Abstract
Background:
Treatment of urethral trauma is currently done after urethral stricture occurs. Stricture therapy after occurrence gives unsatisfactory success rates. Several genes, such as transforming growth factor beta 1 (TGF-β1), matrix metalloproteinase 1 (MMP-1), connective tissue growth factor (CTGF), and plasminogen activator inhibitor 1 (PAI-1), have a proven role in urethral stricture development. The purpose of this study was to assess the effect docetaxel and/or captopril on the RNA expression of those genes.
Methods:
The subjects of this research were 26 male New Zealand rabbits aged 230 ± 20 days weighing 4–5 kg that underwent urethral rupture by endoscopic resection under anesthetized conditions. Subjects were divided into five groups; control, stricture, captopril (captopril 0.05 mg/rabbit/day), docetaxel (docetaxel 0.1 mg/rabbit/day), and docetaxel-captopril (docetaxel 0.1 mg/rabbit/day and captopril 0.05 mg/rabbit/day). Each group consisted of 4–6 rabbits. Each rabbit received a water-soluble transurethral gel containing drug according to its group for 28 days. After the treatment period, rabbits were sacrificed with 200 mg Pentothal, and the corpus spongiosum was then prepared for real-time PCR examination.
Results:
TGF-β1 RNA expression in the stricture group was statistically different from that in the control, docetaxel and docetaxel-captopril groups (
PAI-1 RNA expression in the stricture group differed statistically significantly from the control and docetaxel groups (
Conclusion:
Docetaxel-captopril gel proved to be able to inhibit RNA expression of TGF-β1 and CTGF significantly. Captopril gel proved to be able to inhibit RNA expression of CTGF significantly. Docetaxel gel proved to be able to inhibit RNA expression of TGF-β1, CTGF, and PAI-1 significantly. There were no differences in MMP-1 expression among all study groups. Longer follow up after therapy discontinuation and greater sample size is needed to determine the therapeutic effect.
Introduction
Urethral stricture is a urological condition that occurs mainly in men, with an incidence rate of around 0.3%, and occurs at various ages. Urethral stricture, which greatly affects quality of life and consumes a lot of medical expenses,1,2 is characterized by urethral narrowing due to formation of scar tissue in the corpus spongiosum. Pars bulbaris urethral stricture is the most common type of stricture in developed countries (46.9%).3–6 This contraction of scar tissue causes the urethral caliber to shrink. Signs and symptoms of urethral stricture include weak urine stream, decreased volume of urination, urge urgency, pain during urination, incontinence, pelvic or suprapubic region pain, urethral discharge, swelling of the penis, presence of blood in semen or urine, and darker color of urine.1,4 The etiology of urethral stricture includes trauma, iatrogenic, balanitis xerotica obliterans, hypospadias, radiation, urethral infections, meatal ischemia, and inflammation. Urethral stricture can cause urethritis, bladder stones, fistulas, sepsis, and renal failure.2,3,5,7
Management of urethral stricture is a challenging problem in urology. Stricture characteristics determine urethral stricture management with the aim of creating an adequate urethral caliber. The three main techniques of urethral stricture therapy are urethral dilatation, visual internal urethrotomy, and urethroplasty.3,8 The gold standard technique for handling urethral stricture is urethroplasty because it has the best cure rate compared with other techniques, but this technique requires certain expertise. Stricture urethra recurrence remains high, with the majority within the first 6–12 months. Because of this low success rate, several accompanying techniques have been used to treat wound contractions and to prevent recurrence of strictures, including by installing a Foley catheter, intermittent self-catheterization at home, and urethral stents. This method has several complications and recurrence often occurs unless it is always done for life.2,3,8
The main etiopathology of urethral stricture is fibrosis, scarring of the urethral epithelium, and corpus spongiosum. Fibrosis is a complex multistage process that involves a series of pathophysiological mechanisms. This process is regulated by the release of dissolved mediators such as transforming growth factor beta (TGF-β), angiotensin II, and leukocyte infiltration.3,9,10
TGF-β1 binds to the TGF-β receptor and activates the TGF-β/Smad pathway in fibroblasts. Smad protein translocates to the nucleus and regulates transcription of extracellular matrix (ECM) proteins and alpha smooth muscle actin (α-SMA), which is an important component of myofibroblast contractile microfilament. Myofibroblasts will deposit more ECM molecules, are more contractile than before, and show an intermediate phenotype between fibroblasts and smooth muscle cells. Among various pro- and anti-fibrotic cytokines, the TGF-β isoform appears to play a key role in the development of fibrosis. Three TGF-β isoforms have been identified in mammals: TGF-β1, -β2, and -β3. The effects of various TGF-β isoforms are specific target cells and are context dependent. TGF-β1 is fibrogenic but TGF-β3 has anti-fibrotic properties. TGF-β1 plays an important role in inflammation and fibrosis, and induces hypertrophic changes in cellular function and tissue morphology.11–13
Knowledge of the pathophysiology of organ fibrosis has led to research on several antifibrotic drugs to treat urethral stricture (Figure 1). Docetaxel inhibits the formation of urethral stricture by inhibiting the formation of fibrosis and collagen coagulation. Captopril inhibits fibrosis by inhibiting angiotensin converting enzyme, and is one of the drugs used to treat fibrosis in other organs.3,14,15 Docetaxel and captopril have different working mechanisms. Docetaxel works by inhibiting intracytoplasmic Smads protein. Captopril works by inhibiting angiotensin receptors in the cell membrane. Inhibition through two different pathways is expected to improve the effectiveness and efficiency of therapy. This study aims to show that docetaxel and/or captopril can prevent the urethral stricture development caused by urethral anterior rupture, by using TGF-β1, matrix metalloproteinase 1 (MMP-1), connective tissue growth factor (CTGF), and plasminogen activator inhibitor (PAI-1) RNA expressions as markers. The inhibition of collagen synthesis is expected to maintain urethral elasticity and the urethral lumen diameter.
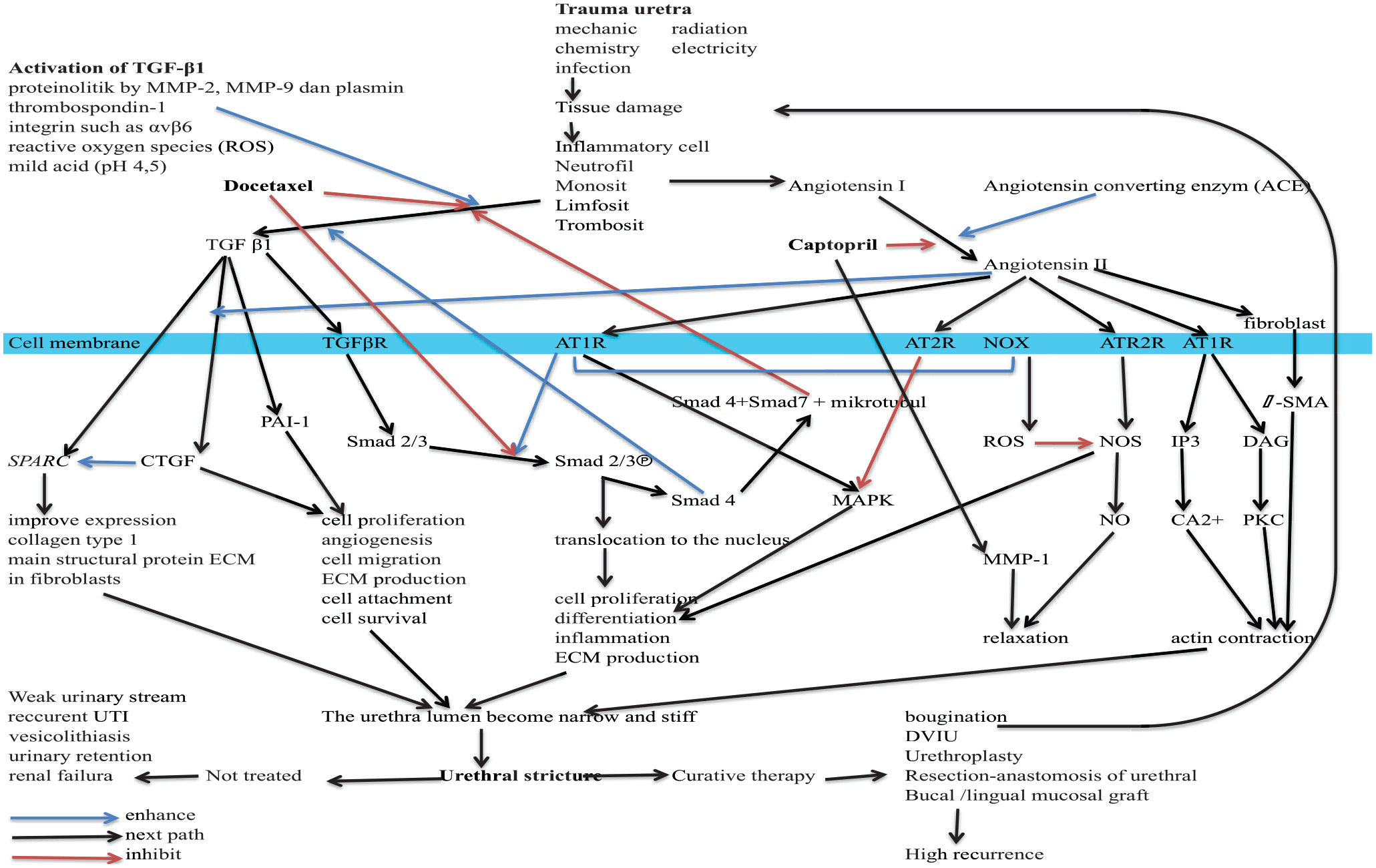
Inhibition mechanism of captopril and docetaxel in mechanism of TGF-β1, MMP-1, CTGF and PAI-1 of urethral fibrosis.
Materials and methods
The subjects of this research were 26 male New Zealand rabbits aged 230 ± 20 days weighing 4–5 kg. Materials needed to make anterior urethral rupture are a set of pediatric resectoscopes, camera sets and endoscope monitors, an electrosurgical unit (ESU), pediatric irrigator sets, infusion poles, feather clippers, 1 and 5 cc syringes, a set of rabbit restraints, stethoscope, preparation table, and applicator with 1 cm barrier from the tip.
Captopril (ab141333) and docetaxel (ab141248) were prepared by Abcam (Cambridge, UK). The intraurethral gel formulation was prepared in Faculty of Veterinary Medicine, Universitas Gadjah Mada (UGM, Yogyakarta, Indonesia). Research was carried out at the Animal House, Department Pharmacology and Therapy; RNA extraction and cDNA making in the Department of Anatomy; and gene expression processing in “Laboratorium Riset Terpadu”, Radioputro Building, Faculty of Medicine, Public Health and Nursing, UGM.
At 1 h before the operation, 300 mg ceftriaxon as prophylactic antibiotics was given intramuscularly. A combination of ketamine (10 mg/kg) and xylazine (3 mg/kg body weight) was given intramuscularly and the rabbit rested for 5–10 min until the onset of anesthesia was reached, then the restraint was released. Hair on the genital region and a region on the dorsal side with a size of 15 × 15 cm was trimmed with a clipper to facilitate sterilization of the operating field, so that the (ESU) negative electrode attaches well. Urethral resection was carried out according to series of the cage number. Sterile water for irrigation was placed 70–80 cm higher than the operating table. Sets of rabbit restraints were placed on the operating table in the supine position. Water-soluble jelly (2 ml) was instilled into the urethra to lubricate the resectoscope.
The resectoscope was inserted into the urethra. Electrocoagulation uses loop-shaped electrodes with a power of 40 W along the 10 mm urethra, starting from the distal verumontanum in sterile conditions. Electrocoagulation was performed circumferentially until the urethra appeared blanching and the mucosa ulcerates.
14
Electrocoagulation was performed in bipolar coagulating mode by the same urologist (WK). After urethral resection was completed, subjects were placed into a cage to recover consciousness and were injected with ketorolac (1 mg/kg body weight). After all rabbits recovered consciousness, randomization was carried out. The research subjects were put into cages according to their respective groups and each cage has prepared
Treatment started on day 1. The control group and stricture group received water-soluble intraurethral gel 0.6 ml/rabbit/day. Captopril group received captopril 0.05 mg/rabbit/day in 0.6 ml water-soluble intraurethral gel. Docetaxel group received docetaxel 0.1 mg/rabbit/day in 0.6 ml water-soluble intraurethral gel. 14 Docetaxel-captopril group received docetaxel 0.1 mg/rabbit/day and captopril 0.05 mg/rabbit/day in 0.6 ml water-soluble intraurethral gel. Instillation uses the urethral sonde, the tip of the sonde 1 cm deep from the external urethral orifice. Drug administrations were carried out in a retrograde transurethral manner every day from the day 1 to day 28 since urethral rupture. 14 The research subjects were euthanized with pentobarbital 200 mg. 16 The urethra was stored in RNA letter solution and placed in a freezer at –20°C. 17
Real-time PCR used specific primer sequences for rabbit TGF-β1, as follows: forward: 5′-AAGGGCTACCACGCCAACTT-3′, reverse: 5′-CGGGTTGTGCTGGTTGTAC-3′. 18 Forward rabbit MMP-1 primer sequence: 5′-TCAGTTCGTCCTCACTCCAG-3′ reverse: 5′-TTGGTCCACC TGTCATCTTC-3′, with a target of 322 bp. 19 Specific primer sequence for CTGF forward: 5′-CGAGGAGTGGGTGTGTGATG-3′, reverse: 5′-GTGTCTTCCAGTCGGTAGGC-3′ with product length of 78 bp. 20 Sequences primers specific to PAI-1 forward: 5′-TTGTTCCGAACCACGGTCAA-3′, reverse: 5′-TCGCTGATCATGCCTTTCGT-3′ with product length 104 bp. 21 Sequence primers specific to GAPDH forward: 5′-GTATGATTCCACCCACGGCA-3′, reverse: CCAGCATCACCCCACTTGAT-3′ with product length 129 bp. 22 TGF-β1, CTGF, PAI-1 and MMP-1 RNA primers were obtained from Macrogen Humanizing Genomics/PT. Indolab Utama (Jakarta Barat, Indonesia).
Statistical analyses
The normality of data distribution was assessed using the Shapiro Wilk statistical test. If the data is normally distributed then it will proceed with the one-way ANOVA test, and if there were significant differences among groups and LSD test were done to trace which pair gave the significant result. If the data is not normally distributed then it will proceed with Kruskal-Wallis Test, and if there were significant differences among groups and Mann-Whitney Test were done to trace which pair gave the significant result.
Results
TGF-β1
Table 1 shows that data for TGF-β1 were not distributed according to the normal curve, so we used a nonparametric statistical analysis, namely the Kruskal–Wallis test, to determine significance among groups (Table 2). There were significant differences among groups and the Mann–Whitney
Real-time PCR of TGF-β1.

CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; PCR, polymerase chain reaction; STR, stricture group; TGF-β1, transforming growth factor beta 1.
Kruskal–Wallis test for TGF-β1.

Kruskal–Wallis test.
Group.
GAPDH, glyceraldehyde dehydrogenase; TGF-β1, transforming growth factor beta 1.
TGF-β1 RNA expression levels in the stricture group were statistically higher than in the control, docetaxel and docetaxel-captopril groups (
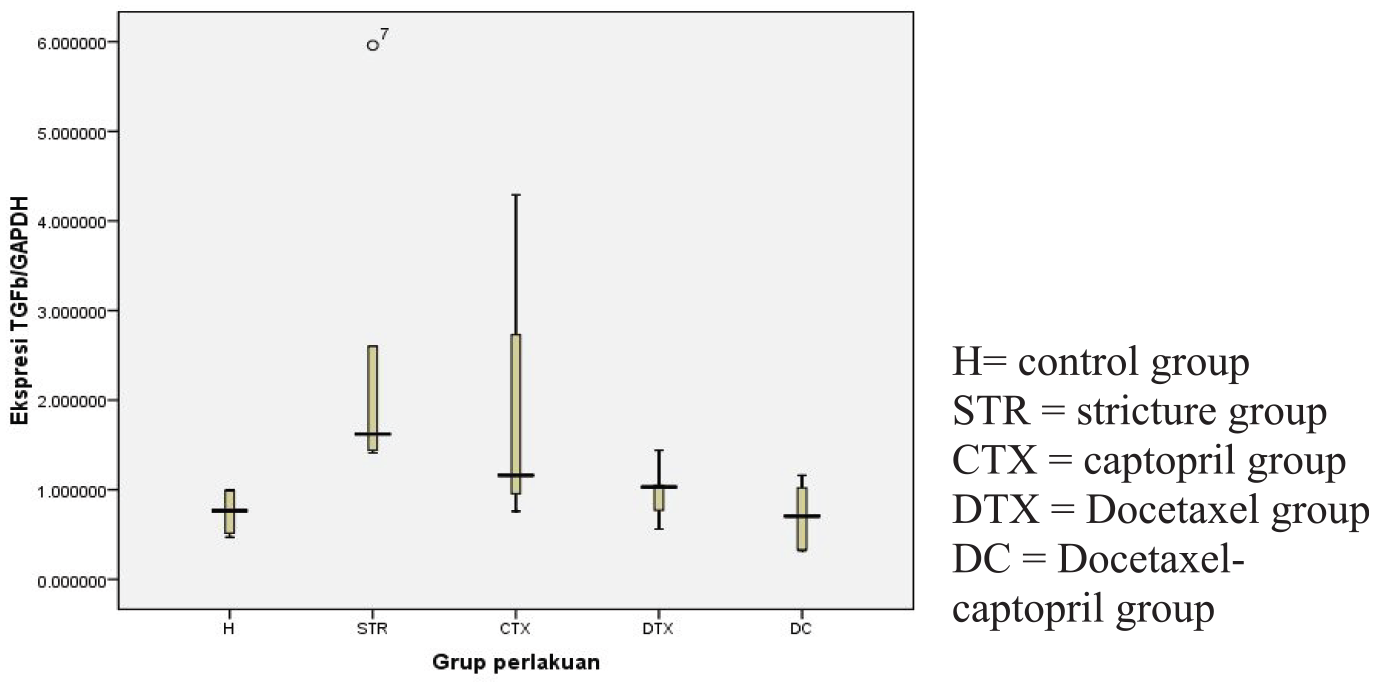
TGF-β1 expression.
Mann–Whitney test for TGF β1.
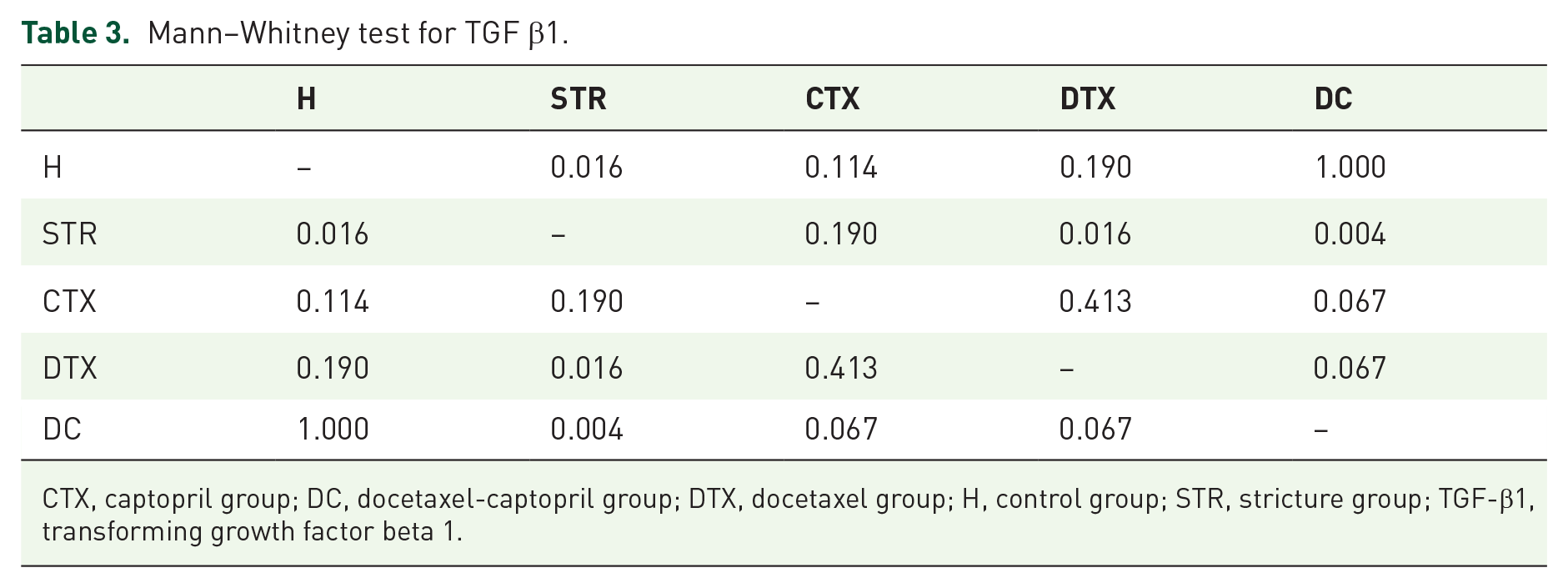
CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; STR, stricture group; TGF-β1, transforming growth factor beta 1.
MMP-1
Table 4 shows that the data for MMP-1 were not distributed according to the normal curve, so we used a nonparametric statistical analysis, namely the Kruskal–Wallis test, to determine significance among groups. MMP-1 RNA expression (Figure 3) did not differ significantly between all groups (
Real-time PCR of MMP-1.
CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; MMP-1, matrix metalloproteinase-1; PCR, polymerase chain reaction; STR, stricture group.

MMP-1 expression.
Kruskal–Wallis test for MMP-1.
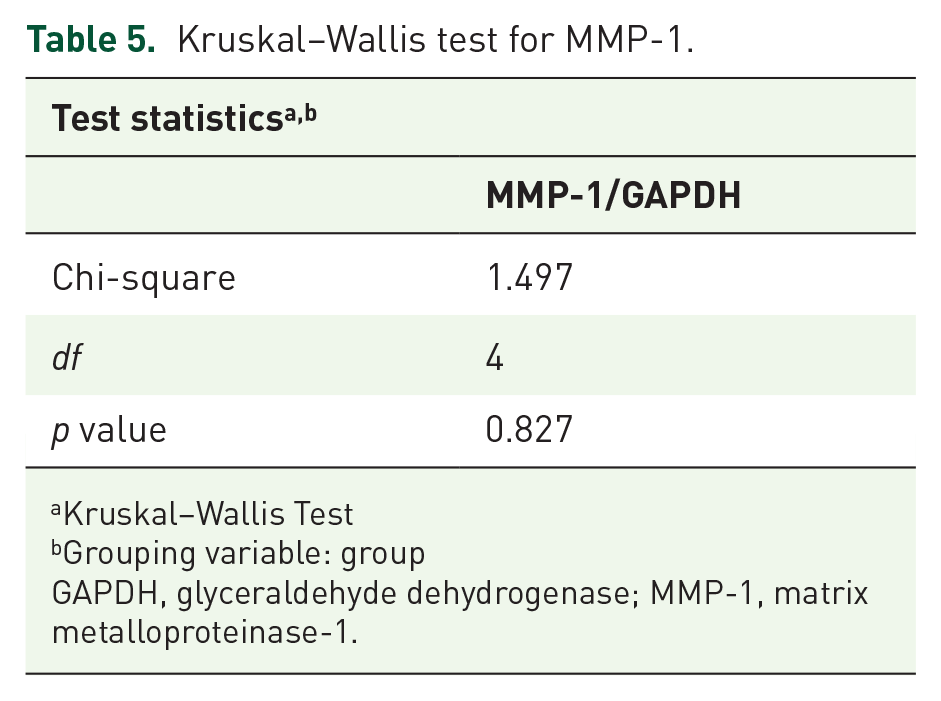
Kruskal–Wallis Test
Grouping variable: group
GAPDH, glyceraldehyde dehydrogenase; MMP-1, matrix metalloproteinase-1.
There were significant differences among groups and least significant difference (LSD) tests were done to trace which pair gave significant results.
CTGF
Results for CTGF expression are shown in Table 6 and Figure 4. All groups were normally distributed, and parametric analysis and one-way analysis of variance (ANOVA; Table 7) were used to determine the significance among groups. Given that there were significant differences among groups, LSD analyses (Table 8) were performed to trace which pair gave significant results. CTGF RNA expression in the stricture group was statistically different from the control, captopril, docetaxel, and docetaxel-captopril groups (
Real-time PCR of CTGF.

CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; CTGF, connective tissue growth factor; PCR, polymerase chain reaction; STR, stricture group.
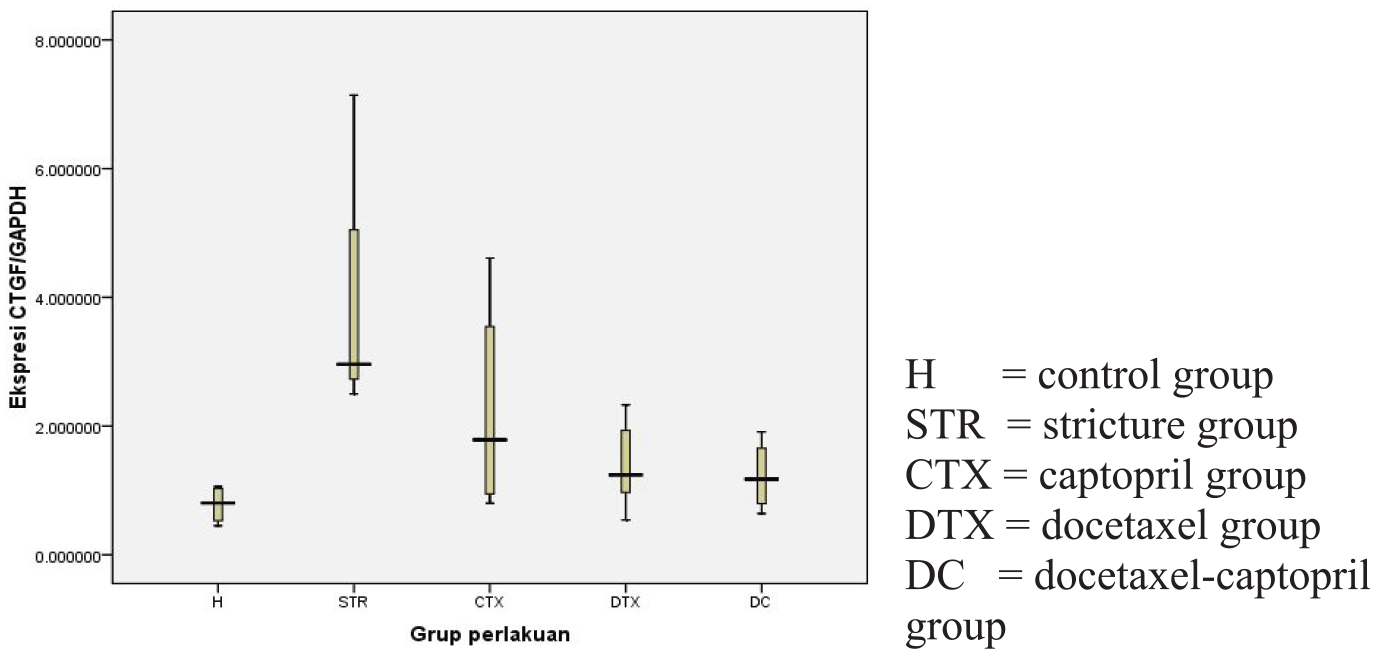
CTGF expression.
One-way ANOVA test for CTGF.
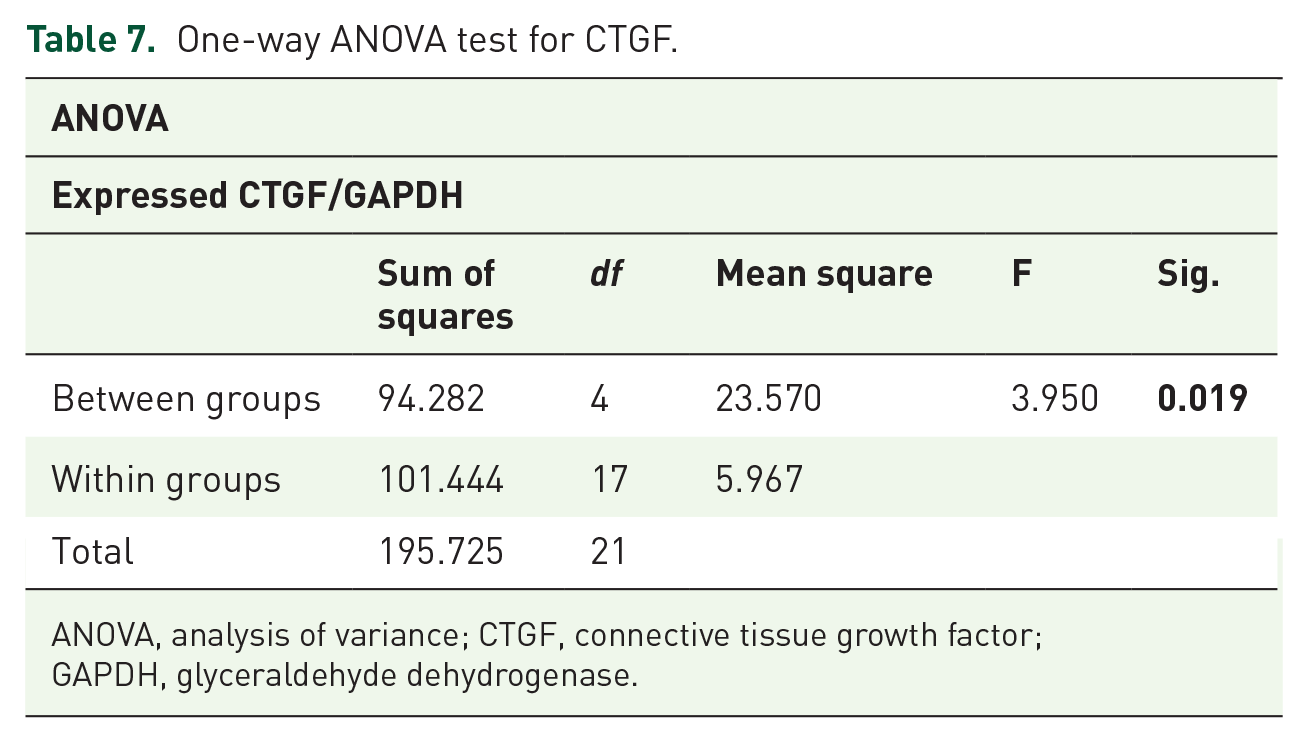
ANOVA, analysis of variance; CTGF, connective tissue growth factor; GAPDH, glyceraldehyde dehydrogenase.
LSD test for CTGF.

CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; CTGF, connective tissue growth factor; LSD, least squares difference; STR, stricture group.
PAI-1
Results for CTGF expression are shown in Table 9 and Figure 5. ANOVA (Table 10) was used to determine the significance among groups. PAI-1 RNA expression in the stricture group were statistically significantly different from the control and docetaxel groups (Table 11;
Real-time PCR of PAI-1.
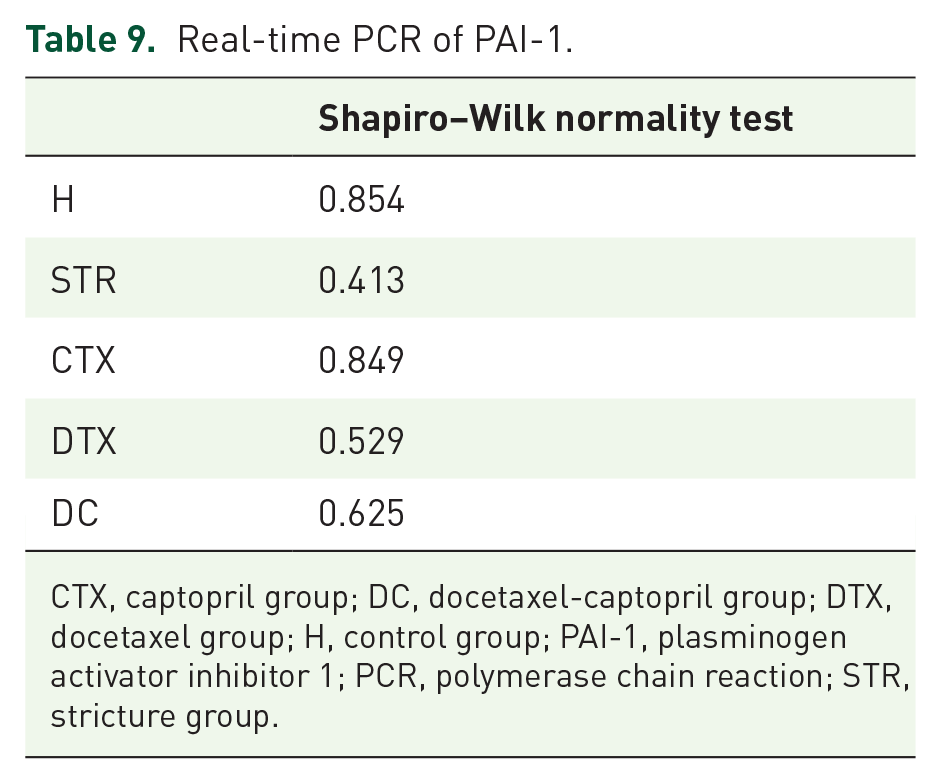
CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; PAI-1, plasminogen activator inhibitor 1; PCR, polymerase chain reaction; STR, stricture group.
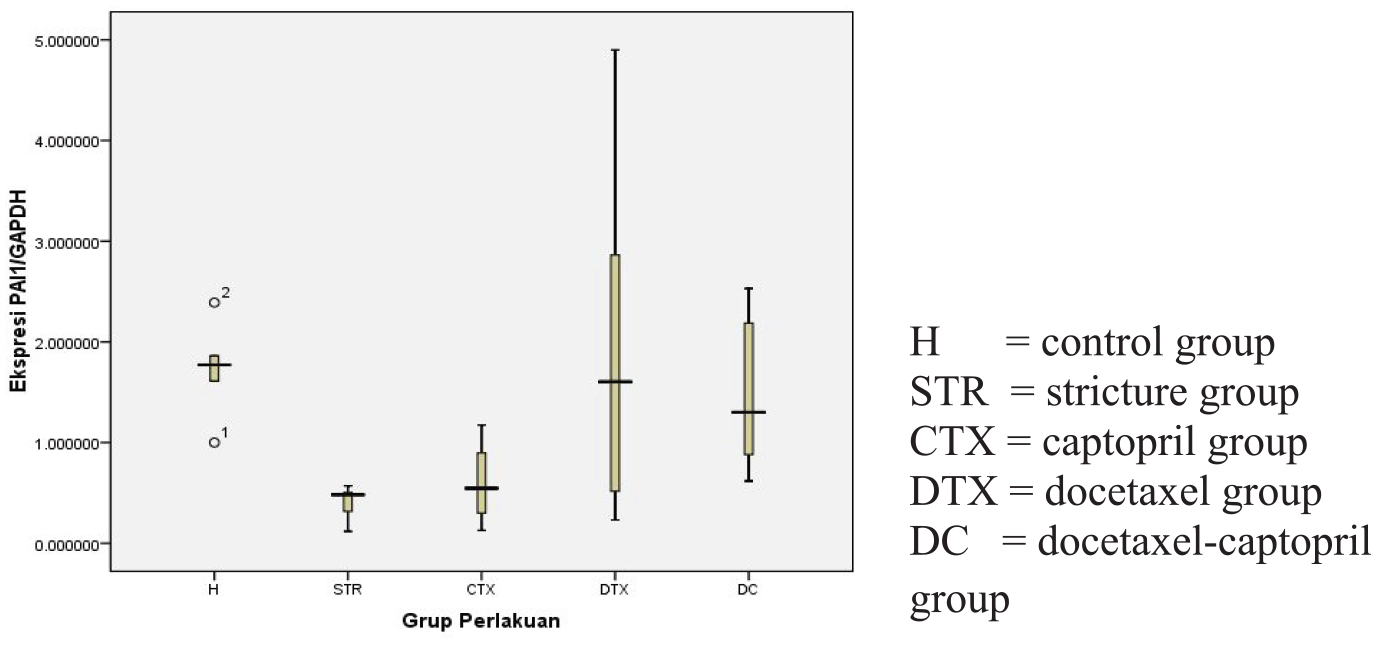
PAI-1 expression.
One-way ANOVA test for PAI-1.
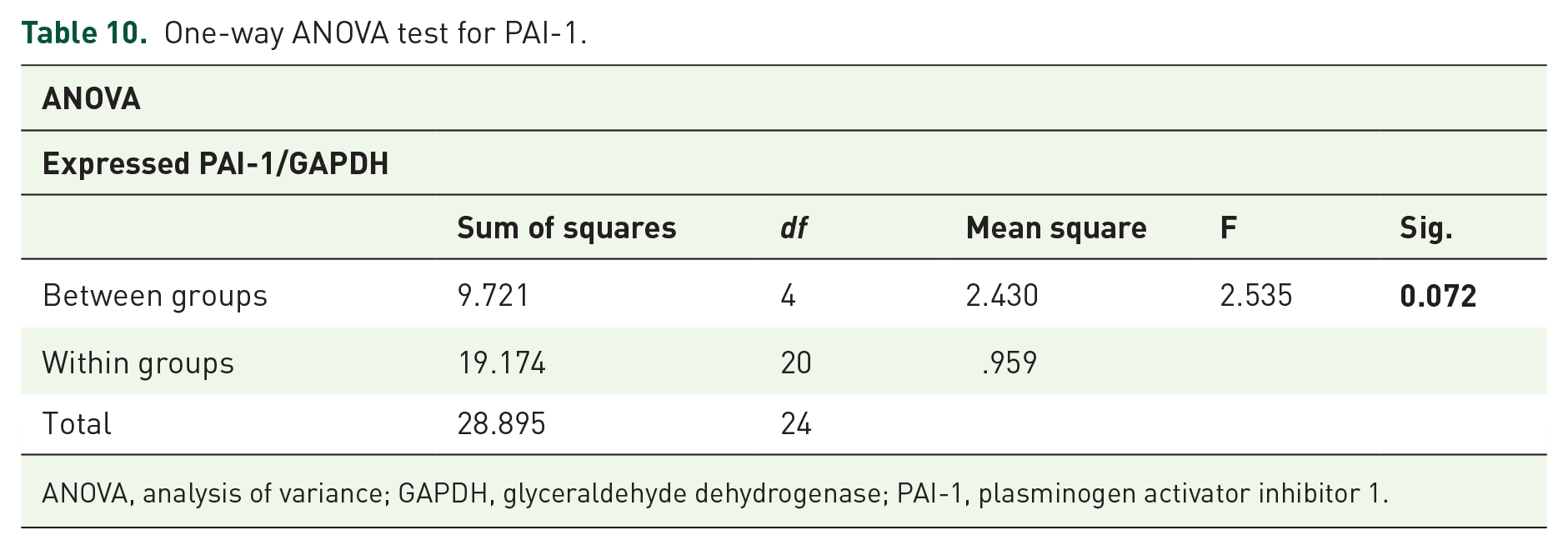
ANOVA, analysis of variance; GAPDH, glyceraldehyde dehydrogenase; PAI-1, plasminogen activator inhibitor 1.
LSD test for PAI-1.
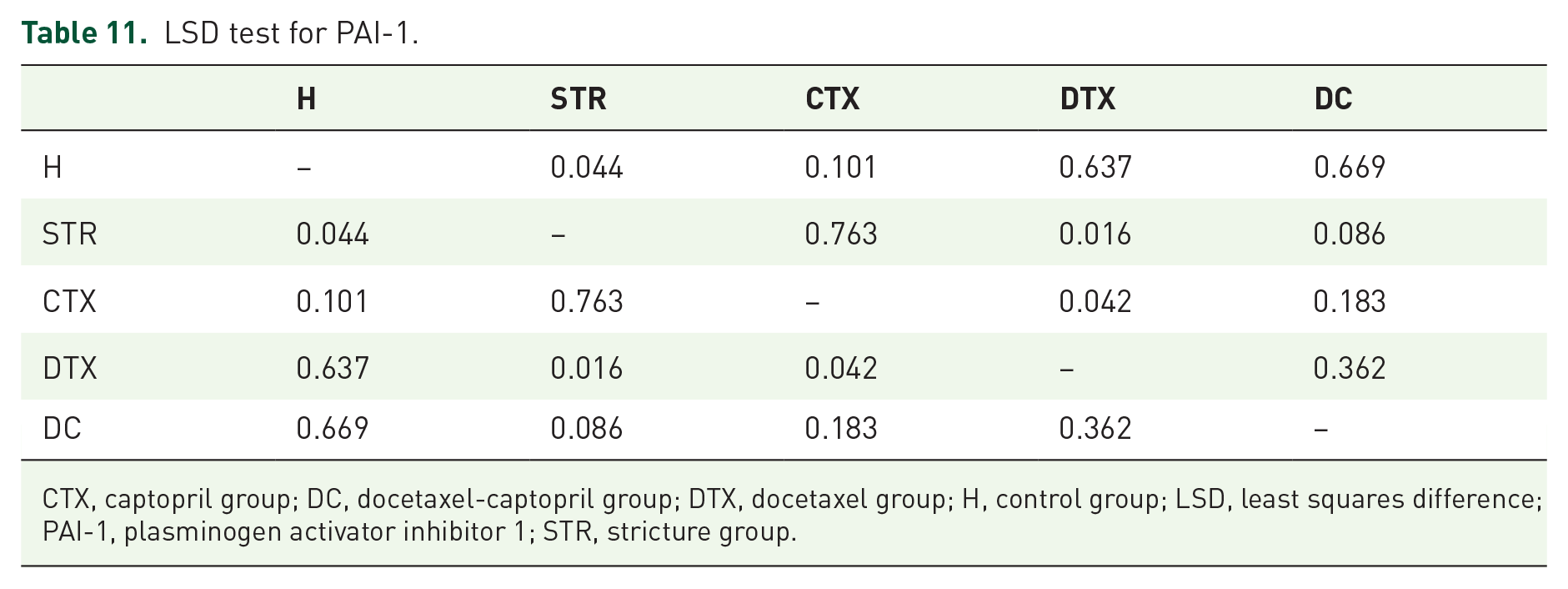
CTX, captopril group; DC, docetaxel-captopril group; DTX, docetaxel group; H, control group; LSD, least squares difference; PAI-1, plasminogen activator inhibitor 1; STR, stricture group.
Discussion
In this study, docetaxel gel and/or captopril intraurethra was used as prophylaxis of anterior urethral stricture in ruptured urethra. Fibrosis is the main pathophysiology of urethral stricture, so, by inhibiting the process of fibrosis, it is expected that urethral stricture can be avoided. These agents have different mechanisms of action in inhibiting the process of fibrosis. Expression of TGF-β1, MMP-1, CTGF, and PAI-1 RNA, as assessed with real-time PCR, was used to assess the fibrosis process that occurred. Expression of these genes was used because it is known that these genes play a central role in the mechanism of fibrosis.
TGF-β1 is a pleiotropic cytokine expressed by almost all human cells. TGF-β1 is synthesized and secreted as an inactive molecule, namely latent TGF-β1. Latent TGF-β1 consists of TGF-β1, which is covalently related to latency related peptide (LAP). Latent TGF-β1 is activated by heat, acid treatment, proteolytic division, and integrin. 23
Transforming growth factor β1 (TGF-β1) is one of the cytokines that is very instrumental in healing wounds and scar tissue in mammals. 24 TGF-β1 serves as an important stimulator for the differentiation of myofibroblasts, with α-SMA as a marker, and increases the production of ECM. 25 TGF-β1 and -β2 play a role in inducing scarring, whereas TGF-β3 inhibits the development of scar tissue. Trans type I (RI) and type II (RII) membrane receptors mediate TGF-β to elicit biological responses. 24
TGF-β induction will increase extracellular matrix production. The TGF-β signal transduction pathway for growth regulation and extracellular matrix production can deviate after TGF-β binds to TβRII and TβRI. Downstream components, such as Smad protein, guanine-nucleotide binding protein (G protein) and mitogen-activated kinase protein (MAPK) affect the TGF-β response that interacts with TβRI. TGF-β response varies depending on cell type and culture conditions. 26
TGF-β1 receptors are on the cell surface. Signaling is initiated by binding to TGF-β, and stabilizing the complex of two type II (TβRII) and two type I (TβRI) receptors. TβRI receptors are activated by TβRII by phosphorylation. These activated TβRI receptors then phosphorylate SMAD2 and SMAD3, which sequentially form trimers with SMAD4. This complex goes to the nucleus to regulate transcription of target genes (e.g. PAI-1) by binding to the co-activator/suppressor and subsequent interaction with the SMAD binding element in the target gene promoter. 23
In this study, we see docetaxel and docetaxel-captopril significantly suppress TGF-β1 RNA expression. This shows that TGF-β1, as the main pro-fibrotic cytokine, can be suppressed significantly by using transurethral docetaxel and docetaxel-captopril. The suppression of TGF-β1 levels is expected to also result in suppression of the development of fibrosis. Suppression of TGF-β1 expression can lead to suppression of sequential fibrosis cascades, in this case PAI-1. Suppression of TGF-β1 expression also results in decreased MMP expression. 27
MMPs are a family of proteolytic enzymes that include collagenase and gelatinase. MMPs are responsible for the degradation of ECM components under various normal and pathological conditions.28,29 MMPs family enzymes are structurally similar to ACE (angiotensin converting enzyme). ACE is a bivalent dipeptidyl carboxyl metalloproteinase. This enzyme comes from a large family of metallopeptidase evolution. ACE and MMP have similar structural homology and inhibitory mechanisms; hence, substances that interact with ACE might have inhibitory activity against MMP. Mammalian MMP inhibition by ACE inhibitors has been confirmed both
MMP is regulated at the level of transcription, secretion, and activation of proenzymes and activated enzymes. MMP mRNA transcription is regulated by growth factors, cytokines, hormones, oncogenes and tumor promoters.
30
MMPs activation can cause breakdown of collagen type II matrix components and aggrecan.
31
MMP-1 expression is increased during physiological and pathological tissue remodeling
In this study, MMP is inhibited through two pathways at once, namely indirect inhibition
CTGF is one of the factors downstream of TGF-β1 in the fibrosis process. TGF-β1 profibrotic activity is largely continued through CTGF activity. 32 Excessive CTGF expression will result in severe fibrosis in various parts of the body. CTGF plays a role in the transformation of fibroblasts into scar-forming myofibroblasts. 33 Suppressing TGF-β1 expression will also suppress CTGF expression. This is expected to occur in the preventive therapy of urethral stricture.
Collagen production is regulated by two ECM proteins, namely SPARC and CTGF. Regulatory mechanisms through SPARC and/or CTGF directly carry out negative feedback transduction of TGF-β signaling, and by binding to TGF-β or TGF-β receptors themselves. SPARC siRNA can reduce the production of type I and III ECM collagen. TGF-β-induced collagen production and/or fibronectin expression can be blocked by blocking CTGF by CTGF siRNA, or antisense oligonucleotide or an appropriate antibody. 32
In this research, treatments give significant decreasing in CTGF RNA expressions in all (captopril, docetaxel and docetaxel-captopril) groups treatment groups compared with the stricture group. This illustrates that administration of these agents topically effectively suppresses CTGF.
PAI-1 controls cell migration and ECM accumulation. PAI-1 is often expressed in conjunction with urokinase (uPA) or tissue plasminogen activator (tPA). In chronic diseases, at the point when uPA or tPA expression decreases, PAI-1 remains high. Therefore PAI is used as a histochemical marker for cell migration or active remodeling, and as an indicator for ECM accumulation in chronic diseases. This process requires the cyclic appearance of tPA or uPA, PAI-1 and uPAR on the cell surface, which regulates proteolysis and local adhesion, which, in turn, regulates cell movement. 34
In this study, PAI-1 levels in control and treatment groups were higher than those in the urethral stricture group. This needs further research; it is likely that the PAI-1 primers used were incorrect, although primer arrangement was obtained from the literature and the primer was prepared by a certified laboratory. This is possible because the results of the control group and stricture group show results that are contrary to theory. Another possibility is the existence of an unknown PAI-1 regulatory mechanism.
This study shows that captopril and/or docetaxel gel applied in a transurethral manner can be used as a preventive agent for urethral stricture with TGF-β1, MMP-1, CTGF, and PAI-1 as markers of fibrosis.
However, due to the small sample size of this study, it is necessary to conduct a large sample, multi-center study in the future.
Conclusion
Docetaxel and/or captopril are promising agents to avoid urethral stricture development in anterior urethral rupture. Captopril alone decreases CTGF expression. Docetaxel alone decreases TGF-β1, CTGF, and PAI-1 expression. The combination of docetaxel and captopril decreases TGF-β1 and CTGF expression. Longer follow up after therapy discontinuation and greater sample size are needed to determine the therapeutic effect. More research is needed to confirm this.
Footnotes
Acknowledgements
This research was carried out after obtaining approval from the Medical and Health Research Ethics Committee (MHREC) Faculty of Medicine Gadjah Mada University- Dr. Sardjito General Hospital Ref: KE/FK/1235/EC/2018 on 22 November 2018.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the LPDP (Lembaga Pengelola Dana Pendidikan/Indonesia Endowment Fund for Education) Ministry of Finance of the Republic of Indonesia
