Abstract
Background:
Technical advancements and intrarenal pressure are synergistic in improving perioperative outcomes during flexible ureteroscopy (FURS). Mismanaged intra-renal pressure (IRP) has negative consequences and it is associated with an increased risk of sepsis, bleeding, pelvicalyceal fluid extravasation, and even collecting system injuries and acute as well as chronic renal failure. The cornerstone of a safe FURS is the ability to continuously monitor IRP to avoid elevation of IRP above the normal range of 10 mmHg.
Objectives:
This scoping review aims to report the current state of real-time IRP monitoring in in vivo clinical studies and the various monitoring methods and technology to understand how this may be best used in daily clinical practice.
Eligibility criteria:
A systematic literature search was conducted. Only in vivo clinical studies published in English documenting IRP measurement methodologies during semirigid or flexible ureteroscopy for urolithiasis management were included.
Results:
Out of 1326 retrieved papers, 17 studies met the inclusion criteria, comprising 2 randomized controlled trials, 2 retrospective studies, and 13 observational studies. Current noninvasive IRP monitoring devices include ureteric catheters placed retrogradely or via percutaneous tubing and connected to pressure transducers, amplified by cardiology-used pressure sensing systems or urodynamic systems, automated pressure-regulating systems, pressure sensing guidewires, and IRP sensing flexible ureteroscopes. The review revealed significant variations in IRP measurement methods, reporting units, and irrigation techniques. Notably, elevated IRP above 30 mmHg was consistently associated with increased postoperative complications, including sepsis.
Conclusion:
Current clinical studies have only ascertained that increased IRP above 40 mmHg positively correlates with infectious complications and postoperative pain. No standardized values are available to predefine safe thresholds in practice. With the availability of noninvasive tools for IRP monitoring, future research should focus on multicenter studies to establish reference ranges and best practices for IRP management, ultimately improving patient outcomes in endourological procedures.
Keywords
Introduction
Technical advancements and intrarenal pressure (IRP) are synergistic in improving perioperative outcomes during flexible ureteroscopy (FURS). Clear vision is crucial for operative success, but due to irrigation tools, this often comes with the drawback of high IRP. The pressurized irrigation flow used during FURS helps maintain optimal vision by ensuring adequate flow during stone lithotripsy and when instruments occupy the channel of the scopes. Mismanaged IRP has negative consequences and is associated with an increased risk of sepsis, bleeding, pelvicalyceal fluid extravasation, and even collecting system injuries and acute as well as chronic renal failure.1–3 Of the proven techniques to manage pressure, the use of a ureteral access sheath (UAS), medical regulation of pressure, and pressure monitoring devices have been employed.1,2
The cornerstone of a safe FURS is the ability to continuously monitor IRP to avoid elevation of IRP above the normal range of 10 mmHg, 4 with ex vivo research on human kidneys showing pyelorenal reflux occurring at 30 mmHg and pyelovenous backflow at 45–75 mmHg.5,6 This can lead to the systemic spread of bacteria and endotoxin from infected stones or the collecting system, resulting in a higher incidence of postoperative urosepsis.
A flexible and navigable suction ureteral access sheath (FANS) could revolutionize the field in modern FURS. 7 Built-in pressure sensors at the ureteroscope tip or connected to FANS can also enable real-time monitoring of IRP during suction aspiration, maintaining IRP during lithotripsy below 30–40 mmHg to minimize complications.8–12
In this scoping review, we report on real-time IRP monitoring in in vivo clinical studies to understand how this may be best used in daily clinical practice.
Methods
Evidence acquisition
This review was conducted following a systematic approach according to the statements of the PRISMA extension for scoping review (PMID: 30178033; Supplemental Material). The PICOS model (Patient Intervention Comparison Outcome Study type) was used to frame and answer the clinical question: P: adult and children undergoing semirigid or flexible ureteroscopy for stone; I: in vivo measurement of IRP; C: none; O: IRP values and postoperative outcomes; and S: prospective and retrospective studies.
The literature search was conducted from inception to 16 August 2024 in several databases, including PubMed, Embase, and Scopus via Boolean operators with the use of the following terms: “intrarenal pressure, OR intrapelvic pressure, OR pressure, and then restricted with keywords (AND) retrograde intrarenal surgery, OR RIRS, OR ureteroscopy” to find studies on IRP during semirigid and flexible ureteroscopy. Only in vivo clinical studies in English with documented pressure monitoring methodology were included. Letters to the editor, meeting abstracts, case reports, and experimental and review papers were excluded. The literature analysis was realized independently by two authors (S.K.K.Y. and V.G.) through Covidence Systematic Review Management® (Veritas Health Innovation, Melbourne, Australia), and discrepancies were solved by consensus. This review was conducted in compliance with PRISMA guidelines (Figure 1).

PRISMA diagram.
Results
The literature search retrieved 1326 papers, of which 417 were duplicates. In all, 909 papers were screened against title and abstract, and 872 papers were excluded because they were unrelated to this review’s aim. The remaining 37 full-text papers were assessed for inclusion, and 20 meeting abstracts were further excluded, leaving 17 studies. There were 2 RCTs, 2 retrospective comparative studies, and 13 prospective or retrospective observational studies. Sixteen studies involved adults; only one involved children (5–18 years old). 8 The sample size ranged from 4 to 189. In 3 studies, IRP monitoring was done during semi-rigid ureteroscopy and 14 during flexible ureteroscopy. Table 1 shows the study characteristics.
Study characteristics.

Variation in reported units and methods in calibration to zero
All except studies by Deng et al. 12 and Huang et al. 11 reported real-time IRP values (Table 2). The most commonly used unit for IRP in clinical studies was mmHg (n = 13), followed by cmH2O (n = 4). Calibration to zero was mentioned in the methodology and performed in 10 of the 17 included clinical studies.
IRP measuring methods and their method of calibration to zero, baseline, and maximum IRPs, safety cutoff IRP value in the study.

Figures are converted from cmH2O to mmHg for easy comparison with the formula: 1cmH2O = 0.73 mmHg.
IRP, intra-renal pressure; SD, standard deviation; UAS, ureteral access sheath.
Variations in IRP values
The mean baseline IRP ranged from 4.38 to 34.8 mmHg (Table 2). IRP peaks as high as 328 mmHg were noted during saline irrigation in FURS. The mean peak IRP following a 10 mL bolus with and without the syphon UAS was 71 versus 104 mmHg, while IRP in automated pressure monitoring integrated irrigation and suction system was consistently below 30 mmHg.
The recorded median IRP varied between different studies, generally being lower than 30 mmHg. The lowest IRP level was at −4.6 ± 2.1 mmHg in automated irrigation and suction platform systems. A total of 30 mmHg was the most frequently used cutoff value for the safety threshold in these studies. The following standard safety cutoff was 40 cmH2O and 40 mmHg.
Variations in IRP monitoring
Suction ureteral access sheath (SUAS) was used in six studies, including an automated system with suction in four studies, general suction UAS in one study, and syphon UAS in one study (Table 3). The size of UAS was 9/11Fr, 10/12Fr, 11/13Fr, 12/14Fr, and 14/16Fr. Pressure guidewire was used in nine studies for the measurement of IRP; a ureteral catheter inserted into the renal pelvis through a ureter or nephrostomy tube connected to either a transducer or a urodynamic machine was adopted in four studies, while irrigation and suction platform which can also record the real-time IRP was used in four studies.
Irrigation and suction settings and ureteral access sheath sizes used in the study.
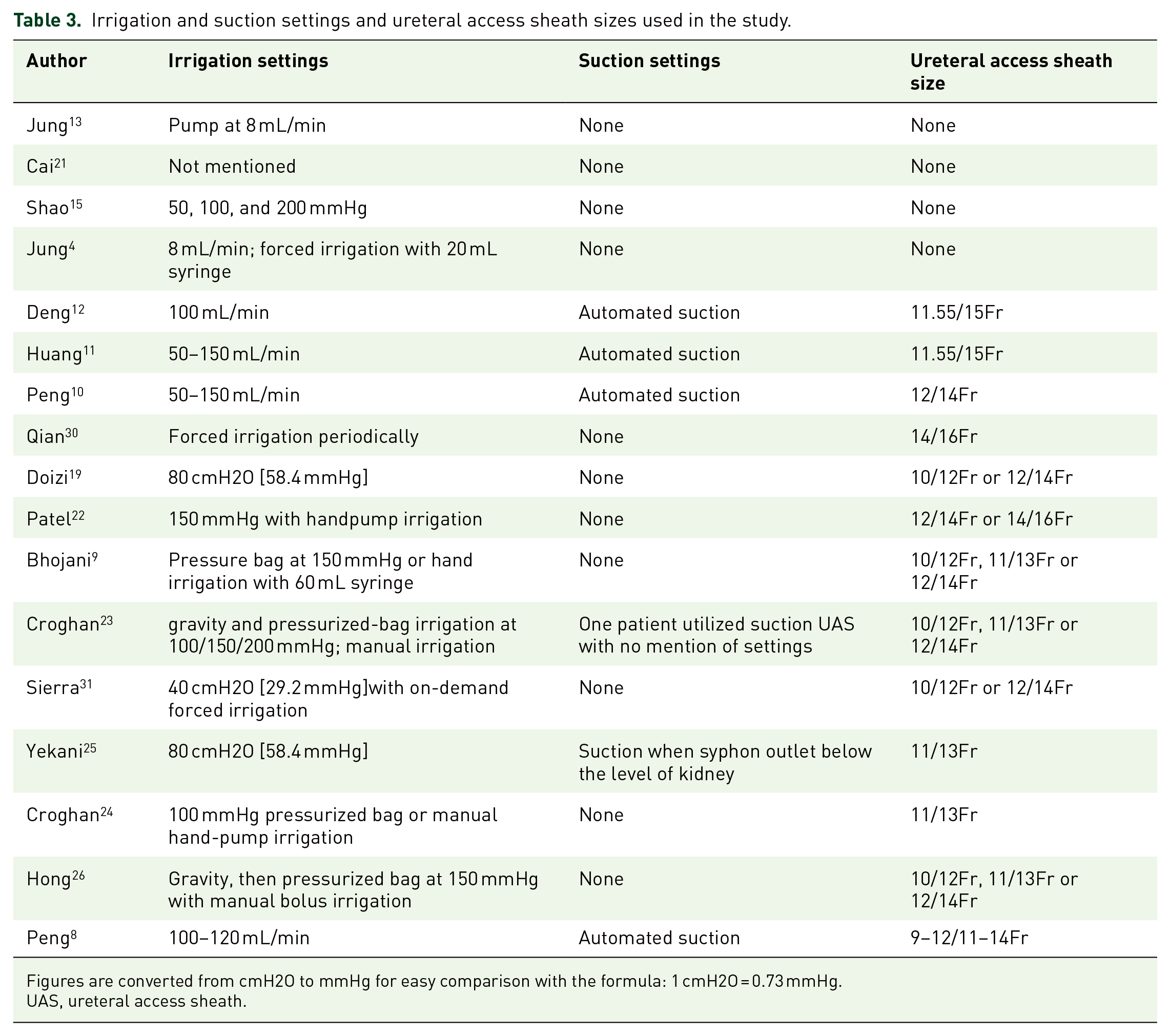
Figures are converted from cmH2O to mmHg for easy comparison with the formula: 1 cmH2O = 0.73 mmHg.
UAS, ureteral access sheath.
Variation in irrigation reporting
A mechanical pump, a hand-held pump, pressure bags, and hand syringe irrigation were used for irrigation (Table 3). Irrigation pressure varied in these studies, ranging from 30 to 150 mmHg, while the flow rate ranged from 8 to 150 mL/min.
In the included studies, 612 patients underwent FURS. The first in vivo study was done in 2008, in which real-time IRP during FURS was measured noninvasively via a retrogradely placed 4Fr ureteric catheter connected to a transducer (Statham Gould #4523551, Dunlap, England) amplifier, and monitor (Medistim CardioMed CM-4008, Oslo, Norway). 13
Nine of the included studies were published from 2021 onwards. Of note, seven of these studies are from Asian centers. Only five studies utilized pressure sensing devices specially developed for urological use, of which four were published by Asian centers.
Discussion
Standardization in reporting IRP monitoring in endourology
Traditionally, in vivo IRP measurement involved pressure transducers connected to nephrostomy tubes14,15 or ureteral catheters,16–18 which were invasive or could potentially impact the accuracy of the readings. Pressure sensor guidewires have been adopted from cardiology and used off-label to measure IRP. 19 Recent technological advancements in the production of semiconductors have helped reduce the size of pressure sensors from the centimeter level in the middle of the last century to the micron level (200 µm) for precise IRP measurement in urology. 20
Tokas et al. 1 attempted in 2019 to report cmH2O as the standard for IRP measurements, as this was also the standard measurement in urodynamics, nonclinical studies, and due to a scarcity of technology. In modern endourology, mmHg is the widely preferred reported unit in 13 of 17 studies.4,8–13,15,21–26 Although the unit is interchangeably converted via simple calculation (i.e., 1 cm H2O = 0.73 mmHg), there is a need to rethink how to report IRP measurement, as outcomes can only be comparable if standardized.
While the manufacturers chose to calibrate the measurement of the latest urologically used pressure sensing devices in mmHg, it also reflects the engineering, type of sensors, and calibration principles.27,28 IRP sensing devices in flexible ureteroscopy are an essential investigational tool that needs well-designed trials that could help meaningfully compare different apparatus in real-world clinical practice. 29 Otherwise, IRP measured and reported differently will pose a heterogeneous bias for clinical studies.
Aside from patient or clinical factors that would affect IRP measurements, engineering factors also play an essential role. According to the working principle, pressure sensors can be divided into piezoresistive, capacitive, optical fiber, resonant, and piezoelectric types. 20 The type of sensor and engineering principle used, how it was engineered in the clinical device, and how it was calibrated to zero before the initiation of measurements are all important factors to be considered. As in all well-designed trials, meaningful results can only be reported if IRP is recorded in a standardized manner across all measuring tools.
Currently available tools and mechanisms of IRP measurement
In Figure 2, we classify the tools broadly into minimally invasive (antegrade) and noninvasive (retrograde) methods. We also provide a device-based subclassification that incorporates all available technology that has been developed.
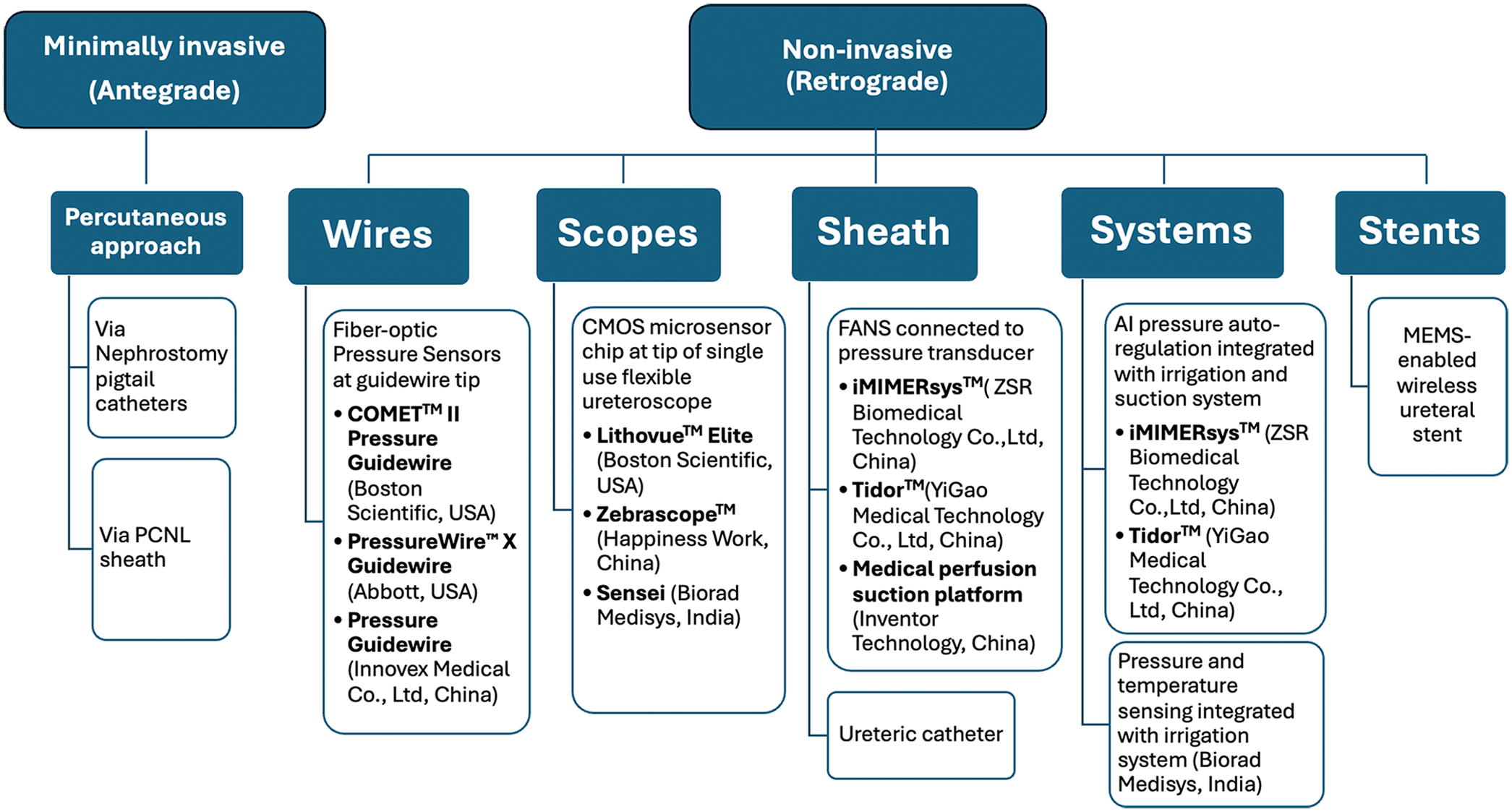
Classification of IRP monitoring devices.
All clinical studies published from 2008 to 2015 utilized ureteric catheters placed retrogradely or via percutaneous tubing and connected to pressure transducers, amplified by cardiology-used pressure sensing systems or urodynamic systems.4,13,15,21 Since 2016, clinical studies have used automated pressure-regulating systems.8,10–12 From 2021 onwards, seven clinical studies were published utilizing the pressure sensing guidewires.19,22–26,30,31 Only one in vivo study has been published using the IRP sensing flexible ureteroscopes. 9 Clinical evaluation of micro-electro-mechanical system (MEMS) wireless ureteral stents and some automated, integrated suction irrigation systems is yet to be thoroughly studied and reported.
While the tools shown in Figure 2 demonstrate a significant positive milestone development toward monitoring IRP, they also reflect the new challenges in providing standardized reporting and measurement and the possible bias of using different systems. We propose that manufacturers use a single unit of measurement to standardize reporting to start. As end users, clinicians could adopt mmHg as most manufacturers now prefer this unit.4,8–13,21–26 This will allow the utility of reporting validation and comparative studies across different IRP devices.
Importance of IRP measurement and clinical relevance
Raised IRP above 40 cmH2O or 30 mmHg and its association with infectious complications, especially sepsis, is well studied.1,32 Recently, Croghan et al. 23 recruited 120 consecutive patients undergoing diagnostic/operative semirigid or flexible ureteroscopy with an irrigation bag positioned 50 cm above the operating table. 23 IRP was measured using a single-use 0.036 cm pressure guidewire (COMET™ II, Boston Scientific©, Marlborough, MA, USA) for intra-coronary and peripheral endovascular use. In this study, the mean IRP was 40.12 ± 2.01 mmHg during FURS. Expectedly, 5% of patients who developed infectious complications had a mean IRP significantly higher than the control group (71.16 ± 36.85 vs 38.62 ± 22.51 mmHg, p = 0.001). This is the first human study to positively correlate the relevance of sepsis and IRP findings >30 mmHg in the FURS. 4 Furthermore, seven patients required readmission for pain; although IRP was higher (49.04 ± 22.25 vs 38.37 ± 22.8 mmHg, p = 0.51), it was not statistically significant.
Congruently, a recent multicenter study of 394 kidney stone patients undergoing FURS with FANS, where suction lowers IRP, 33 demonstrated that day one loin pain measured with visual analog scale was shallow (median 1 (IQR 1–2))], 7 confirming that high IRP is commonly associated with postoperative pain. The same study 7 had zero sepsis rates; only 3.3% of patients developed postoperative fever. These studies show a positive correlation between IRP and pain but require further investigation to determine acceptable thresholds to mitigate the same. Clinically, IRP measurements are directly associated with the postoperative need for analgesia, recovery, quality of life, and overall surgical experience for patients.
Hong et al. 32 attributed the evidence that suction aspiration lowers IRP and prevents pyelocalyceal backflow, intrarenal reflux, and overstretching of the renal capsule, all of which mitigate the risk of sepsis and prevent the release of inflammatory markers and interleukin response associated with overdistention of the pelvicalyceal system. 34 Deng et al., 12 who used an irrigation suction platform with a pressure-sensing SUAS that kept IRP between −2 and 30 mmHg, reported only 5/90 patients experienced low-grade fever for less than 24 h. 12
In addition, the relationship between high IRP and postoperative fever was demonstrated by Qian et al. 30 With a 40 mmHg threshold, 49/136 patients had a higher IRP and had a significantly higher incidence of fever (high-pressure vs low-pressure group 22.45% vs 2.30%, p < 0.001). These studies validate the need to deploy effective measures to lower IRP during FURS.
Figure 3 summarizes the available practical and proven strategies for lowering IRP in FURS. Various measures have been suggested to minimize IRP during the endourological intervention. Some are experimentally proven, and some are clinical hypotheses. 35

Measures to minimize IRP and practice safe flexible ureteroscopy.
UAS is a well-established, simple tool for lowering IRP in FURS. In our review, 12 of the 17 studies reported using UAS of variable sizes. The mean reported IRP was significantly different across these, reiterating the heterogeneity in IRP reporting across various clinical practices. Furthermore, information on the baseline IRP was not well defined for most studies, and the safety cutoff of a 30–40 mmHg threshold was described in only six studies.8–12,30
Unfortunately, we cannot provide a reference range that can be uniformly applicable for FURS when using different scopes, sheaths, and lasing devices. This reiterates that we have the tools but lack the data for specific practical recommendations. There is an urgent need for large multicenter studies.
Medical management
Another way to reduce IRP during flexible ureteroscopy is using intraluminal administration of isoproterenol, a β-agonist. Jung et al. 13 randomized 12 patients undergoing flexible ureteroscopy to irrigation with saline or isoproterenol 0.1 mg/mL using a standardized irrigation rate of 8 mL/min. With similar baseline IRP (12.1 ± 4 mmHg in the saline group and 10.3 ± 4 mmHg in the isoproterenol group), they found that in the saline group, IRP increased to a mean of 33 ± 12 mmHg during ureteroscopy. At the same time, it was significantly lower in the isoproterenol group (19 ± 3 mmHg, p = 0.029). Yet, IRP peaks as high as 328 mmHg were noted during saline irrigation, while the number of pressure peaks >50 mmHg was minimized dramatically during isoproterenol irrigation.
This study’s interpretation cautions urologists that unprecedented, uncontrolled, and unmonitored saline irrigation during FURS will lead to elevated peak pressures. This is why, even though patients may be well intraoperatively, they develop complications postoperatively. This may be worse in high-risk groups prone to sepsis, such as those of old age, multiple comorbidities, and positive preoperative urine culture. 36
Understanding IRP to aid in the safe utilization of flexible ureteroscopy
With a global aging population, FURS is progressively used as a minimally invasive option for unilateral and bilateral renal stones.37,38 A study by Hong et al. demonstrated an inverse correlation between IRP and age during routine flexible ureteroscopy (Pearson correlation r[38]: 0.391, p = 0.013), with the mean and maximum IRPs being higher in younger patients than in the elderly. 26 This becomes imperative to investigate as we extend the utility and indication of FURS in children and even infants.39–41
It is crucial to have precise tools to regulate irrigation fluid and monitor IRP to mitigate any risk of preventable complications.
To understand the influence of the type of irrigation methods and IRP, Croghan et al. 24 randomized 38 patients to undergo FURS with a pressurized bag vis-a-vis hand pump irrigation. They found that hand pump irrigation resulted in significantly higher IRP (62.29 ± 27.45 vs 38.16 ± 16.84 mmHg, p = 0.005). Both irrigation systems were higher than the traditional safety IRP of 30 mmHg. 4 No complication was observed in the pressurized bag cohort. At the same time, one patient in the high-pressure arm had intraoperative bleeding from the collecting system that obscured vision and needed the procedure to be stopped temporarily. One further patient in the high-pressure group developed sepsis. These findings are pertinent as we lack a single irrigation pressure system that can regulate both and is reproducible in all patients. Perhaps this is the future direction in which the industry can focus.
In a recent study that combined FANS and IRP monitoring using a tip flexible pressure controlled UAS in children, no major complications were seen in all 21 patients. 8 Using regulated monitoring, suction, and controlled irrigation at 100–120 mL/min, the median IRP was as low as negative 4.6 ± 2.1 mmHg. This clinical study objectively demonstrated and emphasized the importance of integrated pressure and fluid management to prevent complications due to unprecedented IRP. This should be the future direction for FURS intervention.
Current limitations and future directions
IRP in FURS matters. Mismanagement significantly and negatively influences infectious outcomes post-procedure pain. Yet, we need standardized tools to measure and report IRP, with only isolated small-sample-size studies reporting its clinical use. Compounding this is how it is estimated with IRP scopes or wires as the most common methods deployed.
No head-to-head comparisons have been conducted to determine whether the different systems are comparable and if any variability exists. Discrepancies in surgical techniques, such as ureteroscopy methods, and the heterogenicity of different types of study designs all influence the interpretation of IRP. By collating these results in our review, we hope to help understand the nuances involved and work toward the best approach to manage IRP. In our study, we observe urologists are unable to categorize which laser lithotripsy techniques or what irrigation and suction settings are considered safe without increasing concerns about raised IRP. Information on how different anatomical systems behave to pressure changes and if this will impact FURS outcomes are missing. In addition, patient factors need to be taken into consideration. For patients at a higher risk of postoperative sepsis, we may need to evaluate if keeping the IRP lower than that of the recommended 30–40 mmHg4,5,11,12 is more beneficial especially with the introduction of suction in FURS. Companies that have integrated IRP in scopes do not have adequate clinical data to discuss best practice guidelines while using the tools for IRP management. More information is needed to determine if the prestenting or use of alpha-blockers has any role in mitigating IRP. Optimal IRP strategies in different age groups undergoing FURS need independent understanding. As suction gains momentum and more and more studies document the positive effects of having a balance in suction irrigation and aspiration, we need integrated platforms that can automatically manage irrigation and suction pressures to allow for a continuous low IRP environment that does not impact the ease of performing FURS. Such integrated platforms should have accessible user interfaces and compatibility with existing scopes or sheaths to make them user-friendly and allow for seamless integration.
Currently, to our knowledge, only i-MIMERsys™ (ZSR System Biomedical Technology Co., Ltd, Dongguan, China) is commercially available as a platform for such integrated fluid and pressure management. The Lithovue™ Elite (Boston Scientific Corporation, Marlborough, MA, USA) is the only single-use flexible ureteroscope that has been FDA-approved for IRP monitoring. Still, it does not come with an integrated intelligent fluid management system.
The most significant limitations are the restraints of these systems being differentially available to health systems and the lack of adequate clinical data to measure each one’s efficacy. Also, the integrated pressure systems use their proprietary suction-equipped sheaths, making cross-platform usability impossible.
We have the technology, the experimental knowledge, and the clinical proof that IRP in FURS matters and can be measured. However, it remains to find an effective standardized reporting format that includes objective parameters, such as irrigation and suction pressure and flow rates, to evaluate IRP and effectively determine a safety cutoff during FURS.
One such parameter we propose is the irrigation suction ratio value, which can define the amount of irrigant fluid during aspiration to prevent dangerous fluctuations in IRP and maintain good operative visibility. As suction is a proven game changer 42 and suction UAS continues to evolve and influence FURS, 33 it will become imperative to monitor pressure during FURS.
Last but not least, as the concept of direct-in-scope suction gains traction and its role becomes more defined in FURS, 43 it will become increasingly important to monitor pressure in these cases as well.
Conclusion
Current clinical studies have only ascertained that increased IRP above 40 mmHg positively correlates with infectious complications and postoperative pain. No standardized values are available to predefine safe thresholds in practice. With the availability of noninvasive tools for IRP monitoring, we must aim for comparative studies that can establish reference ranges to allow better control of IRP when using irrigation and suction fluid management systems for FURS in different clinical scenarios.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251314809 – Supplemental material for Current utility, instruments, and future directions for intra-renal pressure management during ureteroscopy: scoping review by global research in intra-renal pressure collaborative group initiative
Supplemental material, sj-docx-1-tau-10.1177_17562872251314809 for Current utility, instruments, and future directions for intra-renal pressure management during ureteroscopy: scoping review by global research in intra-renal pressure collaborative group initiative by Steffi Kar Kei Yuen, Wen Zhong, Yun Sang Chan, Daniele Castellani, Naeem Bhojani, Madhu Sudan Agarwal, Theodoros Tokas, Stefanie Croghan, Helene Jung, Thomas Herrmann, Bhaskar Somani and Vineet Gauhar & The Global Research on Intra-renal Pressure Collaborative Group in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
