Abstract
Background:
Intravenous thrombolysis (IVT) administered as a bridging therapy prior to endovascular thrombectomy (EVT) for acute ischemic stroke may increase the risk of hemorrhagic-transformation (HT). CT-perfusion (CTP) imaging enables quantitative assessment of ischemic core and penumbral tissue and may support individualized hemorrhagic risk stratification.
Objectives:
To assess whether baseline CTP-defined ischemic core parameters are associated with parenchymal-hematoma type 2 (PH-2) following bridging IVT before EVT.
Design:
Observational-cohort-study.
Data sources and methods:
Consecutive patients with large-vessel occlusion treated within 4 h of symptom onset at two tertiary stroke centers between 2017 and 2023 were analyzed. All patients underwent baseline CTP imaging. Outcomes were compared between patients treated with IVT plus EVT and those treated with direct-EVT. HT was assessed on 24-h follow-up noncontrast-CT using ECASS-2 criteria. Multivariable logistic regression was performed to identify independent predictors of PH-2.
Results:
Among 398 patients (50.6% male), 180 received IVT + EVT (mean age 70.2 ± 15.0 years) and 218 underwent direct-EVT (69.5 ± 14.5 years). Baseline characteristics, workflow times, thrombectomy passes, and reperfusion rates were comparable. PH-2 was associated with higher mortality (43.8% vs 11.7%, p < 0.001) and worse 90-day functional outcome (median modified Rankin Score 4 (2–6) vs 3 (1–4), p = 0.002). Bridging-IVT was associated with higher PH-2 rates compared with direct EVT (6.1% vs 2.0%, p = 0.036), without increased symptomatic intracranial-hemorrhage. In sensitivity analyses, bridging-IVT was associated with higher PH-2 rates only in patients with any ischemic core (7.8% vs 1.5%, p = 0.014), core volume >10 mL (8.8% vs 0.8%, p = 0.004), and >20 mL (8.7% vs 1.3%, p = 0.03). Penumbral volume was not associated with PH-2. In multivariable analysis, any ischemic core (odds ratio (OR) 12.67, p = 0.02) and core volume >10 mL (OR 11.12, p = 0.034) independently predicted PH-2.
Conclusion:
Baseline CTP-defined ischemic core volume is strongly associated with severe HT following bridging intravenous alteplase prior to EVT and may inform individualized risk–benefit assessment of bridging therapy.
Plain language summary
When someone has a major stroke caused by a large blood clot blocking blood flow in the brain, doctors have two main emergency treatments. One is giving a medicine through a vein (called “clot-busting” or thrombolysis) to help dissolve the clot. The other is a procedure called thrombectomy, where doctors physically remove the clot using a catheter. Sometimes, both treatments are given together, with the medicine given first while preparing for the procedure. A concern with clot-busting medicine is that it can raise the risk of bleeding in the brain, a complication that can make recovery worse or even be life-threatening. Today, many hospitals routinely perform advanced brain scans (called CT perfusion scans) that show how much brain tissue is already severely injured (the “core”) and how much could still be saved (the “penumbra”). In this study, nearly 400 patients who arrived at two large stroke centers within four hours of symptom onset were analyzed. About half received both the medicine and thrombectomy, and half had thrombectomy alone. The two groups were otherwise very similar. The researchers found that patients who received both treatments were more likely to develop a serious type of bleeding called “parenchymal hematoma type 2” – but only if their brain scans already showed damaged brain tissue (core). This higher bleeding risk was especially clear when the damaged core was larger than 10 milliliters. Importantly, the size of the at-risk but not yet dead tissue (penumbra) did not affect bleeding risk. These results suggest that clot-busting medicine may be safer in patients whose scans show little or no permanently damaged brain tissue, while those with larger damaged areas might do better with thrombectomy alone. Using advanced CT scans may help tailor treatment and avoid harmful side effects.
Introduction
Endovascular thrombectomy (EVT) is indicated for reperfusion therapy in acute ischemic stroke (AIS) due to large vessel occlusion (LVO). 1 The role of intravenous thrombolysis (IVT) as bridging treatment prior to EVT, however, remains debated. While current guidelines continue to recommend IVT in all eligible LVO patients, 1 studies examining bridging treatment, including six randomized control trials and several meta-analyses,2–4 have yielded inconclusive results regarding its added clinical benefit. This uncertainty underscores the need for refined risk–benefit stratification to optimize patient selection for the combined IVT + EVT approach. We sought to improve HT risk stratification following bridging IVT and to delineate subgroups at elevated risk for clinically significant events.
Previously published meta-analyses showed the overall positive effect of bridging IVT on successful recanalization rates.2,4 Generally, the benefit of IVT is greatest within the first hours of symptom onset and diminishes thereafter. 5 However, the current knowledge regarding patient selection that will be adversely affected by bridging IVT is limited. Unfortunately, the longer the brain parenchyma is subjected to ischemia, the more susceptible it becomes to hemorrhagic transformation (HT) in the setting of LVO. 6 Notably, recent studies have shown that initiating IVT bridging more than 2 h after symptom onset is associated with higher rates of HT.7,8
Nevertheless, parenchymal ischemic changes are not entirely time-sensitive and may vary according to various factors such as severity of ischemia, collateral circulation, tissue vulnerability to ischemia, and various systemic metabolic conditions. 9 Observational data suggest that patients with extensive ischemic injury, reflected by lower Alberta Stroke Program Early CT Score (ASPECTS), have an excess risk of symptomatic ICH (sICH) when treated with IVT + EVT compared to EVT alone. 10 Modern CT perfusion (CTP) imaging provides detailed estimates of ischemic core and penumbral tissue, and is increasingly used to guide EVT decision-making in LVO-AIS 11 ; therefore, given its widespread availability, CTP may also enable individualized risk stratification for HT after bridging IVT. In this study, we investigated the predictive value of baseline CTP parameters for HT following bridging IVT.
Methods
Study population
This observational study was conducted in two large academic centers. Both centers prospectively enroll all patients with LVO which underwent EVT. The institutional protocols of our two academic centers allow patients presenting in the early stage of AIS who have an ASPECTS 12 ⩾6 to undergo direct EVT without bridging IVT treatment if the procedure can commence within less than 30 min from arrival to the emergency department. In both centers, as part of institutional protocols, perfusion imaging is performed to all AIS patients regardless of their time of symptom onset or clinical neurological deficit. The study was approved by the institutional review boards of the participating centers with a waiver of informed consent due to its retrospective nature.
In the current study, we analyzed data from consecutive patients that presented within 4 h of symptoms onset during the years 2017–2023 (Figure 1). All patients included were eligible for bridging IVT. The IVT used was only alteplase as tenecteplase was not available during the study period. We compared clinical and radiological outcomes, between the bridging IVT + EVT group and the direct-EVT group. To minimize potential bias, patients who showed major improvement with resolution of their main deficits following IVT, were not taken to the neuro-angiography suite and were excluded from the primary analysis. Notably, for the purpose of the current study patients with uninterpretable CTP imaging were excluded.

Flowchart of included patients.
EVT treatment algorithm
All patients underwent EVT using an approved device or approach, as determined by the treating endovascular team. Procedural variables, including the modified Thrombolysis in Cerebral Infarction (mTICI) score 13 at the procedure’s conclusion and the number of passes required for optimal recanalization, were analyzed. Successful target vessel recanalization was defined as mTICI 2b–3.
Postprocedure, all patients were admitted to intensive care stroke units and managed according to standardized institutional protocols. Blood pressure management included maintaining systolic blood pressure below 140 mmHg for patients with successful recanalization and below 180 mmHg for those who received IV thrombolysis (IVT) but did not achieve recanalization.
Data collection
Time metrics were routinely measured at predefined definitions. Neurological deficits were measured using the National Institutes of Health Stroke Scale (NIHSS) 14 at admission and discharge. Stroke etiology was classified with the TOAST classification. 15 All patients had CT Angiography (CTA) upon admission and a repeat noncontrast head CT (NCCT) 24 h postprocedure. Imaging variables, including admission CTA and CTP and repeated NCCT were routinely assessed by vascular neurologists and neuroradiologist blinded to the clinical scenario. Collateral status was assessed on admission CTA according to modified two phase ASPECT collateral grading scale, 16 with a score of 4−5 defined as good collaterals. HT was assessed both radiologically and clinically according to the ECASS-2 criteria. 17 We used post-EVT NCCT data to classify HT into petechial hemorrhagic infarction and parenchymal hematoma (PH) type 1 or 2, defined as confluent hemorrhage covering less or more than one-third of the infarct volume, respectively. 18 A dual-energy CT protocol was used to enable distinction between HT and contrast extravasation due to blood-brain-barrier (BBB) damage. Further clinical division was made into asymptomatic and sICH. sICH was defined as any apparent extravascular blood in the brain or within the cranium that was associated with clinical deterioration (defined as an increase of 4 points or more in the score on the NIHSS), or led to death, and was identified as the predominant cause of the neurologic deterioration. 19
Functional outcome was assessed with the modified Rankin score (mRS) 20 prior to stroke, upon discharge, and 90 days after stroke. A favorable functional outcome was defined based on mRS-90 as either mRS ⩽2 for patients with baseline mRS ⩽2, or mRS 3 in patients that had baseline mRS 3 prior to admission.
Perfusion parameters
Based on the criteria of the pivotal DEFUSE-3 and EXTEND trials we adopted the following definitions: Core infarct (irreversibly damaged tissue) cerebral blood flow (CBF): <30% of the contralateral hemisphere with markedly reduced cerebral blood volume (CBV). Tissue at risk for ischemia—time to maximum of residual function (Tmax): >6 s. Ischemic penumbra (at-risk but salvageable tissue) is calculated by deducting the territory of CBF: (<30% from the territory with Tmax): >6 s. Although CTP imaging is a valuable tool for assessing AIS, several factors can make it unreliable. Therefore, prior to reviewing CTP images we have decided to exclude unreliable CTP images due to the following criteria: Poor contrast bolus timing—Delay or improper injection of contrast can cause errors in calculating CBF, CBV, and mean transit time. In addition, patient motion artifacts, inadequate scan coverage, and technical errors in postprocessing.
Different CTP image postprocessing analyses were used at both centers. In one center CTP images were processed with Philips IntelliSpace portal software (Philips Healthcare, Best, The Netherlands) and in the other RAPID software was used (iSchemaView Inc., Menlo Park, CA, USA).
We examined the impact of both treatment strategies on the rate of successful recanalization, degree of HT, and 90-day functional outcome in accordance with several perfusion parameters.
Statistical analysis
Statistical analysis was performed using the SPSS software (version 27.0; IBM, Chicago, IL, USA). p < 0.05 was considered significant. Continuous variables were reported as a mean value (±SD), ordinal variables as median (interquartile range (IQR)), and dichotomous variables as percentage of the total. Comparisons or distributions between categories were assessed using Student’s t test for continuous variables, Chi square test for qualitative variables, and the Mann–Whitney or to compare differences between two independent groups when the dependent variable is either ordinal or continuous, but not normally distributed. Multivariate regression models included age, sex, and predictors who were found significant or demonstrated a strong trend in the univariate analysis (p < 0.1), were used to outline modifiers of outcome.
Results
A total of 851 patients were included in our EVT cohort (mean age 71.0 ± 13.9 years; 50.1% male), of whom 567 (66.1%) arrived within 4 h of symptom onset. After excluding 136 patients who underwent EVT before CTP was routinely implemented at our medical centers, 431 patients with available CTP data were identified. Among these, 33 patients (7.7%) had technically inadequate perfusion imaging due to truncation or excessive motion. Ultimately, 398 patients (mean age 70.0 ± 14.6 years; 50.6% male) met the inclusion criteria: admission within the IVT time window and availability of adequate-quality CTP imaging. These patients were included in the final analysis (Figure 1).
Baseline characteristics were similar between patients with (n = 398) and without (n = 33) adequate CTP, except for a lower prevalence of atrial fibrillation (32.6% vs 65.6%, p < 0.001) and lower admission NIHSS scores (median 14 (IQR 10–18) vs 18 (IQR 13–21), p = 0.008) in the CTP group.
Among included patients, 218 (54.8%) patients were treated with direct-EVT, and 180 (45.2%) patients received IVT + EVT (Table 1). Patients who received bridging had less often male (45% vs 52.7%, p = 0.038) and had lower rates of hypertension (60.6% vs 73.8%, p = 0.006), hyperlipidemia (43.0% vs 55.9%, p = 0.012), atrial fibrillation (24.4% vs 40.8%, p < 0.001), congestive heart failure (6.7% vs 16.4%, p = 0.003), active malignancy (4.9% vs 12.7%, p = 0.008), and anti-coagulation treatment (5% vs 29.0%, p < 0.001). No differences were found in the involved vessel, symptoms to door, door to imaging, and door to groin times. Bridging treatment was associated with higher rates of PH2 (6.1% vs 2%, p = 0.036), but not with sICH. Both groups had similar discharge NIHSS, mRS-90, favorable functional outcome, and mortality rates. HT-PH2 was associated with almost four times higher rates of mortality (43.8% vs 11.7%, p < 0.001), worse mRS-90 (4 (2–6) vs 3 (1–4), p = 0.002), and trend toward a lower rate of functional outcome (25% vs 47%, p = 0.083).
Baseline characteristics of AIS LVO patients undergoing direct-EVT versus IVT + EVT.
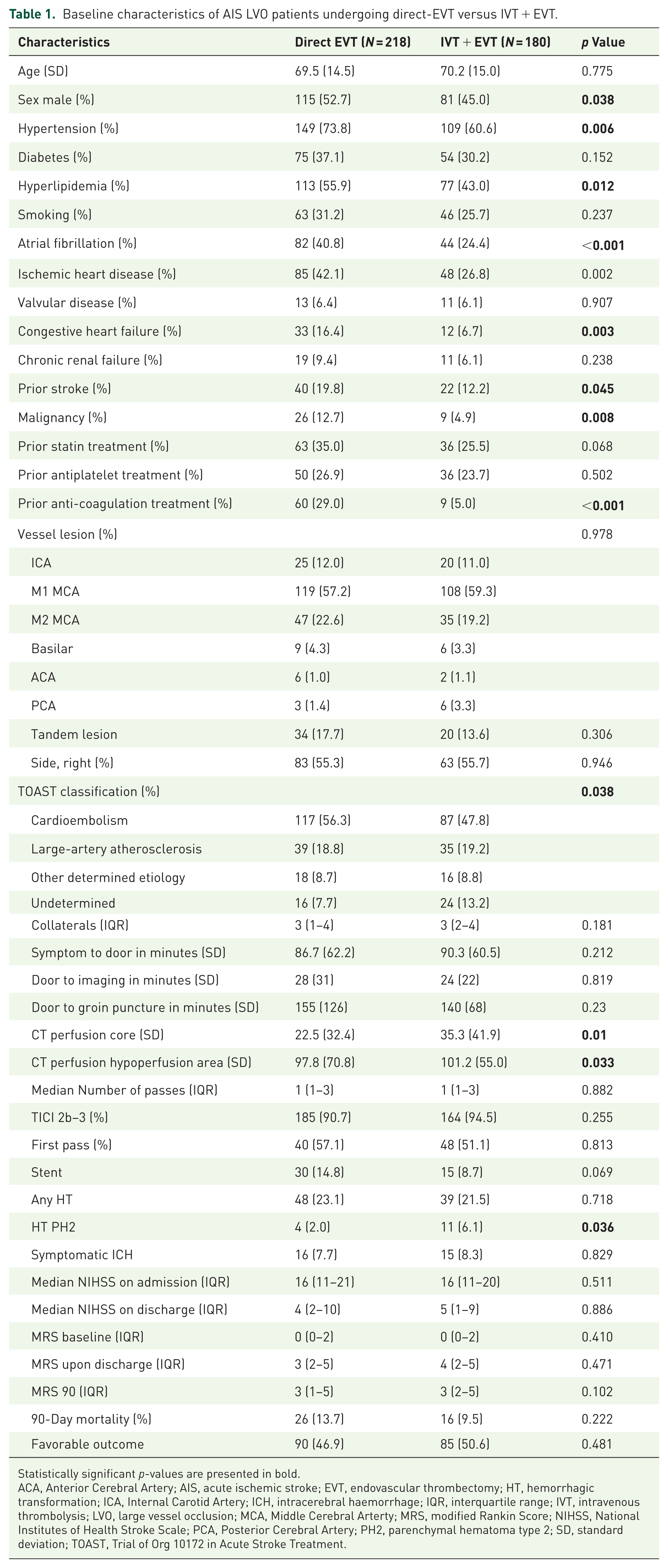
Statistically significant p-values are presented in bold.
ACA, Anterior Cerebral Artery; AIS, acute ischemic stroke; EVT, endovascular thrombectomy; HT, hemorrhagic transformation; ICA, Internal Carotid Artery; ICH, intracerebral haemorrhage; IQR, interquartile range; IVT, intravenous thrombolysis; LVO, large vessel occlusion; MCA, Middle Cerebral Arterty; MRS, modified Rankin Score; NIHSS, National Institutes of Health Stroke Scale; PCA, Posterior Cerebral Artery; PH2, parenchymal hematoma type 2; SD, standard deviation; TOAST, Trial of Org 10172 in Acute Stroke Treatment.
All 398 patients had perfusion mismatches. The average core volume was 28.6 ± 38.0, and the average penumbra volume was 99.32 ± 63.88. Overall, 269 patients (67.4%) had presence of any core, 236 (59.1%) had core volumes above 10 mL, 162 (40.6%) had core volumes above 20 mL, 75 (18.8%) had core volumes above 50 mL, and 48 (12.0%) had core volumes above 70 mL. Compared with patients treated with direct-EVT, patients treated with bridging IVT + EVT had higher penumbra area and core volumes (101 ± 55 vs 98 ± 71 mL, p = 0.033, 35 ± 42 vs 23 ± 32 mL, p = 0.01, respectively).
Continuous analysis of the entire cohort found that higher core volumes were associated with higher rates of PH2 (patients without and with PH2 had mean core volumes of 28.6 ± 36.7 and 39.1 ± 67.9 mL, respectively, p = 0.045). This association became more pronounced when performing an analysis limited to patients that received bridging IVT (patients without and with PH2 had mean core volumes of 34.7 ± 39.1 and 49.7 ± 77.1 mL, respectively, p = 0.036) but was not demonstrated among patients who underwent direct-EVT. Penumbra volumes were not associated with outcome measurements, sICH and PH2 not in the analysis of the entire cohort or analysis by the IVT treated and nontreated cohorts.
In order to further evaluate the association between core volume, bridging IVT, and HT risk, we examined this association by various core cutoff values. In a univariate sensitivity analysis, among patients with presence of any core, age (72.7 ± 10.2 vs 70.7 ± 14.5, p = 0.039), bridging IVT (7.8% vs 1.5%, p = 0.014), and hyperlipidemia (7.5% vs 2.2%, p = 0.037) were associated with PH2, while active malignancy (12.5% vs 4.1%, p = 0.066) and lower rate of first pass recanalization (4.1% vs 11.7%, p = 0.083), demonstrated a trend for association with PH2. Among patients with core volume above 10 mL, age (71.1 ± 9.8 vs 70.8 ± 14.9, p = 0.024), bridging IVT (8.8% vs 0.8%, p = 0.004) were associated with PH2, while hyperlipidemia (7.8% vs 2.4%, p = 0.057), lower rate of first-pass recanalization (4.5% vs 12.5%, p = 0.092), and premorbid mRS (0(0–0) vs 0(0–2), p = 0.076) demonstrated a trend for association with PH2. Among patients with core volume above 20 mL, bridging IVT (8.7% vs 1.3%, p = 0.031) and lower rates of first-pass recanalization (3.9% vs 15.7%, p = 0.046) were associated with PH2, while age (71.1 ± 15.1 vs 70.9 ± 15.1, p = 0.051), active malignancy (18.2% vs 4.6%, p = 0.056), and premorbid mRS (0(0–0) vs 0(0–2), p = 0.052), demonstrated a trend for association with PH2.
Based on the multiple univariate comparisons that were previously described, several multivariate analysis models were performed to explore the predictive role of IVT + EVT for PH2 across various core cutoff values. Included in the model were all variables with p value < 0.1 on their respective univariate analysis. Bridging IVT treatment remained associated with higher rates of PH2 compared to direct-EVT among patients with the presence of any core (odds ratio (OR) 12.67, 95% confidence interval (CI) 1.49–107.95, p = 0.02; Table 2). Similar directionality was found in multivariate analysis for predictors of PH2 looking on core volume above 10 mL (OR = 11.12, 95% CI 1.20–102.67, p = 0.034). Among patients with no evidence of core, no differences were found in PH2 rates between IVT-EVT and direct-EVT. The predictive role of IVT + EVT for PH2 across various core cutoff values is shown in Table 3.
Multivariate analysis for predictors of parenchymal hematoma-2 hemorrhagic transformation among patients with presence of any core on CT perfusion imaging.

Statistically significant p-values are presented in bold.
CI, confidence interval; OR, odds ratio.
Events of PH-2 HT stratified by perfusion core volumes.

Statistically significant p-values are presented in bold. Multiple multivariate models exploring the predictive role of IVT + EVT for HT-PH2 across various core cutoff values.
CI, confidence interval; EVT, endovascular thrombectomy; HT, hemorrhagic transformation; IVT, intravenous thrombolysis; OR, odds ratio; PH2, parenchymal hematoma type 2.
Discussion
In this observational study, we demonstrated that even small volumes of ischemic core on baseline CTP were strong predictors of PH2 among patients treated with bridging IVT, but not among those treated with direct EVT. These findings highlight the potential of CTP-derived ischemic core measurements as critical biomarkers for identifying patients at elevated hemorrhagic risk and refining selection for bridging therapy.
Prior work has consistently shown that perfusion imaging parameters can predict hemorrhagic complications after reperfusion. The DEFUSE and EPITHET trials established that patients with larger ischemic cores had higher rates of sICH following IVT, whereas favorable mismatch profiles balanced treatment benefit against bleeding risk.21,22 Similar associations were observed in the EVT setting, where larger ischemic core volumes correlated with increased postprocedural hemorrhage rates.²³ The novelty of our work is by using CTP parameters for predicting HT following bridging IVT.
In our cohort, the excess risk of PH2 was confined to patients with measurable ischemic core who received bridging IVT, but not to those undergoing direct EVT. This suggests that tissue already undergoing irreversible injury is particularly vulnerable to hemorrhagic conversion when exposed to thrombolysis. Mechanistically, ischemic cores are associated with extensive tissue necrosis and BBB disruption, conditions that facilitate PH formation after IVT. This concept is consistent with prior reports linking elevated BBB permeability on CTP to higher risk of HT following IVT. 24
An important limitation that deserves explicit acknowledgment is the baseline imbalance between the treatment groups. Patients in the bridging IVT + EVT arm had significantly larger CTP-defined core and penumbral volumes at presentation, representing a substrate intrinsically more prone to hemorrhagic conversion after reperfusion. This case-mix difference likely contributed to the higher PH2 rates observed in the bridging group, and parallels findings from low-ASPECTS or large-core populations in which alteplase administration before EVT increased the risk of PH. A previous study demonstrated that bridging IVT in already structurally compromised tissue translated into notably higher sICH and worse functional outcomes, emphasizing that pre-existing infarct burden strongly modifies bleeding risk. 21 Consistent with this, our primary finding—that PH2 is concentrated among patients with any ischemic core receiving alteplase—may partially reflect this underlying imbalance. To address this concern, we examined the association between IVT and PH2 adjusting for baseline core volume, and the results remained directionally similar, although the possibility of residual confounding cannot be excluded.
By contrast, we found no association between penumbral volume and risk of PH2. This aligns with previous studies showing that larger hypoperfusion volumes increase the risk of any HT after EVT, but not specifically PH2 or sICH. 25 We note as a limitation that hypoperfusion index ratio, a perfusion metric recently associated with functional outcomes and modified by pretreatment thrombolysis, was not evaluated in our analysis, despite emerging evidence supporting its prognostic value in LVO patients undergoing reperfusion therapy. 22 The ability of penumbral tissue to recover following timely reperfusion may explain this lack of association with severe hemorrhagic subtypes.
Our results also align with more recent randomized-trial evidence. The individual participant data (IPD) meta-analysis of six RCTs comparing EVT alone with EVT plus IV alteplase reported a modest but statistically significant increase in any ICH and PH when alteplase was administered prior to EVT, despite similar rates of sICH. 23 The pattern we observed—higher PH2 rates in alteplase-treated patients with measurable core—mirrors the directionality of this large pooled analysis and reinforces that even modest acute ischemic damage may amplify the hemorrhagic consequences of IVT.
Because all thrombolysed patients in our cohort exclusively received alteplase, our findings specifically reflect an alteplase-based bridging workflow. Current European Stroke Organisation guidance for LVO stroke now recommends tenecteplase 0.25 mg/kg over alteplase 0.9 mg/kg in eligible EVT candidates, 24 citing evidence of superior early reperfusion with tenecteplase. In the EXTEND-IA TNK randomized trial, tenecteplase significantly increased early recanalization rates compared with alteplase prior to EVT, 25 suggesting that its pharmacodynamic profile may differ meaningfully from alteplase in the bridging setting. Therefore, our conclusions should not be automatically extrapolated to tenecteplase-based workflows, and future studies are needed to determine whether CTP-defined core volumes similarly modify hemorrhagic risk with tenecteplase.
Our findings should also be viewed in light of the IRIS collaboration. The landmark IPD meta-analysis of six randomized trials comparing direct EVT versus EVT preceded by IV alteplase demonstrated higher rates of successful reperfusion with bridging, but no significant differences in sICH or mortality, while any ICH was more frequent in the IVT + EVT arm. 26 More recently, a dedicated IRIS IPD analysis using the Heidelberg classification provided greater granularity: among 2313 patients, bridging therapy was associated with higher rates of PH (PH1/PH2: 7% vs 5%; adjusted OR 1.54, 95% CI 1.02–2.34), and both asymptomatic and sICH correlated with worse functional outcomes on the modified Rankin Scale. 27 Similarly, our study did not demonstrate an association between bridging IVT and sICH. Nevertheless, patients with PH2 suffer from severe neurological deficits and it may have been difficult for the treating clinicians to attribute clinical deterioration solely to the PH and not to the surrounding ischemic changes.
Only a limited number of studies have examined radiological selection parameters for bridging therapy, and thus our findings should be regarded as hypothesis-generating. Advanced imaging modalities such as CTP may eventually inform the choice of EVT strategy. A post hoc analysis of the MR CLEAN-NO IV trial suggested that outcomes may depend on the initial thrombectomy technique employed 26 : in the aspiration-only group, patients undergoing EVT alone fared worse than those receiving IVT followed by EVT, whereas no difference was observed in the stent retriever group between EVT-alone and bridging approaches. Possibly, CTP core volumes may suggest higher risk of significant HT and may guide endovascular approach with less trauma to the vessel wall of the affected LVO.
Previous studies have not demonstrated an association between bridging therapy outcomes and occlusion site, age, or the presence of atrial fibrillation.27–29 Other potential modifiers include hyperlipidemia and prior use of antiplatelets or statins. 30 In our cohort, hyperlipidemia was independently associated with PH2-type HT among patients with core infarction. Prior statin exposure may influence this relationship, as several studies have reported associations between statin therapy and improved outcomes in LVO-AIS, particularly in patients with an atherosclerotic etiology, suggesting a potential protective effect despite an increased hemorrhagic risk.31,32 Certain subgroups, such as patients receiving dual antiplatelet therapy before stroke onset or those with chronic kidney disease, appear to have a heightened risk of sICH following IVT.33,34 Our study was not adequately powered to explore these subpopulations.
Importantly, a recent study investigated perfusion abnormalities on 24-h imaging in patients who achieved complete endovascular reperfusion. 35 It demonstrated that areas of microvascular hypoperfusion may persist despite TICI 3 reperfusion, potentially explaining poor outcomes in some patients and contributing to the so-called “no-reflow phenomenon.” 36 Future research should evaluate whether bridging IVT or adjunctive post-EVT thrombolysis could mitigate these residual perfusion deficits in patients with successful macrovascular recanalization. 37
Future directions should include investigating whether carefully selected adjunct antiplatelet strategies might benefit imaging-defined low-risk patients,38–40 as well as evaluating whether more intensive blood pressure management after reperfusion could reduce PH2 risk in individuals with visible CTP-defined ischemic core. CTP can operationalize adjunct antiplatelet decision-making such as required in the setting of emergent stenting during the EVT procedure, by quantifying ischemic core and BBB vulnerability. Patients with small cores and low permeability burden consistently show lower HT risk after IVT. 41 Additional research is also warranted to determine whether reduced-dose thrombolytic regimens, shorter onset-to-treatment intervals, or early CT hypodensity markers of parenchymal fragility further modify hemorrhagic risk following alteplase-based bridging therapy.
Our findings should be interpreted with caution. Additional studies are warranted to refine patient selection for bridging therapy, including investigations of the interaction between bridging treatment and Onset-to-Door time intervals in larger cohorts, as well as the potential role of advanced imaging parameters.
Our study has several limitations. First, its observational design limits causal inference, and the number of posterior circulation LVO cases was relatively small. Second, because direct EVT was permitted when the neuroendovascular team was available and at the discretion of the attending stroke neurologist, selection bias cannot be excluded, although no significant baseline differences were observed between treatment groups. Third, we were unable to compare patients with witnessed onset versus wake-up stroke owing to insufficient sample size. Finally, we did not assess the time from stroke onset to successful recanalization but instead focused on time metrics up to the initiation of EVT.
Despite these limitations, to the best of our knowledge, our results are the first to show the benefit of CTP in identifying patients at risk for significant HT following bridging IVT. Our results underscore the need for more individualized treatment strategies in the era of EVT and suggest that careful selection for bridging therapy may ultimately improve functional outcomes.
Conclusion
Baseline CTP-defined ischemic core volume is a strong predictor of PH-2 HT among patients treated with intravenous alteplase prior to EVT. The excess hemorrhagic risk associated with bridging thrombolysis was confined to patients with measurable ischemic core and was not observed in those without core involvement or in patients treated with direct thrombectomy alone. These findings suggest that CTP imaging may enable more individualized risk stratification and inform selection for bridging therapy, supporting a precision-based approach to acute reperfusion treatment in large-vessel occlusion stroke.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261425623 – Supplemental material for CT perfusion ischemic core predicts PH-2 hemorrhagic transformation following intravenous alteplase bridging treatment prior to endovascular thrombectomy
Supplemental material, sj-docx-1-tan-10.1177_17562864261425623 for CT perfusion ischemic core predicts PH-2 hemorrhagic transformation following intravenous alteplase bridging treatment prior to endovascular thrombectomy by Asaf Honig, Aviva Alpernas, Yoel Schwartzmann, Hen Hallevi, Issa Metanis, Einor Ben-Assayag, Tali Jonas-Kimchi, Udi Sadeh, Hamza Jubran, Oz Haim, Jose Cohen, Ronen R. Leker and Jeremy Molad in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
