Abstract
Background:
Leucine-rich glioma-inactivated 1 (LGI-1) antibodies are the second most common cause of autoimmune encephalitis, which is characterized by frequent seizures, memory loss, and psychiatric symptoms. Although most patients respond well to immunotherapy, treatment is often delayed or insufficient, resulting in a 14%−35% relapse rate and persistent neurological deficits. Therefore, exploring fast-acting and targeted therapies is crucial.
Objectives:
To present a series of cases involving anti-LGI1 encephalitis treated with efgartigimod, and to assess the efficacy and safety of this therapeutic approach in managing this condition.
Design:
A prospective, multicenter, and observational study.
Methods:
This study prospectively enrolled patients with anti-LGI1 encephalitis treated with efgartigimod and conducted a systematic review of the literature on the use of efgartigimod for anti-LGI1 encephalitis. The clinical outcomes before and 2 weeks after treatment were then compared.
Results:
A total of eight patients and two additional cases from the literature were included in the study, with a mean age of 55.6 ± 15.88 years and a male-to-female ratio of 6:4. The predominant clinical manifestations among the patients included seizures, psychiatric and behavioral abnormalities, and memory decline. Following treatment with efgartigimod, the modified Rankin scale (p = 0.006; 95% confidence interval (CI) 2–2.5), Clinical Assessment Scale in Autoimmune Encephalitis (p = 0.008; 95% CI 2.5–9.5), and Mini-Mental State Examination scores (p = 0.042; 95% CI 0–14) significantly improved. In addition, a significant reduction was observed in both serum IgG levels (p < 0.001; 95% CI 4.66–6.81) and antibody titers (p = 0.004) post-treatment, indicating that the degree of antibody decline is negatively correlated with clinical severity. No adverse events were reported for any of the patients in this study.
Conclusion:
This study suggests that efgartigimod may offer promising efficacy and safety for patients with anti-LGI1 encephalitis. Further randomized controlled trials involving larger cohorts and extended follow-up periods are required to validate these findings.
Plain language summary
Introduction
Leucine-rich glioma-inactivated 1 (LGI1) is a secreted neuronal glycoprotein that is expressed mainly in the hippocampus and temporal cortex. 1 It forms a transsynaptic complex with the presynaptic proteins ADAM11 and ADAM23 and the postsynaptic protein ADAM22, playing a role in the regulation of synaptic transmission and neuronal excitability.2,3 Antibodies targeting the LGI1 protein belong to the IgG4 subtype, and may cause a reduction in Kv1.1 potassium channels and α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptor, leading to the severe impairment of neuronal transmission, plasticity, and memory. 4 Anti-LGI1 autoimmune encephalitis (AE) is the second most common subtype of AE-related to anti-neuronal surface antibodies, resulting in significant neurological and psychiatric symptoms. 5 It has an annual incidence of 0.83 cases per million people, and is more prevalent in males. 1 This condition typically presents with a subacute onset of memory decline, psychiatric behavioral abnormalities, and seizures. 6 Approximately 90% of patients experience seizures, and faciobrachial dystonic seizures (FBDS) are the typical feature of anti-LGI1 encephalitis. 1
The first-line treatments for anti-LGI1 encephalitis typically include high-dose corticosteroids, intravenous immunoglobulin (IVIg), plasma exchange (PE), and immunoadsorption.7,8 The second-line treatments primarily involve anti-CD20 monoclonal antibodies and cyclophosphamide. 9 However, there are several limitations to these treatments. High-dose and prolonged corticosteroids predispose patients to metabolic disturbances, and the incidence of new-onset diabetes can be as high as 15%−40% in treated patients. 10 Treatment with anti-CD20 monoclonal antibodies significantly increases the risk of infection. A retrospective study reported an overall infection rate of 34.3%, 11 with the incidence of serious infections, ranging from 2.2 to 9.8/100 person-years. 12 IVIg and PE rely on limited blood resources; PE and immunoadsorption are invasive procedures and are not typically considered by most patients.13,14 Although the response rate to immunotherapy in patients with anti-LGI1 encephalitis varies between 67% and 92%, 15 treatment is often delayed or insufficient, resulting in a 14%−35% relapse rate1,16 and persistent neurological sequelae, including seizures and cognitive impairment. 17 Therefore, effective, targeted, and well-tolerated treatments for anti-LGI1 encephalitis are urgently needed.
Neonatal crystallizable fragment receptor (FcRn) plays a key role in protecting IgG from lysosomal degradation. 18 Targeting FcRn may offer a promising therapeutic opportunity for autoimmune disease by promoting the rapid clearance and degradation of IgG. 19 Efgartigimod, an FcRn inhibitor, was first approved in 2021 in the USA for the treatment of generalized myasthenia gravis in adults with anti-AChR antibodies. 20 Furthermore, efgartigimod has demonstrated good therapeutic effects in autoimmune diseases such as primary immune thrombocytopenia and chronic inflammatory demyelinating polyradiculoneuropathy. 21 Emerging evidence also indicates that efgartigimod offers a promising therapeutic profile for anti-NMDAR encephalitis.22,23 Nevertheless, investigations into the application of efgartigimod for the treatment of anti-LGI1 encephalitis are currently confined to individual case reports.
This study aimed to document a series of cases involving anti-LGI1 encephalitis patients treated with efgartigimod and to evaluate its efficacy and safety in the treatment of this condition.
Methods
Study design
This study was a prospective observational analysis conducted at multiple centers. Between April 2024 and September 2025, a total of eight patients with anti-LGI1 encephalitis were enrolled at Beijing Tiantan Hospital and the First Affiliated Hospital of Zhengzhou University. Specifically, data were collected from four patients at each institution. The results from the ADAPT+ study 24 demonstrated that clinical improvement began at week 2 following the initiation of efgartigimod treatment, with peak efficacy achieved by week 4. Therefore, we collected clinical data at baseline and at 2 weeks after the treatment.
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, as recommended by the EQUATOR Network.
We carried out a comprehensive and systematic search of the available literature on the use of efgartigimod for anti-LGI1 encephalitis. The literature retrieval platforms included WanFang Data (http://www.wanfangdata.com.cn/), CNKI (https://www.cnki.net/), PubMed (http://www.ncbi.nlm.nih.gov/pubmed/), and Web of Science (https://webofscience.clarivate.cn/wos/alldb/basic-search) for articles up to September 2025 by using the title/abstract keywords “efgartigimod” and “LGI1.” A total of two documented cases of anti-LGI1 encephalitis treated with efgartigimod were identified.25,26
Patients
The inclusion criteria were as follows: (1) patients of any age with a definitive diagnosis of anti-LGI1 encephalitis according to previous diagnostic criteria27,28; (2) a baseline modified Rankin scale (mRS) score ⩾3 before starting efgartigimod treatment; (3) treatment with efgartigimod for 2–4 weeks; and (4) complete and assessable clinical follow-up data. The exclusion criteria included (1) loss to follow-up; (2) positivity for other antibodies, such as anti-NMDAR antibodies and anti-GAD65 antibodies; (3) severe comorbidities, such as heart failure, coagulation disorders, and tumors.
Treatment
All patients received first-line treatments, including intravenous methylprednisolone (IVMP) and/or IVIg. IVMP was started at a dosage of 1000 or 500 mg/day for 3 days, then gradually reduced to oral prednisone at 1 mg/kg body weight and reduced by 5 mg every 2 weeks. IVIg was administered at a dosage of 0.4 g/kg/d for 5 days. Efgartigimod was administered at a dosage of 10 mg/kg once weekly for 2–4 weeks.
Data acquisition and evaluation of treatment efficacy
Demographics and clinical data were extracted from clinical records, including gender, age, clinical symptoms, electroencephalogram (EEG), magnetic resonance imaging (MRI), positron emission tomography (PET), cerebral spinal fluid findings, the level of IgG, antibody titers, serum sodium concentration, neurological functional status, outcomes, and treatment. Neurological functional status and outcomes were assessed using the mRS, Clinical Assessment Scale in Autoimmune Encephalitis (CASE), Mini-Mental State Examination (MMSE), and Montreal Cognitive Assessment (MoCA) at admission and discharge. All baseline assessments, including EEG, MRI, and PET, were performed within the first week of hospitalization and before the initiation of efgartigimod treatment. The comparison of neurological function status, antibody titers, and IgG levels before and after treatment was performed in all 10 patients to assess the efficacy of efgartigimod therapy.
The primary endpoint of this study was the change in CASE scores from baseline to 2 weeks post-treatment. Secondary endpoints included changes in the mRS, MMSE, MoCA scores, LGI1 antibody titers, and serum IgG levels.
Assessment of efgartigimod safety
Efgartigimod safety was evaluated by assessing the incidence, frequency, and severity of all adverse events, such as respiratory and urinary tract infections, hepatorenal dysfunction, allergic reactions, and others.
Statistical analysis
SPSS 24.0 software for Windows was used for statistical analysis (IBM Corp., Armonk, N.Y., USA). Data from eight prospectively enrolled patients and two literature-reported cases were pooled for analysis. The normality of the data distribution of continuous variables was tested by the Shapiro–Wilk test. Normally distributed data were shown as mean ± standard deviation, and group comparisons were performed using the paired t test, with results reported as the mean difference and its 95% confidence interval (CI). Non-normally distributed data were presented as median (interquartile range (IQR)), and group comparisons were performed using the Wilcoxon signed-rank test. For these parameters, the median difference and its 95% CI were estimated using the Hodges–Lehmann method. Categorical variables were compared by the Chi-square test or Fisher’s exact test and are shown as frequencies (proportions). A p-value < 0.05 (two-sided) was considered statistically significant.
Results
Clinical characteristics
A total of 10 patients with anti-LGI1 encephalitis were included in this cohort. The detailed demographic and clinical characteristics of the patients before and 2 weeks after efgartigimod treatment are summarized in Table 1. The mean age of these patients was 55.6 ± 15.88 years (male:female = 6:4). The most common clinical manifestations included seizures (8/10), psychiatric behavioral abnormalities (5/10), and memory decline (4/10). Five patients experienced FBDS. Additionally, hyponatremia was observed in five patients. EEG abnormalities were present in 7 out of 10 patients, predominantly showing slow waves in the frontal (2/7) or temporal region (2/7) or in both the frontal and temporal regions (2/7). Eight patients in our cohort underwent brain MRI scans. Among them, T2/fluid-attenuated inversion recovery hyperintensity in the amygdala and/or hippocampus was observed in seven patients (7/8), and two of these patients (2/7) also exhibited hyperintensity in the basal ganglia. Four patients underwent 18 F-FDG-PET, revealing hypermetabolism in the basal ganglia or temporal lobes.
Clinical characterization of patients with anti-LGI1 encephalitis.

CASE, Clinical Assessment Scale in Autoimmune Encephalitis; CSF, cerebral spinal fluid; EEG, electroencephalogram; F, female; FBDS, faciobrachial dystonic seizures; GTCS, generalized tonic-clonic seizure; IVIg, intravenous immunoglobulin; IVMP, intravenous methylprednisolone; LGI1, leucine-rich glioma-inactivated 1; M, male; MMSE, mini-mental state examination; MoCA, Montreal cognitive assessment; MRI, magnetic resonance imaging; mRS, modified Rankin scale; NA, not available; PET, positron emission tomography.
Efficacy analysis
The median dosage of efgartigimod was 1600 mg (IQR 1600–3200). Both the IgG levels (p < 0.001; 95% CI 4.66–6.81) and serum antibody titers (p = 0.004) decreased significantly after treatment. Clinical neurological function was assessed with the mRS and CASE scores at admission and discharge (Figure 1). The mRS (p = 0.006; 95% CI 2–2.5; Table 2) and CASE (p = 0.008; 95% CI 2.5–9.5; Table 2) scores significantly decreased after treatment with efgartigimod. Cognitive function was assessed with the MoCA or MMSE scores, and the MMSE scores significantly improved after treatment with efgartigimod (p = 0.042; 95% CI 0–14; Table 2). Although no significant difference was observed in the MoCA score (p = 0.068; 95% CI 0–11) between pre- and 2 weeks post-treatment, a downward trend was observed after treatment.
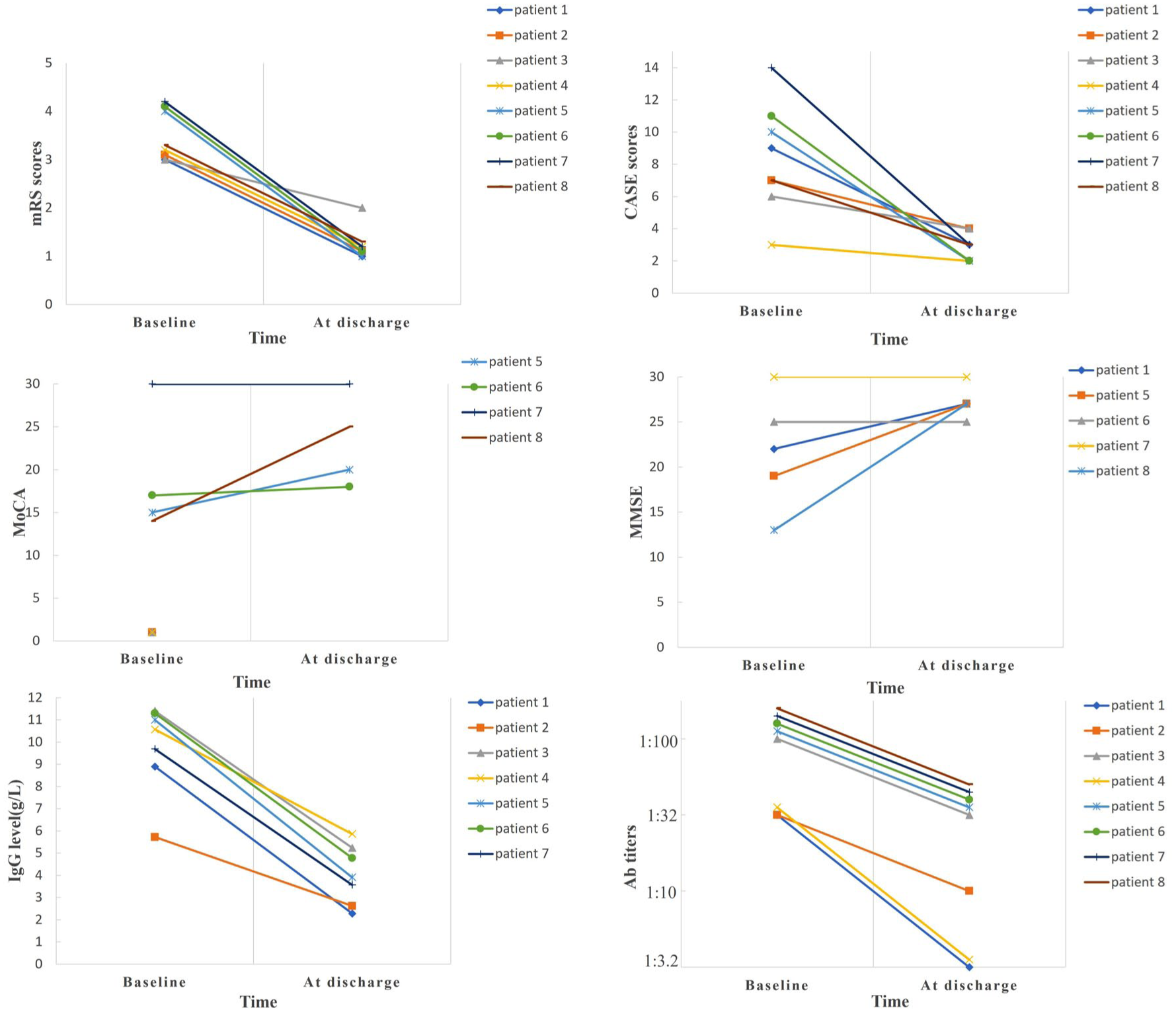
Distribution of mRS, CASE, MoCA, MMSE, IgG level, and antibody titers at admission and at discharge in patients with anti-LGI1 encephalitis.
Efficacy outcomes in all patients.
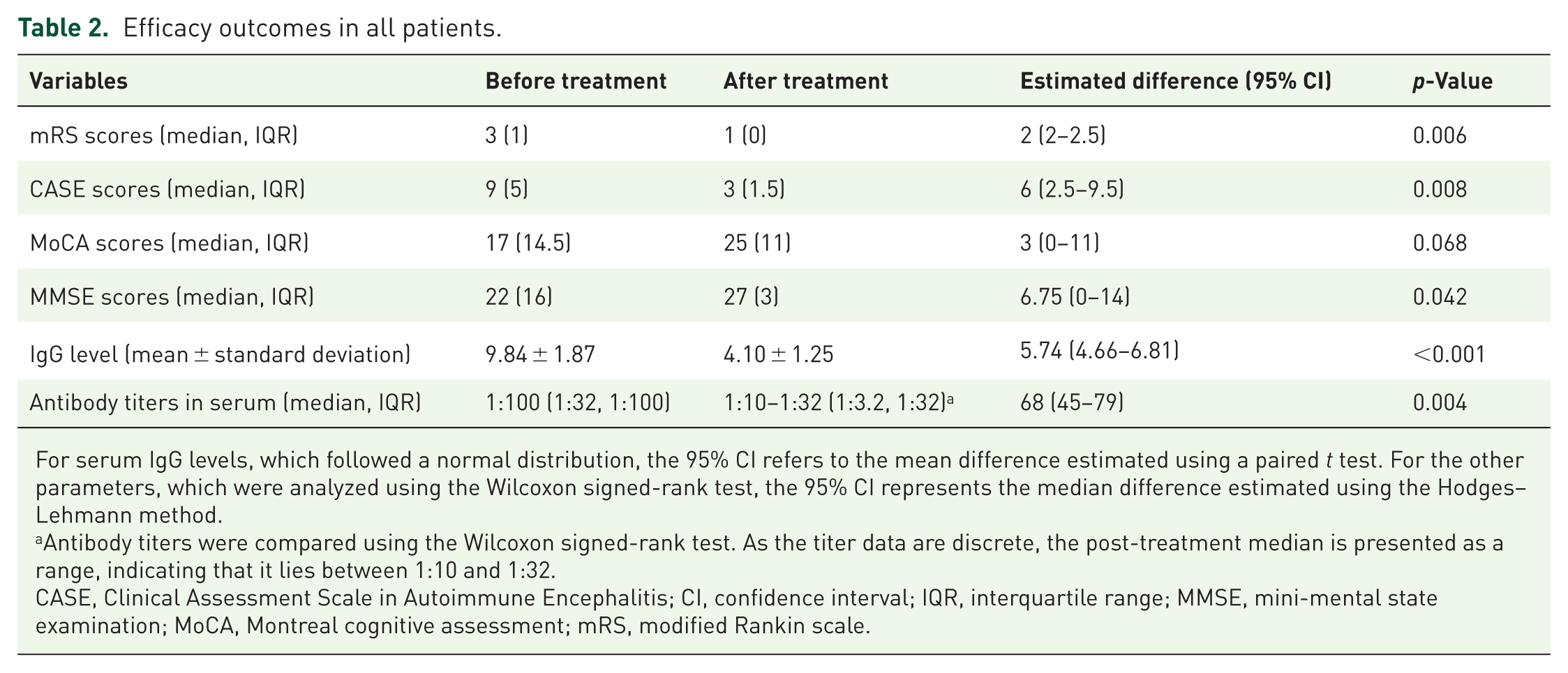
For serum IgG levels, which followed a normal distribution, the 95% CI refers to the mean difference estimated using a paired t test. For the other parameters, which were analyzed using the Wilcoxon signed-rank test, the 95% CI represents the median difference estimated using the Hodges–Lehmann method.
Antibody titers were compared using the Wilcoxon signed-rank test. As the titer data are discrete, the post-treatment median is presented as a range, indicating that it lies between 1:10 and 1:32.
CASE, Clinical Assessment Scale in Autoimmune Encephalitis; CI, confidence interval; IQR, interquartile range; MMSE, mini-mental state examination; MoCA, Montreal cognitive assessment; mRS, modified Rankin scale.
At the 3-month follow-up, three patients remained seizure-free without recurrence of psychiatric and behavioral symptoms, and repeated testing showed no increase in serum antibody titers.
Safety analysis
Throughout the entire follow-up period, no adverse events related to efgartigimod, such as infections, allergic reactions, or hepatic abnormalities, were observed in this study.
Discussion
Anti-LGI1 encephalitis is mediated primarily by the IgG4 antibody subclass.5,29 Short-term memory impairments were observed in 37% of patients during long-term follow-up. 17 Current immunotherapies, including IVMP, IVIg, and PE, aim to reduce inflammation and modulate immune responses.7,9 However, their application is limited by side effects, limited efficacy, or high invasiveness. IVIg demonstrates limited efficacy in IgG4-mediated neurologic autoimmunity, such as MuSK antibody-positive myasthenia,30,31 and may also have a restricted role in anti-LGI1 encephalitis. In contrast, efgartigimod, an Fc segment derived from human IgG1, 21 offers a targeted and less invasive alternative. It exhibits enhanced affinity for FcRn under acidic pH conditions, thereby specifically blocking the binding of FcRn to IgG and promoting the degradation of pathogenic antibodies. Moreover, unlike PE, IVIg, and immunoadsorption, it causes no significant changes in other immunoglobulin classes (IgA, IgM, IgD, and IgE), making it a novel and targeted therapeutic strategy.21,32 It has also been demonstrated that compared with IVIg and PE, efgartigimod reduces the level of IgG4 by 52%, 33 indicating superior efficacy. Additionally, a recent study indicated that efgartigimod can induce B cells to differentiate into plasma cells with a regulatory phenotype, which may be another important mechanism underlying clinical improvement. 34
This study represents the first to report a larger cohort of eight patients, providing preliminary evidence that efgartigimod may offer an additive therapeutic benefit in anti-LGI1 encephalitis when combined with IVMP and/or IVIg. Our findings suggest that the concomitant use of efgartigimod alongside IVMP or IVIg may contribute to rapid and significant clinical improvements, including in mRS scores, CASE scores, and MMSE scores. The lack of significant differences in the MoCA scores may be explained by the limited sample size or missing data. Similar to previous studies,19,35 in our study, efgartigimod effectively and rapidly reduced circulating IgG levels and anti-LGI1 antibody titers. These findings suggest that clearing pathogenic IgG antibodies with efgartigimod may promote faster recovery and a better prognosis. Notably, no adverse events, such as respiratory or urinary tract infections or hepatorenal dysfunction, were observed, demonstrating that efgartigimod is well tolerated in patients with anti-LGI1 encephalitis.
Our findings are consistent with the case reports by Zhang et al. 26 and Zhu et al. 25 By systematically assessing a larger cohort, we provide additional clinical evidence for the effectiveness of efgartigimod in anti-LGI1 encephalitis. Notably, our consistent observation of concurrent reductions in antibody titers and IgG levels, alongside clinical improvement, suggests a direct relationship between the reduction in pathogenic antibodies and clinical recovery in patients with anti-LGI1 encephalitis. Furthermore, efgartigimod has exhibited efficacy in a broader range of patients with AE. A retrospective single-center study demonstrated that the combination of efgartigimod and IVMP resulted in significant improvements in both mRS and CASE scores among patients with AE, particularly within 2 weeks after treatment. 35 Another retrospective study revealed that cognitive deficits were observed in fewer patients receiving efgartigimod, suggesting that it may improve cognitive function more effectively than other treatments. 22 Moreover, a case–control study comparing the treatment of acute-phase anti-NMDAR encephalitis revealed that efgartigimod was superior to IVIG and comparable to immunoadsorption in reducing antibody titers. 23 The efficacy of efgartigimod observed in our cohort is consistent with its reported success in anti-NMDAR encephalitis. Our study further extends the potential applications of efgartigimod. Anti-NMDAR encephalitis is primarily mediated by IgG1, whereas anti-LGI1 encephalitis is mediated by IgG4. These findings suggest that efgartigimod may offer promising therapeutic potential for IgG4-mediated autoimmune diseases, which is related to its mechanism of lowering all IgG subtypes in serum. Notably, the safety profile was favorable, with no significant adverse events observed in this study. These results align with those of previous studies.33,35,36 To further clarify the long-term effectiveness and safety of the treatment, we plan to conduct follow-up assessments of the patients in this cohort at 3, 6, and 12 months after the completion of efgartigimod treatment. The evidence in this research suggests a potential principle: the targeted clearance of pathogenic IgG antibodies represents a promising therapeutic strategy for autoimmune diseases, and FcRn inhibitors may have good therapeutic potential for IgG4-mediated autoimmune diseases.
Currently, in addition to its use as an adjunctive therapy for AE, efgartigimod has demonstrated promising efficacy in multiple-antibody-positive and refractory AE. Xu et al. 37 reported a case with co-existing NMDAR and LGI1 antibodies who failed to respond to initial IVMP and IVIg, but experienced significant improvement in psychiatric symptoms following efgartigimod treatment. Tu et al. 38 similarly reported the successful sequential treatment with ofatumumab followed by efgartigimod for refractory AE with dual anti-NMDAR and anti-GFAP antibody positivity. Furthermore, a recent study showed that efgartigimod may serve as an alternative to both IVIg and immunoadsorption therapies for anti-NMDAR encephalitis during acute attacks, 23 demonstrating its potential value in early-stage disease.
This study has several limitations. First, the sample size was small; therefore, while it offers valuable preliminary clinical evidence, the findings necessitate validation through larger-scale studies. Second, efgartigimod was administered concomitantly with IVMP and/or IVIg in all patients. In addition, no comparative analysis was performed among the three treatment groups: IVMP plus efgartigimod, IVMP plus IVIg, and IVMP alone; therefore, the independent effect of efgartigimod cannot be isolated from the effects of IVMP and IVIg. Third, due to the absence of long-term follow-up, the durability of efficacy and long-term safety of efgartigimod remain unclear. Finally, although incorporating two cases from the literature increased the overall cohort size, the potential for methodological heterogeneity should be noted. Future studies should involve larger, multicenter, prospective randomized controlled trials with extended follow-up periods to better evaluate the efficacy and safety of efgartigimod.
Conclusion
In conclusion, this study suggests the therapeutic potential of efgartigimod in anti-LGI1 encephalitis, indicating rapid clinical improvement and good tolerability when combined with IVIg and/or IVMP. Further randomized controlled trials involving larger cohorts and extended follow-up periods are required to validate these findings. The application of FcRn antagonists is anticipated to extend to IgG4-related autoimmune diseases.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261437004 – Supplemental material for Preliminary observations on the efficacy of efgartigimod in anti-LGI1-associated autoimmune encephalitis
Supplemental material, sj-docx-1-tan-10.1177_17562864261437004 for Preliminary observations on the efficacy of efgartigimod in anti-LGI1-associated autoimmune encephalitis by Jing-Wen Zuo, Ying-Yue Dai, Wen-Jing Liu, Jia-Ying Zhang, Xiao-Qiu Shao, Qun Wang and Rui-Juan Lv in Therapeutic Advances in Neurological Disorders
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
