Abstract
Desmoid tumors are rare soft-tissue masses originating from the proliferation of fibroblasts in the fibroconnective tissues. Intra-abdominal desmoid tumors pose special diagnostic challenge due to multiplicity of differential diagnoses, and difficulty to well characterize the lesions on imaging studies. Desmoid tumors can have atypical presentation making clinical correlation challenging to unsuspecting urologists. Only a few cases have been reported in the urology literature. In this report, we present a case of desmoid tumor presenting with gross hematuria.
Introduction
Desmoid tumors are soft-tissue neoplasms characterized by the proliferation of fibroblasts in the deep fibrous connective tissue. They are rare (2–4 per million per year) with higher incidence in females. Incidence is more common in third and fourth decades of life with more aggressive tumors noted in the younger population. Incidence is usually sporadic, but can be associated with familial adenomatous polyposis (FAP), pregnancy, and surgical trauma. Their benign nature is suggested by non-encapsulated local infiltrating histological pattern, and lack of metastases; however, recurrence rates are high [Wong, 2008].
Common sites of desmoid tumor development are intra-abdominal, abdominal wall, trunk or extremities. Intra-abdominal desmoid tumors originate from the bowel, mesentery or retroperitoneum [Ferenc et al. 2006]. Intra-abdominal desmoid tumors usually present with symptoms of abdominal pain and bowel obstruction due to mass effect. Bowel segments are increasingly being used in the reconstruction of urinary system; however, there are rare reports of development of desmoid tumors in the urinary system [Choi et al. 2010; Karadeniz et al. 2010]. In this report, we present a rare case of gross hematuria due to development of desmoid tumor in a bladder augmentation.
Case report
A 22-year-old White woman presented with acute onset gross hematuria. She had neurogenic bladder due to spina bifida, and underwent bladder augmentation with Indiana augment approximately 14 years ago. On physical examination, a mass was palpable in the right lower abdominal quadrant extending from her pelvis. Computed tomography (CT) scan of her abdomen and pelvis with intravenous and oral contrast revealed a large (11.9 cm × 7.3 cm) hyper-dense pelvic mass with smooth margins and punctate calcifications. Pelvic ultrasound further confirmed solid nature of the mass. Mass was abutting urinary bladder, small bowel, and right adenexa as shown in Figure 1.
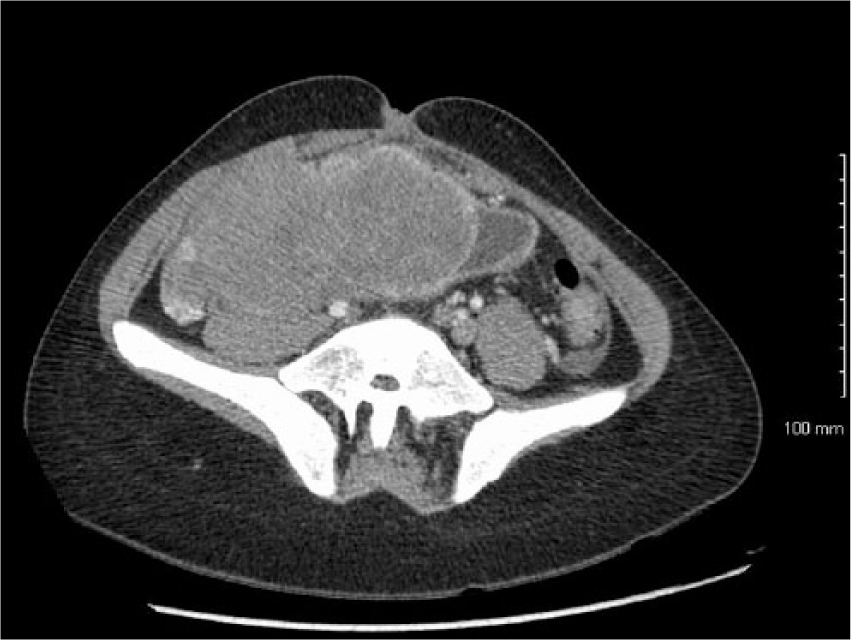
Computed tomography scan of abdomen and pelvis obtained at the initial evaluation shows a large hyper-intense pelvic mass with smooth margins involving the urinary bladder.
After initial evaluation, she underwent cystoscopy with clot evacuation. During cystoscopy, a mass was noted originating from the colonic portion of bladder augmentation. Biopsies of the mass were obtained with a resectoscope and sent for pathological examination. In addition, ultrasound-guided deep-core biopsies were also obtained to further characterize the pelvic mass.
Pathology of the biopsies was consistent with desmoid tumor (Figure 2). Spindled cells and stellate cells with a variable amount of collagen matrix were noted. The tumor cells were diffusely positive for vimentin and focally positive for desmin and actin on immunohistochemistry. Immunostaining for pancytokeratin, melan A and CD34 were negative consistent with the diagnosis of desmoid tumor.

Hematoxylin and eosin staining of biopsies intra-abdominal mass shows spindle and stellate cells with interspersed collagen matrix (10× magnification).
Cystectomy with complete excision of the pelvic mass and ileal conduit urinary diversion was performed. A segment of small bowel with right adenexa was also excised due to involvement by the mass. She had uncomplicated postoperative course and was discharged home on postoperative day 6. Final pathology showed desmoid tumor, and resection margins were negative.
Discussion
Desmoid tumors are rare lesions that develop due to clonal proliferation of fibroblasts in the fibroconnective tissue. Its local infiltrating pattern leads to involvement of multiple organs and can result in unusual symptoms. Concerning our case, desmoid tumor developed from the bowel segment used for Indiana augment and infiltrated in the urinary bladder wall. This resulted in the unusual presentation of gross hematuria.
Intra-abdominal desmoid tumor development is associated with FAP, young age, previous surgical trauma, female gender, and increased estrogenic state [Wong, 2008]. Previous surgery, gender and young reproductive age of our patient put her at increased risk of development of desmoid tumor. On CT scan, desmoid lesions have expansile, homogeneous or heterogeneous appearance with an infiltrative pattern, and calcifications are often noted. Desmoid lesions can be hypo-, iso- or hyper-intense. In our case, the lesion was hyper-intense with calcifications and infiltrative pattern. Intra-abdominal desmoid tumor can be challenging to diagnose on the imaging studies due to possibility of multiple differential diagnosis such as lymphoma, metastases, and gastrointestinal tumors [Kreuzberg et al. 2007]. Because of this diagnostic difficulty on imaging, clinical correlation with symptoms and biopsy of mass is required. Urologists, while evaluating for gross hematuria with CT urogram and cystoscopy, will likely have low suspicion for the desmoid tumor making clinical correlation challenging.
Preoperatively desmoid tumor can be reliably diagnosed on core needle biopsy specimen. The simplicity of core biopsy needle technique and adequate tissue sampling allows high diagnostic accuracy [Dalen et al. 2006]. Our patient underwent ultrasound-guided core needle biopsies in addition to transurethral resection to accurately characterize the nature of mass. Histologically, desmoid tumors are composed of proliferation of bland spindle-shaped cells with interspersed collagen fibers. Cells in desmoid lesions are positive for vimentin, actin and sometimes desmin, as was seen with our case [Kreuzberg et al. 2007; Owens et al. 2007].
Surgical resection is the predominant treatment for resectable desmoid tumors. However, the large size of the tumor and local infiltration into the surrounding organs can make the surgical resection challenging. Preoperative counseling about the possibility of excising adjacent organs that may be involved is imperative. Freedom from recurrence has been noted in up to 80% of patients at 5 years. For the unresectable intra-abdominal desmoid tumor systemic chemotherapy is considered a good alternative. Systemic chemotherapeutic agents such as tamoxifene, doxorubicin, and sulindac have been tried with variable response. Patients with a good response to chemotherapy can be considered for surgical excision if the tumor regresses making it resectable. However, patients with a poor response to chemotherapy should be considered for radiation therapy [Lev et al. 2007]. During surgical resection, attempt should be made to obtain negative margins, however, the significance of negative margins is not well understood as it is not an independent predictor for the recurrence of desmoid tumors. Patients with microscopic positive margins can be observed for recurrence, however, follow-up protocols for desmoid tumors are not well established. The use of adjuvant radiation therapy for grossly positive margins is controversial [Melis et al. 2008].
Our patient did not undergo any follow-up surveillance cystoscopy. She was referred only after development of gross hematuria. Due to concern for development of malignancies, it is widely recommended to perform surveillance cystoscopy and urine cytology 10 years after the enterocystoplasty procedure. However, due to low risk of malignancies and enhanced cost containment, some recent studies have questioned this practice and recommend cystoscopy after development of symptoms such as hematuria, recurrent urinary tract infection (UTI) or bladder pain [Hamid et al. 2009]. In this case, surveillance cystoscopy could have identified the tumor earlier with the possibility of altering the management.
In conclusion, desmoid tumor should be considered in patients with prior urinary tract reconstruction using bowel segments. Surveillance cystoscopy with urine cytology should be offered to the patients with enterocystoplasty 10 years after the augmentation, and should be performed without delay if the patient is symptomatic. Imagining studies with CT scans can help rule out the other etiologies of abdominal mass and metastases. However, core needle biopsies of mass should be obtained to characterize the mass preoperatively. Complete surgical resection of the mass should be attempted followed by surveillance for any recurrence.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, or commercial or not-for-profit sectors.
