Abstract
Everolimus is an oral inhibitor of mammalian target of rapamycin (mTOR-I) and is currently approved for the treatment of metastatic renal cell carcinoma (mRCC) after failure of first-line vascular endothelial growth factor receptor tyrosine kinase inhibitor (TKI).
In this narrative review, we aim to report the available evidence about the use of everolimus as second-line therapy for mRCC. A literature search was performed using PubMed/MEDLINE and abstracts from major conferences on clinical oncology as sources. We report data from prospective as well as retrospective and real world data studies and we analyze the safety and efficacy profile of everolimus as second-line therapy for mRCC.
Although different drugs are currently available for the second-line treatment of mRCC, everolimus represents a feasible and safe option in this setting, especially for patients who have experienced high-grade toxicity or are still carrying TKI-related toxicities from first-line treatment.
Introduction
Renal cell carcinoma (RCC) accounts for 3% of adult solid tumors with growing incidence in recent decades. More than 25% of patients have metastatic disease at the time of diagnosis and about 20–30% will develop metastases during follow up after surgery for localized disease [Cohen and McGovern, 2005; Altekruse et al. 2010].
During recent decades a better knowledge of RCC cytogenetics has led to the development of molecularly targeted agents able to inhibit the major molecular pathways implied in tumor growth: vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), their receptors (VEGFR, PDGFR) and the mammalian target of rapamycin (mTOR) [Banyra et al. 2014; Su et al. 2014].
These pathways can be blocked by monoclonal antibodies against VEGF (i.e. bevacizumab), inhibitors of VEGFR intracellular tyrosine-kinase activity (TKI: sunitinib, pazopanib, sorafenib and axitinib) and agents against TORC-1 and -2 complexes (mTOR-I: temsirolimus and everolimus) (Figure 1).

Mechanism of action of available targeted therapies in metastatic renal cell carcinoma. HIF, hypoxia inducible factor; mTOR, mammalian target of rapamycin; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor.
On the basis of pivotal phase III trials, bevacizumab plus interferon (IFN) α2a [Escudier et al. 2007b; Rini et al. 2010], sunitinib [Motzer et al. 2009], sorafenib [Escudier et al. 2007a] and pazopanib [Sternberg et al. 2010] are recommended by international guidelines [Escudier et al. 2014; Motzer et al. 2015] as first-line treatment for metastatic RCC (mRCC) with favorable and intermediate prognosis according to Motzer’s criteria [Motzer et al. 2000]. However, temsirolimus should be the first choice for patients with a poor prognosis [Hudes et al. 2007].
These drugs have radically changed the natural history of this disease by significantly prolonging both progression-free survival (PFS) and overall survival (OS), which reached 5–11 months and 19–26 months, respectively, and a reduction of the risk of death by 13% compared with cytokines [Iacovelli et al. 2015].
Despite the high activity of these drugs, a substantial number of patients experience progressive disease during first-line treatment. In a retrospective analysis, it was estimated that 52% of patients had access to second-line treatment [Levy et al. 2013], and based on reported clinical benefits, second-line treatment should always be offered to patients with adequate clinical conditions [Escudier et al. 2009]
In this setting there are many different options, including TKIs, sorafenib [Dudek et al. 2009; Sablin et al. 2009; Porta et al. 2011] and axitinib [Motzer et al. 2013], and mTOR-I, everolimus [Motzer et al. 2010] and temsirolimus [Mackenzie et al. 2011], which have all been demonstrated to increase control of the disease. More recently, several analyses have focused on differences between temsirolimus and everolimus [Iacovelli et al. 2014b], and one prospective trial reported a better outcome in favor of sorafenib compared with temsirolimus [Hutson et al. 2014], while no prospective data have compared everolimus with a TKI in second-line therapy. As a result, the best second-line therapy remains controversial and in daily clinical practice the choice is usually based on clinical aspects such as response to first-line treatment, residual toxicities, and clinician’s confidence with each drug.
This article examines the current role of everolimus in the second-line setting, focusing on the therapeutic benefit and safety profile.
Methods
This paper was designed to be a narrative review: sources of documents considered were PubMed/MEDLINE, and published abstracts from major conferences on clinical oncology, such as the American Society of Clinical Oncology and the European Society of Clinical Oncology meetings, as well as bibliographies reported in selected papers. Only papers investigating the role of everolimus in the second-line setting were included. The cutoff for the research was January 2015.
Mechanism of action of everolimus
About 65–70% of clear-cell RCCs are associated with loss of function of the Von Hippel-Lindau (VHL) oncosuppressor gene located on chromosome 3p [Kim and Kaelin, 2004]. One of the VHL protein’s targets are members of the hypoxia-inducible factor (HIF) α family, which are upregulated in normal cells when levels of oxygen are low. HIF in fact induces the expression of several genes involved in glucose uptake and metabolism (Glut-1), angiogenesis (VEGF) and cell growth (PDGFβ, transforming growth factor α) [Chen et al. 1995]. VHL normally inhibits HIF activity by the attachment of polyubiquitin tails, which allows the protein to be degraded by proteasome. When VHL is mutated or nonfunctioning, HIF is constantly active and favors the transcription of a cadre of genes involved in short-term and long-term adaptation to hypoxia.
By contrast, the mechanism of mTOR-I is quite distinct from that of bevacizumab and TKIs. As part of mTORC1 and mTORC2 complexes, mTOR has a central role in the regulation of cell growth: it is a downstream effector of the phosphatidylinositide 3-kinases/AKT (PI3K/AKT) pathway that receives input from multiple signals, including hormones, nutrients and other mitogens, and stimulates synthesis of different proteins implicated in cell growth and angiogenesis. It probably also leads to increased expression of HIF [Sarbassov et al. 2005; Thomas et al. 2006].
mTOR-I temsirolimus and everolimus are rapamycin analogues that induce an allosteric inhibition of mTORC1. In fact, these molecules bind to an intracellular protein called FKBP-12, which in turn ties mTORC1, thus blocking the mTOR serine/threonine kinase signaling. The interruption of mTOR signaling suppresses the production of proteins implicated in cell growth and angiogenesis [Sarbassov et al. 2005; Thomas et al. 2006; Brugarolas, 2007; Populo et al. 2012].
Prospective clinical studies
In a phase II study [Amato et al. 2009], 41 patients with mRCC were recruited: 34 (83%) were previously treated with systemic therapy, which for the large majority (61%) was immunotherapy such as interleukin 2 (IL-2), IFN or both, and nine received angiogenesis inhibitors or chemotherapy. All patients received everolimus at a dosage of 10 mg/day continuously and the primary endpoint of the study was PFS.
At the cutoff date, PFS was 11.2 months [95% confidence interval (CI) 1.7–36.2] with 57% of patients progression free for at least 6 months. Treatment was globally well tolerated with mostly grade 1 and 2 toxicities. The most common treatment-related adverse events (AEs) were anorexia (38%), nausea (38%), diarrhea (31%), stomatitis (31%) and rash (26%). Low-grade hematological and biochemical alteration including hyperglycemia, hypercholesterolemia and hypertriglyceridemia were also reported. Noninfectious pneumonitis occurred in 19 patients, and in seven of them this was grade 3, resulting in dose delays of 7–14 days; in four cases the dose was later re-escalated to 10 mg daily.
This spectrum of toxicities was consistent with those reported in previous phase I studies and in the experiences of patients who had received a transplant [O’Donnell et al. 2008; Tabernero et al. 2008; Ganschow et al. 2014] and considered as a class effect of mTOR-I.
Positive results of this study allowed a subsequent phase III study [Motzer et al. 2010], in which 410 patients were randomly assigned in a 2:1 ratio to everolimus 10 mg/day or placebo. All patients had histological-proven mRCC and had received at least one prior systemic treatment with a TKI targeting the VEGFR.
The results showed a median PFS of 4.9 months (95% CI 3.7–5.5) for everolimus and 1.9 (95% CI 1.8–1.9) for placebo (p < 0.0001), thus increasing the probability of being progression free at 6 months from 2% to 26%. Because of the preplanned possibility of crossover, 111 of 139 patients allocated to the placebo group also received everolimus; as a result, no significant difference in terms of OS was seen (14.8 months for everolimus versus 14.4 months for placebo) [Motzer et al. 2010]. This study also confirmed the safety profile of everolimus, with a very low proportion of grade 3 and 4 toxicities and few patients requiring dose interruption (34%) or reduction (5%). Noninfectious pneumonitis occurred in 37 patients, but only in 10 patients (three with grade 2 and seven with grade 3 disease) was treatment discontinued.
The majority of patients enrolled in this trial (79%) received everolimus after more than one previous systemic anticancer treatment, so that only 89 patients received everolimus as a pure second-line treatment. When stratified by previous therapy, 56 patients had received sunitinib and 30 had received sorafenib as the only previous treatment. A preplanned prospective subanalysis also confirms the efficacy of everolimus in this subgroup of patients, with significantly prolonged PFS from 1.8 to 4.6 months in the sunitinib group and from 1.9 to 3.8 months in the sorafenib group [Calvo et al. 2012]. This evidence suggests that everolimus provides a longer PFS in patients who have received only one previous line of therapy, thus it is recommended as a second-line treatment.
Another prospective study testing everolimus in patients with mRCC is RECORD-3. This was a phase II, randomized trial in which 471 patients were randomly assigned to the sequence of sunitinib followed by everolimus at tumor progression or the inverse [Motzer et al. 2014]. The primary endpoint was to assess the noninferiority in terms of PFS of first-line everolimus compared with sunitinib. The results of the study reported a median PFS of 7.9 and 10.7 months for everolimus and sunitinib, respectively [hazard ratio (HR) 1.4, 95% CI 1.2–1.8]; thus the primary endpoint was not met. Interestingly, the median combined PFS, defined as the time from randomization to death or progression, was 21.1 months for the sequence everolimus–sunitinib and 25.8 months for the inverse treatment (HR 1.3, 95% CI 0.9–1.7). Moreover, the final data reporting OS were in favor of the sequence sunitinib–everolimus (32.0 versus 22.4 months, HR 1.2, 95% CI 0.9–1.6), confirming that the sequence sunitinib–everolimus is superior and also showing that it can achieve an extremely important result in terms of OS [Motzer et al. 2013] comparable to the sequence sunitinib followed by sorafenib reported in the SWITC trial (30.2 months) [Michel et al. 2014].
More recently, the updated results of a prospective noninterventional study confirmed the activity of everolimus after VEGF-targeted therapy [Bergmann et al. 2015]. The study included more than 300 patients with a median follow up of 5.2 months. Of these, 63% of patients received everolimus as second-line treatment. The median treatment duration was 6.5 months and in patients who received everolimus as second-line treatment the median time to progression and PFS were 7.1 and 6.9 months, respectively.
Available data on PFS and OS from prospective studies are summarized in Table 1.
Prospective studies of everolimus in second-line treatment.

NIS, noninterventional trial; NR, not reached; OS, overall survival; PFS, progression-free survival; TKI, tyrosine kinase inhibitor.
Retrospective clinical studies and real-world data
Although clinical trials are obviously necessary, the patient populations involved in these studies are selected and often do not completely reflect ‘real life’ patients, as recently demonstrated in mRCC [Heng et al. 2014a]. Therefore, retrospective evaluation and expanded access programs can give information more reflective of clinicians’ daily practice [Rizzo et al. 2014].
In 2010, Vickers and colleagues retrospectively analyzed 216 patients receiving second-line therapy with anti-VEGFR TKI or mTOR-I after first-line treatment with anti-VEGFR TKI [Vickers et al. 2010]. In this study, the sequence TKI–TKI had longer median time to treatment failure (defined as the time from initiation of second-line treatment to interruption due to toxicity, progression or death) than the sequence TKI–mTOR-I (4.9 versus 2.5 months, p = 0.014); conversely, the median OS from the start of second-line treatment did not differ (14.2 versus 10.6 months). Nevertheless it is worth stressing that, beside the lack of any difference in terms of OS, the group of patients receiving mTOR-I as second-line treatment only included 24 patients (21 with temsirolimus and 3 with everolimus). Moreover, there were imbalances between the two groups in terms of non-clear cell histology (13% versus 2.8%, p = 0.045) and sarcomatoid features (13% versus 1%, p = 0.01), which may have negatively affected the outcomes of the mTOR-I group.
A German study retrospectively evaluated 108 patients receiving TKIs as first-line treatment (78.7% sunitinib and 21.3% sorafenib) [Busch et al. 2011]. Among them, 42 received another TKI as second-line treatment and 62 received everolimus. In this analysis, both approaches appeared to be equally efficacious in terms of second-line PFS (4.0 months for TKI versus 3.6 for everolimus, p = 0.455) and entire sequence duration (11.0 months for TKI–TKI versus 18.5 for TKI–everolimus, p = 0.051); instead, OS was significantly longer for everolimus (29.0 months versus 43.0, p = 0.034). Safety data were consistent with those from previous reports and predictably target related.
An interesting exploratory analysis of RECORD-1 data showed that everolimus can be safely used also in older patients [Porta et al. 2012]. Among the 416 patients enrolled in the RECORD-1 study, 36.8% were at least 65 years old and 17.5% were at least 70 years old; in this particular population, PFS was significantly increased compared with placebo (5.4 versus 2.2 for ⩾65 years and 5.1 versus 1.9 for ⩾70 years). In addition to these data, toxicities in older patients did not differ from those in the overall population. Stomatitis was the most common toxicity, numbers of grade 3 or 4 AEs were low (with anemia, lymphopenia, infection and hyperglycemia being the most common) and, surprisingly, noninfectious pneumonitis did not occur more frequently in this population. Patients at least 70 years old had a greater occurrence of dose reduction or interruption, and a higher mean number of concomitant therapies. Although several limitations may affect this analysis, such as its retrospective nature and the absence of patient stratification by age, it showed that everolimus is active in this particular population without jeopardizing its safety profile. This result is quite different from that of other retrospective analyses in which TKIs seem to be less well tolerated in older patients [Eisen et al. 2008; Van der Veldt et al. 2008].
The result reached by the RECORD-1 trial was also confirmed by another German retrospective analysis [Maute et al. 2014] of 81 patients receiving everolimus as second- (32) or third-line treatment (49), with a median treatment duration of 4.5 months.
Further data about sequences have recently been reported by the AVATOR study, in which 42 patients received bevacizumab with or without interferon as first-line treatment and everolimus as second-line treatment. Efficacy and safety were analyzed [Thiery-Vuillemin et al. 2015]. The median PFS for second-line treatment was 17.0 months (95% CI 5–Not Reached), while the median OS was not reached. These excellent results are also supported by a positive safety profile consistent with that from the RECORD-1 trial. Although dose reductions were more frequent than in the RECORD-1 trial (26% versus 7%), it should be noted that in the AVATOR study patients were included consecutively without strict selection; therefore patients usually ineligible for randomized clinical trials (such as patients with brain metastases, multiple sclerosis, ECOG-Performance Status 3, pulmonary embolism) were analyzed.
An Italian retrospective analysis reviewed 89 patients receiving mTOR-I or TKI as second-line treatment after sunitinib [Iacovelli et al. 2014a]. No significant differences between the two strategies were found and the PFS for 65 patients treated with everolimus was 4.3 months and the OS was 35.8 months, with a disease control rate of 60%. The results in terms of OS were consistent with other data reported by the same author in a previous study [Iacovelli et al. 2013].
More recently, another observational retrospective cohort study conducted in Italy [Rizzo et al. 2015] analyzed 100 consecutive patients whose disease had progressed on a prior anti-VEGFR TKI and were treated with everolimus. In this analysis, the overall disease control rate was quite high (81%), with a median PFS of 8 months. Although 20% of patients had to reduce everolimus because of adverse reactions, grade 3 toxicities were reported in only 15% of subjects and no grade 4 events were described.
The REACT study (RAD001 Expanded-Access Clinical Trial in RCC) was a global, open-label EAP that included over 1300 patients with mRCC from 34 different countries worldwide [Grünwald et al. 2012]. Over 60% of patients received at least two prior systemic therapies and a quarter of patients were heavily pretreated, having undergone more than three different lines of treatment. Despite the inclusion of patients with poor prognosis and non-clear-cell histology, the effectiveness and safety of everolimus were both confirmed. The disease control was reached in more than half of patients (53.3%) and also in those patients who had had rapid progressive disease on their last regimen (45.2%). From the point of view of tolerability, no new safety issues were identified and dose reduction or dose interruption because of AEs were similar to those observed in the RECORD-1 trial (48.1% versus 45%). Stomatitis, hematological, biochemical toxicities and dyspnea were confirmed to be the most frequent toxicities, while the incidence of any grade and high-grade noninfectious pneumonitis was 6.1% and 2.7%, respectively.
From large phase III trials to today, different experiences have been reported with everolimus and more and more information has been gathered. Nevertheless, there is still no clear indication about which second-line treatment is the best for patients with mRCC whose disease has progressed after first-line treatment. In a recent systematic review and meta-analysis of observational studies and ‘real-world’ experiences about second-line treatment, the authors conclude that it is not possible to determine whether TKI or mTOR-I is currently the best option [Heng et al. 2014b].
Considering the lack of strengthened data in favor of one sequence compared with the other, in clinical practice it is possible to use and inverse algorithms based on the safety profile [Bracarda et al. 2014]. Everolimus is characterized by a typical mTOR-oriented spectrum of toxicities with very few and mild toxicities overlapping with TKIs. This permits everolimus to also be effectively used in patients with residual TKI-related collateral effects or in those intolerant to TKIs [Bracarda et al. 2012]. In a subgroup analysis performed on subjects enrolled in the RECORD-1 trial, patients who switched to second-line everolimus because of unacceptable toxicity reached a median PFS of 5.4 months with a reduction of risk of disease progression by 68% compared with placebo (HR 0.32, 95% CI 1.8–3.7, p = 0.004) without unexpected toxicities and very low rates of grade 3 and 4 AEs. Available data on PFS and OS from retrospective studies are summarized in Table 2.
Retrospective studies of everolimus in second-line treatment.
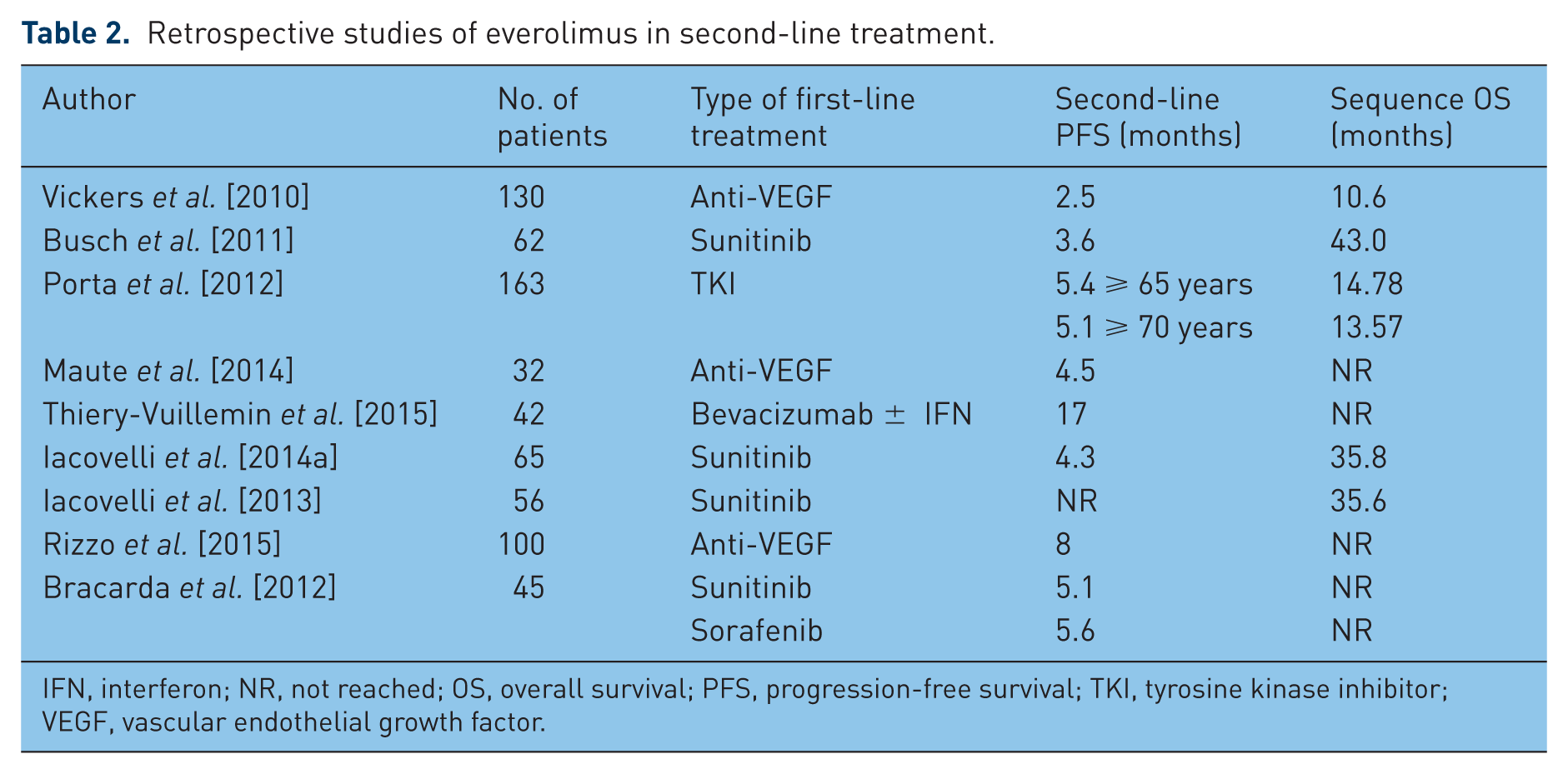
IFN, interferon; NR, not reached; OS, overall survival; PFS, progression-free survival; TKI, tyrosine kinase inhibitor; VEGF, vascular endothelial growth factor.
Conclusion
Besides mRCC, everolimus is also currently indicated for breast cancer and neuroendocrine pancreatic tumors on the basis of randomized phase III trials [Yao et al. 2011; Piccart et al. 2014]. These results showed the wide activity of this small molecule and the necessity of obtaining more and more information on its possible uses in medical oncology. Therefore, many different clinical trials are currently active and recruiting worldwide, especially in patients with renal cancer, which are investigating possible applications of this drug. These studies aim to evaluate everolimus as neoadjuvant [ClinicalTrials.gov identifier: NCT00831480] or adjuvant treatment [ClinicalTrials.gov identifier: NCT01120249], as a combined [ClinicalTrials.gov identifier: NCT02089334; NCT02077933; NCT01582009] or sequential approach [ClinicalTrials.gov identifier: NCT01545817], and many other indications.
Several prospective studies have shown the significant activity of everolimus as second-line treatment for patients with mRCC whose condition previously failed to respond to VEGF-targeted therapies and its safety has been confirmed by the results of various retrospective experiences.
In addition to this information, it would be of great interest to identify possible predictive factors of response to this drug in order to select patients who would benefit the most from it. Some data seem to show that expression levels of different proteins such as phospho-mTOR and phospho-S6RP may be potential predictive biomarkers for efficacy of everolimus in patients with mRCC [Li et al. 2014], but these findings still need prospective confirmation. Therefore, currently patient selection can only be based on clinical parameters.
Although different drugs are available as second-line treatment for mRCC, there is no doubt that everolimus represents a feasible and safe option, especially for patients who have experienced high-grade toxicity or are still carrying TKI-related toxicities from first-line treatment.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
