Abstract
This narrative review synthesizes current evidence on the medical management of nocturnal polyuria, including antidiuretic replacement therapy as well as other emerging modalities, with particular emphasis on areas of active investigation and future research directions. Relative to earlier formulations, the pharmacological profiles of novel desmopressin acetate nasal spray and orally disintegrating tablet formulations appear favorable in optimizing the balance between efficacy and safety. Additionally, several highly selective small-molecule arginine vasopressin 2 receptor agonists are under active development, while appropriately timed short-acting diuretics, pharmacotherapy for hypertension, nonsteroidal anti-inflammatory drugs, and sex hormone replacement therapy are also a focal point of extensive ongoing nocturnal polyuria research. Emerging laboratory technologies now make feasible a sub-stratification of nocturnal polyuria patients into substrate-based phenotypes for individualized treatment. An increasingly refined understanding of the pathogenesis of nocturnal polyuria, and arginine vasopressin dysregulation in particular, has also introduced new opportunities for point-of-care testing in patients with nocturnal polyuria.
Introduction
Nocturia is one of the most common and bothersome lower urinary tract symptoms (LUTS) in the general population.1,2 Nocturia is associated with significant morbidity, having been identified as an age-independent risk factor for falls and hip fractures, direct mediator of poor sleep, and prognostic factor for all-cause mortality,3–5 and successful treatment of nocturia is correspondingly accompanied by significant improvements in overall health-related quality of life. 6 The pathogenesis of nocturia is multifactorial, 7 but medical therapy is frequently a staple of treatment, particularly in the absence of identifiable contributory comorbidities or behavioral factors – as is often the case in the real-world outpatient urology setting.8,9 Historically, pharmacotherapy for nocturia centered on agents directed at lower urinary tract dysfunction, such as those commonly used in the treatment of benign prostatic hyperplasia (BPH) and overactive bladder syndrome (OABS). Problematically, agents for BPH and OABS often fail to treat the primary pathophysiology of nocturia and, thus, inconsistently translate to meaningful benefit for patients in the real-world clinical setting.10–12 However, several recent advancements in pharmacotherapy for nocturnal polyuria – the most common cause of nocturia in adults of all ages and backgrounds – have the potential to reshape management and raise the standard of care for patients suffering from nocturia.13,14 Accordingly, this narrative review aims to synthesize available evidence on the efficacy and safety of current pharmacotherapies, as well as and progress toward individualized treatment for patients with nocturnal polyuria.
Antidiuretic replacement therapy
Mechanism
Desmopressin (1-deamino-8-
Early formulations and endpoints
In healthy patients, the act of sleeping through the night without urinating requires that the nocturnal urine volume not exceed functional bladder storage capacity. 26 When nocturnal urine volume does exceed the functional storage threshold, the requisite end result is nocturnal urination – nocturia if the sensation of bladder fullness causes the patient to awaken and, if not, enuresis must ensue. 10 Further, in view of the mechanism of action of antidiuretic replacement therapy, the primary role of desmopressin in alleviating both nocturia and nocturnal enuresis is accomplished via a significant reduction in nocturnal water diuresis.22–25 Correspondingly, initial trials of desmopressin in patients with nocturia employed oral tablet doses with proven efficacy in the treatment of monosymptomatic nocturnal enuresis (100–400 mcg).27–29
Despite the congruence between the pathogenesis and expected mechanism of treatment response between nocturia and nocturnal enuresis, 30 there were several pertinent methodological differences between enuresis and nocturia trials, as well as other fundamental distinctions between these two entities seemingly not accounted for in the dosing schema for nocturia. 31 The standard primary outcome variable in trials of desmopressin for primary monosymptomatic nocturnal enuresis was change in the frequency of wet nights. 32 In view of the production-storage mismatch central to the pathogenesis of nocturnal micturition, a binary treatment outcome (i.e., “dry night” or “wet night” in enuretic children) requires maximal suppression of nocturnal urine production to a level below the functional storage threshold. Among adults with nocturia, the analogous outcome would be complete symptom resolution (i.e., zero nocturnal voids), which was indeed the criterion for “full response” in the dose titration phase of early pivotal trials for nocturia.22,23 While complete resolution of nocturia (without adverse events) would indeed reflect the optimal response to treatment for nocturia, the number of nightly voids is associated positively with nocturia-related impairments in quality of life, such that nocturia severity has been increasingly described to exist on a graded continuum. 33 Current understanding of nocturia as a continuous measure is of direct clinical relevance because treatment that successfully reduces nocturnal voiding frequency is associated with significant improvements in overall health-related quality of life – even in patients who do not experience complete symptom resolution. 34
Many of the consequences of nocturia on health-related quality of life have been increasingly recognized to be mediated by the direct adverse effects of nocturia on sleep outcomes. 7 Multiple studies have implicated nocturia as the most common cause of nocturnal awakenings in adults,35,36 and most patients with nocturia experience their first nocturnal voiding episode within the first 2–3 h of sleep. 37 This time period is particularly meaningful because it coincides with the greatest concentration of slow-wave (i.e., N3 or “deep”) sleep in the normal sleep cycle. Slow-wave sleep, which is clustered toward the beginning of the night,38,39 is considered the most restorative stage of sleep. 40 Disrupted slow-wave sleep may not be recovered later in the night upon return to sleep and independently contributes to significant daytime fatigue and discomfort, even when sleep efficiency and total sleep duration are preserved.41,42 Patients with nocturia demonstrate an inverse relationship between the total number of nocturnal voids and the duration of time from sleep onset until the first nocturnal awakening to void [i.e., first uninterrupted sleep period (FUSP)]. 3 Consistently, a shorter FUSP is associated with lower whole-night sleep quality, sleep efficiency, and total sleep duration, corresponding to significant impairment in daytime function. 37 Furthermore, improvement in the actual number of nocturnal voids is accompanied not only by a prolongation in the FUSP, but also in the total duration of sleep and overall sleep quality.43,44 Treatment aimed at extending FUSP in particular may be central to mitigating the vast deleterious sequelae of nocturia, as was exemplified in a recent study of blood sugar in nocturia patients, which reported significant improvement in blood glucose upon successful prolongation of FUSP. 45
Consideration of more refined clinical endpoints, with a particular emphasis on graded treatment response, sleep, and the broader sequelae of nocturia, has facilitated a more holistic approach to nocturia evaluation and management. By the same token, there now exist several lines of evidence to suggest that the optimal dose range of desmopressin for nocturia may actually lie below the dose range initially surmised from the management of enuresis. While the pharmacokinetics of desmopressin are comparable between children and adults, the pharmacodynamic effects of desmopressin differ significantly between these two populations.46–48 In the treatment of adults with nocturia, there appears to be a large discrepancy between the median effective dose and the dose required to achieve maximal antidiuretic response, such that progressively increasing dose levels may produce minimal additional effect.49,50 Consistently, administration of desmopressin in line with the median effective dose range retains most of its therapeutic effect, lending to ⩾33–50% improvement in nocturia severity in a sizable proportion of patients, with improvements in FUSP and health-related quality of life comparable with earlier studies of patients maximally exposed to desmopressin.49,50 Moreover, a plasma desmopressin concentration which exceeds the threshold required for a positive antidiuretic effect may actually prolong the duration of antidiuresis, exerting unintended effects on fluid handling beyond the hours of sleep.51,52 Higher drug levels are also associated with hyponatremia – the most important risk factor associated with desmopressin. 27 Taken together, lower-dose formulations of desmopressin for nocturia are thought to offer a significant benefit in optimizing the balance between therapeutic effect and tolerability in patients of all ages. 53
Recent formulations
Desmopressin has been approved in more than 80 countries since the 1980s for the treatment of symptomatic nocturia. 54 In the United States (US), two new formulations, desmopressin acetate nasal spray and orally disintegrating tablets, are currently indicated for patients with nocturnal polyuria who void ⩾2 times nightly.55,56
Desmopressin acetate nasal spray
In March 2017, desmopressin acetate nasal spray became the first desmopressin formulation to be approved by the US Food and Drug Administration (FDA) in the treatment of nocturia, indicated for patients with nocturnal polyuria who void ⩾2 times nightly. 57 Desmopressin acetate nasal spray is an intranasal formulation of desmopressin with cyclopentadecanolide (CPD) – a novel cyclic fatty acid excipient designed to improve permeation, enhancing absorption across the nasal mucosa and bioavailability of the active compound. 58 The proposed advantages of this formulation include potentially more consistent and predictable dosing, more rapid onset of the drug effect, and decreased likelihood of prolonged drug activity. 54
Desmopressin acetate nasal spray is available in doses of 0.83 mcg and 1.66 mcg per 0.1 ml nasal spray, bioequivalent to 0.75 mcg and 1.5 mcg desmopressin free base per spray, respectively. 57 The 1.66 mcg/0.1 ml dosage is recommended for patients <65 years without risk factors for hyponatremia, whereas patients ⩾65 years or at increased hyponatremia risk should be started at the 0.83 mcg/0.1 ml dose, and, if needed, advanced to 1.66 mcg/0.1 ml after 1 week provided that serum sodium remains within normal limits. 55 Patients should be instructed to administer one spray in either nostril approximately 30 min prior to bedtime. Consistent with other desmopressin products, the main safety finding with this agent is hyponatremia, which can be mitigated by following warnings and precautions outlined in the prescribing information as well as scheduled serum sodium monitoring. 55 Serum sodium should be measured before treatment, within 7 days and approximately 1 month after initiation or dose escalation, and then periodically during treatment, with more frequent monitoring in patients ⩾65 years or at increased risk of hyponatremia. 55
FDA approval for desmopressin acetate nasal spray was based on data from two independent North American phase III double-blind randomized placebo-controlled trials [ClinicalTrials.gov identifiers: NCT01357356 and NCT01900704] of patients ⩾50 years of age with ⩾2 nightly voids for more than 6 months and ⩾2.16 average nightly voids during a 2-week screening period (Table 1). 59 Eligible patients were randomized equally to receive 1.66 mcg/0.1 ml or 0.83 mcg/0.1 ml (or 1.11 mcg/0.1 ml in one of the studies only) or placebo for 12 weeks. Co-primary endpoints were mean change in nocturia frequency and the percentage of patients reporting a ⩾50% response in nocturia frequency, as determined from nighttime voiding diaries maintained for three consecutive nights, which were completed by participants at baseline and then 1, 2, 4, 6, 8, 10, and 12 weeks post-randomization. Secondary endpoints were duration of the FUSP, percentage of nights with ⩽1 or 0 nocturnal voids, change in nocturnal urine volume, and impact on quality of life [as assessed by validated questionnaire (one study only)]. Safety was assessed based on the incidence of adverse events, physical examination and electrocardiogram, and serum sodium monitoring at baseline and then at 2-week intervals post-randomization.
Phase III randomized control trials on desmopressin acetate nasal spray and orally disintegrating tablet formulations for nocturia.

Several pertinent sex-based differences observed on post hoc analysis (see main text).
Refractory to behavioral counseling and a 1-week placebo run-in period.
FUSP, first uninterrupted sleep period (time from sleep onset until first nocturnal voiding episode); QoL, quality of life; US, United States.
On pooled intent-to-treat analysis (n = 1333), both doses of desmopressin acetate nasal spray conferred significant benefit compared with placebo for mean change in nocturia frequency [−1.4 with 0.83 mcg/0.1 ml and −1.5 with 1.66 mcg/0.1 ml compared with −1.2 with placebo (p < 0.0001 for both)] as well as ⩾50% response rate [37.9% with 0.83 mcg/0.1 ml and 48.7% with 1.66 mcg/0.1 ml compared with 30.3% with placebo (p = 0.0227, p < 0.0001, respectively)]. Additionally, both doses also conferred significant benefit compared with placebo in change in FUSP duration [+96 min with 0.83 mcg/0.1 ml and +108 min with 1.66 mcg/0.1 ml compared with +72 min with placebo (p = 0.0121, p < 0.0001, respectively)], and statistical significance was observed for all other secondary endpoints. A total of 5.9% of all patients discontinued the study due to adverse events, with study drug-induced adverse events observed in 3.5% of patients receiving 0.83 mcg/0.1 ml, 5.6% receiving 1.66 mcg/0.1 ml, and 4.0% receiving placebo. Upper respiratory symptoms (e.g., nasal discomfort, congestion, rhinorrhea, sinus pain, or throat irritation) were the adverse event most commonly leading to study discontinuation. The incidence of hyponatremia, defined as serum sodium ⩽125 mmol/l or <130 mmol/l with symptoms, was 1.1% with 1.66 mcg/0.1 ml, 0% with 0.83 mcg/0.1 ml, and 0.2% with placebo. Differences in other adverse events were similar across treatment groups. Treatment efficacy was maintained with no overall changes in the safety profile at follow up of up to 2 years. 57
Desmopressin acetate orally disintegrating tablets
The FDA approved desmopressin acetate orally disintegrating tablets in June 2018, indicated for patients with nocturnal polyuria who void ⩾2 times nightly. 56 Orally disintegrating tablets are unique in that they are offered at sex-specific dosages, thus facilitating individualized treatment. Orally disintegrating tablets are available in doses of 27.7 mcg and 55.3 mcg, equivalent to 25 mcg and 50 mcg of desmopressin, respectively. 56 The desmopressin 25 mcg tablets are recommended for women and the desmopressin 50 mcg tablets are recommended for men. 56 Patients should be instructed to administer one tablet sublingually without water 60 min before bedtime. Consistent with other desmopressin products, the main safety finding with this agent is hyponatremia, which can be mitigated by following warnings and precautions outlined in the prescribing information for orally disintegrating tablets as well as scheduled serum sodium monitoring. 56 Serum sodium should be measured before treatment, within 7 days and approximately 1 month after initiating treatment, and then periodically during treatment, with more frequent monitoring in patients ⩾65 years or at increased risk of hyponatremia. 56
Foundational evidence regarding the efficacy and safety of orally disintegrating tablets has been derived from three independent multicenter phase III double-blind randomized placebo-controlled trials involving both men and women [ClinicalTrials.gov identifier: NCT00477490], 34 specifically men [ClinicalTrials.gov identifier: NCT01262456], 60 and specifically women [ClinicalTrials.gov identifier: NCT01223937]. 61 The trial involving both men and women was a 4-week parallel group study designed to test the relative efficacy and tolerability of low doses of orally disintegrating tablets using a step-down approach. 34 Eligible for inclusion were men and women ⩾18 years of age with ⩾2 voids nightly as confirmed by 3-day voiding diary analysis during screening. Included patients were randomized accounting for age and nocturnal polyuria into equal groups to receive 10, 25, 50, or 100 mcg desmopressin orally disintegrating tablets or placebo for 28 days. Co-primary endpoints were mean change in average nocturnal voiding frequency and the percentage of patients reporting a ⩾33% response in nocturia frequency from baseline to the final visit. Secondary endpoints included change in nocturnal and total urine volume, duration of the FUSP, and change in nocturia-related quality of life. Safety was assessed based on the incidence of adverse events, physical examination with comprehensive blood and urine analyses, and serum sodium monitoring performed at baseline, on days 4 and 8, and then weekly for the remainder of the study period.
A total of 757 patients were included in the final intent-to-treat analysis. Among all patients, the 50 and 100 mcg doses conferred significant benefit for mean change in nocturia frequency [−1.18 with 50 mcg and −1.43 with 100 mcg compared with −0.86 with placebo (p = 0.020, p ⩽ 0.0001), respectively] and the 100 mcg dose conferred significant benefit for 33% responder rate [71% with 100 mcg compared with 47% with placebo (p < 0.0001)]. For secondary endpoints, change in nocturnal urine volume compared with placebo was significant with 25, 50, and 100 mcg doses, change in 24-h in total urine volume was significant with the 100 mcg dose, and change in duration of the FUSP was significant with 25, 50, and 100 mcg doses. In total, 24 (3%) of patients were found to have serum sodium levels <130 mmol/l, of whom 9 (1.1%) had reductions to <125 mmol/l.
Although not factored into the primary study design, post hoc analyses revealed several pertinent outcome differences on the basis of sex. Among males, the 100 mcg dose conferred significant benefit compared with placebo for mean change in nocturia frequency [−1.38 with 100 mcg compared with −0.84 with placebo (p = 0.005)] as well as for 33% responder rate [65% with 100 mcg compared with 50% with placebo (p = 0.050)]. For secondary endpoints, change in nocturnal urine volume compared with placebo was significant with 50 and 100 mcg doses, change in 24-h in total urine volume was significant with the 100 mcg dose, and change in duration of the FUSP was significant with the 100 mcg dose. Among females, 25, 50, and 100 mcg doses conferred significant benefit compared with placebo for mean change in nocturia frequency [−1.22 with 25 mcg, −1.23 with 50 mcg, and −1.51 with 100 mcg compared with −0.88 with placebo (p = 0.020, p = 0.009, p < 0.0001, respectively)] as well as for 33% responder rate [62% with 25 mcg, 59% with 50 mcg, and 77% with 100 mcg compared with 42% with placebo (p = 0.020, p = 0.040, p < 0.0001, respectively)]. For secondary endpoints, change in nocturnal urine volume compared with placebo was significant with 10, 25, 50, and 100 mcg doses, change in 24-h in total urine volume was significant with the 25 mcg dose, and change in duration of the FUSP was significant with 25, 50, and 100 mcg doses.
This parallel study of men and women was particularly noteworthy in that it provided a pivotal foundation for establishing sex-specific dosing recommendations. These data from Weiss and colleagues, in conjunction with results from an open-label extension trial [ClinicalTrials.gov identifier: NCT00615836] and two separate phase I studies, were subsequently operationalized by Juul et al. to characterize sex differences in antidiuretic response to desmopressin therapy. 50 Juul and colleagues observed a significant sex difference in nocturnal urine volume with desmopressin, characterized by a significantly greater decrease (i.e., desmopressin sensitivity) with 10 and 25 mcg doses in women compared with men, which could not be explained by pharmacokinetic differences. Regarding tolerability, no hyponatremia events (defined as serum sodium <130 mmol/l) were identified in women ⩽50 years or men ⩽65 years of age, but a dose-response relationship was observed in patients beyond 50 years of age, wherein the incidence of hyponatremia was approximately twofold greater in women at dose levels ⩾25 mcg. Taken together, Juul et al. concluded that a dose of 50−100 mcg was optimal in balancing therapeutic benefit with tolerability in men, while a dose of 25 mcg is efficacious in women with no observed incidences of clinically significant hyponatremia. Notably, these findings were consistent with well-controlled physiologic evidence of sex differences in the endogenous regulation of AVP secretion and sensitivity, 63 thought to be mediated by sex hormones or differential expression of AVPR2 receptor mRNA and protein on the X chromosome due to the process of X-inactivation in females.64–66
In view of growing evidence favoring sex-specific dosing for orally disintegrating tablets, Weiss et al. subsequently investigated the efficacy and safety of 50 mcg and 75 mcg orally disintegrating tablets compared with placebo in men ⩾18 years with ⩾2 voids nightly, as confirmed by 3-day voiding diary analysis during screening. 60 Eligible patients underwent age-stratified equal randomization to receive 50 mcg or 75 mcg of desmopressin or placebo for 12 weeks. Co-primary endpoints were mean change in nocturia frequency and the percentage of patients reporting a mean change in average nocturia frequency of ⩾33%, as determined from longitudinal analysis of 3-day voiding diaries, which were completed immediately pre- and post-randomization and then at weeks 1, 4, 8, and 12. Secondary endpoints were mean change in average nocturnal voiding frequency and ⩾33% response rate at the 3-month mark, duration of the FUSP, and change in nocturnal urine volume. Safety was assessed based on the incidence of adverse events, physical examination with comprehensive blood and urine analyses, and serum sodium monitoring performed at baseline and then 4, 7, 30, 60, and 90 days after treatment initiation.
On final analysis (n = 385), both doses of desmopressin acetate orally disintegrating tablets conferred significant benefit compared with placebo for adjusted mean change in nocturia frequency [−1.25 with 50 mcg and −1.29 with 75 mcg compared with −0.88 with placebo (p = 0.0003, p < 0.0001, respectively)] as well as for adjusted odds of ⩾33% response rate [odds ratio (OR) 1.98 with 50 mcg versus placebo; OR 2.04 with 75 mcg versus placebo (p = 0.0009, p = 0.004, respectively)]. Additionally, statistical significance was observed for secondary endpoints at 3 months, and both doses conferred significant benefit compared with placebo for change in FUSP duration [+112 min with 50 mcg and +116 min with 75 mcg compared with +73 min with placebo (p = 0.006, p = 0.003, respectively)]. The most common adverse events were dry mouth and headache, and a total of 4.7% of all patients experienced an adverse event leading to discontinuation. The incidence of hyponatremia was 1.9% with 50 mcg, 3.3% with 75 mcg, and 0% with placebo using a serum sodium threshold of ⩽125 mmol/l, and 1.9%, 7.4%, and 0%, respectively, using a serum sodium threshold of <130 mmol/l.
Contemporary to the desmopressin trial involving men, Sand et al. investigated the efficacy and safety of 25 mcg orally disintegrating tablets compared with placebo in women ⩾18 years of age with ⩾2 voids nightly, as confirmed by 3-day voiding diary analysis during screening. 61 Core features of the study design, including randomization, primary and secondary outcomes, measurement and timing of outcomes, and safety monitoring, were analogous to those employed by Weiss and colleagues. 60 On final analysis (n = 261), desmopressin conferred significant benefit compared with placebo for adjusted mean change in nocturia frequency (−1.46 with desmopressin compared with −1.24 with placebo, p = 0.028) as well as for adjusted odds of ⩾33% improvement (OR 1.85, p = 0.006 compared with placebo). Statistical significance was also observed in the change in average nocturnal voiding frequency at the 3-month mark, as well as in the duration of the FUSP and nocturnal urine volume from baseline, although no significant difference was observed in 3-month 33% responder rate. The most common adverse events were urinary tract infection, headache, and upper respiratory tract infection, and a total of 1.9% of all patients experienced adverse events leading to discontinuation. The incidence of hyponatremia according using a serum sodium threshold <130 mmol/l was 2.2% in the desmopressin group and 0% in the placebo group. No patients in either study arm were found to have serum sodium ⩽125 mmol/l.
More recently, the concept of sex-specific dosing was also further explored through two independent multicenter phase III randomized placebo-controlled trials investigating orally disintegrating tablets in Japanese males [ClinicalTrials.gov identifier: NCT02904759] and females [ClinicalTrials.gov identifier: NCT02905682]. 62 Patients ⩾20 years of age were eligible if they were found to have persistent nocturia (⩾2 nocturnal voids) due to nocturnal polyuria after completing a 1-week placebo run-in period with behavioral counseling. Females received either 25 mcg or placebo and males received 25 mcg, 50 mcg, or placebo. The primary endpoint was change from baseline nocturnal voiding frequency during 12 weeks of treatment. Secondary endpoints included changes in duration of the FUSP, nocturnal urine volume, nocturnal polyuria index (defined as nocturnal urine volume/24-h urine volume), and nocturia-related quality of life. Safety was assessed based on the incidence of adverse events, physical examination with comprehensive blood and urine analyses, and serum sodium monitoring performed at baseline and then on days 3, 7, 14, 30, 60, and 90 after treatment initiation.
A total of 525 patients were included in the full analysis set. Among males (n = 338), both 25 and 50 mcg dose levels conferred significant benefit in mean nocturia frequency compared with placebo [−0.96 with 25 mcg and −1.21 with 50 mcg compared with −0.76 with placebo (p = 0.0143, p < 0.0001, respectively)]. Among females (n = 187), 25 mcg improved mean nocturia frequency with borderline statistical significance compared with placebo (−1.11 versus −0.95, respectively, p = 0.0975). While the primary endpoint did not meet statistical significance according to conventional standards, duration of the FUSP, nocturnal urine volume, and nocturnal polyuria index all significantly improved beyond placebo for all dose levels in females as well as in males. Further, 25 mcg tablets in females, as well as 25 and 50 mcg tablets in males, demonstrated long-term efficacy and reasonable tolerability in a subsequent long-term trial designed partly as an extension study for subjects completing the phase III trials [ClinicalTrials.gov identifier: NCT03051009]. 67 Treatment-emergent hyponatremia of <130 mmol/l was observed in none of the females and in 0.9%, 1.8%, and 0% of males with 25 mcg, 50 mcg, and placebo, respectively. (Yamaguchi, Juul and Falahati, 2020) No patients of either sex were found to have serum sodium ⩽125 mmol/l. (Yamaguchi, Juul and Falahati, 2020)
Sodium monitoring
Owing to significant regional regulatory variation, current guidelines from the International Continence Society advocate for comprehensive monitoring when serum sodium checks are clinically warranted, but do not offer a consensus as to whether baseline serum sodium must be assessed before starting desmopressin in young patients with no risk factors for hyponatremia. 68 However, more definitive recommendations have been detailed in current European Association of Urology (EAU) guidelines for non-neurogenic male LUTS, as well as US FDA prescriber information for both desmopressin acetate nasal spray and orally disintegrating tablets. EAU guidelines specify that serum sodium should be assessed at the time of medication initiation or dose titration, and then 3, 7, and 30 days after baseline, followed by periodic monitoring across the course of treatment. 69 According to these guidelines, maintenance serum sodium checks can be scheduled every 3 months for patients with stable serum sodium during the initial monitoring sequence, but should occur more frequently in patients ⩾65 years or with risk factors for hyponatremia, and the initial monitoring regimen should be restarted following dose escalation. 69 As outlined in the sections above, FDA prescriber information for both desmopressin acetate nasal spray and orally disintegrating tablets specifies safe use to include serum sodium monitoring for all patients prior to medication initiation, restart, or dose escalation, and again within 7 days and approximately 30 days from baseline, with subsequent periodic monitoring and more frequent checks in patients ⩾65 years or at increased risk of hyponatremia.55,56 Indeed, the risk of hyponatremia in patients ⩾65 is highlighted by the inclusion of desmopressin in the 2019 American Geriatrics Society Beers Criteria describing pharmaceuticals to be used with particular caution in both men and women of this age group. 70
Notwithstanding the logistical considerations of routine serum sodium monitoring, recent research contends that a conservative approach to sodium testing may reliably prevent clinically significant hyponatremia in patients on desmopressin replacement therapy. Using pooled data from 1443 patients included in phase III trials for desmopressin acetate orally disintegrating tablets,34,60,61 Juul et al. performed a meta-analysis to identify specific risk factors for clinically significant treatment-induced hyponatremia, defined as serum sodium <130 mmol/l. 27 In both sexes, patient characteristics independently associated with significant hyponatremia included age ⩾65 years and diminished renal clearance (<60 ml/min). 27 Baseline serum sodium <135 mmol/l, as well as exposure to desmopressin above the sex-specific minimum effective dose (25 μg for females and 50 μg for men), were also identified as independent risk factors for clinically significant hyponatremia, and a majority of all hyponatremic events occurred within the first 3 weeks of treatment.
Consistent with these observations, Juul et al. devised a structured serum sodium monitoring plan for preventing desmopressin-induced hyponatremia. One approach proposed by the investigators called for a baseline serum sodium check in all patients, with repeat monitoring at 7 and 30 days in patients ⩾65 years or concurrently taking other medications associated with hyponatremia. Notably, for patients exposed to the minimum effective dose of desmopressin, adherence to this sodium monitoring plan with immediate discontinuation following a serum sodium level <135 mmol/l would have successfully identified all subsequent cases of clinically significant hyponatremia for exclusion from further treatment. This particular sodium monitoring schedule was also noteworthy because of its significant overlap with current FDA prescriber guidelines for desmopressin (albeit slightly less stringent due to the researcher’s omission of 7- and 30-day repeat checks in healthy younger patients). While such analyses have not been repeated using desmopressin acetate nasal spray, nearly half the incident cases of serum sodium <130 mmol/l in pivotal trials for that formulation were likewise observed within the first 2 weeks following treatment initiation. 54
Current trends in antidiuretic replacement therapy
Based on a recent update from the EAU non-neurogenic male LUTS guidelines panel, desmopressin for nocturia in men <65 years is the only medication supported by Level 1a evidence, as well as the only specific pharmacotherapy to receive a Grade A recommendation in the treatment of nocturia. 71 At present, real-world nocturnal polyuria patients are being actively recruited for long-term specified-use prospective observational trials, which stands to further corroborate current evidence regarding the efficacy and safety of currently-approved desmopressin formulations [ClinicalTrials.gov identifier: NCT04329975]. In the clinical setting, there exists significant investigative interest in phenotyping the population of patients most likely to respond to antidiuretic replacement therapy, including our own work on differential trajectories of intra-nocturnal urine production between distinct subsets of patients with nocturnal polyuria.72–78
While desmopressin is the only antidiuretic peptide available to treat nocturia and other conditions characterized by AVP deficiency, several alternative small-molecule AVP2R agonists are in various stages of development. A phase II trial of ASP7035 for nocturnal polyuria is listed in the European Clinical Trials Database as ongoing at some study sites (EudraCT2013-003701-25). Recently completed trials include a phase II study of SK-1404 for nocturnal polyuria [ClinicalTrials.gov identifier: NCT03116191] and a phase IIB/III study of fedovapagon (VA106483) for nocturia in men with BPH [ClinicalTrials.gov identifier: NCT02637960]. While results of these trials are not widely available, a summary of key results was included in a prior evidence briefing on fedovapagon from the National Institute for Health Research, which reported significant improvement in primary study endpoints pooled across the entire treatment period. 79 Even more recently, a series of newly discovered truncated AVP analogs were reported to retain AVP2R potency with increased selectivity, and thus hold the potential to further improve the pharmacological profile of antidiuretic replacement therapy. 80 Two of these new compounds, FE 202217 and FE 201836, were selected for clinical development, and a response-adaptive dose-finding phase II trial in nocturnal polyuria patients has been completed for FE201836 [ClinicalTrials.gov identifier: NCT03201419].
Additional pharmacotherapies for nocturnal polyuria
While medications prototypically used for lower urinary tract dysfunction have been increasingly recognized to affect systems and processes beyond the bladder,81–83 their potential effects on physiologic fluid handling and urine production are highly complex,84–86 and possible benefit in reducing nocturnal urine production has not been consistently demonstrated.87–91 Further, these agents have been studied most extensively in carefully selected LUTS patient subsets, among which they generally reduce nocturia by 0.2−0.4 or fewer voids per night beyond placebo. 71 As such, desmopressin remains the only medical therapy in the clinician’s standard toolkit for LUTS from which clinically meaningful improvement in nocturia due to nocturnal polyuria may be reasonably expected.
However, beyond antidiuretic replacement therapy, several other emerging pharmacotherapies may also be effective in targeting excess nocturnal urine production. At present, timed diuretic therapy holds a Grade C recommendation from the EAU guidelines panel for male LUTS, but does find support through Level 1B evidence. 71 Evidence supporting the role of pharmacotherapy for hypertension, nonsteroidal anti-inflammatory drugs (NSAIDs), and sex hormone replacement therapy in treating nocturnal polyuria is variable in quality. Moreover, the specific physiologic mechanisms by which these agents contribute to a positive treatment response in patients with nocturia have not been completely characterized, but such knowledge gaps are the focal point of extensive ongoing nocturnal polyuria research.75,92 As was the case for antidiuretic replacement therapy, diuretic therapy for nocturia should be accompanied by regular screening for hyponatremia, 71 while all above-mentioned modalities require careful patient selection and shared decision-making to optimize the balance between therapeutic effect and risks associated with treatment.93–96
Diuretics
The relationship between nocturia and diuretics is rather paradoxical and highly dependent on the specific situation at hand. Several population studies have identified an association between nocturia severity and diuretic use,97–99 and yet, multiple double-blind placebo-controlled trials involving healthy adults have found afternoon administration of potent diuretics to significantly improve nocturia. Because diuretics are a core tenet in the management of congestive heart failure and other volume overload states, and daytime third-space fluid sequestration, in and of itself, is an important pathophysiologic mechanism of nocturnal polyuria in some patients, 100 the population-level association between nocturia and diuretics is likely explained, in large part, by the status of diuretics as a marker of volume overload states.101,102 In contrast, among otherwise healthy adults, administration of short-acting diuretics in the afternoon (i.e., more than 4–6 h prior to bedtime) is thought to improve nocturia by restoring the circadian rhythm of sodium homeostasis. 103
In a double-blind randomized placebo-controlled crossover study, adults regularly experiencing ⩾2 nocturnal voids in a general practice setting were administered 2 weeks of bumetanide 1 mg and of placebo, taken 4−6 h prior to bedtime. 104 Of the 28 patients who completed the study, bumetanide conferred a significant benefit compared with placebo in median number of weekly nocturnal voids (10.0 versus 13.8, p < 0.05). A total of 33 patients were initially enrolled in the study, including 2 excluded for nonadherence and 3 excluded for adverse events, although the time frame and severity of the adverse events was not made clear.
These results were later extended through the use of voiding diaries in a subsequent 4-week double-blind randomized placebo-controlled study of furosemide. 105 Reynard et al. randomized 49 men ⩾50 years with nocturnal polyuria to receive afternoon furosemide 40 mg or placebo following a 2-week placebo run-in period. Nighttime and daytime voiding frequency and voided volumes were compared between groups using 7-day voiding diaries completed in the second week of the run-in period and then during the fourth week of treatment. Of the 43 (88%) men who completed the study, furosemide conferred a significant benefit in nocturia frequency (−0.5 versus 0, p = 0.014) and the proportion of subjects responding by ⩾1 nightly void (36.8% versus 5.0%, p = 0.02). While the change in nocturnal urine volume observed between groups was not statistically significant (−120 versus +9 ml, p = 0.064), subjects in the experimental group were found to experience a significant increase in daytime voided volume with no change in 24-h urine output, corresponding to a significant improvement in the nocturnal polyuria index with furosemide (−0.18 versus 0, p = 0.001). No adverse events were reported.
The efficacy of furosemide in treating nocturia has been also been assessed in two studies involving combination pharmacotherapy. One trial published in Chinese randomized 64 LUTS patients to receive afternoon furosemide 40 mg combined with doxazosin 4 mg or doxazosin monotherapy. 106 Combination therapy conferred significant improvement in nocturia frequency (−1.22 versus −0.47 voids, p = 0.003) and nocturnal urine volume (−138 versus −43 ml, p < 0.001) relative to alpha-blocker monotherapy. 106 In another prospective randomized control trial of adults >60 years with nocturia, afternoon furosemide staggered with bedtime desmopressin therapy was found to significantly improve nocturia severity compared with placebo, 107 but it should be noted that loop diuretics remain a contraindication to desmopressin in current FDA prescriber guidelines.55,56
A more recent multicenter open-label randomized crossover trial compared the efficacy of furosemide and an gosha-jinki-gan (GJG) − an East Asian blended herbal medicine which is popular in some countries for nocturia and other LUTS – in patients ⩾50 years of age with nocturia (⩾2 nightly voids) due to nocturnal polyuria. 108 Furosemide 20 mg was administered once daily in the afternoon, while GJG 7.5 mg was administered 3 times daily, and patients sequentially received each treatment for a total of 4 weeks. Of the 36 patients who completed the study, analysis of 3-day voiding diaries revealed significant improvement in mean nocturnal voiding frequency with both interventions relative to baseline [3.3 with furosemide and 3.9 with GJG, compared with 4.4 voids at baseline (p < 0.001, p = 0.020, respectively)], as well as significantly greater benefit with furosemide compared with GJG (p < 0.001). Mean nocturnal urine volume was also lower with furosemide compared with GJG and baseline [793 ml with furosemide, 897 ml with GJG, and 991 ml at baseline (p < 0.001 between furosemide and both GJG and baseline)]. All adverse events were graded as mild and no serum electrolyte imbalances were observed.
Antihypertensive therapy
Disruption in the normal circadian rhythm of blood pressure has been increasingly recognized as an important mediator of the widely described population-level association between nocturia and hypertension.99,102,109,110 Blood pressure follows a well-defined circadian pattern, characterized by a 10–20% decline during the main sleeping period relative to daytime. The absence of this expected nocturnal blood pressure decline, or “dip,” is referred to as non-dipping hypertension – a phenomenon affecting upwards of 40–50% of hypertensive adults.111,112 In the Nagahama cohort study of community-dwelling adults in the Japanese general population, frequent nocturnal urination was found to be independently associated with a smaller drop in nocturnal blood pressure relative to daytime blood pressure, as determined from scheduled home blood pressure recordings. 113 Similar results were observed using multi-day ambulatory blood pressure monitoring in the HEIJO-KYO cohort study of community-dwelling older adults in Japan, which found nocturia to be significantly associated with higher nighttime blood pressure and blunted nocturnal dipping, independent of several potential confounding variables including antihypertensive medication utilization and circadian rhythm parameters. 114
The pathophysiology of nocturia owing to non-dipping hypertension has not yet been fully characterized, but likely involves dysregulation of the renin-angiotensin-aldosterone pathway, corresponding to a relative excess in aldosterone and enhanced nocturnal natriuresis.115,116 In a well-controlled study of healthy Danish men with occasional nocturia, the occurrence of nocturia was characterized by both greater nocturnal urine volume and higher nighttime mean arterial blood pressure compared with nights without nocturia, supporting the role of excess nocturnal urine production as a key mediator in the relationship between nocturia and nocturnal blood pressure. 117 Non-dipping hypertension on ambulatory blood pressure monitoring was also found to be significantly more prevalent in nocturic subjects with versus without nocturnal polyuria in a recent convenience sample of patients with LUTS. 118
Current evidence suggests that medical optimization of hypertensive disease may also be an effective intervention in the clinical management of nocturnal polyuria. In a prospective clinical study, Cho et al. reported significant improvement in nocturia frequency, nocturnal urine volume, and nocturnal polyuria index after 4 weeks of afternoon hydrochlorothiazide 25 mg adjunctive therapy in men with nocturia refractory to alpha blocker monotherapy. 119 Although blood pressure was not taken into account, it should be noted that thiazide diuretics exert a particularly pronounced antihypertensive effect, as evidenced by the fact that thiazide-like diuretics are often warranted as first-line management for hypertension. 120 In a non-controlled exploratory analysis of elderly patients (73.3% with nocturia) with losartan-refractory hypertension started on hydrochlorothiazide 12.5 mg, Kojima et al. observed significant improvement in nocturia frequency as well as systolic and diastolic blood pressure after 1 month of combination therapy, although statistical significance did not persist for nocturia relative to baseline at the 3-month mark. 121 While nocturia response to antihypertensive treatment remains under-studied, it is one of the primary aims of an active pilot study on treatment for nocturnal hypertension in African-American men [ClinicalTrials.gov identifier: NCT03319823], designed as a foundation for future grant-funded comprehensive research on this topic.
Nonsteroidal anti-inflammatory drugs
NSAIDs inhibit the synthesis of prostaglandins, which are known to increase and maintain renal blood flow and subsequent sodium and water excretion, and may also indirectly counteract renal free water conservation via inhibition of endogenous AVP activity at the renal collecting duct.122,123 In a 1-month double-blind randomized placebo-controlled study, 80 men with ⩾2 nocturnal voids and LUTS with BPH were assigned to receive nightly 100 mg celecoxib – a selective cyclooxygenase-2 inhibitor – or placebo. 124 Patients in the exposure group experienced significant benefit in mean nocturnal voiding frequency, whereas no such effect was observed among controls [5.17−2.5 with celecoxib and 5.30−5.12 with placebo (p < 0.0001, p = 0.980, respectively)]. Four (10%) patients in the experimental group experienced mild gastric discomfort and no serious adverse events were reported in either group.
More recently, Lee et al. published data from a 14-day multicenter phase II double-blind randomized placebo-controlled 4-arm parallel study of a novel immediate/sustained-release formulation of combination acetaminophen-ibuprofen pharmacotherapy [ClinicalTrials.gov identifier: NCT02646826]. 125 A total of 86 patients with ⩾2.5 nightly voids and overactive bladder symptoms were assigned one of three dose levels or placebo. The active medication conferred significant benefit in reducing average nocturnal voiding frequency compared with placebo at all dose levels [−1.1 with low-dose, −1.4 with mid-dose, and −1.3 with high-dose compared with −0.3 with placebo (p = 0.002, p = 0.003, p = 0.001, respectively)]. There were no adverse events deemed treatment-related or severe in any group.
Although both aforementioned studies were limited by the absence of voiding diary data to quantify changes in nocturnal urine production, extended-duration diaries were obtained through a 2-week double-blind randomized placebo-controlled crossover study of diclofenac. 126 Addla et al. reported that diclofenac 50 mg tablets taken nightly by 26 adult patients with nocturia (⩾2 nightly voids) due to nocturnal polyuria conferred significant benefit compared with placebo, with nocturia frequency improving from 2.7 to 2.3 voids (p < 0.004) and nocturnal polyuria index improving from 0.44 to 0.39 (p < 0.001), as well as borderline significance observed for nocturnal urine volume (802–745 ml, p = 0.05). 126 Notably, the significant shift in 24-h urine production toward the daytime period (i.e., decreased nocturnal polyuria index) is consistent with well-controlled physiologic evidence on the effect of diclofenac on renal function, characterized by a rapid and persistent reduction in free water clearance owing to a 35% decrease in renal plasma flow and glomerular filtration rate with a nadir at 2 h and evidence of recovery starting at 3 h. 127 Several institutional studies have also demonstrated efficacy in treating nocturia with loxoprofen, a non-selective cyclooxygenase inhibitor marketed in some countries,128–132 with evidence supporting reductions in nocturnal urine volume as the primary mediator of nocturia treatment response.128,130,131
Hormone replacement therapy
During and after the transition to menopause, nocturia may arise in the context of the genitourinary syndrome of menopause (GSM), 133 which is a broad symptom complex encompassing multiple lower urinary tract symptoms and genital/sexual signs and symptoms associated with a decrease in sex hormones. 134 A wide range of pharmacologic treatment options are available for GSM, including multiple formulations of systemic and vaginal estrogen products, as well as newer non-estrogen medications such as the selective estrogen receptor modulator ospemifene. 94 Evidence derived from patients with OABS and secondary analyses of pivotal trials on the systemic effects of estrogen replacement therapy identified a significant therapeutic benefit of hormone replacement therapy in improving nocturia severity.135,136 Retrospective data from postmenopausal women with symptoms consistent with OABS suggest that ospemifene may also be effective for treating nocturia.137,138
Although the direct role of GSM pharmacotherapy in the management of nocturia has classically received relatively less attention, nocturia has recently been recognized not only as one potential facet of the GSM, but rather as one of the most pervasive complaints in this population. 133 The direct relevance of nocturia in patients with GSM is also underscored by a recent prospective observational study of systemic hormone replacement therapy from Pauwaert and colleagues, which was highly novel in that it considered nocturia the primary outcome variable, and found oral estradiol-progesterone combination therapy to indeed confer significant benefit for nocturia. 139 A prospective observational cohort study led by Pauwaert et al. to assess the effect of 10 different hormone replacement therapies on nocturia is also in the active recruitment phase [ClinicalTrials.gov identifier: NCT04433897].
Nocturnal polyuria is thought to be the most common cause of nocturia in patients of all ages and backgrounds, but its prevalence further increases with age, such that a disproportionate burden of nocturnal polyuria would be expected in postmenopausal women compared with younger nocturia cohorts. 14 Consistently, exploratory data have identified decreased urine output as a potential central mechanism in nocturia treatment response following hormone replacement therapy in postmenopausal women. 140 Well-controlled physiologic evidence demonstrates that administration of estradiol is associated with increased plasma AVP concentration, volume expansion, and a lower osmotic threshold for AVP release, as well as enhanced renal sodium reabsorption despite a stable plasma aldosterone concentration, suggesting that synthetic estrogens may also directly influence electrolyte handling at the level of the kidney. 65 Taken together, current evidence suggests that estrogens as well as progesterones play an important role in the central regulation of whole-body fluid balance and sodium regulation. 141 For an excellent review on the hormone changes and fluid regulation during menopause, see Stachenfeld. 141
Emerging technologies: individualizing nocturnal polyuria diagnosis and treatment
In recent years, the nocturia research community has grown increasingly aware of the extensive breadth of pathophysiologic mechanisms that may underlie nocturia. 102 Parallel to the diversity in nocturia pathophysiology, nocturia patients are similarly heterogenous with respect to the composition of urine produced during the hours of sleep. Compared with nocturia patients without nocturnal polyuria, the volume of urine produced during the night by those with nocturnal polyuria is not only greater in volume, but also significantly more dilute during the early hours of sleep. 74 Consistently, our own analysis of the volume and osmolality of a single urine sample from the early hours of the night revealed that these parameters combined can predict a diagnosis of nocturnal polyuria with a reasonably high degree of sensitivity and specificity. 142
While the volume of a single void can be measured easily, urine osmolality – the gold standard parameter for quantifying urine concentration – was historically measured using a freezing point depression technique, which is time-intensive and not widely available in the routine diagnostic laboratory setting. 143 However, urine osmolality was recently demonstrated to be reliably estimated from an assessment of standard urinalysis parameters (e.g., creatinine, glucose, and specific gravity) in conjunction with urine conductivity – a dilution parameter which is relatively easy to obtain using meters that can now be integrated into standard urinalysis equipment.143–145 Accordingly, current technology now makes feasible the analysis of single-void nocturnal urine samples in a general diagnostic laboratory setting to obtain clinically meaningful data for diagnosing nocturnal polyuria. This potential diagnostic alternative is particularly noteworthy in view of growing investigative interest in adjunctive strategies to voiding diary analysis in order to facilitate a more patient-centered approach to nocturia evaluation and management.146–151
Even among patients who share a common diagnosis nocturnal polyuria, the composition of their nocturnal urine is highly variable,117,152,153 such that most nocturnal polyuria patients can be further phenotyped as having (a) excess free water diuresis, (b) excess solute diuresis, or (c) mixed water/solute diuresis74,154 (Figure 1). Phenotyping patients by the composition of their excess urine volume – the relevant substrate for pharmacologic interventions directed at nocturnal polyuria – carries the potential to directly facilitate individualized treatment. 155 For example, in view of the mechanism of antidiuretic replacement therapy, patients with nocturnal polyuria owing to water diuresis or a mixed phenotype would be expected to demonstrate the most robust response to treatment with desmopressin. 156 Conversely, normalization of circadian homeostasis with appropriately timed diuretic/natriuretic would likely be of particular benefit in patients with a solute-predominant phenotype. 103 Urine composition-based phenotyping may thus also be useful in guiding future research on antihypertensives, NSAIDs, and sex hormone replacement therapy in the treatment of nocturnal polyuria.
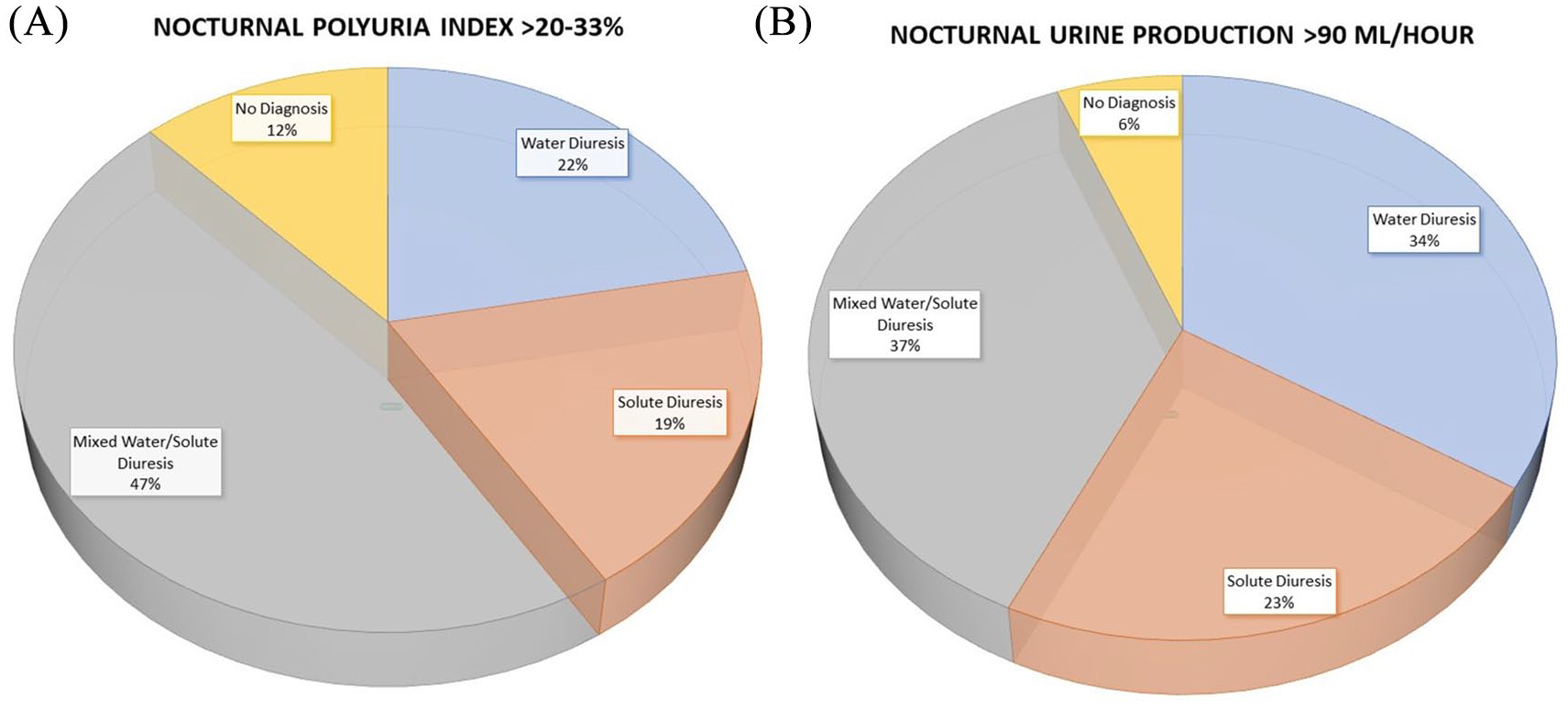
Observed distribution of nocturnal polyuria phenotypes following different definitions for nocturnal polyuria. 74 (A) Nocturnal polyuria defined as nocturnal polyuria index (nocturnal urine volume/24-h total urine volume) >0.20-0.33. (B) Nocturnal polyuria defined as nocturnal urine production >90 ml/h. Modified with permission from Goessaert et al. 74
Beyond substrate-guided nocturnal polyuria phenotypes, contemporary advancements in our understanding of the pathogenesis of nocturnal polyuria, and AVP dysregulation in particular, have introduced several novel opportunities for point-of-care testing in patients with nocturnal polyuria. Urinary AVP from early-morning urine samples has been recognized as a potential biomarker for nocturnal polyuria, and, more specifically, impaired circadian rhythmicity in endogenous AVP activity.157–159 Additionally, pre-treatment plasma copeptin levels have been found to be predictive of desmopressin response in younger patients with nocturnal polyuria, 160 and this concept is also the subject of a large ongoing prospective observational study currently in the recruitment phase [ClinicalTrials.gov identifier: NCT02621736]. Measurement of arginine-stimulated plasma copeptin has been identified as an effective and relatively safe alternative to the water deprivation test in the diagnosis of the polyuria-polydipsia syndrome,161,162 and it stands to reason that this technology may also be relevant in identifying nocturnal polyuria owing to blunted nocturnal endogenous AVP activity. 163
Future directions
In general, evidence supporting the efficacy and safety of non-antidiuretic therapies for nocturnal polyuria remains limited, and combination pharmacotherapies targeting excess nocturnal urine production have likewise not been well studied. Further research is also needed to better characterize the patients most likely to respond to antidiuretic replacement therapy and other pharmacologic modalities targeting excess nocturnal urine production. While this narrative review highlights mechanisms underlying the effects of several medications on nocturnal urine production in particular, nocturia treatment response with medications other than diuretics has not been completely characterized, and these agents may also alter other factors relevant to nocturia such as bladder contractile function and sleep architecture.164–170 The utility of urinary phenotypes, as well as copeptin assays and other potential modalities for point-of-care testing, requires evaluation in larger study samples of real-world patients with nocturnal polyuria.
Footnotes
Authors’ contributions
Conceptualization, T.F.M. and A.J.W.; Investigation, T.F.M.; Writing – Original Draft, T.F.M.; Writing - Review & Editing, T.F.M., J.P.W., K.E. and A.J.W.
Conflict of interest statement
Thomas F. Monaghan has no direct or indirect commercial incentive associated with publishing this article and certifies that all conflicts of interest relevant to the subject matter discussed in the manuscript are the following: Jeffrey P. Weiss is a consultant for Ferring and the Institute for Bladder and Prostate Research, outside the submitted work. Karel Everaert is a consultant and lecturer for Medtronic and Ferring and reports institutional grants from Allergan, Ferring, Astellas, and Medtronic, outside the submitted work. Alan J. Wein has served as an advisor/consultant for Bulkamid, Medtronic, Serenity, Urovant, and Velicept, outside the submitted work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
