontinence, and were also associated with the risk of recurrence.20,21 Furthermore, there were no large-scale studies evaluating the efficacy and safety of marsupialization for Skene’s gland cysts. 22 In our case, the markedly different oncologic versus cyst-oriented surgical fields precluded a definitive operative plan while malignancy remained plausible. Thus, upfront surgery was deemed inappropriate. Given the acute urinary retention and the possibility of a PET/CT false positive, 17 we selected to perform the needle puncture biopsy, both to relieve obstruction and to secure a definitive diagnosis. At last, the outcome demonstrated that needle puncture biopsy, which balanced the need to rule out malignancy to minimize damage, was highly beneficial for the patient.
Last but not least, our case had some limitations. First, we had to acknowledge that, constrained by the brief 1-month follow-up, our case cannot address long-term recurrence risk or potential complications. Moreover, a single imaging modality offered only limited evidence of treatment response, so additional investigations, such as ultrasonography, were advisable.
Conclusion
Our clinical experience showed that puncture biopsy in conjunction with drainage was a safe and effective technique for suspected periurethral and urethral masses, especially those with unknown malignant potential, meeting both diagnostic and therapeutic needs at the same time.
