Abstract
Urgency is the prevalent and most bothersome symptom of overactive bladder (OAB) and the treatment of urgency is the primary objective in the management of OAB. Urgency has a major impact on other symptoms of OAB and culminates in an increased frequency of micturition and reduced volume voided, which may contribute to shorter intervals between the need to void. Antimuscarinic agents and mirabegron, a β3-adrenoceptor agonist, constitute the main oral pharmacotherapeutic options for the treatment of urgency and other OAB symptoms. The reduction of urgency and other OAB symptoms significantly improve health-related quality of life. This review will explore the distinct mechanisms of action and effects of antimuscarinic agents and mirabegron, in relation to their effect on the pathophysiology of urgency. The review will also provide an overview of the various validated measurements of urgency and the numerous clinical trials regarding antimuscarinic agent monotherapy, mirabegron monotherapy, or combination treatment with mirabegron added on to the antimuscarinic agent solifenacin. A narrative review of the literature relating to pathophysiology of urgency, the validated measurements of urgency, and clinical trials relating to the pharmacological treatment of urgency. Antimuscarinic agent monotherapy, mirabegron monotherapy, or combination treatment with mirabegron added on to the antimuscarinic agent solifenacin statistically significantly reduce the symptoms of urgency compared with placebo. Combination therapy with mirabegron added on to solifenacin also statistically significantly reduces the symptoms of severe urgency compared with antimuscarinic agent monotherapy. A critique of the clinical benefits of combination therapy is also provided. Combination therapy provides an alternative treatment in patients with OAB that includes urgency who respond poorly to first-line monotherapy and who may otherwise often move on to more invasive treatments.
Introduction
Overactive bladder (OAB) is a prevalent disorder and is defined as urinary urgency, usually accompanied by daytime frequency and nocturia with or without urgency incontinence, in the absence of urinary tract infection or other obvious pathology.1,2,3 The International Continence Society (ICS) defines urgency as “the complaint of a sudden compelling desire to pass urine, which is difficult to defer” and urinary urgency incontinence as any involuntary leakage of urine preceded by urgency. 2 Urinary urgency is the pivotal and most bothersome OAB symptom1,4,5 and the treatment of urgency is the primary objective in the management of OAB. 1 The prevalence of urgency increases with advancing age. 6 Urgency has a significant negative impact on health-related quality of life (HRQoL) and the reduction of urgency symptoms significantly improves HRQoL in patients with OAB.7,8
This review will explore the distinct mechanisms of action and effects of antimuscarinic agents and the β3-adrenoceptor agonist, mirabegron, in relation to their effect on the pathophysiology of urgency, the key symptom of OAB.
Pathophysiology of urgency
Urgency has a major impact on other symptoms of OAB and culminates in an increased frequency of micturition and reduced volume voided, which may contribute to shorter intervals between the need to void (Figure 1). 9 While urgency impacts urgency incontinence, urinary leakage is controlled by a number of factors including urethral sphincter strength/weakness, the mobility of the individual, access to a toilet, and other environmental factors. 9 The reduction in the intervoidal interval caused by urgency may be instrumental in the development of nocturia, which occurs in 33% of patients with OAB. The cycle of urgency, urgency incontinence, and increased necessity to void leads to a worsening of symptoms and a continual cycle of OAB symptoms.

Impact of urgency on other overactive bladder symptoms. 9
The bladder wall consists of three main layers: the urothelium, the lamina propria, and the detrusor muscle (Figure 2), 10 all potentially involved in sensory signaling. 11 Bladder function is controlled by a series of reflexes during the filling and voiding phases (Figure 3). 12 Activation of afferent nerve fibers is involved in initiating urgency symptoms. 13 The afferent neurons of the bladder consist of myelinated Aδ and nonmyelinated C fibers which are conveyed in the pelvic, hypogastric, and pudendal nerves. The Aδ-fiber endings are located in the detrusor muscle and are activated when the bladder is filling. This constitutes the sympathetic storage reflex [pelvic-to-hypogastric reflex, Figure 3(a)]. Within the spinal cord, sympathetic firing from the lumbar region is initiated, which, by effects at the ganglionic level, decreases excitatory parasympathetic input to the bladder. Postganglionic adrenergic neurons release noradrenaline, which, by stimulating β3-adrenoceptors in the bladder wall, causes relaxation of the bladder, and keeps the outflow region (bladder neck and urethra) contracted via α1-adrenoceptor stimulation. Thus, the Aδ afferent and the sympathetic efferent fibers constitute a vesico-spinal–vesical storage reflex, which maintains the bladder in a relaxed mode while the proximal urethra and bladder neck are contracted. In response to a sudden increase in intra-abdominal pressure, such as during a cough, laugh, or sneeze, a more rapid somatic storage reflex [pelvic-to-pudendal reflex, Figure 3(a)], also called the guarding or continence reflex, is initiated. The evoked afferent activity travels along myelinated Aδ afferent nerve fibers in the pelvic nerve to the sacral spinal cord, where efferent somatic urethral motor neurons, located in Onuf’s nucleus, are activated and contract the striated muscle sphincter.

The bladder wall. 10

Human lower urinary tract innervation. 12
C fiber nerve endings are located in urothelium and lamina propria and are activated when the bladder is filled to functional or anatomical capacity. Theories based on experimental studies in animals and humans suggest that signals conveying urge are transported via Aδ fibers and those carrying urgency sensations are conveyed by C fibers, leading to distinct patterns of central nervous system (CNS) activation.5,14
The pathophysiology of urgency has been extensively reviewed.5,12,14 –17 Pathophysiological triggers that contribute to the sensation of urgency can be located in the CNS as well as peripherally. In the latter, two signaling pathways can be defined: the myogenic and the urothelial. The bladder smooth muscle is organized in units or modules that exhibit spontaneous, myogenic contractile activity that can be recorded in isolated muscle strips or in isolated whole bladders where it has been termed ‘autonomous bladder micromotions’. 18 The contractions of the different units are not coordinated and as one unit may contract while the other relaxes, no significant intravesical pressure is generated, and no bladder emptying occurs. However, these muscle contractions, which are enhanced when the bladder is filled, can generate afferent activity, leading to normal sensations of bladder filling. In bladder dysfunction (such as partial denervation of the detrusor), they may be partly coordinated and enhanced, which can lead to urgency and increased frequency.19 –21 Even if there is no parasympathetic outflow from the spinal cord during bladder filling (which is required for the voiding contraction), it can be assumed that local factors, such as prostaglandins, or even acetylcholine (Ach), generated from non-neurogenic sources, can stimulate the myogenic activity and lead to increased afferent activity. Another possibility influencing the contractions is via efferent, short postganglionic cholinergic nerves. 22 These nerves are inhibited by the sympathetic nervous system via β3-adrenoceptors (Figure 4). 23 The spontaneous contractions and the factors influencing them are considered targets for drugs meant for treatment of OAB.
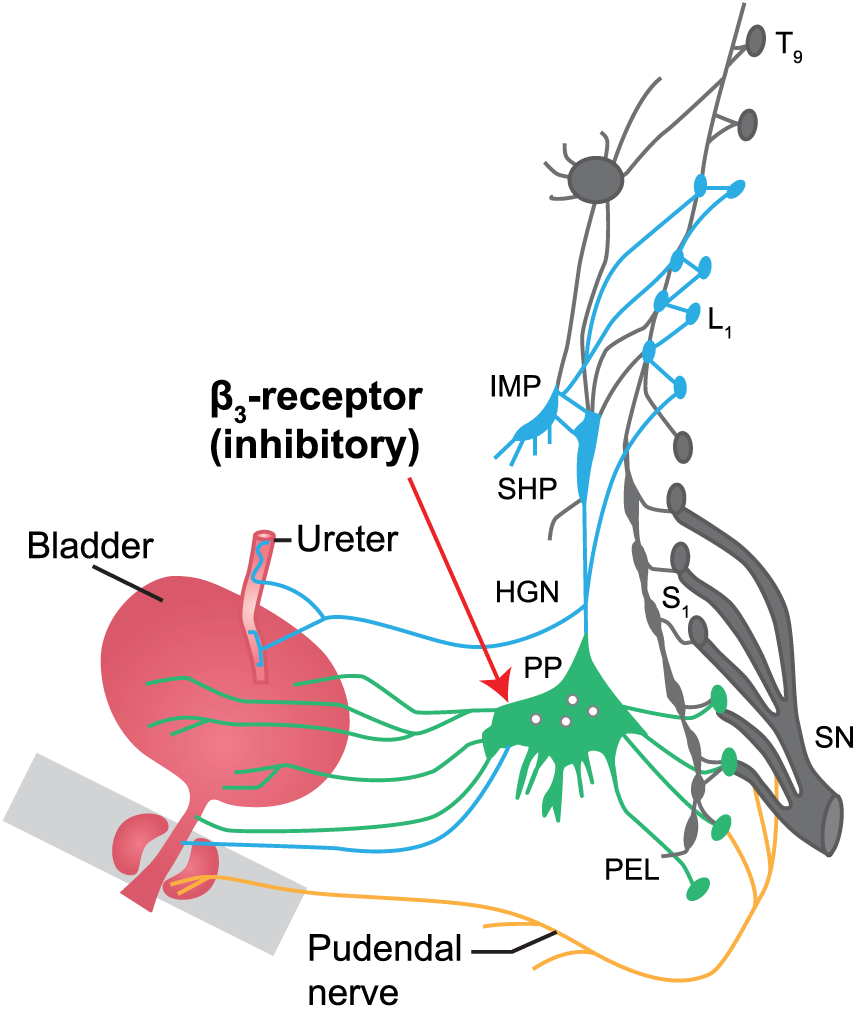
The pelvic plexus. 23
The urothelial signaling pathways are dependent on the interaction between the urothelium and afferent nerves. 11 During bladder distension, urgency may be evoked by stimulation of the suburothelial neural plexus in the peripheral neural system. The release of chemical mediators, such as adenosine triphosphate, neurotropic factors, serotonin, prostaglandins, histamine, and bradykinin, may initiate signaling within and between urothelial cells, bladder nerves, and other cells within the bladder. 15 Inappropriate signaling may alter reflexes, leading to defective bladder control and distension, resulting in urgency.12,16 Urothelial cells in the aging bladder have been associated with bladder dysfunction and the urgency sensation.15,24 –28 Muscarinic receptor stimulation may affect urothelial cell signaling by enhancing afferent signaling and detrusor contractions within the bladder (Figure 5).15,29,30 Changes in neuronal and non-neuronal Ach within urothelial cells in the aging bladder have been associated with bladder dysfunction and the urgency sensation.14,31

Mechanisms of action for antimuscarinic and β3-adrenoceptor agonists in the urothelium and lamina propria afferent nervous system. 30
Urgency is a perception dependent on afferent nervous system signals or generated in the CNS without changes in the peripheral system. Urgency can thus be initiated by defective signaling anywhere in the afferent circuit connecting to the central nuclei of the CNS limbic system and the prefrontal cortex, which controls negative feedback of the micturition center. 16 Brain imaging studies of bladder control involving individuals with normal bladder control and those with OAB have indicated that cortical brain responses in individuals with OAB are exaggerated and this is presumed to induce the urgency sensation. 17
Validated measures of urgency
The assessment of urgency as the ultimate driver of all OAB symptoms is highly important. 9 However, urgency has frequently been overlooked as an outcome measure in clinical trials. 1 Measurement of urgency is difficult because of the subjective nature of the symptoms where a patient feels urgency but cannot objectively measure it. 32 The Patient Perception of Intensity and Urgency Scale (PPIUS) is recommended for assessing the severity of urgency and urgency incontinence felt by patients at each micturition. 32 The PPIUS rates urgency as 0, no urgency, no need to empty the bladder but bladder was emptied for other reasons; 1, mild urgency, could postpone voiding for as long as was needed; 2, moderate urgency, could postpone urgency for a short while without bladder release; 3, severe urgency, could not postpone voiding and had to rush to the toilet; 4, urgency incontinence, leakage before arriving at the toilet. 33 The PPIUS is validated and provides reliable results for known severity groups and is recommended for use for both clinical trials and clinical practice.32,33 The Total Urgency and Frequency Score (TUFS) is derived from the PPIUS and measures both total urgency and frequency of micturition scores. 34 TUFS was validated in patients with OAB and the TUFS score is calculated by adding PPIUS scores recorded by the patient in a urinary diary and dividing this sum by the number of days of the diary record. 34 The Indevus Urgency Severity Scale (IUSS), which defines severe urgency as extreme urgency discomfort that interferes with other activities or tasks, Urgency Perception Scale (UPS), and urgency perception score have all been validated in clinical trials and in each scale up to five grades of urgency are assigned a score.34 –37 The overactive bladder questionnaire (OAB-q) evaluates symptom bother with eight questions, two of which relate to the ICS definition of urgency symptoms. 1 The visual analog scale to measure urgency bother asks patients to determine the severity of urgency symptoms over the previous 7 days by placing a mark on a 0–100 mm line (0 mm, no bother and 100 mm, worst possible bother). 1
Mechanism of action of monotherapies to treat overactive bladder symptoms
Muscarinic receptor antagonists are widely used for the treatment of patients with urgency and other OAB symptoms.30,38 Traditionally, the mechanism of action of muscarinic receptor antagonists was thought to be through reducing detrusor muscle contractions by blocking muscarinic receptors on the cell surface. 30 However, antimuscarinics also block muscarinic receptors in, for example, the urothelium and suburothelial myofibroblasts (Figure 5), which may decrease bladder afferent activity in both Aδ and C fibers, and reduce urgency sensation and other OAB symptoms.30,39 –43 Furthermore, it seems possible to relieve symptoms without significantly reducing detrusor contractility. 44 Interestingly, when given to OAB patients, Ach esterase inhibitors (preventing the degradation of Ach) enhance bladder overactivity and initiate urgency. 45
In the human lower urinary tract, β3-adrenoceptors can be found in bladder detrusor, but also in urothelium46,47 (Figure 5), and a recent study demonstrated that β3-adrenoceptors are abundantly located in Ach-containing nerve fibers in the human bladder.48,49 Assuming that Ach release from cholinergic terminals during bladder filling contributes to OAB symptoms, the finding that activation of prejunctional β3-adrenoceptors may downregulate Ach release resulting in an inhibitory control of parasympathetic activity (Figure 4) may be of importance.22,47
Mirabegron is a selective β3-adrenoceptor agonist whose preclinical pharmacological profile has been well described in vitro and in vivo.50,51 In animal models, mirabegron increases intervoidal intervals, bladder compliance, and reduces nonmicturition contractions in the bladder, while preserving active voiding function. 51 Mirabegron is an alternative therapeutic option for the treatment of urgency and even if it has a different mode of action from antimuscarinic agents, both classes of drugs decrease bladder afferent activity. 52 Its clinical efficacy is well documented (see below).
Monotherapy clinical trials: antimuscarinics
A number of placebo-controlled clinical trials have shown that fesoterodine, imidafenacin, propiverine, solifenacin, and tolterodine were statistically significantly more effective at reducing the mean number of urgency episodes/24 h compared with placebo (Figure 6).53 –62 Pooled differences in mean changes from baseline varied between 0.4 and 1.7 episodes per day compared with placebo. Trospium chloride extended release was also statistically significantly more effective at reducing the mean number of severe urgency episodes/24 h (IUSS) compared with placebo. 63 There was some evidence favoring daily doses of solifenacin 5 mg and 10 mg compared with tolterodine 4 mg, and solifenacin 10 mg compared with propiverine 20 mg. 64

Antimuscarinic agent monotherapy trials: mean change from baseline in urgency episodes/24 h.
Treatment with antimuscarinic agents also significantly reduces the incidence of severe urgency in subjects with OAB and increases the warning time to the onset of an urgency episode compared with placebo.53,65 Clinically meaningful improvement in symptoms of urgency and severe urgency may begin within the first few days of antimuscarinic treatment. 66 Dose escalation of antimuscarinic agents may further improve urgency symptoms compared with lower doses of the same agent; however, the antimuscarinic burden is more likely to increase (dry mouth, constipation, etc.) resulting in a decreased rate of persistence.67,68 This is an important consideration, especially when treating elderly OAB patients.
Few studies report data for severe urgency episodes/24 h (grade 3 or 4 PPIUS), although, in the studies that reported severe urgency, antimuscarinic monotherapy statistically significantly reduced severe urgency episodes/24 h (grade 3 or 4 PPIUS) for fesoterodine (−1.1 episodes/24 h), tolterodine (−0.6 episodes/24 h), and solifenacin (−0.8 episodes/24 h) compared with placebo (Figure 6).53,56 While up to 85% of patients receiving antimuscarinic treatment experienced a significant improvement in severe urgency symptoms after 12 weeks of treatment compared with placebo, in studies that reported on resolution of urgency, antimuscarinic monotherapy only completely resolved urgency in approximately 40% of patients after 12 weeks.54,59,61
It is important to note that escalating the antimuscarinic dose to obtain a better response also affects muscarinic receptors outside of the bladder, therefore there is an increase in systemic antimuscarinic adverse events (AEs).
Monotherapy clinical trials: β3-adrenoceptor agonists
Mirabegron has a different mechanism of action from antimuscarinic agents and has similar efficacy compared with antimuscarinic agents. Mirabegron has a lower incidence of antimuscarinic AEs for example, dry mouth and constipation that are commonly associated with antimuscarinics and often a reason for treatment discontinuation.69,70
A number of placebo-controlled clinical trials have shown that mirabegron was statistically significantly more effective at reducing the number of severe (PPIUS grade 3 or 4) urgency episodes/24 h compared with placebo (Figure 7).70 –74 In a pooled analysis of phase III clinical trial data, mirabegron 50 mg monotherapy (−0.64 episodes/24 h) statistically significantly reduced severe urgency episodes/24 h (grade 3 or 4 PPIUS) compared with placebo. 70 In the BEYOND study, mirabegron 50 mg demonstrated similar efficacy to solifenacin 5 mg at reducing the number of severe urgency episodes/24 h (PPIUS grade 3 or 4) episodes after 12 weeks of treatment (−4.61 versus −4.84 episodes/24 h, respectively, from baseline values of 7.7 and 7.7 episodes/24 h, respectively) and there was no statistically significant difference (−0.23, p = 0.16) between the two active comparator arms. 71 Despite mirabegron having a different mechanism of action from antimuscarinic agents, the onset of action of mirabegron in reducing urgency compared with placebo occurs as early as week 2, which is similar to the onset of action of antimuscarinic agents. 75 The significant reductions in severe urgency and other OAB symptoms with mirabegron treatment were also accompanied by statistically significant improvements in HRQoL (OAB-q Symptom Bother score, HRQoL Total score, and Coping and Concern subscales). 75 A number of large studies have shown that patients prescribed mirabegron stayed on treatment longer with better adherence compared with antimuscarinic agents.76 –79

Mirabegron monotherapy trials adjusted mean change from baseline to end of treatment in urgency/24 h (grade 3 or 4) on the Patient Perception of Intensity and Urgency Scale.
Significance of mirabegron for the management of urgency in clinical practice: combination therapy
There are a number of potential benefits associated with combination therapy that utilizes two different classes of drugs. Targeting two distinct molecular mechanisms in the regulation of detrusor activity is anticipated to have an additive effect on OAB symptoms, which may be particularly beneficial in hard-to-treat patients, for example, those with urgency refractory to antimuscarinic monotherapy. In this instance, adding mirabegron to an antimuscarinic agent may be more effective in controlling urgency compared with antimuscarinic monotherapy alone.
An initial phase II dose-ranging trial in 1306 patients with OAB (the Symphony study), evaluated the efficacy and safety of solifenacin + mirabegron combination therapy (solifenacin 2.5, 5, or 10 mg + mirabegron 25 or 50 mg) compared with each treatment as monotherapy or placebo over 12 weeks. 80 Compared with solifenacin 5 mg monotherapy, combination therapy demonstrated significant improvements in mean volume voided (primary endpoint), micturition frequency and number of urgency episodes, without increasing the bothersome adverse effects associated with antimuscarinic therapy (with the possible exception of constipation).
A more difficult-to-treat population than in Symphony was included in the BESIDE study, which investigated whether the addition of mirabegron to solifenacin 5 mg (combination therapy) was more effective than solifenacin monotherapy in reducing the symptoms of OAB in incontinent OAB patients. 81 The reduction in the mean number of incontinence episodes/24 h was statistically significantly greater with combination therapy (−1.80) compared with solifenacin 5 mg monotherapy (−1.53). There was a statistically significant difference in change from baseline to the end of treatment in the mean number of severe urgency episodes/24 h (PPIUS scale grade 3 or 4) with combination therapy (solifenacin 5 mg + mirabegron 50 mg; reduction in severe urgency episodes/24 h −2.95 episodes, baseline 5.84 episodes/24 h) compared with solifenacin 5 mg (reduction −2.41 episodes/24 h, baseline 5.68 episodes/24 h) or 10 mg (reduction −2.54 episodes/24 h, baseline 5.79 episodes/24 h) monotherapy. At the end of treatment, the difference between combination therapy and solifenacin 5 mg was −0.54 episodes/24 h, p < 0.001 and solifenacin 10 mg −0.40 episodes/24 h, p = 0.007 (Figure 8). Combination therapy was superior to solifenacin 5 mg as early as week 4 (the first timepoint measured) and was superior to solifenacin 10 mg monotherapy from week 8 to the end of treatment in improving the mean number of urgency episodes (PPIUS grade 3 or 4)/24 h. There were statistically significantly more responders to combination treatment and statistically significant improvements in HRQoL reported for patients receiving combination therapy compared with monotherapy. 82

The positive impact of combination therapy with solifenacin and mirabegron compared with solifenacin monotherapy on the adjusted mean change from baseline to end of treatment in urgency/24 h (Patient Perception of Intensity and Urgency Scale grade 3 or 4) from the BESIDE study.
The SYNERGY study evaluated combination treatment with solifenacin 5 mg + either mirabegron 25 mg or 50 mg versus the respective monotherapies or placebo in a large OAB patient population with urinary incontinence. 83 Combination treatment did not achieve a statistically significant effect (p = 0.052) versus mirabegron 50 mg in one of the coprimary endpoints (change from baseline to end of treatment in incontinence episodes/24 h). For the second coprimary endpoint, adjusted change from baseline to end of treatment in micturitions/24 h was greater in the combination therapy groups versus monotherapies, with nominal p values <0.05. For change from baseline to end of treatment in mean number of urgency episodes (PPIUS grade 3 and/or 4)/24 h, solifenacin 5 mg + mirabegron 50 mg demonstrated improvements compared with solifenacin 5 mg [adjusted mean difference (standard error) of −0.45 (0.18), with 95% confidence intervals (CIs) of −0.82, −0.09; p = 0.014], mirabegron [adjusted mean difference −0.87 (0.19), 95% CIs −1.24, −0.51; p < 0.001] and placebo [adjusted mean difference −1.44 (0.18), 95% CIs −1.81, −1.08; p < 0.001]. Solifenacin 5 mg + mirabegron 25 mg also demonstrated significant improvements compared with placebo [adjusted mean difference −1.32 (0.18), 95% CIs −1.68, −0.96; p < 0.001], mirabegron [adjusted mean difference −0.65 (0.18), 95% CIs −1.01, −0.28; p < 0.001] but the improvements did not reach statistical significance when compared with solifenacin 5 mg [adjusted mean difference −0.33 (0.18), 95% CIs −0.69, 0.03; p = 0.074]. For change from baseline to end of treatment in mean number of urgency incontinence episodes/24 h, solifenacin 5 mg + mirabegron 50 mg demonstrated improvements compared with solifenacin [adjusted mean difference of −0.23 (0.11), with 95% CIs of −0.45, −0.02; p = 0.043], mirabegron [adjusted mean difference −0.32 (0.11), 95% CIs −0.54, −0.10; p = 0.019] and placebo [adjusted mean difference −0.61 (0.11), 95% CIs −0.83, −0.39; p < 0.001]. Solifenacin 5 mg + mirabegron 25 mg also demonstrated improvements compared with mirabegron [adjusted mean difference −0.37 (0.11), 95% CIs −0.59, −0.15; p < 0.001] and placebo [adjusted mean difference −0.62 (0.11), 95% CIs −0.84, −0.40; p < 0.001] but improvements versus solifenacin were not statistically significant [adjusted mean difference −0.24 (0.11), 95% CIs −0.46, −0.02; p = 0.134]. Analysis of the mean number of urgency and urgency incontinence episodes showed that the effect size was larger for those who had received OAB treatment prior to entering the study than for those who were treatment naïve. For both urgency episodes and urgency incontinence episodes, the 95% CIs for the differences of combination versus both monotherapy components excluded zero in the group of previously treated patients.
The measurement and reduction of urgency symptoms is highly important because urgency is the central system and driver of all OAB symptoms. 9 Although combination therapy has clear benefits for patients, the pathophysiology of OAB is multifaceted, with common symptoms such as urgency being mediated by both myogenic and neurogenic mechanisms. 16 Hence, combination therapy may not be completely successful in reducing symptoms as the full spectrum of pathophysiological pathways behind the clinical syndrome are not being addressed.
Combination treatment with solifenacin and mirabegron will also need to be evaluated in real-life clinical practice as an alternative to the current approaches, which include dose escalation of antimuscarinic monotherapy or switching to more invasive techniques, for example, intravesical botulinum toxin use which can come with an increased AE burden, especially urinary tract infections and intermittent self-catheterization or sacral neuromodulation.81,84 –86 The BESIDE study mirrored a real-life setting in a hard-to-treat population who remained incontinent after 4 weeks of daily solifenacin 5 mg monotherapy. 81 Adding mirabegron 50 mg to solifenacin 5 mg was superior to solifenacin 5 mg monotherapy in improving urgency, urinary urgency incontinence, and other OAB symptoms in patients who had an insufficient response to solifenacin 5 mg monotherapy. Furthermore, significantly more patients receiving combination therapy achieved clinically meaningful improvements in incontinence, micturition frequency, and HRQoL. 82 The improvements in OAB symptoms in patients receiving combination therapy resulted in significant improvements in HRQoL compared with those receiving solifenacin monotherapy. Urgency is the key symptom of OAB and often drives other symptoms, such as nocturia, which can have serious consequences in older patients by increasing the risk of falls and chronic insomnia that affect daytime daily functioning. 87 The severity of urgency, especially urgency urinary incontinence, affects HRQoL and improvements in urgency and urinary urgency incontinence with combination therapy versus monotherapies were associated with significant improvements in OAB-q Symptom Bother score, HRQoL Total score, and patient perception of bladder condition (PPBC). 82 In a phase IV open-label study, mirabegron added on to solifenacin (2.5 mg or 5 mg) combination therapy was well tolerated with no new safety signals observed and significant improvement in OAB symptoms at end of treatment compared with baseline. 88
Other considerations
The US Food and Drug Administration does not recognize urgency as an outcome measure; as a consequence, urgency is reported as a secondary outcome in most clinical trials. The subjective nature of urgency means that some patients are affected differently or respond differently to treatment. Older patients have more severe irresistible ‘urge’ which is believed to be a major pathological mechanism in the generation of symptoms in the white matter of the brain.87,89 Older patients are less mobile and more prone to urgency being a greater burden.
From clinical trial data, it is clear that not all patients with urgency respond to antimuscarinic agents and β3-adrenoceptor agonists. One explanation for this is that the perception of urgency may be generated in the CNS and may therefore be outside the mechanism of actions of antimuscarinic and β3-adrenoceptor agonists. However, theoretically, most of the afferent pathway response generated through the sympathetic or parasympathetic system could be blocked by OAB treatment. Indeed, while the peripheral nervous system effect on the perception of urgency is well established, there are still several mechanisms that are not well understood and require further research to fully understand the mechanisms involved in generating urgency symptoms. Nevertheless, antimuscarinic agents, mirabegron, or combination treatment with mirabegron added on to solifenacin 5 mg statistically significantly reduces the symptoms of severe urgency (PPIUS grade 3 or 4) by week 12 compared with placebo. Combination therapy with mirabegron added on to solifenacin 5 mg also statistically significantly reduces the symptoms of severe urgency compared with solifenacin monotherapy. Combination therapy offers an alternative treatment in patients with OAB, including urgency refractory to first-line monotherapy, who may often move on to more invasive treatments.
Footnotes
Acknowledgements
All authors contributed to the concept and design of the review and provided major editorial input on the earlier and final versions of the manuscript. The authors would like to thank Matthias Stölzel of Astellas Pharma Global Development for his helpful contributions to the manuscript. Medical writing support was provided by John Clarke of Envision Scientific Solutions and funded by Astellas Pharma Global Development, Inc.
Funding
The review was funded by Astellas Pharma Global Development, Inc.
Conflict of interest statement
KEA has received personal fees from Astellas, Allergan, and Bayer. JNC has received honoraria and consultancy fees from Astellas. PVK has received personal fees from Astellas, Ferring, and Medtronic. NC, MH, CK, and ES are employees of Astellas Pharma, Europe BV.
