Abstract
Background:
Lower urinary tract symptoms (LUTS) secondary to benign prostatic hyperplasia (BPH) are a bothersome frequent symptom in adult males. This systematic review analyzed the available evidence on the pharmacokinetic and pharmacodynamic features of alfuzosin, and its clinical efficacy both as monotherapy and in combination with other drugs for the treatment of male LUTS/BPH.
Methods:
A systematic review of the last 10 years was performed using the MEDLINE, EMBASE and Cochrane libraries in March 2020. The protocol for this systematic review was registered on PROSPERO (Central Registration Depository: CRD42020136120) and is available in full on the University of York website.
Results:
Alfuzosin is a quinazoline derivative and, although a nonspecific α1-blocker, exhibits a selective concentration in the prostate compared with plasma in patients with BPH. Three registration trials assessed the safety and efficacy of alfuzosin. The 10 mg daily formulation has a three-layered matrix containing the active substance between two inactive coats allowing a drug release over 20 h. Alfuzosin showed high tolerability, few vasodilatory effects and a low rate of ejaculation disorders over older alpha-blocking compounds thanks to the high uroselectivity of alfuzosin and its preferential concentration at urinary level. Six randomized clinical trials (RCTs) assessed efficacy and safety of alfuzosin
Conclusions:
Alfuzosin is an effective drug for the treatment of LUTS/BPH, with a lower rate of sexual disorders compared with other alpha-blockers. Alfuzosin is also safe with low adverse events in case of concomitant antihypertensive therapy and in patients with cardiovascular morbidity. Safety and efficacy of alfuzosin has been reported also in case of combination therapy with antimuscarinic agents and PDE5i.
Keywords
LUTS/BPH: definition, epidemiology and standard of care
Benign prostatic hyperplasia (BPH) is characterized by an increased proliferation of both epithelial and stromal tissue, especially in the periurethral zone of the prostate, and is one of the most common conditions in elderly men. 1 The prevalence of BPH increases substantially with advanced age, with a reported prevalence ranging from 8% to 60% in the adult population.2,3 BPH can cause bothersome lower urinary tract symptoms (LUTS), including storage, voiding and post-micturition symptoms variously combined together.4,5 LUTS secondary to BPH (LUTS/BPH) seriously affects quality of life 3 and moreover impacts greatly on public health due to the possible consequences of untreated BPH, such as acute urinary retention (AUR), urinary tract infections and worsening of the renal function.6,7
According to current international guidelines,3,4 pharmacological therapy represents the first therapeutic approach in patients with moderate or severe LUTS/BPH. In this context, α1-adrenoceptor antagonists (also known as α1-blockers) play a key role for the treatment of LUTS/BPH. To date, several α1-blockers with different pharmacokinetic and pharmacodynamic profiles have been developed for clinical use.
Aim of this systematic review is to analyze the available evidence on the pharmacokinetic and pharmacodynamic features of alfuzosin, and its clinical efficacy both as monotherapy and in combination with other drugs for the treatment of male LUTS/BPH.
Evidence acquisition
Literature search
A systematic review of the English literature was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and the Cochrane Handbook for Systematic Reviews of Interventions.8,9 A PRISMA checklist is provided as Supplementary Table 1. We searched MEDLINE, Scopus and Web of Science to investigate the efficacy and the treatment profile of alfuzosin in men treated with alfuzosin in monotherapy and in combination therapy for BPH between 1 January 2000 and 28 February 2020. After a first screening based on study title and abstract, all papers were assessed based on full text and excluded with reasons when appropriate. Two reviewers (A.M. and A.M.) carried out this process independently. Disagreement was solved by a third party (C.D.N.). The following string terms were used:
Eligibility criteria
As proposed by the PRISMA guidelines, we used the Population, Intervention, Comparator, Outcome and Study design approach to specify the inclusion criteria. Reports were considered relevant when they included men affected by benign prostatic hyperplasia and lower urinary tract symptoms (P), undergone medical treatment with alfuzosin (I) and compared with patients with comparable symptoms for BPH and treated with other alpha-blockers, finasteride, dutasteride or treated with endoscopic or open or minimally invasive surgery for the removal of the prostatic adenoma (C). These patients were compared to determine the incidence of AUR and BPH-related surgery during the medical treatment, the change in the International Prostate Symptom Score (IPSS), and the secondary outcome measures were changes in individual subjective symptom scores, quality of life score, and peak flow rate (Qmax) from baseline at uroflowmetry. Only randomized clinical trials and cohort prospective studies according to the latest version of the evidence medicine pyramid were included for qualitative analysis. 10 Conversely, retrospective studies, low-quality prospective studies, review articles, editorials, comments and meeting abstracts were excluded. References of included manuscripts were scanned for additional studies of interest.
Data extraction
The titles and abstracts of all references retrieved in the searches were screened for potential eligibility, and the full texts of articles identified as possibly being relevant were obtained and evaluated. After full text evaluation, data were independently extracted by two authors (A.M. and A.M.) for further assessment of qualitative analyses. All extracted variables were crosschecked to ensure their reliability. Furthermore, we searched for methods and important confounders to establish comparability. All discrepancies regarding data extraction were generally resolved by consensus or finally decided by a third author (C.D.N.). A flow chart for the article selection process according to PRISMA-P 2015 checklist is detailed in Figure 1. The protocol has also been registered in the International Prospective Register of Systematic Reviews database (PROSPERO: CRD42020136120).

Flow chart for article selection process to analyze functional urinary outcomes in men affected by benign prostatic hyperplasia and lower urinary tract symptoms undergone medical treatment with alfuzosin.
Alfuzosin: pharmacology and pharmacodynamics
The pharmacology and pharmacodynamic properties of alfuzosin are well established and were extensively analyzed in two reviews in
Alfuzosin is a quinazoline derivative characterized by the presence of a diaminopropyl spacer and the absence of a piperidine moiety (Figure 2). These biochemical properties determine a selective and competitive antagonist effect on the contraction of prostatic smooth muscle mediated by the α1-adrenoceptors.

Chemical structure of alfuzosin. Molecular Formula: C19H27N5O4•HCl. Chemical Name: N-[3-[(4-Amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-2-furancarboxamide monohydrochloride.
The receptor selectivity and pharmacokinetics parameter of alfuzosin compared with tamsulosin and silodosin are summarized in Table 1. The different safety and efficacy profile of various α1-blockers depend on their distinct selectivity to the various subtypes of α1-adrenoceptors (α1-ARs) expressed in numerous human tissues. In particular, three different α1-ARs subtypes (α1-A, α1-B and α1-D) with distinctive features in terms of distributions and mediated pharmacological and physiological effects have been identified. 13 In detail, α1-A receptors are the predominant subtype expressed in the human prostate tissue with an increased (nearly 9-fold increase) expression in patients with BPH. 14
Receptor selectivity and pharmacokinetics parameter of alfuzosin compared with tamsulosin and silodosin.

Ishiguro M, Futabayashi Y, Ohnuki T,
Schwinn DA and Roehrborn CG. Alpha1-adrenoceptor subtypes and lower urinary tract symptoms.
Akiyama K, Hora M, Tatemichi S,
AUC∞, area under the plasma concentration-time curve from time zero to infinity; Cmax, maximum plasma concentration; t1/2, elimination half-life; tmax, time to reach the Cmax; uroselective receptors, α1A and α1D; vascular epithelium, α1b.
Similar to the other α1-blockers, alfuzosin also counteracts the effect of noradrenaline on smooth muscle cells in the prostate and consequently the symptoms due to bladder outlet obstruction. 13 It has been largely demonstrated both in human and animal studies that the activity and side effects of α1-blockers rely on their uroselectivity rather than the α1-AR subtype affinity as previously reported.15,16
In this scenario, alfuzosin, although a nonspecific α1-blocker, 17 exhibits a selective concentration in the prostate, compared with plasma, in patients with BPH. In detail, the selective concentration of alfuzosin, tamsulosin, doxazosin and terazosin were compared and, in particular, their affinity for prostatic and renal α1-adrenoceptors in intact enzymatically isolated myocytes of the human prostate and renal artery was assessed. Alfuzosin showed a significantly higher concentration in the prostatic tissue compared with other α-blockers. 18 Also, in clinical practice, the blood and prostatic concentrations of alfuzosin were determined in patients with BPH treated with alfuzosin 5 mg twice daily prior to surgery in an open trial, and high diffusion of the drug into the prostate in BPH patients was found. 19
Several formulations of alfuzosin for the treatment of LUTS/BPH have been developed. Initially, a 2.5 mg immediate-release formulation (administered three times daily) was developed; afterwards, a 5 mg (administered twice daily) sustained-release formulation was produced. 20 Lastly, a 10 mg alfuzosin with a controlled release over the dosage interval has been developed. As the drug is highly soluble in water, the major challenge in making a controlled-release formulation was to control its release from the dosage forms. A three-layered matrix containing the active substance between two inactive coats, one swellable and one erodible, was developed allowing a drug release over 20 h with a near constant dissolution rate between 2 and 12 h. 20 The absolute bioavailability of 10 mg alfuzosin HCl extended-release tablets under fed conditions is 49%. The time to maximum concentration is 8 h. Cmax and AUC0–24 are 13.6 (SD = 5.6) ng/mL and 194 (SD = 75) ng·h/mL, respectively. 20 The absorption extent is 50% lower under fasting conditions. For this reason, it should be taken with food and with the same meal each day. 20
The distribution volume in healthy male middle-aged volunteers was 3.2 L/kg after intravenous administration.
21
Furthermore,
Alfuzosin is mostly metabolized by the liver, while only 11% of the molecules of the administered dose are excreted without modifications in the urine. 22 Oxidation, O-demethylation and N-dealkylation are the three metabolic processes which alfuzosin undergoes. 22 The most important hepatic enzyme isoform involved in its metabolism is the Cytochrome P450 3A4 (CYP3A4).
Although it is not indicated to check the activity of the CYP3A4, repeated administration of 400 mg of ketoconazole, a potent inhibitor of CYP3A4, following a single 10 mg dose of alfuzosin, increased 2.3-fold alfuzosin Cmax. 20 Alfuzosin use is also contraindicated in the case of patients taking telaprevir and boceprevir that are used for the treatment of hepatitis C virus infection and induce a potent, reversible inhibition of the CYP3A4/5 enzyme families. 23
The properties of once-daily formulation of alfuzosin were studied in an elderly population without finding any pharmacokinetic difference in 16 healthy volunteers between 66 and 78 years. 24 Despite 35% greater concentration of alfuzosin in patients aged ⩾75 years, 22 clinical trials showed no overall differences in safety or effectiveness between subjects ⩾65 years, ⩾75 years and younger men. 22
As regards the pharmacodynamic profile of alfuzosin in special populations, the presence of chronic heart failure has not been found to significantly affect the pharmacokinetics of 10 mg daily alfuzosin. Concerning patients with reduced renal function, an evaluation of the effects of the treatment was performed on 26 volunteers, of which there were eight with normal renal function, six with mild, six with moderate, and six with severe chronic kidney disease.
25
The study showed that the mean apparent total body clearance was decreased 30–40% in all groups with renal impairment.
25
However, the product monograph highlighted that in clinical trials the safety profile of patients with mild (
Alfuzosin: clinical efficacy as monotherapy
Registration trials
Results from registration trials are summarized in Table 2. The first randomized placebo-controlled trial assessing the safety and efficacy of alfuzosin (2.5 mg TID) as monotherapy for the treatment of LUTS/BPH was published in 1991 by the BPH-ALF group.
26
In this trial, alfuzosin was superior to placebo in terms of increased urinary flow parameters [including post-voided residual volume (PVR)] and was also associated with a lower AUR rate (0.4%
Description of characteristics and reported outcomes of randomized registration clinical trials of alfuzosin.

LE: level of evidence; PVR: post-voided residual; RCT: randomized clinical trial.
Lately, in 1997, the ALGEBI group reported the results of a randomized controlled trial (RCT) comparing the safety and efficacy of alfuzosin 5 mg BID
In 2000, van Kerrebroek
Efficacy
Efficacy of alfuzosin as monotherapy for the treatment of BPH/LUTS has been reported in several prospective series. Lukacs
Afterwards, the ALF-ONE study group reported the 2-year results of a large open-label study with alfuzosin 10 mg once daily enrolling 839 European patients followed by GPs.
31
At the endpoint, the IPSS improved by 7 points (–38.5%) from baseline (
In a pooled analysis of the data from the three pivotal phase III registration trials studies conducted to test the efficacy of alfuzosin 10 mg once-daily (
Overall, alfuzosin 10 mg was well tolerated, and the most common AE related to vasodilatation was dizziness/postural dizziness (3.1%). As previously reported, ejaculatory disorders were uncommon (0.3%) and, importantly, vasodilation-related symptoms were rare also in elderly men and those under antihypertensive therapy. These results were confirmed also at the 3-year follow-up of the same study, emphasizing the lasting effectiveness and safety of alfuzosin 10 mg for the treatment of LUTS/BPH. 33
Indeed, the same study group investigated the effect on sexual function of alfuzosin 10 mg in 2434 sexually active patients. After 1 year of therapy in those men with baseline sexual dysfunction, alfuzosin was proved to improve the rigidity of erection, the amount of ejaculate and pain/discomfort on ejaculation (all
In another randomized clinical trial conducted in 52 US centers, the effects on sexual function of extended-release alfuzosin 10 mg once daily were evaluated. 35 A slight improvement in erectile function after the administration of alfuzosin was found in patients with erectile dysfunction at baseline; conversely, in all the other patients extended-release alfuzosin significantly improved erectile function in men with LUTS/BPH only while maintaining a stable ejaculatory function. 35 Indeed, sexual dysfunction and LUTS have a close correlation. 36 Although the mechanisms by which alfuzosin decreases sexual dysfunction have not been fully understood, possible explanations could be related to the improvement in the patients’ quality of life, 3 to the direct effects of the blockade of the overall α1-adrenergic receptor signal transduction pathway on erectile function, 37 and to the limited effects on α1a receptors that induce a decrease of the volume of ejaculate by relaxation of bladder neck and/or reduce seminal vesicle contraction and sperm progression. 38
The Italian Alfuzosin Co-Operative Group published the results of a multicentric study on safety and efficacy of alfuzosin 2.5 mg TID administered for 1 year. 39 Both objective (uroflowmetry and residual urine volume) and subjective [IPSS and Quality of Life (QoL) questionnaires] outcomes gradually improved from baseline throughout the treatment period and only 4.3% of patients dropped out due to AEs.
Almost in the same period, the results of a large (4018 patients) multicenter observational study conducted in Spain were published; in particular, the authors focused on the cardiovascular safety profile of alfuzosin 5 mg BID for the treatment of LUTS/BPH.40,41 In this study, alfuzosin was well tolerated independently of the age of the patient. In particular, the distribution of vasodilatory/non-vasodilatory AEs was similar in all age groups, and the incidence of asymptomatic orthostatic hypotension was low (0.58%) and not affected by the age of the patients, thus highlighting the uroselectivity of alfuzosin irrespective of patient age. 41
Finally, the effectiveness and safety of alfuzosin 10 mg OD has been reported in the open-label extension study of the aforementioned ALFORTI study group. 42 Among the 311 patients included, IPSS and uroflowmetry parameters significantly improved from baseline as well as QoL index. Only 4.4% of patients experienced AEs (mainly represented by slight dizziness), and ejaculation disorders were also rare (0.6%). 42
Side effects and safety profile: the role of uroselectivity
Beside the excellent cardiovascular profile together with the low rate of ejaculation disorders that emerged from the above reported studies, alfuzosin showed high tolerability and few vasodilatory effects over older alpha-blocking compounds.
43
The adverse rate of alfuzosin, tamsulosin and silodosin are summarized in Table 3. A systematic review of the literature (RCTs of alfuzosin
Adverse effects of alfuzosin, tamsulosin and silodosin compared with placebo.

Tamsulosin [package insert] Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc, 2003.
Alfuzosin [package insert] Bridgewater, NJ: Sanofi-Synthelabo, Inc, 2004.
Cho HJ and Yoo TK. Silodosin for the treatment of clinical benign prostatic hyperplasia: safety, efficacy, and patient acceptability.
Conversely, the ALF-ONE study group examined the impact of age, cardiovascular comorbidity and antihypertensive co-medication on the tolerability of 10 mg alfuzosin; 46 among 6523 enrolled patients, 6.4% withdrew from the study mainly for AEs, while 229 (3.5%) experienced serious AEs. Overall, the most commonly reported AE was dizziness (4.8%), whereas hypotension was uncommon (0.7%). Of note, age, cardiovascular comorbidity and antihypertensive co-medication had no impact on the safety profile of 10 mg alfuzosin.
Similarly, in a large multicenter open-label study, 335 patients with LUTS/BPH were randomized to receive alfuzosin 10 mg alone or alfuzosin 10 mg + antihypertensive combination therapy. 47 The authors found no differences in terms of efficacy and safety between the two groups, although in patients with uncontrolled or untreated hypertension, alfuzosin 10 mg alone or in combination with antihypertensive therapy appeared to decrease systolic and diastolic BP.
An interesting study by Park
A potential association between presence or absence of metabolic syndrome and response to alfuzosin therapy has been explored in a recent double-blind, randomized, placebo-controlled study; 49 after 12 weeks of treatment, all patients did benefit from therapy, independently from baseline characteristics and presence/absence of metabolic syndrome in particular.
Moreover, α1-blockers have been traditionally related to the onset of so-called intraoperative floppy iris syndrome (IFIS) during cataract surgery with potential influences on postoperative outcomes.
50
A well-conducted multicenter study evaluated the effects of tamsulosin or alfuzosin
Comparison with other alpha-blockers
Six RCTs have assessed efficacy and safety of alfuzosin
Characteristics and results of studies assessing the adverse events and outcomes of alfuzosin

same individuals were treated with two different agents during different time frames.
Nordling J. Efficacy and safety of two doses (10 and 15 mg) of alfuzosin or tamsulosin (0.4 mg) once daily for treating symptomatic benign prostatic hyperplasia.
Buzelin JM, Fonteyne E, Kontturi M,
de Reijke TM and Klarskov P. Comparative efficacy of two $α$1;-adrenoreceptor antagonists, doxazosin and alfuzosin, in patients with lower urinary tract symptoms from benign prostatic enlargement.
Manohar CMS, Nagabhushana M, Karthikeyan VS,
Manjunatha R, Pundarikaksha HP, Madhusudhana HR,
Senkul T, Yilmaz O, Iseri C,
Lapitan MCM, Acepcion V and Mangubat J. A comparative study on the safety and efficacy of tamsulosin and alfuzosin in the management of symptomatic benign prostatic hyperplasia: a randomized controlled clinical trial.
IPSS: International Prostate Symptom Score; NS: non significant; PVR: post-voided residual; RCT: randomized clinical trial.
Nordling
52
reported the results a multicenter experience including 625 patients who were randomized to receive alfuzosin 10 mg or 15 mg, tamsulosin 0.4 mg or placebo. Alfuzosin (10 mg or 15 mg) significantly improved urinary symptoms and uroflowmetry parameters compared with placebo, similarly to tamsulosin, but with a lower rate of sexual function AEs (placebo, 0%; alfuzosin 10 mg, 3%; 15 mg, 1%; tamsulosin, 8%) compared with tamsulosin. Similar results were reported by Buzelin
Alfuzosin: clinical efficacy in combination treatment
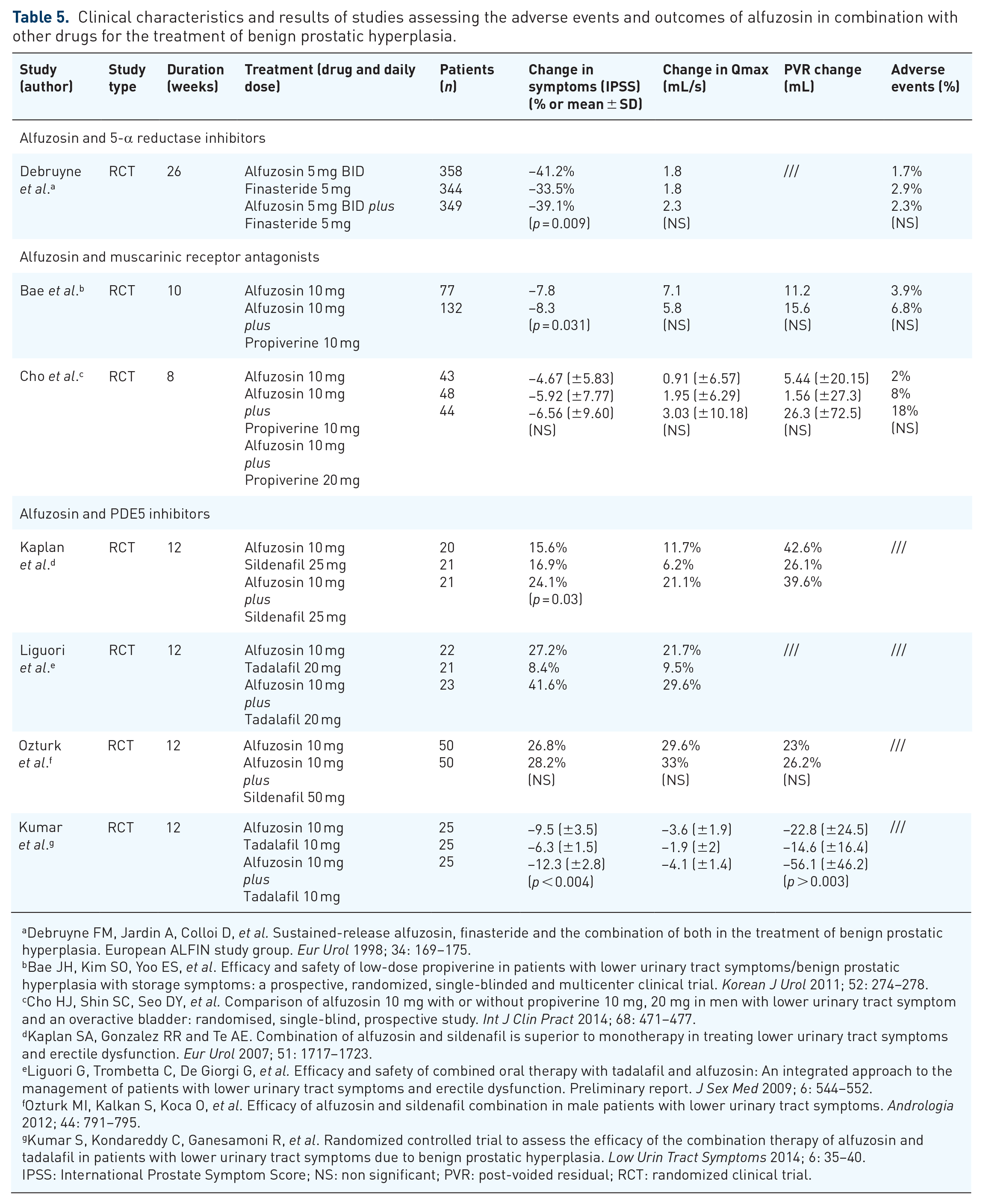
Several studies have investigated the clinical efficacy and safety of alpha-blockers variously combined with other classes of drugs active on LUTS/BPH. The underlying rationale is based on the possibility of obtaining a synergistic effect from the combination of drugs with different pharmacodynamic and pharmacokinetic characteristics. In detail, seven RCTs have tested the association of alfuzosin and different classes of drugs, as shown in Table 5.
Clinical characteristics and results of studies assessing the adverse events and outcomes of alfuzosin in combination with other drugs for the treatment of benign prostatic hyperplasia.
Debruyne FM, Jardin A, Colloi D,
Bae JH, Kim SO, Yoo ES,
Cho HJ, Shin SC, Seo DY,
Kaplan SA, Gonzalez RR and Te AE. Combination of alfuzosin and sildenafil is superior to monotherapy in treating lower urinary tract symptoms and erectile dysfunction.
Liguori G, Trombetta C, De Giorgi G,
Ozturk MI, Kalkan S, Koca O,
Kumar S, Kondareddy C, Ganesamoni R,
IPSS: International Prostate Symptom Score; NS: non significant; PVR: post-voided residual; RCT: randomized clinical trial.
The ALFIN study group reported the results of a large multicenter RCTs aiming to compare the efficacy and safety of alfuzosin alone
Two RCTs have investigated the results of the association of alfuzosin with a muscarinic receptor antagonist (propiverine) in men that met the symptom criteria for overactive bladder with symptomatic outlet obstruction.58,59 In both studies, the association of alfuzosin with propiverine did not provide additional benefits in terms of IPSS voiding symptom score, QoL, Qmax, and PVR; on the contrary, storage symptoms scores significantly improved in case of combination therapy. These results may suggest the use of antimuscarinic agents in combination with an alpha-blocker in patients with marked irritative LUTS.
Moreover, four RCTs have investigated the effects of the combination of alfuzosin with PDE5-inhibitors (PDE5i), two trials with tadalafil (10 or 20 mg)60,61 and two trials with sildenafil (25 or 50 mg).62,63 In general, the results of these trials have demonstrated additional benefits from the association of alfuzosin with PDE5i in terms of both urinary and sexual symptoms without an observed increase rate of AEs. Therefore, alfuzosin + PDE5i may be a safe and effective option in patients with LUTS/BPH and concomitant erectile dysfunction also thanks to the aforementioned uroselectivity of alfuzosin and its low rate of sexual disorders.
Conclusion
Alfuzosin is a safe and effective drug for the treatment of LUTS/BPH, as proven by several clinical trials. Compared with other alpha-blockers, mostly thanks to its high concentration in the prostatic tissue, alfuzosin seems to be associated with a lower rate of sexual dysfunction. Moreover, in terms of safety profile, alfuzosin has been found to be safe also in the case of concomitant antihypertensive therapy and in patients with cardiovascular morbidity. Furthermore, safety and efficacy of alfuzosin has been reported also in case of combination therapy with antimuscarinic agents and PDE5i.
Supplemental Material
sj-pdf-1-tau-10.1177_1756287221993283 – Supplemental material for Alfuzosin for the medical treatment of benign prostatic hyperplasia and lower urinary tract symptoms: a systematic review of the literature and narrative synthesis
Supplemental material, sj-pdf-1-tau-10.1177_1756287221993283 for Alfuzosin for the medical treatment of benign prostatic hyperplasia and lower urinary tract symptoms: a systematic review of the literature and narrative synthesis by Andrea Mari, Alessandro Antonelli, Luca Cindolo, Ferdinando Fusco, Andrea Minervini and Cosimo De Nunzio in Therapeutic Advances in Urology
Footnotes
Contribution
Conception and Design: Mari, Antonelli, Cindolo, Fusco, Minervini, De Nunzio.
Acquisition of data: Mari, Antonelli, Minervini.
Analysis and interpretation of data: Mari, De Nunzio.
Drafting of the manuscript: Mari, Cindolo, Fusco, Minervini
Critical revision of the manuscript for important intellectual content: Antonelli, Cindolo, Fusco, Minervini, De Nunzio
Statistical analysis: NA
Obtaining funding: Antonelli, Cindolo, Fusco, Minervini, De Nunzio
Administrative, technical, or material support: Mari, Antonelli, Cindolo, Fusco, Minervini, De Nunzio
Supervision: De Nunzio
Conflict of interest statement
Ferdinando Fusco: Speaker at symposiums and seminaries for Recordati, Astellas, Glaxo Smith Kline. Cosimo de Nunzio: Consultancy for Pierrefabre, Janssen, Sanofi, Glaxo Smith Kline. Other authors have no additional conflict of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial support in the preparation of this publication was provided and funded by Sanofi. Sanofi’s review of this article has been strictly limited to all references to alfuzosine to enhance consistency within the current labeling of the product and its appropriate use. The authors, individually and collectively, are responsible for all content and editorial decisions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
