Abstract
Purpose:
To evaluate if prostate-specific antigen density (PSAD) predicts incidental prostate cancer (iPCa) in patients undergoing robot-assisted simple prostatectomy (RASP) for benign prostatic hyperplasia (BPH).
Methods:
A total of 100 consecutive patients undergoing RASP for BPH were analyzed. Patients were stratified into low-risk and higher-risk groups based on their iPCa risk: 60 patients (PSAD ⩽ 0.1 ng/mL/cc) and 40 patients (PSAD > 0.1 ng/mL/cc), respectively. Outcomes included iPCa detection rates, preoperative imaging/biopsy utilization, and postoperative complications. A multivariable logistic regression and an univariate linear regression analysis were conducted to assess whether PSAD can predict the incidence of PCA.
Results:
iPCa was detected in 8% of cases. Five patients had <5% tumor material in their final pathology (pT1a), while three had more than 5% (pT1b). iPCa was detected in eight patients, six with International Society of Urological Pathology (ISUP) 1 and 2 with ISUP > 2. Patients with ISUP 1 were managed with active surveillance; only one chose robot-assisted radical prostatectomy, and the two with ISUP 2 and 3 opted for external radiation. Seven iPCa cases occurred in the low-PSAD group (11.7%), and one in the high-PSAD group (2.5%). In multivariate logistic regression, only a prior negative prostate biopsy was the strongest predictor of iPCa (odds ratio = 5.2, p = 0.01). PSAD > 0.1 ng/mL/cc was not associated (p = 0.09). A univariate linear regression using PSAD as a continuous variable showed no significant association (p = 0.27).
Conclusion:
PSAD, whether dichotomized (threshold of >0.1 ng/mL/cc) or continuous, didn’t predict iPCa in men with large prostates. To optimize cancer detection, patients with large prostates may profit from prostate MRI before bladder outlet surgery, especially those with a history of prior prostate biopsy. Further research, including larger multicenter studies, is needed to validate our results.
Plain language summary
Background: Prostate-specific antigen density (PSAD)—a blood test that adjusts PSA levels for prostate size—is often used to detect prostate cancer. However, its accuracy in men with large prostates (a common feature of benign enlargement, or BPH) remains unclear. What We Did: We studied 100 men with large prostates (>80 mL) who underwent robotic surgery (RASP) for BPH. Patients were grouped by PSAD levels (low: ⩽0.1 ng/mL/cc vs. high: >0.1 ng/mL/cc) to see if PSAD predicted hidden ("incidental") prostate cancer found during surgery. Key Findings: PSAD did not predict cancer risk. Surprisingly, 88% of hidden cancers (7 of 8 cases) occurred in the low-PSAD group (⩽0.1 ng/mL/cc), while only 1 case appeared in the high-PSAD group. Prior prostate biopsies were the only predictor. Men with a previous negative biopsy were 5.8× more likely to have hidden cancer. Prostate size mattered. Cancers were found in larger prostates (avg. 182 mL vs. 140 mL in non-cancer cases), suggesting PSA may be "diluted" in big glands. Why This Matters: PSAD, a widely used tool, may miss cancers in men with large prostates due to PSA dilution or inflammation. MRI before surgery could help best detect hidden cancers, as biopsies and PSAD alone were unreliable. Conclusion: PSAD alone is not reliable for predicting hidden prostate cancer in men with large prostates. Clinicians should consider additional tests (like MRI) to avoid missing cancers during BPH surgery
Introduction
Benign prostatic hyperplasia (BPH) continues to be a common issue in older adult men, 1 often necessitating surgical intervention for refractory lower urinary tract symptoms (LUTS). 2 Robot-assisted simple prostatectomy (RASP) has emerged as a minimally invasive alternative to other techniques in large prostates, offering superior perioperative outcomes. 3 Authors reporting on RASP typically concentrate on postoperative outcomes, often overlooking oncological results. 4 However, detecting incidental prostate cancer (iPCa) during RASP for presumed BPH presents a substantial clinical challenge, with reported rates ranging from 2.5% to 16%.5–7 iPCa is a tumor found incidentally after surgery for benign prostate hyperplasia, autopsy, or radical cystoprostatectomy for bladder pathologies. Pathology reports classify incidental PCa after bladder outlet surgery into two groups based on tumor percentage: <5% (T1a) and >5% (T1b). 8
Preoperative assessment of iPCa risk is vital for effective counseling, surgical planning, and preventing missed oncological care management opportunities.6,8,9 Di Bello et al. 10 found that patients with high-grade iPCa who are not actively treated have significantly worse OS than those who are actively treated. They also suggest waiving active treatment for iPCa patients in whom other causes of mortality surpass cancer-specific mortality. 11 Current screening strategies, including prostate-specific antigen (PSA) thresholds and digital rectal examinations, lack specificity, especially in large prostates, 12 where PSA elevation may reflect prostate volume rather than malignancy. 13 Prostate-specific antigen density (PSAD), which normalizes PSA to gland volume, has shown promise in prostate cancer detection14,15 but remains underexplored in the RASP population. 16 Esparto et al. 5 found PSAD only to correlate with International Society of Urological Pathology (ISUP) grade in their BPH patients who had iPCA. A PSAD threshold of 0.15 ng/ml/cc was suggested to enhance the prediction of clinically significant prostate cancer (csPCa). 17 Yusim et al. 18 found patients with PSAD values under 0.09 ng/ml are unlikely to have clinically significant prostate cancer. However, their research encompassed patients with a variety of prostate sizes, while this study is solely focused on individuals with enlarged prostates. It suggests a validated PSAD cutoff of 0.1 ng/mL/cc for larger prostates, lower than for smaller ones, and evaluates PSAD as a predictor of iPCa in BPH patients undergoing RASP.
Methods
Study design and surgical approach
This single-center cohort study included 100 consecutive cases without any exclusions who underwent RASP for BPH between September 2019 and November 2024 at a referral urological department. All RASP procedures were performed by a single surgeon experienced in robotic surgery, with a caseload of more than 2500 robot-assisted procedures using the Da Vinci X® platform (Intuitive Surgical, California, USA) under institutional protocol.
All patients underwent a multiport transperitoneal robotic approach with a standardized 4-port configuration. Adenoma Enucleation was performed via transcapsular or transvesical approach, depending on gland anatomy and bladder pathology. Patients with median lobes, a history of transurethral resection of the prostate, bladder stones, or diverticula underwent transvesical simple prostatectomy. All other patients underwent the transcapsular approach. The Madigan urethral preservation technique was tested on some patients, but since antegrade ejaculation was not a priority and long-term outcomes are unknown, it was not routinely performed. All patients left the surgical room with a bladder irrigation, which was continued postoperatively in case of gross hematuria. The Foley catheter was removed after a cystogram confirmed no leakage. No drain was used. Postoperative complications within 90 days after surgery were graded by Clavien-Dindo. 19 The enucleated specimens underwent standardized histopathologic evaluation by genitourinary pathologists. The enucleated prostate adenomas were examined like radical prostatectomy specimens, except for the surgical margins. For incidental prostate carcinomas, the Gleason score percentage was estimated based on the tumor’s proportion within the entire specimen. 20
Participants
Participants in the study had to have a prostate volume exceeding 80 mL, as assessed by transrectal ultrasound (TRUS), and demonstrate refractory lower urinary tract symptoms (LUTS) or related complications, like urinary retention, which required the use of either a transurethral or suprapubic catheter. Some patients underwent surgery due to recurrent infections and bladder stones. Patients with a prior prostate MRI showing a prostate imaging reporting and data system (PIRADS) score below three or those who had a negative prostate biopsy proceeded directly to RASP. Similarly, patients with a PSAD below 0.1 ng/ml/cc who did not have an MRI or biopsy were referred directly to RASP. For patients without an MRI or biopsy and a PSAD above 0.1 ng/ml/cc, an MRI was recommended to be performed, or alternatively, a systematic prostate biopsy was conducted. Patients were divided based on their PSAD in two groups: group 1 (n = 60): low risk for iPCa with PSAD under 0.1 ng/ml/cc and group 2 (n = 40): higher risk with PSAD above 0.1 ng/ml/cc.
Variables and measurements
Baseline characteristics such as age, American Association of Anaesthesiologists (ASA) score, body mass index (BMI), prostate volume, and PSA levels, along with PSAD calculated as serum PSA (ng/mL) divided by prostate volume (mL) via TRUS were included (Table 1). Additional factors considered were anticoagulation status, catheter presence, International Prostate Symptom Score (IPSS), International Index of Erectile Function (IIEF-5), previous prostate MRI, biopsy results and BPH medication. The primary endpoint was iPCa on final pathology. 19
Baseline characteristics entire cohort and stratified by prostate-specific antigen density.

PSAD (serum PSA (ng/mL) divided by prostate volume (27) on transrectal ultrasound). Continuous variables are presented as mean ± SD. Categorical variables are presented as number (%).
ASA, American Society of Anesthesiologists; BMI, body mass index; IIEF-5, International Index of Erectile Function-5; IPSS, International Prostate Symptom Score; PSAD, prostate-specific antigen density.
Statistical analysis
Analyses were conducted using SPSS® v27 (IBM Corp., Armonk, New York, USA). For the matched-pairs analysis, parametric and non-parametric variables were compared using the independent T-test and Mann–Whitney U tests. Multivariable logistic regression identified predictors of iPCa; the included four covariates (PSAD, prior biopsy, prostate volume, and age). A univariate linear regression model using PSAD as a continuous variable to predict iPCa was also conducted. Adjusted odds ratios (aORs) with 95% CIs are reported.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 21 The STROBE checklist is found in Supplemental Material 2.
Results
Baseline characteristics
Patients in Group 1 were 3 years younger than those in the other group, but this difference was not statistically significant (p = 0.06). Overall, patients were relatively comorbid, with 34% classified as ASA 3, yet both study groups had a balanced distribution (p = 0.7). They reported severe LUTS and erectile dysfunction (ED) symptoms, with no statistical differences observed (p = 0.2 and 0.1). A total of 40% patients in group 1 and 50% in group 2 had a preoperative urinary catheter due to urinary retention (p = 0.3). Patients in group 2 had significantly higher serum PSA levels than in group 1 (14.2 vs 6.5 ng/mL, p < 0.001), although their prostate volumes were significantly smaller than those of patients in group 1 (121 vs 158 mL, p < 0.001). A total of 57% of men in our study underwent prostate MRI, biopsy, or both, to rule out PCa before undergoing RASP. The undertaking of this preoperative diagnostic was significantly more done in patients with PSAD 0.1 ng/ml (Table 1). Overall, 21 out of 100 men in our study were under 5a-reductase therapy either alone or in combination with a-blocker, with similar distribution among study groups (p = 0.8).
Intra and postoperative results
Console time was 7 minutes longer in group 2 patients (82 vs 75 min), without statistical difference (p = 0.1; Table 2). Postoperative catheter days were also longer in group 2 patients, who required 6.2 versus 4.5 days (p = 0.001). The same applies to adenoma weight, which was higher in group 2 (107 vs 84 g, p = 0.02). Also, patients in group 2 had a higher prevalence of concomitant conditions such as bladder stones, bladder diverticula, and inguinal hernias that required management during surgery (47.5% vs 16.7%, p = 0.001). Minor and major complications in the first 90 days after the initial surgery were comparable between the study groups, p = 0.3 and 0.45, respectively. The most frequent minor complications were prolonged Gross hematuria, urinary tract infections (UTIs), and acute urinary retention (AUR). Two patients developed urinoma, treated with percutaneous drainage (CDC IIIa). Two men underwent surgery for bowel obstruction (CDC IIIb). The first patient among them developed abdominal adhesions very fast after RASP and had to be operated on during hospital stay. The second one developed what our surgeons described as an ileus caused by an internal hernia of the bowel. One patient had upper urinary tract obstruction from ureteral edema, which resolved with stenting. One port hernia was repaired (CDC III b), and one patient needed transurethral re-resection for residual adenoma 3 months following the initial procedure RASP. The readmission rate was also comparable between groups (12.5% in group 2 vs 6.7% in group 1, p = 0.3).
Intraoperative and postoperative outcomes of the entire cohort stratified by PSAD.
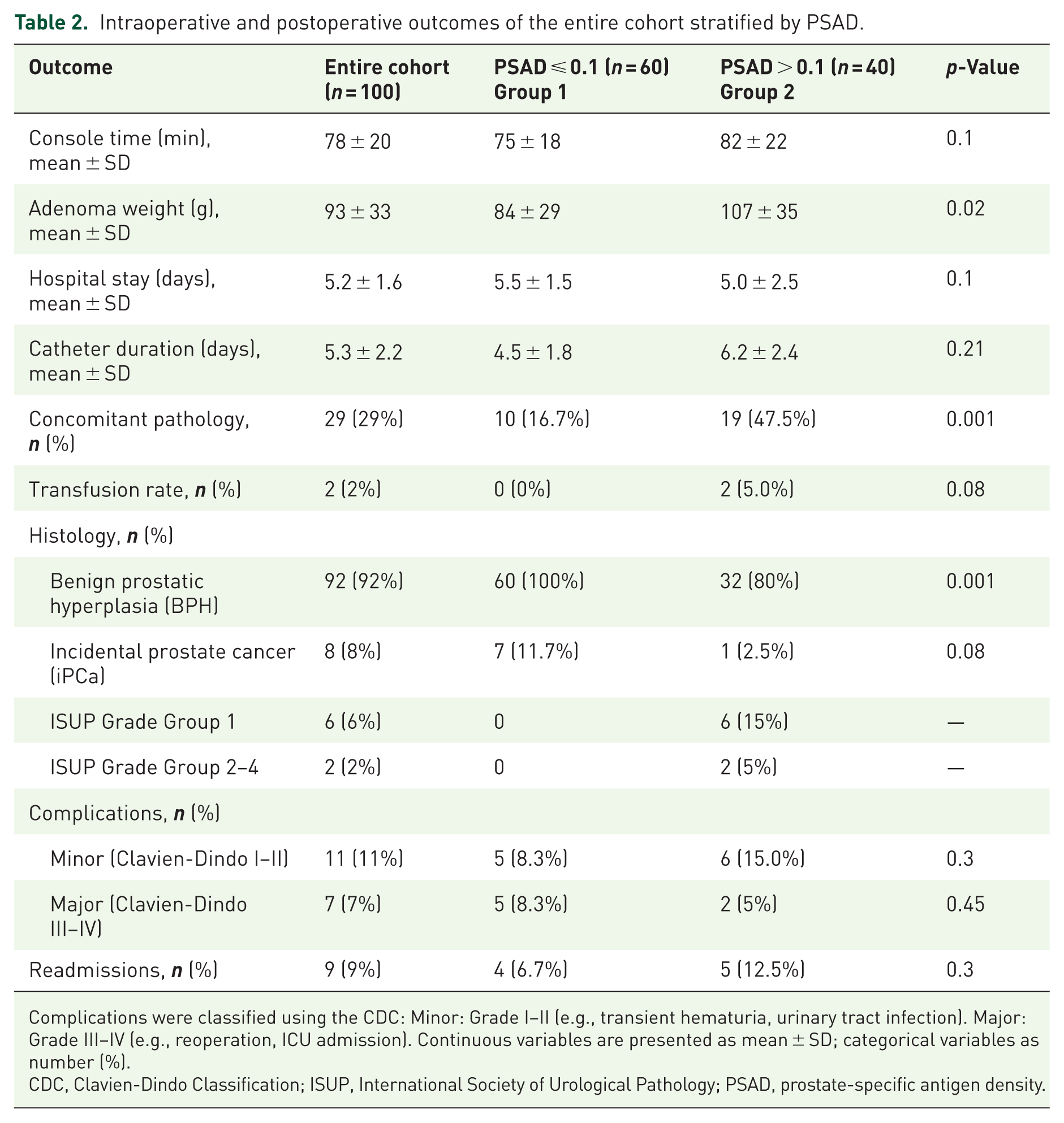
Complications were classified using the CDC: Minor: Grade I–II (e.g., transient hematuria, urinary tract infection). Major: Grade III–IV (e.g., reoperation, ICU admission). Continuous variables are presented as mean ± SD; categorical variables as number (%).
CDC, Clavien-Dindo Classification; ISUP, International Society of Urological Pathology; PSAD, prostate-specific antigen density.
Overall, 8 men out of 100 had prostate carcinoma in the final pathology (8%). Seven of them (88%) were found in group 1, patients with PSAD under 0.1 ng/ml/cc. Overall, 6 had ISUP 1, and 2 had clinically significant carcinomas, ISUP 2 or 3 in their final histology. Five patients had <5% PCa, while three had ⩾5% (pT1b). Patients with ISUP ⩽ 1 were managed with active surveillance; only one chose robot-assisted radical prostatectomy, and the two patients with ISUP 2 and 3 opted for external radiation. Among the eight iPCa cases, 50% (4/8) underwent only preoperative MRI, and 25% (2/8) had only prostate biopsies. Two patients (25%) had both MRI and biopsy, yet iPCa was undetected preoperatively—further details in Table 3.
Analysis of incidental prostate cancer predictors.
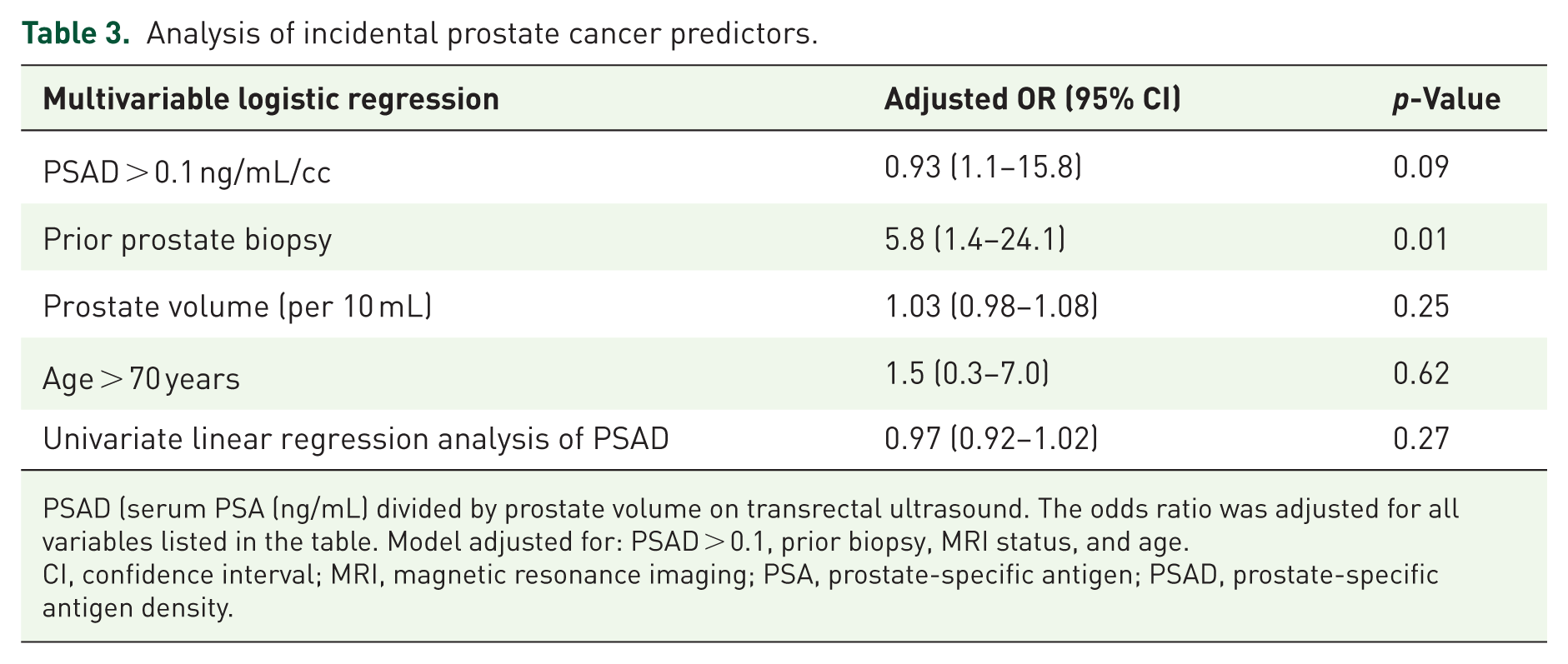
PSAD (serum PSA (ng/mL) divided by prostate volume on transrectal ultrasound. The odds ratio was adjusted for all variables listed in the table. Model adjusted for: PSAD > 0.1, prior biopsy, MRI status, and age.
CI, confidence interval; MRI, magnetic resonance imaging; PSA, prostate-specific antigen; PSAD, prostate-specific antigen density.
Predictors of incidental prostate cancer
We conducted a multivariable logistic regression analysis to determine if PSAD, prior prostate biopsy, prostate volume, and age can predict iPCa. The results revealed that the PSAD = 0.1 ng/ml/cc threshold failed to predict iPCa. The same was true for prostate volume and age (p = 0.25 and 0.62). Only biopsy history was linked to 5.8 times greater odds of iPCa (p = 0.01). Furthermore, we ran a univariate linear regression model using PSAD as a continuous variable to predict iPCa. The result showed no significant association (OR = 0.97 per 0.01 ng/mL/cc increase, 95% CI: 0.92–1.02, p = 0.27). Details are given in Table 3.
Additionally, when comparing the PSAD between patients with carcinoma and those without, the mean PSAD for carcinoma patients was 0.07 ng/ml/cc, lower than 0.1 ng/ml/cc for patients with BPH. Their prostate volume was also significantly higher, 182 versus 140 ml (p = 0.02). Refer to Supplemental Table 1 for further details.
Discussion
The key finding of our study is that a PSAD threshold of 0.1 ng/mL/cc failed to predict iPCa (OR = 0.093, p = 0.09). PSAD was unable to differentiate between patients with large prostates who may harbor iPCa (Supplemental Table 1). In a comparable cohort of patients with large prostates, Herlemann et al. found that increased patient age and higher PSAD were associated with iPCa. 22 Their patients exhibited an average prostate volume of 80 ml, with an iPCa incidence of 17%. In our current study, we could not confirm the practicality of PSAD as a predictive tool for iPCa within the BPS population with larger prostates than 80 ml.
In our practice, while we utilized PSAD-driven triage for patient management, men with low PSAD (⩽0.1 ng/mL/cc) proceed to surgery without further diagnostics. Men exhibiting high PSAD (>0.1 ng/mL/cc) should ideally undergo a prostate MRI and proceed to prostate biopsy if indicated, to avoid the incidental finding of clinically significant prostate cancer. Based on this study, we cannot, however, provide scientific evidence to substantiate our practice. While PSAD has diagnostic and predictive value in the BPH population with smaller prostates,14,23 it may not provide the same accuracy in men with larger prostates, possibly due to PSA being diluted by the significant volume of the prostate or concomitant prostate inflammation. Our findings correlate with those of Omri et al., 16 who found that PSAD correlated with csPCa only in small and medium-sized prostates, but not in large glands.
In their multicenter review of incidental prostate carcinoma in patients undergoing tissue-ablative surgery for benign prostatic hyperplasia, Yoo and associates reported performing prostate biopsies on all patients with a PSA level of ⩾4.0 ng/ml or with abnormal digital rectal examination (DRE). 24 They suggested PSAD as a predictor of iPCa. This research predates the use of prostate MRI and was based on standard practices at the time. In their study, only 4.8% of men were diagnosed with iPCa; however, in our cohort, the rate was 8%, even though over half (57%) of men in our study and all patients with iPCa had an MRI, a biopsy, or both before RASP. In our study, seven out of eight patients diagnosed with iPCa had a PSAD below 0.1 ng/ml/cc. The two men diagnosed with csPCa exhibited PSAD values of 0.03 and 0.01. This finding challenges the use of a PSAD cutoff. Our results can’t support either a PSAD cutoff of 0.10 ng/ml in biopsy-naïve patients or 0.15 ng/ml in those with a prior negative biopsy. 23 Furthermore, univariate linear regression with PSAD as a continuous variable showed that PSAD failed as a predictor of iPCa, even when analyzed continuously. The analysis confirms that PSAD, whether dichotomized or continuous, didn’t predict iPCa in men with very large prostates.
The sole significant predictor of iPCa identified in our study was a history of prior prostate biopsy (OR = 5.2, p = 0.01). Nguyen et al. 17 concluded that a PSAD cutoff exceeding 0.15 ng/ml/cc in patients with PIRADS three lesions on prostate MRI would result in a 71% reduction in the number of required biopsies; however, this approach would lead to the omission of 15% of cases involving csPCa. In contrast to our results, their study found a negative correlation between a history of prostate biopsy and csPCa. However, a cost-effectiveness approach that reduces unnecessary biopsies in low-risk patients while prioritizing advanced diagnostics for high-risk individuals, like those with higher PSAD and previous prostate biopsy history, allows them to have a prostate MRI before RASP. Our results may also be relevant to patients with enlarged prostates undergoing other procedures.
Despite its limitations, PSAD remains a clinically useful and cost-effective triage tool. At our institution, Prostate MRI involves considerable out-of-pocket costs (€600) and prolonged waiting times (2–3 months), while biopsies, although covered by insurance, carry non-trivial risks. 8 Nearly half of the patients (44%) had catheters in place preoperatively, creating an urgency to avoid diagnostic delays. Furthermore, elderly patients (e.g., ⩾80 years) may opt for watchful waiting even if cancer is detected.
In our study, several limitations must be acknowledged: the retrospective nature, single-center design, and modest sample size may limit the generalizability of our findings. Without conducting power analysis to determine the appropriate sample size, we included all patients who underwent RASP at our institution within the study period, without applying any exclusion criteria. This approach was adopted to reflect real-world scenarios. In our cohort, 21 patients received 5α-reductase therapy, with 19 also on combination therapy including an α-blocker. We avoided discussing the impact on PSA levels and PSAD because most didn’t have the treatment for enough time (e.g., 6 months) and chose prompt surgery to relieve symptoms. This factor may impact the accuracy of our ongoing study, necessitating that we interpret our results with caution. Additionally, our multivariable model, incorporating four predictors (PSAD > 0.1, prior biopsy, prostate volume, and age), is at a high risk of overfitting. According to the typical rule of at least 10 events per predictor variable, around 40 events would be needed for our model, but we fell short of this requirement. This overfitting suggests the model’s estimates may be overly optimistic. We chose to dichotomize PSAD at the clinically relevant threshold of 0.1 ng/mL/cc based on our institutional protocol to improve clinical interpretability. We acknowledge, however, that this approach decreases statistical power. The major complication rate (7%) is within the range reported in other studies.25,26 It’s essential to highlight that we recorded all complications within 90 days after surgery. A total of 34% of our patients were classified as ASA 3, indicating higher comorbidity. The transfusion rate was 2%, which is favorable compared to other studies [Dotzauer, 2021 #300]. Additionally, we only placed DJ stents or ureteral catheters in 3/100 cases during the procedures. Moreover, 44% of patients underwent surgery while having a preoperative catheter, and 40% of them had concomitant pathologies, which could complicate the operation due to catheter-associated bacteriuria. Lastly, a learning curve bias must still be acknowledged, even though the single surgeon involved in this study has performed almost 2500 robot-assisted surgeries. MRI availability varied due to financial constraints, potentially underestimating its utility. Prospective multicenter validation of the PSAD’s usefulness, integrating PIRADS scores and targeted biopsy techniques, is warranted.
Conclusion
PSAD, whether dichotomized (threshold of >0.1 ng/mL/cc) or continuous, didn’t predict iPCa in men with large prostates. This is probably due to PSA dilution in large glands, which restricts the utility of PSAD. A history of prior prostate biopsy suggests an elevated risk for iPCa in BPH patients undergoing bladder outlet surgery.
Prostate MRI is crucial in addressing the diagnostic challenges associated with large prostate glands. Multicenter validation is warranted to enhance preoperative risk stratification.
Supplemental Material
sj-pdf-1-tau-10.1177_17562872251386996 – Supplemental material for Incidental prostate cancer: is prostate-specific antigen density a valid predictor? A case–control study
Supplemental material, sj-pdf-1-tau-10.1177_17562872251386996 for Incidental prostate cancer: is prostate-specific antigen density a valid predictor? A case–control study by Mahmoud Farzat and Florian M. Wagenlehner in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
