Abstract
Recent advances in ultrasonography (US) technology established modalities, such as Doppler-US, HistoScanning, contrast-enhanced ultrasonography (CEUS), elastography, and micro-ultrasound. The early results of these US modalities have been promising, although there are limitations including the need for specialized equipment, inconsistent results, lack of standardizations, and external validation. In this review, we identified studies evaluating multiparametric ultrasonography (mpUS), the combination of multiple US modalities, for prostate cancer (PCa) diagnosis. In the past 5 years, a growing number of studies have shown that use of mpUS resulted in high PCa and clinically significant prostate cancer (CSPCa) detection performance using radical prostatectomy histology as the reference standard. Recent studies have demonstrated the role mpUS in improving detection of CSPCa and guidance for prostate biopsy and therapy. Furthermore, some aspects including lower costs, real-time imaging, applicability for some patients who have contraindication for magnetic resonance imaging (MRI) and availability in the office setting are clear advantages of mpUS. Interobserver agreement of mpUS was overall low; however, this limitation can be improved using standardized and objective evaluation systems such as the machine learning model. Whether mpUS outperforms MRI is unclear. Multicenter randomized controlled trials directly comparing mpUS and multiparametric MRI are warranted.
Introduction
Prostate cancer (PCa) is the second most frequent cancer and the fifth highest cause of cancer death among males worldwide in 2020. 1 Transrectal ultrasound (TRUS) prostate biopsy (PBx) has been the gold standard for diagnosing PCa. 2 Although TRUS has several strengths including availability, cost-effectiveness, familiarity to urologists, and ability to real-time guidance, TRUS alone cannot reliably detect PCa. 3 Thus, more accurate diagnostic method is needed.
Currently, magnetic resonance imaging (MRI) enables anatomical/functional imaging of the prostate and visualizes the majority of clinically significant prostate cancer (CSPCa).4–7 Interpretation of multiparametric magnetic resonance imaging (mpMRI) sequences is standardized by the Prostate Imaging Reporting and Data System (PIRADS). 8 Using software to fuse previously obtained mpMRI and real-time TRUS images, the MRI/TRUS fusion PBx integrates the advantages of both MRI, with its ability for lesion detectability, and TRUS, with its real-time imaging guidance.6,9 MRI-visible lesion can be precisely sampled by MRI/TRUS fusion targeted PBx, and each biopsy trajectory with spatial coordinates in the prostate is recorded in the MRI/TRUS fusion software for review. In fact, MRI/TRUS fusion PBx gained popularity and mpMRI prior to PBx for all men with a suspicion for PCa is recommended by guidelines (i.e. European Association of Urology, American Urological Association, and Society of Abdominal Radiology). mpMRI, however, has several limitations including availability, the expensive cost, the difficulty for real-time imaging, and low inter-reader agreement.4,5 In addition, patient/prostate movement, prostate deformation, and the registration error between MRI and TRUS images may have impact on the tumor detection and localization.10,11
Recent advances in ultrasonography (US) technology established US modalities, such as Doppler-US, HistoScanning, contrast-enhanced ultrasonography (CEUS), elastography, and micro-ultrasound (micro-US).12–17 Although these new modalities have some limitations including need of specialized equipment, inconsistent results, lack of standardizations, and external validation, the early results of multiparametric US have been promising (Table 1).18–23 The ‘Multiparametric Ultrasound (mpUS)’ is attracting attention in the field of Urology and Radiology. The European Association of Urology (EAU) guidelines on PCa mentioned mpUS as a promising imaging approach for PCa diagnosis, but they also pointed out that lack of standardization and lack of large-scale evaluation were drawback. 24 Currently, it is not stated on guidelines whether mpUS should be a supplemental or standalone imaging technique on PCa diagnosis. Herein, we review studies focusing on the role of new US modalities and mpUS in PCa diagnosis and describe future directions.
Features of US modalities.

B-mode, brightness-mode; CEUS, contrast-enhanced ultrasonography; FT, focal therapy; mpUS, multiparametric ultrasonography; PCa, prostate cancer; PZ, peripheral zone; TRUS, transrectal ultrasound; TZ, transitional zone; US, ultrasonography.
Ultrasound modalities in use for PCa diagnosis
Brightness-mode TRUS
Conventional US images are usually generated on the brightness (B) mode. 25 The characterization of tissues in B-mode US images is possible through the evaluation of acoustic parameters such as attenuation and backscattering coefficients obtained from radiofrequency (RF) echo signals. 26 B-mode US images display maps of echo-signal amplitude on a monitor with gray-scale, pixel-brightness values that are a function of the video signal. 27 First, the probe emits a short US pulse that will penetrate deep into the tissue or, when encountering a tissue with a different acoustic impedance, be reflected toward the transducer. Images are then created by detecting these backscattered US waves. The length of the ultrasonic waves and the various acoustic impedances impact the generation of a B-mode image.28,29
B-mode imaging has limitations in PCa detection as the backscatter signals from PCa, and normal prostate can be similar (Figure 1(a)). There also tends to be heterogeneity in the signals generated from the transition zone in this mode of sonographic imaging. 30 Furthermore, when different settings are utilized to generate B-mode images of the same tissue, such as gain and time-gain compensation settings, diverse images are expected to be visualized. Using the same imaging settings between different operators may help mitigate this the most. 28

Representative images of B-mode, CDUS, and CEUS. A 55-year-old man with PSA 6.8 ng/ml on active surveillance for GG1 cancer. Follow-up biopsy revealed GG2 cancer on right peripheral zone from apex to base. 2 cm × 1.5 cm hypoechoic lesion (a) with flow surrounding the lesion and diffuse flow within the lesion on CDUS (b) was confirmed (arrowhead). On CEUS imaging, (c) a faster contrast uptake in the right apex of the prostate and regular uptake to the rest of the prostate (arrowhead).
Traditionally, B-mode sonographic imaging has been utilized as a part of the primary detection method of PCa. TRUS B-mode imaging has been extensively studied to assess its diagnostic capacity for detecting PCa. Its advantages include providing real-time images and being the most accessible, economical, and least harmful medical imaging device. 26 The sensitivity and specificity of conventional B-mode TRUS are limited and range from 40 to 50% in PCa detection from most studies, however. 30 According to Postema et al., 29 TRUS Standard B-mode has sensitivity in PCa detection ranging only from 11 to 35% and a positive predictive ranging from 17 to 57%. The utility of B-mode ultrasound, however, has been seemingly undermined according to Steinkohl et al., 31 who showed that the B-mode TRUS imaging of the prostate could detect up to 62% of mpMRI-visible lesions, which is currently the most accurate imaging modality. In addition, B-mode has been regarded as insufficiently accurate for tumor detection, making systematic US-guided biopsies necessary. Systematic biopsy is a widely accepted practice for PCa detection in which samples from pre-defined anatomical zones of the prostate are retrieved in a nontargeted manner. 26 Given the randomness of this approach, however, it causes an increase in false-negative rates up to 35%. 31
Doppler-US
Color Doppler ultrasonography (CDUS) has been proposed as a tool to improve the accuracy of TRUS using other parameter than echogenicity on regular B-mode US. 12 The vascularity of a suspicious hypoechoic area on regular gray-scale B-mode US can be identified and characterized by CDUS or Power Doppler ultrasonography (PDUS). 32 Although PDUS is more sensitive than CDUS in detecting slow blood flow, PDUS has not shown better PCa detection than CDUS. 32 In addition, PDUS does not depict the direction of the blood flow. 22
Angiogenesis is one of the pathologic hallmarks for the growth of tumors. A Doppler shift is defined as the wavelength difference caused by the movement of an object. The principle behind Doppler-US is to help identify the wavelength changes reflecting the moving cells within the bloodstream. 33 Therefore, an increased number of shifts is associated with neovascularization in that tissue.22,17
The Doppler flow patterns between malignant and benign lesions were assessed by Ashi et al. 12 In that series, the median velocity of PCa lesions was 1.35 cm/s compared with a median of 0.36 cm/s for their benign counterparts. In addition to that, the flow was continuous and phasic for malignant lesions versus irregular on benign ones. The flow characteristics seen on PCa areas are possibly related to the higher number of vessels, increased diameter, and reduced flow resistance. The lack of smooth muscle on neovascularized vessels explains the reduced resistance to flow. Furthermore, the increased cell volume within the lesion can constrict these vessels, increasing flow velocity. CDUS can detect vessels as small as 1 mm, yet microvessels in prostate malignancies are as little as 10 microns, representing a diagnostic challenge for this tool. Nevertheless, PDUS can be used to increase the sensitivity to smaller vessels (less than 2 mm).17,34
When evaluating a PCa lesion with CDUS, different flow patterns have been described: (1) focal within the lesion, (2) flow surrounding the lesion, and (3) diffuse flow within the lesion, the latter being the most common pattern (Figure 1(b)). 32
Kuligowska et al. 35 reported PCa diagnostic performance of CDUS using 544 PBx patients who underwent sextant biopsy and targeted biopsy of US abnormalities. Sensitivity/specificity for PCa detection was 41%/85% for TRUS alone, 43%/66% for CDUS alone, and 57%/61% for the combination. There may be a room for further improving the performance of Doppler-US. Recently, Zeng et al. 36 investigated diagnostic performance of three-dimensional (3D) power Doppler ultrasound (3D-PDUS) for CSPCa detection with the virtual organ computer-aided analysis technique. A total of 99 participants with suspicion for PCa prospectively underwent TRUS + PDUS + 3D-PDUS. Using detection of CSPCa on PBx as the reference standard, vascularization index determined by 3D-PDUS achieved 86% of sensitivity and 87% of specificity. The vascularization index was calculated by the percent of color-coded voxels within the volume of interest. CSPCa detection by vascularization index was 82.1% and was statistically higher than TRUS (69.5%) and PDUS (63.4%).
The sensitivity for PCa detection increases when CDUS is utilized, improving diagnostic performance of TRUS. On the contrary, specificity decreases. This can be explained by the fact that prostatitis can be interpreted as a malignant lesion plus the preferential identification of larger and higher-grade lesions with Doppler, in which angiogenesis occurs more frequently.37,38 Therefore, inflammatory changes or conditions within the prostate can be easily confused with a malignancy without the proper clinical correlation. CDUS can also be used in other situations such as surveillance comparisons on repeat biopsies or as a marker of not viable tissue after local treatments for PCa by measuring the absence of Doppler signals. 38 Major limitations of CDUS are operator-dependency and lack of standardization.
Superb microvascular imaging (SMI) is a novel technology that visualizes slow microvascular blood flow. 33 While conventional Doppler-US may depict clutter signals caused by tissue movement, SMI suppress the clutter to specifically detect blood flow signal owing to its machine algorithm. 39 Compared with conventional Doppler-US, SMI may visualize slow blood flow with high frame rates, high resolution, high sensitivity, and less motion artifact. Zhu et al. 33 found SMI detected blood vessels in 97.3% of patients with PCa, and there was a positive correlation between the quantity of microvascular on SMI and biopsy Gleason score. Interestingly, Ohashi et al. 40 reported a case in which slow blood flow in the prostate stromal sarcoma (PSS) was detected by SMI. Of note, the intratumoral blood flow was not detected by conventional CDUS. They performed targeted biopsy toward the lesion with the blood flow under SMI-guidance, and viable tumor cells were successfully sampled. They emphasized the usefulness of SMI to target the lesion with intratumoral blood flow indicating viable cells, as PSS often includes a large necrotic area. Furthermore, some researchers performed SMI pre- and post-focal ablation for localized PCa.39,41 Using SMI, they confirmed presence of tumor blood flow before ablation, and then confirmed disappearance of the blood flow after ablation to define technical success for the ablation.39,41 Although the impact of SMI on oncological outcomes has not been proven yet, SMI-guided target PBx and focal ablation seems feasible.
HistoScanning
HistoScanning is an ultrasound-based imaging system used to improve accuracy for detection, localization, tumor volume, and suggestive PCa changes, consisting of the scanning and sequentially evaluation of the prostate tissue using a rectal probe. 13 This technology allows a software analysis of unprocessed 3D-reconstructed ultrasound RF data and its comparison with a database preloaded in the system.13,42 HistoScanning software proposes three different algorithms linked to tissue heterogeneity, cell density, and vascularity to analyze the signals before displaying the results on the screen. 43
HistoScanning technology is performed in a three-step stepwise manner. First, a TRUS probe connected externally to a motor (motorized TRUS) generates a complete 3D-prostate scan. Second, the operator outlines the regions of interest (ROIs) using the HistoScanning software platform. Finally, a computerized algorithm analysis provides red-coded areas suspicious for PCa and the corresponding tumor volume in a non-real-time manner.44,45 Yet, in cases in which low-quality imaging data are encountered, purple-coded areas are displayed in the analysis.44,45
In a similar fashion to MRI–US fusion modalities, HistoScanning true targeting (HS-TT) allows for the conversion of HistoScanning results to real-time TRUS targets. 13 Following imaging and algorithm analysis, an additional software system provides the operator with commands on how to maneuver the TRUS probe fitted with a needle guide to sample cores from specific, red-coded areas that have been identified.7,44,45
In addition, just as the perineal PBx route has gained popularity, the HistoScanning perineal biopsy route has evolved as well. HistoScanning perineal-guided biopsy is performed with the patient in dorsal lithotomy. The perineal-guided biopsy is performed using a triplane US probe, and the HistoScanning report system, and the brachytherapy template grid as a guide. Therefore, template-guided HistoScanning targeted biopsy can be performed without supplementary procedures. 7
There has been controversy in the literature regarding HistoScanning performance detecting PCa.42,46 Some earlier studies show a high HistoScanning accuracy performance characteristic detecting PCa lesions of ⩾0.5 cm3 with sensitivity and specificity up to 100% and 80%, respectively. 47 Despite these early encouraging results, subsequent studies yield controversial findings, with lower sensitivity (37–70.3%) and specificity (14.7–73%).42,46,48,49 Similarly, a study in which a lower PCa detection lesion ⩾ 0.1 cm3 reported even lower results (60% and 66%, respectively). 50 Moreover, several studies concluded that HistoScanning accuracy is determined by objective factors such as prostate volume and subjective factors including operator’s experience in performing the motorized TRUS that is essential for 3D-reconstructed US RF data. 13 Thus, HistoScanning may represent a valuable tool for experienced operators.
CEUS
Joyner et al., 51 in 1967, described an ultrasonic contrast study while performing an echocardiogram. Instilling saline solution through an intracardiac catheter formed mini bubbles seen as clouds of echoes during the test. CEUS consists of the imaging technique involving the administration of intravenous ultrasound contrast agents to improve the visualization of structures of interest. 52 Specifically, for PCa, this strategy aids in the detection, diagnosis, and follow-up of suspicious lesions while performing targeted PBx with TRUS.
Conventional gray-scale TRUS is limited to detecting PCa or distinguishing ablated prostate from normal prostate with high accuracy. Agents, however, containing microbubbles that improve the visualization of the prostate microvasculature increases the chances for detection of a malignant lesion as prostatic adenocarcinoma is characterized by angiogenesis with increases of microvasculature density (Figure 1(c)). 53 CEUS brings several potential advantages in the management of PCa including diagnosis, facilitating targeted PBx, real-time evaluation and confirmation of adequate tissue ablation after focal therapy (FT), and identification of post-treatment recurrence during post-ablation surveillance by achieving better vascular imaging resolution. 52
Doppler ultrasound can accurately assess macrovessels but not vessels less than 200 μm as those typically seen with angiogenesis. Ultrasound contrast containing microbubbles measuring 2–6 μm are pure intravascular as they do not cross into the interstitial space, they are small enough to pass through the pulmonary circulation and enter the systemic circulation, but large enough not to escape the endothelium, and excellent to see microvasculature typical of prostate adenocarcinoma.54,55 The following ultrasound-enhancing agents (UEAs) are approved for use: Lumason® (sulfur hexafluoride lipid-type A microspheres), SonoVue®, Definity®, or Luminity®. 56 UEA do not require laboratory assessments prior to the procedure, and do not have any nephrotoxicity, hepatotoxicity, or cardiotoxicity known. A multi-institutional study of 5576 patients undergoing contrast-enhanced echocardiography reported an adverse event rate of 0.27%, with all adverse events being mild and transient. 57
CEUS can assess perfusion within the small microvessels (40 μm) seen in PCa with a positive predictive value up to 91.7%, sensitivity up to 79.3%, and accuracy of 83.7%. The addition of CEUS-guided targeted PBx may be associated with significantly improved cancer detection rate compared with 12-core systematic biopsy.14,58,59,60 Also, it provides immediate intraoperative visualization of ablated area with clear and sharp margins, therefore confirming that the targeted area of suspicious was indeed treated as planned. Of note, CEUS represents a compelling strategy for the evaluation and diagnosis of other urologic malignancies such as kidney and bladder cancer.61,62
Proprietary software can process raw data acquired by CEUS. 63 Time-intensity curve (TIC) is depicted by the software plotting echo mean in dB (X-axis) against time (Y-axis). Quantitative parameters such as peak intensity (PI), wash-in slope (WIS), and time-to-peak (TTP) can be acquired from TIC. By comparing TIC extracted from PCa suspicious ROI and contralateral normal ROI, PCa exhibits higher PI, steeper WIS, and earlier TTP.64,65 Ablated tissue by high-intensity focused ultrasound (HIFU) can be confirmed as a flat curve with minimal slope on TIC (Figure 2). 52

Representative images of pre-/post-HIFU imaging with CEUS. A 62-year-old man with PSA 8.2 ng/ml. Prebiopsy multiparametric MRI revealed PCa suspicious lesion on left side peripheral zone from the base to apex gland level (PIRADS score 5). Systematic 12-core biopsy plus 6-targeted core biopsy revealed GG2 cancer from the left side of the prostate. The patient wished to undergo left hemi-HIFU as a definitive treatment for PCa. Prior to HIFU, CEUS was performed to evaluate vascularity of the bilateral prostate lobes (arrowhead shows contrast enhancement on cancerous area). Time-intensity curves analysis showed higher peak intensity, steeper wash-in slope, and earlier time-to-peak on the left side (yellow circle and yellow graph) of the prostate than those on right side (green circle and green graph). Immediately after HIFU, a second CEUS evaluation was performed to confirm the ablated area (arrow). While the treated side lobe successfully lost vascularity, the contralateral side maintained vascularity. At this point, the procedure was terminated. Postoperatively, PSA dropped to 0.26 ng/ml, follow-up imaging (mpMRI, B-mode TRUS, CDUS, and CEUS) did not detect PCa recurrent suspicious lesion. One year after HIFU, follow-up systematic biopsy confirmed no cancer on both treated and untreated side of the prostate.
Elastography
Ultrasound elastography, an imaging technique that detects and quantifies tissue stiffness, is helpful in diagnosing PCa as malignant tissue has greater stiffness than benign tissue. The two most common types of elastography used on the prostate are strain elastography (SE) and shear-wave elastography (SWE). 66 SE measures the strain seen in the tissue under mechanical stress induced by the transrectal probe. To avoid the risk of applying excessive compression and falsely increasing tissue stiffness, an inflated balloon (with water, for example) may be used to separate the probe and the rectum. 67 SWE measures the propagation of mechanical waves through a tissue, which can be altered by its stiffness. 68
SE in the prostate has shown improved accuracy over TRUS in detecting PCa. A meta-analysis with 508 patients comparing SE with histopathology following radical prostatectomy (RP) showed a sensitivity of 72% and specificity of 76% for PCa detection. 69 A systematic review including 1840 patients showed an increase of 7–15% in overall PCa detection when targeted biopsy by TRUS elastography was combined with systematic biopsy compared with systematic biopsy alone. 70 Schiffmann et al., 71 however, did not report similar results. Results of the study involving 679 males and 4074 prostate biopsies from 6 different regions of the prostate demonstrated that SE-targeted biopsy had an overall high value of specificity (90%) and negative predictive value (NPV, 87%) and a low value of sensitivity (19%) and positive predictive value (PPV, 25%). Low sensitivity and PPV suggest that SE may not identify sextants that indeed present PCa.
A systematic review including 2227 patients from 16 SWE studies shows promising results. 72 In nine studies analyzed, systematic biopsy was the reference standard at the per-sample level (core, sextant level). Pooled sensitivity and specificity were 85% and 85%, respectively. When histopathology of RP was used as the reference standard, pooled sensitivity and specificity were 71% and 74%, respectively. Fu et al. 73 also demonstrated positive results in their prospective study of 221 patients that compared SWE with MRI of the prostate. Sensitivity, specificity, PPV, NPV, and accuracy were 78.97%, 90.67%, 71.30%, 93.66%, and 88.03%, respectively. Between SWE and MRI, there was no statistically significant difference in PCa diagnosis (p = 0.259). SWE’s diagnostic ability, however, was marginally superior to MRI for CSPCa (p = 0.013). Different results were reported by Xiang et al. 74 SWE alone was shown to have a lower diagnostic value than MRI alone, but the combination of both imaging modalities demonstrated higher sensitivity than any of the methods alone.
Despite having favorable outcomes, elastography is not without its limitations. First, the manual compression of the prostate by the SE operator may change the tissue’s elasticity, limiting the accuracy of the diagnosis.75,76 This reduces results’ reproducibility, as it is dependent on the examiner’s skill and experience. Also, there is no method to compress the prostate uniformly. Second, stiff lesions do not necessarily indicate cancer, and cancerous lesions are not always stiff. 77 Confounding factors such as increased prostate volume, calcifications, and fibrous tissue may produce false-positive results. Finally, considerable limitations include the small box, slow frame rates, and penetration issues of SWE. 66
Elastography of the prostate can be considered as an additional method to detect PCa and guide biopsy. TRUS-targeted biopsy using elastography seems promising, but prospective multicenter studies should be performed to confirm its usefulness. Technique standardization and validation to allow further studies comparison are needed.
Micro-US
Conventional B-mode TRUS performed at 8–12 MHz allows for adequate penetration depth for the prostate; however, the spatial resolution may not be enough to differentiate physiological glandular ducts, acini of the prostate, and malignancies.78–80 To improve the limited resolution of conventional TRUS, micro-US utilizing 29 MHz transducer has been developed.78,81 Compared with the conventional TRUS, this higher frequency system can visualize the prostate with three times higher spatial resolution (70 µm). 17 A trade-off, however, exists between the spatial resolution and the penetration depth, because attenuation is proportional to frequency. 82 Thus, the penetration depth of mpUS system is reduced to 50 mm, which still covers the entire prostate in the standard size.81,83 Current micro-US system only provides sagittal plane that is optimum view for transperineal PBx. The lack of axial images, however, makes MRI-guided software fusion PBx unachievable, although MRI-guided cognitive fusion PBx still can be performed. 84
Using ultrasonographic appearance of PCa areas on micro-US, a 5-point scoring system for suspicious lesions was created, allowing for more consistent interpretation of prostate images. 81 This grading system is called PRI-MUS (Prostate Risk Identification using Micro-Ultrasound), which is analogous to the PIRADS grading for mpMRI. PRI-MUS 1 and 2 are likely benign. PRI-MUS 3 is associated with intermediate risk of cancer and could be supported by targeted or systematic biopsy. PRI-MUS 4 and 5 lesions are highly correlated to significant disease, and targeted biopsy is indicated.81,85 Using this grading system, a multi-institutional randomized controlled trial comparing first-generation micro-US with conventional ultrasound-guided PBx was conducted by Pavlovich et al. 86 Systematic 12-core transrectal PBx results were used as the reference standard. The trial was split into pre- and post-image interpretation training. The PRI-MUS was developed by the data from the pretraining group, and then used by the post-training group. Improved per-core sensitivity of micro-US in the post-training group compared with the pretraining group was confirmed (60.8% versus 24.6%, p < 0.01), and the sensitivity was significantly higher than conventional TRUS (versus 38.0%, p < 0.001). Moreover, per-patient detection of CSPCa in the micro-US arm improved by 7% after training (32–39%, p < 0.03). The per-patient CSPCa detection of micro-US, however, was not better than conventional TRUS (34.6% versus 36.6 for the entire cohort, and 39.0% versus 39.0% for the post-training cohort).
As the first pilot study on the accuracy of micro-US was reported, 78 several studies have compared micro-US with conventional B-mode TRUS and mpMRI.16,85–88 Zhang et al. 89 conducted a meta-analysis of seven studies including 769 patients to analyze the accuracy of micro-US for PBx. They revealed that micro-US had a pooled sensitivity, specificity, diagnostic odds ratio, and area under the summary receiver operating characteristic (ROC) curve of 0.91, 0.49, 10, and 0.82, respectively. Sountoulides et al. 90 compared the PCa detection rate of micro-US versus mpMRI targeted PBx (TBx) in his meta-analysis of 13 studies containing 1,125 patients. The detection ratio (DR) was estimated as the micro-US TBx detection rate divided by the mpMRI-TBx detection rate. The pooled DR for grade group (GG) 1, ⩾1, ⩾2, and ⩾3 PCa were 0.94, 0.99, 1.05, and 1.25, respectively. Therefore, Sountoulides et al. 90 concluded micro-US and mpMRI-TBx showed similar detection rates across all PCa grades. Furthermore, another meta-analysis of 15 studies including 2967 patients by Dariane et al. 91 evaluated the added value of micro-US-guided PBx compared with systematic biopsies (SBx). They found micro-US TBx identified more CSPCa [DR = 1.18, 95% confidence interval (CI) = 0.83–1.68] and significantly less GG1 PCa (DR = 0.55, 95% CI = 0.41–0.73) than SBx. The first multicenter prospective study, including 1040 patients, compared diagnostic performance of micro-US with those of mpMRI. Micro-US TBx and mpMRI TBx were taken from PRI-MUS >3 and PIRADS >3 lesion. In the comparison with mpMRI TBx, micro-US TBx showed significantly higher sensitivity (94% versus 90%, p = 0.03) and NPV (85% versus 77%, p = 0.04) with similar specificity (22% versus 22%, p = 0.45) and PPV (44% versus 43%, p = 0.32) for the detection of CSPCa. 92 The results of the OPTIMUM (Optimization of prostate biopsy–Micro-Ultrasound versus MRI) study, an ongoing three-arm randomized controlled trial, will determine whether micro-US can be used as an alternative to MRI/TRUS fusion biopsy. 93
Multiparametric US
As shown above, new US modalities have demonstrated promising results. It, however, is unclear whether one new US modality alone can achieve satisfactory diagnostic performance for PCa. These modalities visualize different aspects of the prostate. The concept of mpUS is similar to mpMRI. Therefore, mpUS is useful for lesion volumetry, guidance for PBx and FT as well. As dynamic contrast imaging on MRI can do, Doppler and CEUS can detect vascularity in PCa tissues. Elastography depicts the stiffness of the prostatic tissue, which may correspond to cancerous cell density. Considering the success of mpMRI for PCa diagnosis, the combination of multiple US modalities also has potential to achieve more reliable performance. Considering several US modalities showing feasibility of guidance for PBx and FT, mpUS may be useful for guidance for PBx and FT as well.13,39–41,52
A few groups studied mpUS defined as the combination of 3 or more US modalities.18–23 In the past 5 years, however, mpUS has gain popularity in urology and radiology field.30,94–101
Zhang et al. 94 evaluated malignant features of US modalities using 12-core systematic PBx in 40 patients with benign histology and 38 men with a localized PCa as the reference standard. They demonstrate that when US modalities are combined (‘⩾3 malignant features’ on TRUS or ‘asymmetric distribution’ on SWE or ‘nonsynchronous wash-in/out, unequal enhancement, and heterogeneous distribution’ on CEUS), the mpUS achieves high PCa detection performance [sensitivity: 97.4%, specificity: 77.5%, PPV: 80.4%, NPV 96.9%, accuracy: 87.2%, area under the receiver operating characteristic curve (AUROC): 0.874] which was compatible with mpMRI (sensitivity: 94.7%, specificity: 60.0%, PPV: 69.2%, NPV 92.3%, accuracy: 76.9%, AUROC: 0.774). Targeted biopsy, however, was not performed, and therefore, the performance of per lesion was not evaluated. SWE or CEUS is usually evaluated in conjunction with B-mode findings. As the performance of SWE or CEUS (AUROC: 0.860 and 0.859) were similar to mpUS (AUROC: 0.874), the added value of third US modality seems to be limited.
Mannaerts et al. 95 prospectively evaluated the CSPCa diagnostic performance of combination of three US modalities (B-mode, SWE, and CEUS) in 48 men undergoing RP as the reference standard. Uniquely, they used an automated RP histopathological correlation method to precisely evaluate CSPCa lesion localization. US modalities were evaluated by three readers using a 5-point Likert-type scale to score 12 anatomical ROI. When Likert-type scale ⩾3 was used as threshold, ROI-specific sensitivity, specificity, PPV, and NPV for CSPCa diagnosis using mpUS were 74%, 59%, 65%, and 70%, respectively. The sensitivity was significantly higher than all single US modalities alone (B-mode: 55%, SWE: 55%, and CEUS: 59%). On the other hand, the specificity was not significantly different from all single US modalities alone (B-mode: 61%, SWE: 61%, and CEUS: 63%). In their subgroup analysis for mpUS performance, ROI-specific sensitivity for the lesion on peripheral zone (PZ) was higher than that on transitional zone (TZ) (80% versus 67%). While the sensitivity of SWE alone for PZ tumor was less than B-mode or CEUS alone, B-mode or CEUS alone detects less TZ tumors than SWE. These findings may justify the concept of mpUS as combination of these US modalities. Index lesion (defined as the highest Gleason grade lesion on RP histology) detection rate of ROI1 and ROI2 were significantly higher with mpUS than all single US modalities (mpUS: 88% versus CEUS: 73%, B-mode: 72%, or SWE: 70%). Interobserver agreements that were evaluated by the Krippendorff α were not high [mpUS (cut-off value Likert-type ⩾3): 0.33 and mpUS (cut-off value Likert-type ⩾4): 0.48]. Unfortunately, comparison with mpMRI findings was not performed in this study, and the sample size was small. Of note, all participants had PCa that was eventually treated by RP; therefore, population bias likely existed. Compared with single US modalities, mpUS detected more CSPCa on both PZ and TZ. This is a clear advantage of mpUS.
Postema et al. 96 evaluated the CSPCa (any GG ⩾3 and GG 2 larger than 0.5 ml) diagnostic performance of B-mode, CEUS, contrast ultrasound dispersion imaging (CUDI), and mpUS (combination of these three US modalities) with 133 men undergoing RP as the reference standard. CUDI, a computer-aided quantification technique, was generated from the CEUS recordings. Likelihood of presenting CSPCa for each imaging modality (B-mode, CEUS, and CUDI) was scored on a 1–5 Likert-type scale by five observers. In their multicenter study, sensitivity/specificity/AUROC for CSPCa diagnosis was 81%/64%/0.78 for CEUS, 83%/55%/0.79 for CUDI, and 83%/55%/0.78 for mpUS. Using a weighted Fleiss Kappa statistic, poor interobserver agreement of US modalities was shown in the study (CEUS: 0.20, CUDI: 0.18, combination: 0.18).
More recently, Grey et al. 102 conducted a multicenter prospective paired-cohort study to compare diagnostic performance for CSPCa (any area with GG ⩾3 or maximum cancer core length ⩾6 mm) between mpUS (B-mode + CDUS + elastography + CEUS) versus mpMRI. mpMRI evaluation was based on Likert-type system instead of PIRADS, and each US imaging was evaluated with standardized Likert-type scoring method. The overall lesion score was determined at the discretion of the reporter. Using three-core mpUS or mpMRI-targeted PBx as the reference standard, they found CSPCa detection by mpUS alone, mpMRI alone, and the combination of mpUS and mpMRI were 26%, 30%, and 32%, respectively. As a result, 7% of CSPCa were exclusively detected by mpUS alone, whereas 20% of CSPCa were exclusively detected by mpMRI alone. The authors concluded mpUS could be an alternative to mpMRI as a diagnostic test for patients with high risk of PCa particularly in the case of that mpMRI cannot be performed.
This inconsistent performance and limited interobserver agreement of mpUS may be due to the lack of well-structured or standardized evaluation system. Wildeboer et al. 97 assessed the performance of US radiomic features that extracted from B-mode, SWE, and CEUS for the localization of PCa using machine learning methods (a random forest classification algorithm) in their analysis of 48 men with biopsy-confirmed PCa. The RP specimens of these 48 patients were used as the reference standard. While the best-performing single radiomic feature such as contrast velocity achieved a region-wise AUROC of 0.69 for any PCa and 0.76 for CSPCa (defined as PCa > GG2), respectively, multiparametric combination of radiomic features achieved outperforming AUROC of 0.75 for any PCa and 0.90 for CSPCa, respectively. The radiomic features of perfusion-, dispersion-, and elasticity-related features were most frequently selected as effective parameters for PCa classification by machine learning model. Importantly, they also showed that effective radiomic parameters derived from B-mode, SWE, and CEUS were not correlated with each other; therefore, these three modalities may be cumulative. As the selected radiomic parameters substantially differed between the PZ and TZ, they emphasized the necessity for accurate zonal segmentation for US evaluation.
Morris et al. 98 evaluated the feasibility of using B-mode, acoustic radiation force impulse imaging (ARFI), SWE, and quantitative ultrasound (QUS) midband fit (MF) to provide image guidance for targeted PBx. ARFI is a US modality to assess elasticity using acoustic radiation force. While SWE provides quantitative stiffness of tissue, ARFI reflects a relative stiffness of tissue. Computing normalized spectra, spectral-based QUS methods quantify the scattering properties of tissues. MF is a most common parameter in QUS analysis which is generated using a linear fit to the normalized backscattered spectra. They acquired B-mode, ARFI, SWE, and MF from 35 men with biopsy-confirmed PCa, and combined them with a linear support vector machine (SVM) method. The SVM was trained and validated by a subset of data from 20 patients and tested by a remaining 15 patients’ data. All participants underwent prostatectomy after imaging, whole mount prostates were histologically analyzed and PCa lesions were assigned on a 27-region model as the reference standard. They evaluated contrast and contrast-to-noise ratio (CNR) as lesion visibility metrics and generalized contrast-to-noise ratio (gCNR) as a metric to assess the overlap in the distributions of the two ROIs. mpUS statistically significantly outperformed B-mode and SWE with respect to contrast, CNR, and gCNR, MF with respect to contrast and CNR, and ARFI with respect to CNR. They pointed out that both calcifications and the distance from US probe limited the performance of mpUS.
Future directions
Currently, accurate diagnosis and appropriate stratification of PCa is essential for patient-specific PCa management. 103 Recent advances in PCa imaging technology including mpMRI enabled more precise PCa localization. Whether these new imaging modalities improve clinical outcomes such as overall survival has not been determined yet. 101 Precise imaging, if well validated, may reduce unnecessary PBx and the risk for overdiagnosis and overtreatment. 104 FT based on accurate real-time imaging is expected to achieve better oncological/functional outcomes. 52 Therefore, establishing more precise imaging is important.
Overview of the most up-to-date literature focusing on US for PCa diagnosis is shown on Table 2. Although sample size of the recent studies on mpUS was still small and most of studies were single center, aggregated effectiveness of multiple US modalities likely exists when particular modalities were combined (i.e. B-mode + SWE + CEUS).94,95,97,98,102 Variation of the combined US modalities are limited so far. Inclusion of state-of-the-art US modality such as SMI or micro-US may further improve the performance of mpUS.
Overview of the most up-to-date literature on the finding.

ANNA/C, computerized artificial neural network analysis; ARFI, acoustic radiation force impulse imaging; AUC, area under the curve; B-mode, brightness-mode; CDUS, color Doppler ultrasonography; CEUS, contrast-enhanced ultrasonography; CNR, contrast-to-noise ratio; CSPCa, clinically significant prostate cancer, CUDI, contrast ultrasound dispersion imaging; gCNR, generalized contrast-to-noise ratio; GG, grade group; GS, Gleason score; MCCL, maximum cancer core length; MF, midband fit; mpMRI, multiparametric magnetic resonance imaging; mpUS, multiparametric ultrasonography; MRI, magnetic resonance imaging; NPV, negative predictive value; OR, odds ratio; PBx, prostate biopsy; PCa, prostate cancer; PDUS, power Doppler ultrasonography; PHS, prostate HistoScanning; PIRADS, Prostate Imaging Reporting and Data System; PPV, positive predictive value; PRI-MUS, prostate risk identification using micro-ultrasound; PSA, prostate-specific antigen; PSAD, prostate-specific antigen density; PZ, peripheral zone; ROI, region of interest; RP, radical prostatectomy; SBx, systematic biopsy; SMI, superb microvascular imaging; STARD, standards for reporting diagnostic accuracy studies; SVM, support vector machine; SWE, shear-wave elastography; SWEI, shear wave elasticity imaging; TBx, targeted biopsy; TCCL, total cancer core length; TRUS, transrectal ultrasound; TT, true targeting; TTPM, template transperineal mapping; TZ, transitional zone; US, ultrasonography.
Whether mpUS even outperform mpMRI is unclear, because a few studies directly compared these methods so far.94,99,102 Multicenter and randomized controlled trials to compare them with large sample size are still needed. Some aspects including lower cost, real-time imaging, applicability for some patients (i.e. claustrophobia or hip prosthesis) who cannot undergo mpMRI, and availability in the office setting are clear advantage of mpUS, compared with mpMRI, however.
How to integrate multifactorial findings on each US modality can strongly affect the performance of mpUS; therefore, it is important to consider. Some investigators just added targeted biopsy to PCa suspicious lesion on any US modalities.18,19 This strategy increases sensitivity and NPV, while it decreases specificity and PPV. 22 Other investigators scanned the prostate with strained elastography first to detect ROI, and they further screened whether the ROI is cancer suspicious on CEUS. 21 The strategy results in the increased specificity/PPV and the decreased sensitivity/NPV. 22 To optimize the performance of mpUS, some researchers used Likert-type scoring methods,95,96,99 and others evaluated the existence of some imaging features. 94 Even when Likert-type scoring was performed, however, relatively low inter-reader agreement of mpUS was still a drawback.95,96,118 It may be partly due to the nonstandardized mpUS reading process and the lack of well-validated and objective evaluation system. Currently, machine learning approach enables automated image recognition and providing quantitative assessments of massive number of complex radiographic characteristics. 119 Deep learning, a subset of machine learning, has already shown the equivalent diagnostic performance to that of health-care professionals. 120 To achieve more accurate and reproducible ultrasonographic assessments, as Wildeboer et al. 97 and Morris et al. 98 showed the feasibility in their studies, applying machine learning method on multifactorial radiomic features of mpUS is a promising approach.119,121 Of note, as the complexity of machine learning algorithm increases, the developed model typically increases the performance but becomes less understandable algorithm. To overcome the limitation of machine learning method, the learning process should be interpretable by human, the number of radiomic features to be evaluated should be minimum, and the biological interpretation/validation of the radiomic features are needed. 122 While 25% of CSPCa can be missed by mpMRI, 123 mpUS does detect CSPCa that was missed by mpMRI. 94 Integrated evaluation of both mpUS and mpMRI findings can be a fascinating future concept (Figure 3).
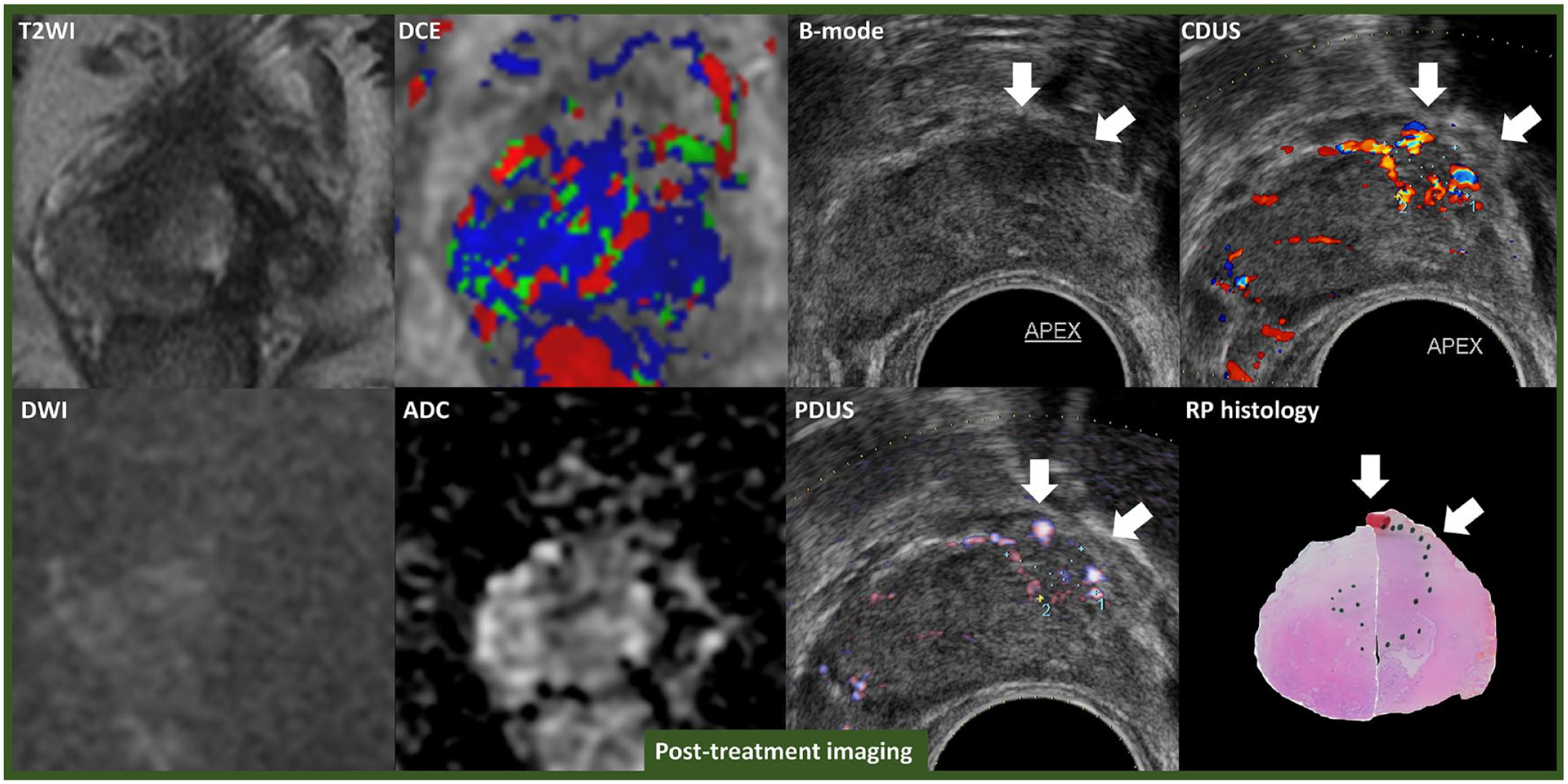
Representative images of negative on mpMRI but positive on mpUS. A 46-year-old man with PSA 7.0 ng/ml on active surveillance for GG1 cancer. Although surveillance mpMRI did not revealed PCa suspicious lesion, B-mode TRUS, CDUS, and PDUS detected 1.3 cm × 0.7 cm of PCa suspicious lesion on the left base of the prostate. Early enhancement was not observed on CEUS. Systematic biopsy + targeted biopsy to the PCa suspicious lesion detected GG2 PCa on the left base to mid area of the prostate. The patient selected left hemi-HIFU as a definitive treatment for PCa. Postoperatively, PSA dropped to 3.6 ng/ml, follow-up mpMRI did not show lesions suspicious for CSPCa (left figures). B-mode TRUS, CDUS, and PDUS detected 1 cm × 0.7 cm suspicious HEL on the left apex anterior area of the prostate (arrow on right figures), however. Targeted biopsy to the HEL revealed GG2 PCa recurrence. He underwent salvage robotic RP. GG2 PCa lesion with extraprostatic extension on the left anterior area was confirmed on RP histology specimen (green dots shows PCa mapping).
Conclusion
In the past 5 years, mpUS has exponentially gained popularity in urology and radiology field. Several latest evidence showed the potential of mpUS to provide more reliable performance for CSPCa detection and guidance for PBx and FT. Furthermore, some aspects including lower cost, real-time imaging, applicability for some patients who have contraindication for mpMRI, and availability in the office setting are clear strengths of mpUS. Machine learning approach and integration of radiomic features may improve the diagnostic performance of mpUS. Multicenter randomized controlled trials are warranted to directly compare the diagnostic performance of mpUS and mpMRI.
