Abstract
Background
For over 100 years white-light cystoscopy has remained the gold-standard technique for the detection of bladder cancer (BCa). Some limitations in the detection of flat lesions (CIS), the differentiation between inflammation and malignancy, the inaccurate determination of the tumor margin status as well as the tumor depth, have led to a variety of technological improvements. The aim of this review is to evaluate the impact of these improvements in the diagnosis of BCa and their effectiveness in clinical practice.
Methods
A systematic literature search was conducted according to the PRISMA statement to identify studies reporting on imaging modalities in the diagnosis of NMIBC between 2000 and 2017. A two-stage selection process was utilized to determine eligible studies. A total of 74 studies were considered for final analysis.
Results
Optical imaging technologies have emerged as an adjunct to white-light cystoscopy and can be classified according to their scope as macroscopic, microscopic and molecular. Macroscopic techniques including photodynamic diagnosis (PDD), narrow-band imaging (NBI) and the Storz Professional Image Enhancement System (IMAGE1 S, formerly known as SPIES) are similar to white-light cystoscopy, but are superior in the detection of bladder tumors by means of contrast enhancement. Especially the detection rate of very mute lesions in the bladder mucosa (CIS) could be significantly increased by the use of these methods. Microscopic imaging techniques like confocal laser endomicroscopy and optical coherence tomography permit a real-time high-resolution assessment of the bladder mucosa at a cellular and sub-cellular level with spatial resolutions similar to histology, enabling the surgeon to perform an ‘optical biopsy’. Molecular techniques are based on the combination of optical imaging technologies with fluorescence labeling of cancer-specific molecular agents like antibodies. This labeling is intended to favor an optical distinction between benign and malignant tissue.
Conclusions
Optical improvements of the standard white-light cystoscopy have proven their benefit in the detection of BCa and have found their way into clinical practice. Especially the combination of macroscopic and microscopic techniques may improve diagnostic accuracy. Nevertheless, HAL-PDD guided cystoscopy is the only approach approved for routine use in the diagnosis of BCa by most urological associations in the EU and USA to date.
Introduction
Urothelial carcinoma of the bladder (BCa) is a prevalent malignancy with an estimated incidence of 76,960 cases and 16,390 cancer-specific deaths in the United States alone in 2016. 1 Approximately 75% of BCa patients present with a non-muscle invasive disease (NMIBC) confined to the mucosa (Ta, CIS) or submucosa (T1). 2 Due to its heterogeneous natural history, the clinical behavior of these tumors varies, ranging from low-grade variants to very aggressive and potentially deadly high-grade subtypes with a very high risk of tumor recurrence and progression. 3 Therefore, an early diagnosis is essential for accurate therapeutic management. Based on its endoluminal localization, the detection of BCa traditionally has relied on cystoscopy for over 100 years. 4 In 1878 the first working cystoscope was presented by Maximilian Carl-Friedrich Nitze, a German urologist, and Joseph Leiter, a Viennese instrument maker. 5 Even if the Nitze/Leiter cystoscope was a landmark discovery, it was by no means perfect. The biggest limitation was the tungsten wire used for lighting. Hence, the greatest efforts have been made to improve the optical quality of cystoscopy, leading to modern-quality white-light cystoscopy (WLC).
Today, WLC is the gold-standard technique for the detection of BCa and can be performed as a flexible or rigid approach for initial identification of suspicious lesions. 2 Despite the now achieved optical quality, there are some unresolved shortcomings. While there is a sufficient detection rate for papillary tumors, the reliability in the case of flat lesions falls dramatically to 58–68%.6–8 Additionally, smaller or satellite tumors may be missed.6 –8 Moreover, the inaccurate determination of the tumor margin can lead to incomplete tumor resection, resulting in BCa residuals in up to 40% of cases.2,9,10 Finally, an inadequate assessment of the tumor invasion may lead to clinical understaging, prompting inadequate disease management.11,12
To overcome these shortcomings, different optical imaging technologies have emerged as an adjunct to WLC to improve BCa detection and resection. According to their scope, those technologies can be classified as macroscopic, microscopic and molecular. Macroscopic techniques including ‘photodynamic diagnosis’ (PDD), ‘narrow-band imaging’ (NBI) and the ‘Storz Professional Image Enhancement System’ (IMAGE I S) are similar to WLC, but are superior in the detection of bladder tumors by means of contrast enhancement. 8 Especially the detection rate of very mute lesions in the bladder mucosa (CIS) could be significantly increased by the use of these methods. Microscopic imaging techniques like ‘Confocal laser endomicroscopy’ and ‘Optical coherence tomography’ permit real-time high-resolution assessment of the bladder mucosa at a cellular and sub-cellular level with spatial resolutions similar to histology, enabling the surgeon to perform an ‘optical biopsy’. Molecular techniques are based on the combination of optical imaging technologies with fluorescence labeling of cancer-specific molecular agents like antibodies. This labeling is intended to favor an optical distinction between benign and malignant tissue.
All these technological advances have the potential to improve the diagnostic procedure as well as the endoscopic management of BCa, and shall be described in more detail in this review.
Methods
A systematic literature search was conducted according to the PRISMA statement 13 to identify studies reporting on imaging modalities in the diagnosis for NMIBC between 2000 and 2017. The PubMed database was searched along with a free-text hand search using one or several combinations of the following items: ‘bladder cancer’, ‘non-muscle invasive bladder cancer’, ‘cystoscopy’, ‘imaging modalities’, ‘diagnosis’, ‘detection’, ‘fluoresces’, ‘blue-light’, ‘white-light’, ‘photodynamic diagnostic’, ‘PDD’, ‘5-ALA’, ‘Hexvix’, ‘hexyl 5-aminolevulinate’, ‘HAL’, ‘narrow-band imaging’, ‘Storz Professional Image Enhancement System’, ‘SPIES’, ‘molecular imaging’, ‘optical coherence tomography’, ‘confocal laser endomicroscopy’, ‘Raman spectroscopy’, ‘multiphoton microscopy’, ‘scanning fiber endoscopy’, ‘ultraviolet autofluorescence’, ‘molecular imaging’. The selection process was conducted at two stages; the first stage was performed via initial screening of the title and abstract to identify eligible publications. The second stage was done via full-text reading, including a manual search of publications in journals not listed in PubMed to further avoid missing any eligible study. For this systematic review, we excluded (1) non-English articles; (2) editorials or case reports; and (3) repeated publications on the same cohort to avoid publication bias. After completion of the systematic search, a risk of bias assessment was conducted according to the Cochrane Handbook for Systematic Reviews of Randomized Studies 14 and the Newcastle–Ottawa scale for retrospective studies (Figure 1). 15

CONSORT diagram showing selection process of data included in meta-analysis.
Macroscopic imaging modalities
Photodynamic diagnosis
PDD, also known as fluorescence cystoscopy or blue-light cystoscopy, enables a wide-field fluorescence imaging of the bladder wall. PDD requires preoperative intravesical application of photosensitizing agents, which will be taken up by the urothelium and incorporated into the heme-biosynthesis of the cells, resulting in an intracellular accumulation of photoactive porphyrines, especially in abnormal cells.8,10,16–25 After exposure to blue light (380–480 nm), the dysplastic cells emit a characteristic red fluorescence, which can be easily discriminated against the blue background of healthy urothelium (Figure 2). 16 Two protoporphyrin analogues, 5-aminolevulinic acid (5-ALA) and its ester derivative hexaminolevulinate (HAL), have been approved for PDD of bladder tumors. Both are practically free from side effects.

Intraoperative image of a lesion suspicious for BCa located on the right bladder wall: (a) white-light cystoscopy; (b) fluorescence-guided cystoscopy using PDD (HAL).
The efficacy of PDD with 5-ALA to increase BCa detection rates has been proven in a large number of clinical trials.8,10,16–25 Unfortunately, some of its biochemical characteristics, such as the hydrophilicity at physiological pH, low solubility in fats leading to poor bioavailability and rapid disappearance of tissue fluorescence (photobleaching) during cystoscopy, have limited the use of 5-ALA in everyday practice. 2 These limitations have been overcome with the introduction of HAL (Hexvix®), which is characterized by higher fat solubility as well as rapid and uniform absorption by urothelial cells. Within the cells, HAL is converted into 5-ALA by an esterase, allowing PDD to be performed. 16
More than 300 studies on fluorescence cystoscopy have been published, demonstrating a superiority of PDD either with HAL or 5-ALA compared to WLC in terms of BCa detection rates, especially CIS, as well as its positive impact on cancer recurrence and progression.8,10,16–25
In detail, numerous prospective trials (Table 1) have demonstrated PDD to be significantly more effective than WLC in terms of BCa detection, even if it does not allow distinguishing tumor grade. 2 The sensitivity for PDD ranges from 76% to 97% compared to 46% to 80% for WLC. 22 These findings were reflected within two meta-analyses (I: n = 900, 8 studies; II: n = 2807, 27studies) identifying 20% greater sensitivity for PDD over WLC.8,23 In contrast, only two studies did not find higher tumor detection rates for PDD.24,25
Comparison of recurrence-free survival rates between photodynamic diagnosis and white-light cystoscopy in selected studies in patients with non-muscle-invasive bladder cancer.

5-ALA, 5-aminolevulinate acid; HAL, hexyl aminolevulinate; PDD, photodynamic diagnostic; RFS, recurrence-free survival; WLC, white-light cystoscopy.
In addition, the superiority of PDD using HAL in the detection of flat lesions, particularly CIS, seems undisputed. The detection rates for PDD range from 49% to 100% versus 5% to 68% for WLC alone.2,8 Overall, three meta-analyses concluded that detection rates for CIS were found to be approximately 25–30% higher under PDD guidance.8,17,18
Moreover, several studies have suggested that the use of PDD is associated with a more complete tumor resection and significantly prevents cancer recurrence. 2 The majority of studies reported on recurrence-free survival rates between 3 and 24 month with PDD compared to WLC. Recurrence-free rates at 12 and 24 months were higher in up to 27% and 24% of cases, respectively.8,18 It is noteworthy that patients with multifocal tumors seem to benefit most from PDD guidance. 10
Finally, a recent study supports the assumption that the detection and resection of NMIBC under PDD guidance reduces the risk of progression. 19
Therefore, HAL-PDD guided cystoscopy is the only approach approved for routine use in the diagnosis of BCa by most urological associations in the EU as well as in the USA, and is particularly recommended for the diagnosis of primary NMIBCs. 2
However, PDD is limited by false-positive rates in up to 10–12% of cases due to autofluorescence by endogenous tissue fluorophores, and the high capability of immune cells to accumulate the photosensitizing agent. 21 Hence, PDD should not be used for patients who received intravesical immunotherapy or chemotherapy within the previous 90 days. 2
Narrow-band imaging
NBI is a high-resolution wide-field endoscopic technique that improves BCa detection by filtering out the red spectrum from white light without the need for intravesical contrast administration. The resultant blue (415 nm) and green (540 nm) spectra are absorbed by hemoglobin, thus highlighting the contrast between capillaries and mucosa and facilitating rapid real-time evaluation of suspected lesions.31,32 The filter technology has been incorporated into standard flexible and rigid cystoscopes, enabling facile toggling between WLC and NBI. 31 Several studies have been published examining the effectiveness of NBI (Table 2).31 –39 Based on the results of a large meta-analysis (n = 1022), NBI is superior in BCa detection compared to WLC (sensitivity: 95% versus 75%). 36 Especially detection of CIS can be improved significantly by using NBI + WLC compared to WLC alone (sensitivity: 90–100% versus 50–83%).32 –35 Additionally, the current body of evidence based on some single-center studies allows the admission of a positive impact on recurrence-free survival for patients undergoing Narrow Band Imaging Guided Transurethral Resection of the Bladder (NBI-TURB) compared to WLC. In one randomized trial (n = 254) with a 2-year follow up, a reduced recurrence rate (22% versus 33%) and improved recurrence-free survival (22 versus 19 months) was reported by using NBI rather than WLC alone. 35 These results were confirmed by a meta-analysis (n = 1024). 39 However, it must be noted that some of the included studies were underpowered. A large prospective international study of the Clinical Research Office of the Endourological Society (CROES) recently finished recruitment. 38 Therefore the effect of NBI on therapeutic outcomes has not yet been confirmed. However, it seems obvious that better visualization would offer better local control, even if NBI does not allow for determination of tumor invasiveness or tumor grade prediction.
Comparison of detection rates for bladder cancer by narrow-band imaging and white-light cystoscopy in selected studies.
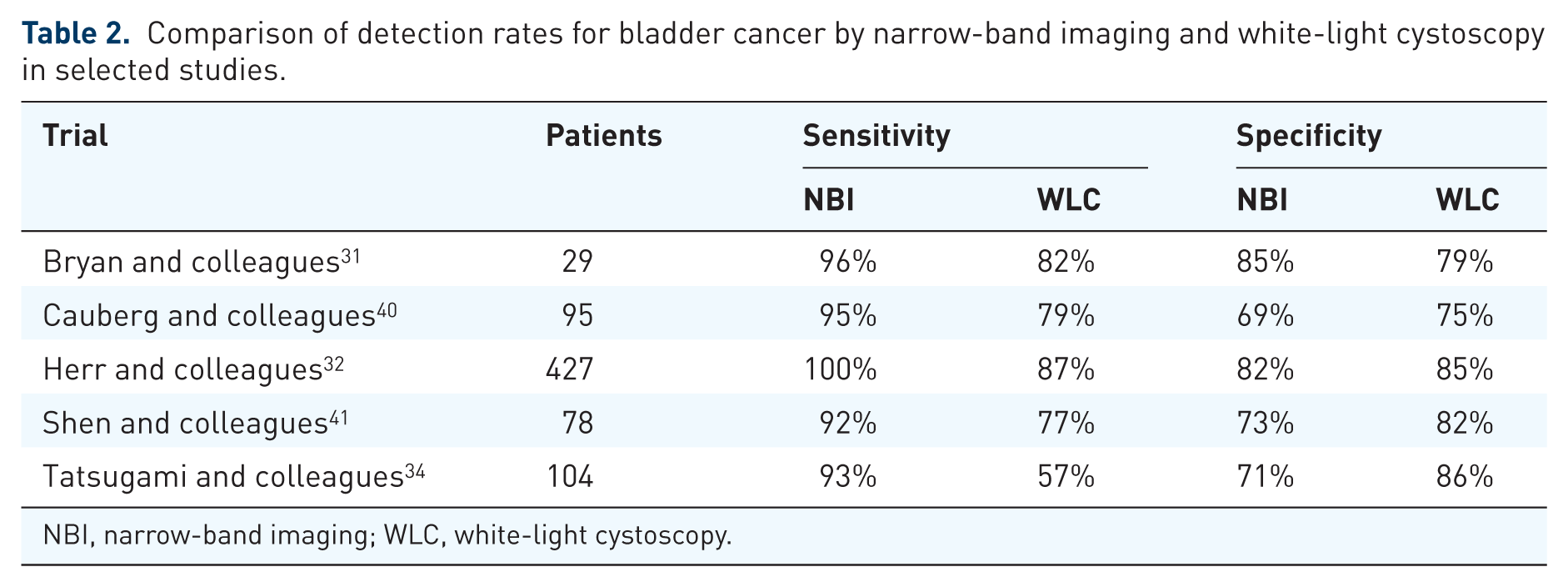
NBI, narrow-band imaging; WLC, white-light cystoscopy.
Storz Professional Image Enhancement System® (IMAGE1 S, formerly known as SPIES)
Based on a similar technology to NBI, Storz® has developed the Storz Professional Image Enhancement System (IMAGE1 S)®, which was recognized as Innovation of the Year in 2014 by the Society of Laparoendoscopic Surgeons. This system offers several image enhancement modalities, containing a white light (WL), spectra A, spectra B, chroma and clara mode. Spectra A and B are based on color tone shift algorithms to increase the contrast (Figure 3). Chroma enhances the sharpness of the displayed image. Clara uses a local brightness adaptation in the image to achieve greater visibility of darker regions within the image. Additionally, the system is able to provide a standard WL image in a side-by-side viewing mode with the IMAGE1 S image simultaneously (Figure 4). Currently, the IMAGE1 S technology is being investigated in a large prospective international trial by CROES. 42 Recruitment is still ongoing.

Intraoperative image of a lesion suspicious for BCa located on the right bladder wall: (a) white-light cystoscopy; (b) Storz IMAGE1 S – spectra A mode; (c) Storz IMAGE1 S – spectra B mode.

Intraoperative image of a lesion suspicious for BCa located on the left bladder wall in the side-by-side viewing mode with IMAGE1 S: (a) white-light cystoscopy; (b) Storz IMAGE1 S – chroma mode.
Microscopic imaging modalities
Optical coherence tomography
The optical coherence tomography (OCT) technique provides real-time, high-quality, cross-sectional images of superficial tissues utilizing waves of the near-infrared light spectrum (890–1300 nm). 2 Depending on the back-scattering properties of the different tissue layers, a cross-sectional image with 2 mm depth of penetration and 10–20 μm spatial resolution is thus produced. Under OCT, normal urothelium appears as a weakly scattering dark layer, the lamina propria as a light layer with the highest intensity of scattering and the muscularis propria again as a less scattering layer. In cancerous tissues, these anatomical layers got lost, thereby destroying the contrast. Based on this layering, OCT was able to determine the invasiveness of a bladder lesion (staging) in up to 90% of the cases, and differentiate between healthy and abnormal urothelium with a sensitivity and specificity of 90–100% and 65–89%, respectively.20,21 However, prediction of tumor grade is not possible. Despite these promising findings, OCT is limited by false-positive results, possibly due to disruptions of the bladder wall in benign lesions, and the fact that scanning of the entire bladder is a laborious task, complicating its use in everyday practice. 43 Therefore PDD might be used as an adjunct to OCT to reduce these false-positive findings and simplify the procedure.
Confocal laser endomicroscopy
Confocal laser endomicroscopy (CLE) is an optical biopsy technology that provides microscopic, real-time, histopathologic information in vivo. 44 For this technique a fiber-optic probe (diameter ranging from 0.85 mm to 2.6 mm; compatible with any standard cystoscope) and fluorescein, an FDA-approved contrast agent that can be administered intravesically or intravenously with minimal toxicity, needs to be used to resolve microarchitectural and cellular features as well as physiological processes such as vascular flow.44 –47 Due to the high spatial resolution (1–5 μm), CLS is comparable to conventional histopathology and allows a distinction between high-grade and low-grade bladder cancer. 45 The clinical feasibility has been shown, inter alia, by Sonn and colleagues in vivo and ex vivo. 33 Additionally, an imaging atlas has been published, facilitating the diagnosis and grading of bladder cancer. Although a recent study demonstrated only a moderate inter-observer variability in image interpretation, larger studies examining the diagnostic accuracy of CLE for real-time cancer diagnosis are pending. 48 Moreover, this technique is limited by a very small field of view with limited tissue penetration.45,49
Raman spectroscopy
Raman spectroscopy (RS) (named after the Indian physicist C.V. Raman) is the spectroscopic investigation of the inelastic scattering of light on molecules or solid bodies (Raman scattering) without the requirement of an exogenous contrast agent. By means of infrared light (785–845 nm), intrinsic chemical bonds are excited and an optical contrast is generated depending on the tissue properties. On the basis of the resulting individual ‘tissue fingerprint’, RS was able to distinguish BCa from the normal bladder surface with a sensitivity of 85–86% and a specificity of 79–100%.50,51 By combining so-called surface-enhanced Raman scattering (SERS) nanoparticles and cancer-specific antibodies, an additional signal amplification can be generated to perform a molecular mapping. 52 This technique is limited by a narrow viewing area, weak signals and possible time shift up to 5 s. 52
Multiphoton microscopy
Multiphoton microscopy (MPM) is a laser-scanning microscopy technique to visualize autofluoresces of cells caused by the simultaneous absorption of at least two near-infrared photons (700–800 nm). 53 MPM uses intrinsic tissue fluorophores such as NADH, FAD or collagen. 53 One well-conducted in vitro study reported on an 88% accuracy of MPM imaging regarding the differentiation between benign and neoplastic lesions. The in vivo approach is still under investigation. This technique is limited by its shallow penetration of 0.5 mm, leading to a lack of visualizations of nuclear details and tumor invasion, thus not allowing formal cancer staging.53 –56
Ultraviolet autofluorescence
Similar to MPM, ultraviolet autofluorescence makes use of the different presence of endogenous fluorophores (e.g. NAD and tryptophan) to distinguish between normal, inflammatory and cancerous urothelium.57,58 In contrast to the MPM, however, an exposure with UV laser radiation is necessary. While feasibility has already been shown in a small series of cases, the reproducibility and reliability, as well as the effects of the UV-induced toxicity, must be further investigated. 58
Scanning fiber endoscopy
Scanning fiber endoscopy (SFE) is an ultra-light, flexible endoscope that is capable of analyzing the tissue using laser scanning (red, green and blue) to produce a wide-angle, full-color, high-resolution image. 59 SFE has been successfully used in ex vivo bladder models, but its viability within an in vivo approach is still under investigation. 60,62
Molecular imaging
Molecular imaging (MI) is the combination of macroscopic optical imaging techniques and the administration of fluorescent-labeled elements (FLEs) like antibodies. These FLEs selectively bind to cancer cells, allowing an enhanced differentiation between cancer and benign tissue. One promising FLE is the CD47 antibody (anti-CD47), a surface marker, which is expressed on more than 80% of BCa cells.2,44 An ex vivo study on 25 intact bladders derived from radical cystectomy demonstrated the use of the fluorescent-labeled anti-CD47 to detect BCa by combined CLE and PDD techniques. In all cases anti-CD47 binding to cancer lesions was between 95- and 1100-fold greater than to benign urothelium. 44 Additionally, the sensitivity and specificity for CD47-targeted imaging using PDD were 82.9% and 90.5%, respectively (n = 21). 61 Nevertheless, further in vivo evaluation is required for anti-CD47 regarding its toxicity and manageability.
Conclusions
Optical improvements to the standard WLC have proven their benefit in the detection of BCa and have found their way into clinical practice. As a future prospect, especially the com-bination of macroscopic and microscopic techniques may improve diagnostic accuracy. Nevertheless, HAL-PDD guided cystoscopy is the only approach approved to date for routine use in the diagnosis of BCa by most urological associations in the EU, as well as in the USA. Further technologies are on the horizon that may further enhance our ability to detect, accurately stage and treat BCa.
Footnotes
Funding
G. Gakis, A. Stenzl: Research funding from IPSEN.
Conflict of interest statement
The authors declare that there is no conflict of interest.
