Abstract
Background:
Ofatumumab (OFA), a humanized anti-CD20 monoclonal antibody, shows potential in myasthenia gravis (MG) with antibody against acetylcholine receptor (AChR-Ab), but comparative efficacy across AChR and muscle-specific kinase (MuSK) subtypes remains uncharacterized.
Objectives:
To evaluate OFA’s clinical and immunological effects in AChR-MG versus MuSK-MG, identify predictors of minimal symptom expression (MSE), and explore the dynamics of B-cell repopulation.
Design:
A retrospective study in a single center.
Methods:
Twenty-one refractory MG patients (13 MuSK+, 8 AChR+) received individualized OFA induction (Regimen 1: two 20 mg doses ⩾2 weeks apart; Regimen 2: single 20 mg dose) and maintenance (20 mg upon CD19 + B counts ⩾5 cells/μL repopulation or clinical exacerbation). Outcomes included time-to-MSE, Quantitative Myasthenia Gravis Score (QMGS) dynamics, prednisone reduction, and serial immunophenotyping (B-cell activating factor (BAFF), a proliferation-inducing ligand (APRIL), soluble CD40L (sCD40L), microphage migration inhibitory factor (MIF), and immunoglobulin).
Results:
MuSK-MG achieved faster MSE (median time: 6 vs 15 months, log-rank p = 0.164, age-adjusted Cox p = 0.027), and a higher 3-month improvement (92.3% vs 50.0%; p = 0.047). Earlier achievement of MSE was associated with shorter disease duration (p = 0.038) and concomitant corticosteroid therapy (p = 0.030). Prednisone doses decreased significantly over 6 months (p = 0.029) without difference between groups (p = 0.109). MuSK-MG showed synchronized BAFF/APRIL declines while AChR-MG presents transient BAFF elevation and sustained APRIL. BAFF levels showed a significant positive correlation with QMGS in MuSK-MG, while IgM/IgG positively correlated with QMGS in both subtypes. Median B cell-repopulation time was 47.7 days.
Conclusion:
OFA demonstrates superior efficacy in MuSK-MG with distinct immunological modulation. AChR-MG requires combinatorial strategies targeting compensatory pathways.
Keywords
Introduction
Myasthenia gravis (MG) is a paradigm of T cell-dependent, B cell-mediated chronic autoimmune disorder of neuromuscular junction, characterized by muscle weakness and fatigability that can progress to life-threatening respiratory failure. While 80%–85% of MG patients have antibodies (Ab) against acetylcholine receptor (AChR), 4%–8% possess antibodies to muscle-specific kinase (MuSK), 1 a subtype associated with prominent bulbar and respiratory involvement. 2
Standard MG therapies, including acetylcholinesterase inhibitors, corticosteroids, and immunosuppressants, are effective but often cause significant long-term side effects. 1 About 15% of patients exhibit poor or no response to conventional treatments, meeting the criteria for refractory disease. 3 Recently, molecular therapies such as B cell-depleting agents, complement inhibitors, neonatal Fc receptor (FcRn) inhibitors, and chimeric antigen receptor (CAR) T cell-based therapies have emerged, transforming the treatment landscape 3 ; however, therapeutic selection must consider disease heterogeneity. FcRn inhibitors promote IgG degradation and complement inhibitors block membrane attack complex formation, both targeting downstream pathways, whereas B cell depletion (e.g., anti-CD20 antibodies) acts upstream and remains pivotal. 3
Rituximab (RTX), a chimeric anti-CD20 monoclonal antibody targeting B cells (excluding stem, progenitors, and plasma cells), has demonstrated efficacy in MuSK-MG but yields inconsistent responses in AChR-MG.4–8 Its murine components increase immunogenicity, causing infusion-related reactions and potential long-term treatment resistance. 9 Ofatumumab (OFA), a fully humanized anti-CD20 antibody approved for multiple sclerosis (MS), 10 shows promise for AChR-MG via the MS dosing regimen.11,12 However, this approach imposes a substantial cost burden, and its long-term efficacy remains uncertain due to limited follow-up (only 3 months in current studies), leaving retreatment strategies undefined. 11 While RTX has been studied in both subtypes, comparative data on OFA between AChR-MG and MuSK-MG are lacking, a significant gap given the distinct pathophysiological mechanisms of these subtypes and their potential for differential treatment responses.
B-cell activating factor (BAFF), a proliferation-inducing ligand (APRIL), and a soluble CD40 ligand (sCD40L) share structural homology with TNF and play crucial roles in MG pathogenesis. These cytokines regulate B cell maturation, survival, immunoglobulin class switching, and antibody production. Zhou et al. 13 observed elevated serum BAFF levels post-RTX treatment. Emerging evidence further implicates the microphage migration inhibitory factor (MIF)-CD74 axis in modulating B cell proliferation, differentiation, and survival in MG patients.14,15 Despite these advances, differential cytokine responses to OFA treatment in AChR-Ab versus MuSK-Ab MG subtypes remain unexplored.
This exploratory study evaluates the clinical efficacy of OFA in AChR-Ab and MuSK-Ab MG subtypes using an individualized, B cell-guided dosing regimen and examines its differential effects on serum BAFF, APRIL, sCD40L, MIF, and immunoglobulins levels between subtypes. These findings offer preliminary insight into subtype-specific treatment responses, potentially guiding personalized MG therapy.
Methods
Standard protocol approvals
This was a single-center, retrospective observational study, approved by the Ethical Committee of Xiangya Hospital, Central South University (No. 202408003). Moreover, we have provided patients with detailed information about disease and obtained the patients’ consent to repeated OFA treatment, while written informed consent was obtained from all patients.
Patients
Twenty-one refractory MG patients treated with OFA between January 2022 and June 2025 were retrospectively enrolled. Inclusion criteria comprised: (1) age ⩾18 years; (2) definitive diagnosis of generalized AChR-MG or MuSK-MG confirmed by exertional weakness and seropositivity for AChR/MuSK antibodies. Pharmacological testing and repetitive nerve stimulation were performed when clinically indicated but were not mandatory for seropositive patients; (3) Myasthenia Gravis Foundation of America clinical classification (MGFA-CC) of II–V; (4) refractory MG defined as by meeting 1 of the following criteria: suboptimal response (unchanged or worsening clinical status) despite conventional immunosuppressive treatment with prednisolone plus ⩾1 immunosuppressant at least 3 months, intolerance to immunosuppressive therapies, inability to taper steroids without relapse or dependence on maintenance plasmapheresis (PE) or intravenous immunoglobulin (IVIG). 16 (5) no RTX exposure within 6 months; (6) minimum 3-month follow-up; (7) exclusions of potentially acute or chronic viral or bacterial infections (HIV, latent hepatitis B, tuberculosis, etc.). Clinical data were retrospectively retrieved from the electronic medical records of Xiangya Hospital.
Study design
The OFA protocol involved induction followed by maintenance therapy (Figure 1). To enhance feasibility and cost-effectiveness in this off-label MG cohort, a response-guided induction strategy was applied over a 2-month window. All patients received an initial 20 mg dose, with a second dose administered if CD19+ B-cell depletion was suboptimal after approximately 2 weeks.

Repeated ofatumumab treatment protocol.
Maintenance therapy (20 mg) was initiated upon CD19+ B-cell repopulation (⩾5 cells/μL) or clinical exacerbation, defined as either a ⩾3-point increase in Quantitative Myasthenia Gravis Score (QMGS) from the prior assessment or a ⩾2-point increase in MG-ADL score from the best-recorded score since last treatment. The stricter repopulation threshold (vs ⩾1%) was adopted based on rheumatoid arthritis (RA) studies, anticipating faster B-cell recovery with OFA to preempt relapse.17–19
For patient who received rapid-clearance antibody therapy (RCAT) prior to OFA initiation, OFA was initiated following specific intervals after prior rescue therapies: at least 2 months after IVIG, at least 2 weeks after efgartigimod (EFG), and at least 1 day after PE (Table S1), based on the pharmacokinetic and pharmacodynamic profiles of each therapy.20–22 Concurrent nonsteroidal immunosuppressants were discontinued upon OFA initiation, while corticosteroid doses were adjusted clinically.
Patients underwent 2–3 monthly outpatient visits for QMG and ADL scoring and blood tests (complete blood counts, B-cell subsets, and immunoglobulins) as part of routine clinical care. Not all samples were available at each timepoint due to clinical constraints, but all collected samples were analyzed. Patients who did not come for follow-up visits but remained contactable by phone received telephonic MG-ADL assessments.
Serum MuSK-Ab (radioimmunoassay; positive >0.05 nmol/L) and AChR-Ab (ELISA; positive >0.45 nmol/L) titers were quantified. BAFF, APRIL, and sCD40L levels were assessed by commercial ELISA kits (mlbio, Shanghai, China), and MIF by KE00248 Human MIF ELISA. Immunoglobulin and light chains were quantified by immunoturbidimetry, using the following normal ranges: IgG 7.0–16.0 g/L, IgM 400.00–2800.00 mg/L, IgA 700.00–5000.00 mg/L, Kappa chains 1.87–4.05 g/L, and Lambda chains 1.11–2.57 g/L.
Outcome measures
The primary outcome was time to first achieve minimal symptom expression (MSE; MG-ADL ⩽1) 23 post-OFA initiation, compared between AChR-MG and MuSK-MG cohorts. Secondary outcomes included between-group differences and longitudinal changes in QMGS, prednisone doses, and immunological biomarkers at baseline, 3 months, and 6 months. To control for the effect of recent rapid antibody-clearing therapies (PE, EFG, or IVIG), longitudinal analysis of QMGS also incorporated the peak scores from the 3 months pretreatment. The proportion of achieving “improved” status (defined as a ⩾3-point reduction in QMGS from the pre-3-month peak if peak ⩾16, or a ⩾2-point reduction if peak <16) at 3 months post-OFA was compared between antibody groups. 24 Exploratory analyses assessed predictors of time to MSE and B-cell repopulation, the influence of antibody subtype and time postinjection on immunological parameters, and correlations between these parameters and QMGS.
Statistical analysis
Continuous data are presented as mean ± standard deviation (normally distributed) or median (interquartile range (IQR); nonnormal), and categorical variables as counts (%). Group comparisons used t tests, Mann–Whitney U, Wilcoxon signed-rank, or Fisher’s exact tests as appropriate.
Time to first MSE was analyzed with Cox regression. Optimal cut-points for continuous variables were determined using maximally selected rank statistics. Multiple Cox models were compared to address potential baseline imbalances; a model adjusted for treatment age was selected based on optimal Akaike and Bayesian Information Criterion (AIC and BIC) values. The robustness of antibody effect was further validated through bootstrap resampling (n = 1000) and sensitivity analyses incorporating additional covariates. Prognostic factors were identified through univariable Cox regression, with age-adjusted analyses for age-associated variables (p < 0.2). Stratified univariable Cox analyses were additionally conducted within each antibody subgroup to explore subtype-specific determinants of treatment response.
Longitudinal trajectories of continuous outcomes were analyzed using linear mixed-effects models with patient-specific random intercepts. Missing data were handled under a missing-at-random assumption via full maximum likelihood. Model selection was based on likelihood ratio tests and AIC, comparing nested models that included fixed effects for antibody type, time, their interaction, and age at treatment. p-Values for fixed effects were obtained using Satterthwaite’s approximation, and post hoc pairwise comparisons were performed with Bonferroni correction. Effect sizes (Cohen’s d) were calculated with the denominator as the pooled standard deviation (combining random intercept and residual variances). Confidence intervals (CIs) for Cohen’s d were derived using the delta method and t distribution with Kenward–Roger degrees of freedom.
The within-subject dynamics analysis served as the primary method to evaluate longitudinal associations (patients with ⩾2 measurements) between immunological markers and clinical symptoms by modeling person-mean-centered deviations using linear regression. As a secondary analysis, linear mixed-effect models (LMMs) with a random intercept for patients were employed to validate the primary findings. The strength of the correlation was quantified with the Spearman rank correlation coefficient (rs).
All analyses were performed in R v4.4.1, using two-sided tests with statistical significance set at p < 0.05.
Results
Baseline characteristics
A total of 21 patients were identified (13 MuSK-MG, 8 AChR-MG; mean age 38.1 ± 15.6 years; 17 women and 4 men) with a median follow-up of 8.0 (IQR 6.0, 15.0; range 3–36) months. The median OFA injections were 3.0 (IQR 2.0, 5.0; range: 1–11; Table 1).
Baseline characteristics.
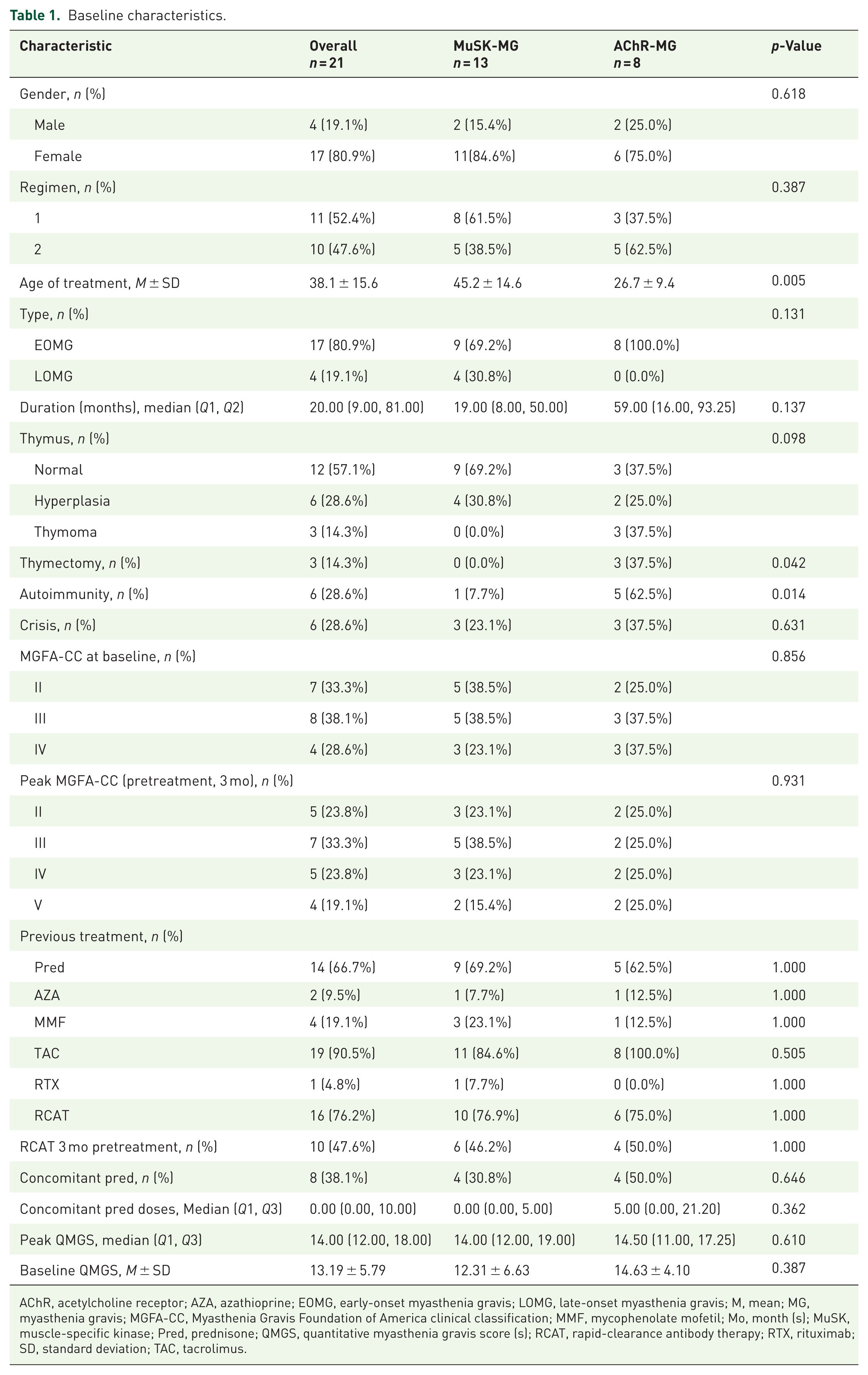
AChR, acetylcholine receptor; AZA, azathioprine; EOMG, early-onset myasthenia gravis; LOMG, late-onset myasthenia gravis; M, mean; MG, myasthenia gravis; MGFA-CC, Myasthenia Gravis Foundation of America clinical classification; MMF, mycophenolate mofetil; Mo, month (s); MuSK, muscle-specific kinase; Pred, prednisone; QMGS, quantitative myasthenia gravis score (s); RCAT, rapid-clearance antibody therapy; RTX, rituximab; SD, standard deviation; TAC, tacrolimus.
AChR-MG and MuSK-MG groups demonstrated comparable gender distribution, treatment regimens, disease subtypes, thymic status, baseline/peak MGFA-CC, prior therapies, crisis history, and concomitant prednisone doses. Peak QMGS at 3-month pretreatment and QMGS at baseline were also similar. Significant intergroup differences emerged in: (1) treatment initiation age (MuSK-MG 45.2 ± 14.6 vs AChR-MG 26.7 ± 9.4 years, p = 0.005), (2) thymectomy rates (0% MuSK-MG vs 37.5% AChR-MG, p = 0.042), and (3) autoimmune comorbidity prevalence (7.7% MuSK-MG vs 62.5% AChR-MG, p = 0.014). Comorbidities included hyperthyroidism (n = 5) and atopic dermatitis (n = 1) in AChR-MG, and nephritis (n = 1) in MuSK-MG.
Achieve improved or MSE
MSE was achieved by 9 (69.2%) MuSK-MG and 3 (37.5%) AChR-MG patients. The median time to first MSE attainment was 6 months for MuSK-MG versus 15 months for AChR-MG (log-rank p = 0.164, univariate Cox HR = 0.440, 95% CI: (0.118–1.635), p = 0.220; Figure 2(b), Table S2).
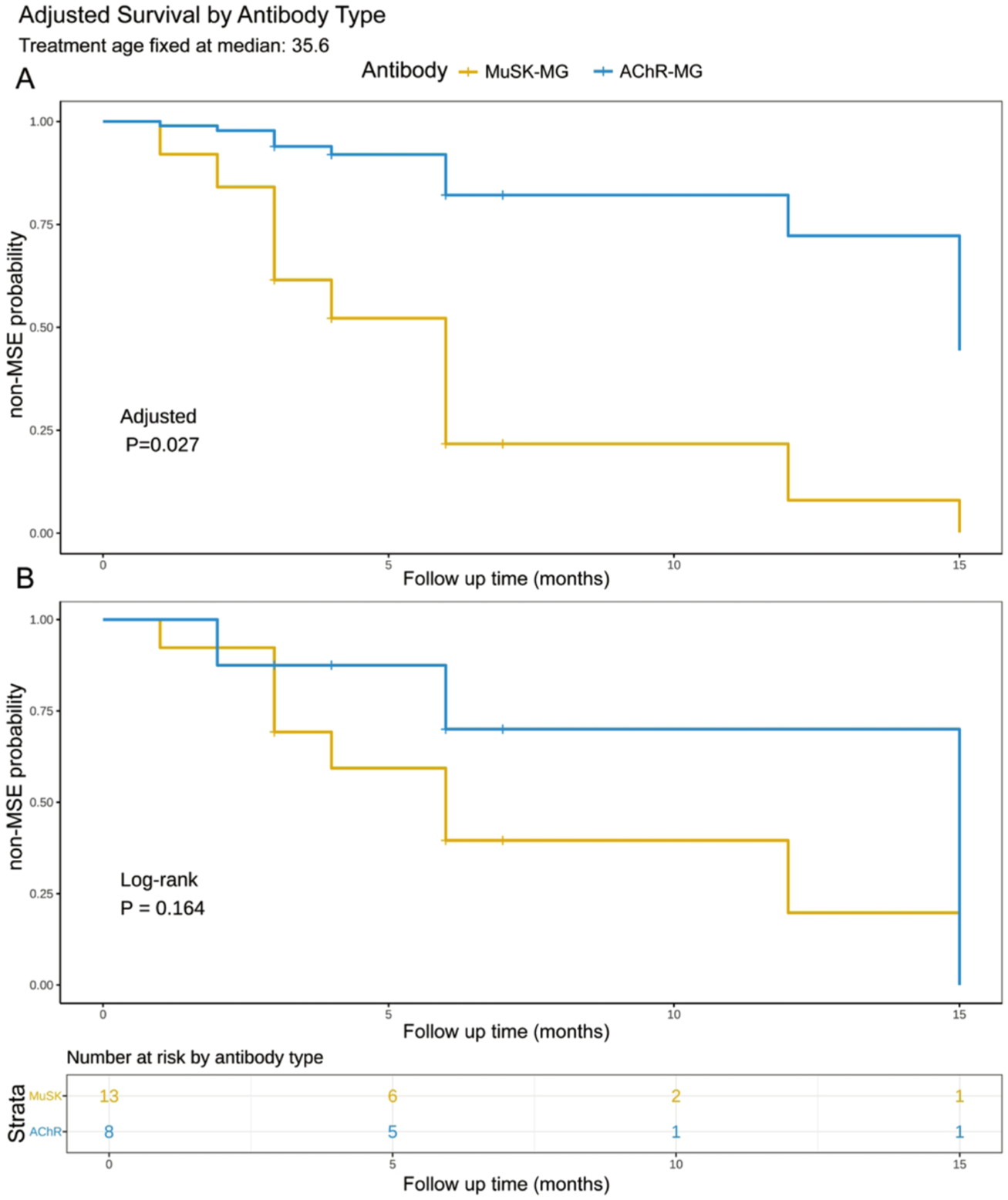
Time to first MSE by antibody type. (a) Adjusted survival curves showing the probability of not achieving MSE over time, stratified by antibody type (MuSK-MG vs AChR-MG), with adjustment for age of treatment using Cox proportional hazards regression. Age of treatment was fixed at the median value of 35.6 years. The analysis demonstrates that antibody type remains significantly associated with time to first MSE achievement after adjusting for age (p = 0.027). (b) Unadjusted Kaplan–Meier survival curves with corresponding risk table showing the number of patients at risk at each time point. The log-rank test indicates a marginally significant difference in time to first MSE between antibody types (p = 0.164).
To address potential confounding, we compared multiple Cox models and selected the treatment age-adjusted model based on optimal AIC (50.9) and BIC (51.8). In this model, MuSK-MG was independently associated with faster MSE attainment (adjusted HR = 0.129, 95% CI: (0.021–0.794), p = 0.027; Figure 2(a), Table S2). Bootstrap validation (n = 1000 iterations) confirmed the robustness of this association (median HR = 0.094, 95% CI: (0–0.717)). Specifically, 98.2% of resampled datasets showed a protective effect (HR < 1; Figure S1). Sensitivity analyses showed the effect persisted when further adjusting for thymectomy (Model 5; HR = 0.086, p = 0.020) but was attenuated when adjusting for autoimmune comorbidity (Model 4; HR = 0.318, p = 0.278; Table S2). Clinically, more MuSK-MG patients achieved symptom improvement within 3 months (92.3% vs 50.0%, p = 0.047).
Predictive analyses identified shorter disease duration (⩽9 months; HR = 0.261, p = 0.038) and concomitant prednisone use (unadjusted HR = 4.702, p = 0.030; age-adjusted HR = 5.733, p = 0.027) were associated with earlier MSE attainment (Table S3). After adjusting for age, the comorbid autoimmunity also emerged as a significant predictor (HR = 0.135, p = 0.036). In subgroup analyses, younger treatment age (p = 0.036) and prednisone use (p = 0.044) predicted earlier MSE specifically in MuSK-MG patients. No significant predictors were identified within the AChR-MG cohort, likely due to the limited statistical power of this small subgroup (n = 8; Table S4).
Effectiveness of OFA on study outcomes
Clinical measurement
LMM revealed differential responses to OFA between antibody subtypes. For overall QMGS, a significant antibody × time interaction was also observed (p < 0.05 at months 3 and 6), reflecting faster improvement in MuSK-MG (Table S5). While baseline scores were comparable, MuSK-MG patients exhibited significantly greater reductions at month 3 (mean difference (MD): −7.65, 95% CI: (−14.05 to −1.24), p = 0.020) and month 6 (MD: −7.45, 95% CI: (−14.00 to −0.90), p = 0.027; Table 2, Figure 3(a)). Longitudinal analysis revealed significant improvement in MuSK-MG patients, with QMGS decreasing by 5.63 from baseline to month 3 (95% CI: (2.31–8.95), p = 0.017) and by 7.46 from baseline to month 6 (95% CI: (4.27–10.65), p < 0.001), while AChR-MG showed minimal changes, with a nonsignificant 0.56-point increase at month 3 and a 1.47-point decrease at month 6. Since 10 patients received RCAT within 3 months before OFA, which can provide rapid alleviation of symptoms albeit with a transient effect, we also analyzed peak QMGS 3 months pretreatment, which also yielded consistent findings (Figure 3(b)).
Comparison of MuSK-MG and AChR-MG patients: Marginal means, between-group differences, and effect sizes for QMGS and immunological parameters.

AChR, acetylcholine receptor; APRIL, a proliferation-inducing ligand; BAFF, B-cell activating factor; CI, confidence interval; M, estimate marginal means; MD, mean difference between estimated marginal means; MIF, microphage migration inhibitory factor; MG, myasthenia gravis; Mo, month (s); MuSK, muscle-specific kinase; Pred, prednisone; QMGS, quantitative myasthenia gravis score (s); sCD40L, soluble CD40L; SE, standard error.

Longitudinal dynamics of clinical and laboratory parameters in MG after OFA treatment. (a–j) Longitudinal dynamics of clinical and laboratory parameters in MG over 6 months. (k–n) Levels of BAFF, APRIL, sCD40L, and MIF after OFA. Serum BAFF, APRIL, sCD40L, and MIF levels in untreated MG and in OFA-treated MG patients stratified into three groups based on time since last treatment: ⩽30, >30 to⩽90, and >90 days.
Subscore analysis indicated marginally significant antibody × time interactions for ocular and axial QMGS over the 6 months (Table S5, Figure S2(B) and (E)). Ocular scores were significantly lower in MuSK-MG patients at month 3 (MD: −3.12, 95 %CI: (−5.54 to −0.70), p = 0.012) and at month 6 (MD: −2.74, 95% CI: (−5.23 to −0.26), p = 0.031; Table S6), while axial scores showed a similar nonsignificant trend with substantial effect sizes. Analyses of axial and limb QMGS trajectories from pretreatment peak to month 6 revealed significant antibody time interactions, indicating greater improvement in MuSK-MG patients (Tables S7 and S8, Figure S3(E) and (F)).
Prednisone doses decreased significantly over time with a significant main effect of time (p = 0.029). Throughout treatment, AChR-MG patients consistently required higher doses than MuSK-MG patients (Cohen’s d = 1.50), although between-group differences were not statistically significant (p = 0.125; Table 2, Figure 3(c)).
Laboratory results
LMM revealed significant antibody × time interactions for BAFF, APRIL, and IgM/IgG levels (Table S9). Baseline BAFF was markedly elevated in MuSK-MG patients (p < 0.001), while APRIL levels were comparable (p = 0.814; Table 2, Figure 3(d) and (e)). After OFA initiation, their trajectories diverged: BAFF declined progressively in MuSK-MG but showed a transient increase at month 3 in AChR-MG, with no intergroup difference by month 6 (p = 0.154). Conversely, APRIL levels were significantly higher in the AChR-MG group at both follow-up time points (month 3: MD: 901.06, 95% CI: (278.77–1523.34) pg/mL, p = 0.006; month 6: MD: 894.32, 95% CI: (220.18–1568.45) pg/mL, p = 0.010). Levels of sCD40L and IgM/IgG showed a significant declining trend over 6 months (Figure 3(f) and (h)). There was no significant intergroup difference in the levels of sCD40L, MIF, IgM/IgG, IgM, IgG, Kappa, and Lambda chains at each time point over 6 months (Tables S9 and S10, Figures 3 and S4).
Antibody titers exhibited distinct longitudinal patterns. MuSK-Ab titers decreased significantly over time (main effect of time, p = 0.006), with a confirmed reduction at month 6 compared to baseline (MD: −0.58, 95% CI: (−1.02 to −0.14), p = 0.008; Figure 3(i), Tables S11 and S12). In contrast, AChR-Ab titers did not show a statistically significant main effect of time, and post hoc comparisons revealed no significant changes between specific time points (Figure 3(j)).
Due to the heterogeneous retreatment intervals, the OFA-treated cohort was stratified by time since last OFA dose (untreated, ⩽30 days, 31–90 days, >90 days). LMM revealed distinct temporal dynamics of BAFF and APRIL levels between antibody subtypes. At baseline, BAFF was significantly higher in MuSK-MG but declined progressively posttreatment, eliminating the initial group difference (Tables S13 and 14, Figure 3(k)). In contrast, APRIL levels were comparable at baseline (p = 0.721) but became significantly lower in MuSK-MG during the 31–90 day (p = 0.017) and >90-day intervals (p = 0.004; Figure 3(l)). No significant intergroup differences were observed for sCD40L, MIF, IgM/IgG, IgM, IgG, light chain levels between MuSK-MG and AChR-MG across intervals (Tables S13 and S14, Figures 3 and S5).
To assess the relationship between immunological parameters and clinical disease severity, we performed longitudinal within-person analysis using person-mean centered linear regression. Within-person increases in BAFF levels were associated with worsening QMGS specifically in MuSK-MG (β = 0.008, 95% CI: (0.002–0.014), p = 0.014), explaining 12.6% of the variance in clinical severity fluctuations, which was also reinforced by LMM (Figure 4(a)–(d)). Conversely, rising antibody titers correlated with clinical worsening only in AChR-MG by within-person analysis (β = 0.905, 95% CI: (0.058–1.753), p = 0.037; Figure 4(i) and (j)), but this pattern was not discernible in the LMM (Figure 4(k) and (l)). Interestingly, within-person increases in IgM/IgG were significantly associated with elevating QMGS both in MuSK-MG (β = 55.45, 95% CI: (16.63–92.36), p = 0.006) and AChR-MG patients (β = 90.80, 95% CI: (43.22–138.39), p < 0.001), explaining 12.14% and 33.61% of the variance in clinical severity fluctuations, respectively, which was consistent in LMM (Figure 4(e)–(h)). IgM showed a pattern similar to IgM/IgG, whereas APRIL, sCD40L, MIF, IgG, Kappa, and Lambda chains did not correlate with QMGS in within-person analysis (Figure S6).

Correlation between immunological parameters and QMGS. Within-subject longitudinal analysis showed a positive correlation between serum BAFF levels and QMGS in MuSK-MG (rs = 0.473, ps < 0.001; LRM estimate: 7.951e−03, 95% CI: (0.002–0.014), p = 0.014) (a), but not in AChR-MG (rs = −0.017, ps = 0.937; LRM estimate: −7.528e−04, 95% CI: (−0.020 to 0.019), p = 0.938) (b). LMM showed a positive correlation between BAFF levels and QMGS in MuSK-MG (rs = 0.318, ps = 0.029; LMM estimate: 0.008, 95% CI: (0.002–0.015), p = 0.019) (c), but not in AChR-MG (rs = 0.203, ps = 0.341; LMM estimate: 0.004, 95% CI: (−0.016 to 0.025), p = 0.687) (d). Within-subject longitudinal analysis showed a positive correlation between serum IgM/IgG and QMGS in MuSK-MG (rs = 0.455, ps < 0.001; LRM estimate: 54.497, 95% CI: (16.633–92.362), p = 0.006) (e) and in AChR-MG (rs = 0.623, ps < 0.001; LRM estimate: 90.803, 95% CI: (43.218–138.387), p < 0.001) (f). LMM also showed a significant correlation between IgM/IgG and QMGS in MuSK-MG (rs = 0.204, ps = 0.112; LMM estimate: 41.022, 95% CI: (4.278–78.043), p = 0.026) (g) and AChR-MG (rs = 0.107, ps = 0.559; LMM estimate: 66.289, 95% CI: (14.172–116.526), p = 0.006) (h). Within-subject longitudinal analysis showed a positive correlation between serum AChR-Ab levels and QMGS in AChR-MG (rs = 0.546, ps = 0.007; LRM estimate: 0.905, 95% CI: (0.058–1.753), p = 0.037) (j), but not in MuSK-MG (rs = 0.052, ps = 0.713; LRM estimate: 0.322, 95% CI: (−1.928 to 2.572), p = 0.775) (i). LMM also showed no significant correlation between antibody titers and QMGS in either MuSK-MG (rs = 0.354, ps = 0.009; LMM estimate: 0.555, 95% CI: (−0.296 to 0.458), p = 0.645) (k) or AChR-MG (rs = 0.055, ps = 0.802; LMM estimate: 0.024, 95% CI: (−0.296 to 0.458), p = 0.884) (l). (A simple linear regression line is plotted with a shadow indicating 95% CI. Each point corresponds to the deviation of immunological parameter and QMGS from the respective mean for the same patient at the same time point in the first two columns; each point represents a patient’s immunological parameter and QMGS at a specific time point in the 3–4 columns.)
Exacerbation and retreatment
Nine exacerbations occurred in this study (Table S15). Two MuSK-MG patients exacerbated within the induction window (MuSK-9 and MuSK-6), while four exacerbations (three MuSK-MG: MuSK-10, MuSK-12, and MuSK-13; one AChR-MG: AChR-7) coincided with B-cell repopulation during maintenance. Interestingly, the patient (MuSK-9) who restarted OFA treatment after stabilized by EFG, experienced exacerbation again (QMG rose from 3 to 5 and ADL from 1 to 3) with CD19+ B-cells 1.22/µL. Although MuSK-13 and AChR-7 received OFA after exacerbation, they continued worsening. AChR-7 showed no response with QMGS increasing from 15 to 28 after the third dose.
Most retreatments were triggered by CD19+ B-cell repopulation (⩾5 cells/μL) without clinical exacerbation. Predictive performance analysis of B-cell threshold of 5 cells/μL indicated that its sensitivity and specificity were 0.667 and 0.340, respectively, and the positive and negative predictive values (PPV and NPV) were 0.103 and 0.900 (Figure S7). The conventional ⩾1% threshold showed higher specificity (0.642) with equal sensitivity (0.667; PPV = 0.174, NPV = 0.944). The memory B-cell (>0.01% of peripheral blood mononuclear cells (PBMCs)) threshold demonstrated the highest sensitivity (0.833) but the lowest specificity (0.113; PPV = 0.096, NPV = 0.857).
To characterize repopulation kinetics, linear modeling was applied for clinical utility with CD19+ B counts (Figure S8) and CD19+ B/lymphocyte ratios (Figure S9) analyzed against days since last treatment (excluding the initial 14-day postinduction clearance phase). Median time to CD19 + B ⩾ 5 cells/μL was 47.70 (range 17.1–227.4) days, and to CD19 + B/lymphocyte ⩾1% was 89.3 (range 25.8–775.7) days (Table S16). Predictive analyses revealed trends toward delayed repopulation in MuSK-MG (p = 0.053), previous mycophenolate mofetil (MMF) exposure (p = 0.065), and those without autoimmune comorbidity (p = 0.055; Table S17). Prior MMF exposure significantly prolonged time to reach the ⩾1% threshold (p = 0.013). Lower baseline IgG correlated with longer repopulation time (r = −0.496, p = 0.032; Table S18).
Discussion
This study demonstrates ofatumumab’s therapeutic efficacy in both AChR-MG and MuSK-MG, providing the first comparative analysis of clinical and immunological effects between subtypes after OFA.
Current evidence supports B-cell repopulation-guided retreatment to balance efficacy and overtreatment.25,26 Given the exploratory use of OFA in MG and its faster B-cell repopulation than other anti-CD20 therapies as well as its monthly administration in MS maintenance, 17 we adopted a proactive threshold of ⩾5 cells/μL threshold, aligning with RA protocols. 19 In our cohort, the conventional CD19 + B cell/lymphocyte ⩾1% threshold showed moderate sensitivity (0.667), consistent with previous literature (predicting only 33%–57% relapses).5,25 The ⩾5 cells/μL threshold displayed equal sensitivity (0.667) but lower specificity (0.340); its high NPV (0.900) prioritizes prevention. This stringent approach likely contributed to sustained remission, particularly in the responsive MuSK-MG group, though it limits direct comparability with studies using conventional criteria.
A key observation was that B-cell levels between 5 cells/μL and 1% defined a safe window, beyond which relapse risk increased. Noteworthily, despite receiving OFA at 1.8% B cells and achieving depletion later, patient’s (MuSK-13) exacerbation progressed, requiring rescue therapy. This suggests that by the time B-cells surpass 1%, plasmablasts generated during repopulation may have already entered a therapy-resistant effector stage, supporting earlier intervention within the identified safe window. Although memory B-cell thresholds (>0.01% PBMCs) has prognostic value in RTX-treated MG with high sensitivity, its low PPV (28.0%) limits its utility, 27 a finding corroborated by our study (PPV 0.096), indicating it may signal broad immune reconstitution rather than specifically predicting imminent relapse.
Baseline imbalances reflected established phenotypic distinctions between MuSK‑MG and AChR‑MG, including the bimodal age distribution and higher prevalence of thymic pathology and autoimmune comorbidity in AChR-MG.2,28–31 While MuSK-MG was robustly associated with earlier MSE attainment in our primary age-adjusted model, this association attenuated upon further adjustment for autoimmune comorbidity. This observation warrants a nuanced interpretation beyond residual confounding. Autoimmune comorbidity is not a random confounder but an intrinsic element of the AChR-MG phenotype.30,32 Consequently, statistical adjustment for this phenotype-defining feature in a limited sample may inadvertently partially correct for the very biological construct under investigation. Therefore, the attenuation likely underscores the strong collinearity between antibody subtype and its constitutive clinical phenotype. Our findings indicate that the superior treatment response profile is a characteristic of the MuSK-MG phenotypic cluster. This insight reframes the clinical question and underscores the need for future studies in larger cohorts to delineate the specific contributions within this phenotype.
Our primary outcome demonstrated superior efficacy profile of OFA in MuSK-MG compared to AChR-MG, with faster attainment of MSE (age-adjusted) and a higher 3-month improvement rate—consistent with the established differential response to RTX.4,6–8,33,34 The fully humanized nature and subcutaneous administration of OFA may offer practical advantages, though direct comparison with RTX was not performed. Mechanistically, this differential efficacy may stem from the cellular origins of autoantibodies: MuSK antibodies are produced mainly by short-lived plasmablasts (a subset of which are CD20+) that are replenished by CD20+ memory B cells, whereas AChR antibodies are sustained by long-lived plasma cells lacking CD20 expression.35–37 We also observed great heterogeneity in the time to B-cell repopulation (1–8 months), suggesting that OFA’s convenient subcutaneous protocol could enhance cost-effectiveness in patients with slower repopulation. Trends toward delayed repopulation in certain subgroups (e.g., MuSK-MG, previous MMF exposure) warrant future validation. Earlier MSE was associated with shorter disease duration, supporting early immunotherapy to limit damage and prevent the establishment of resistant plasma cell pools.
OFA treatment significantly reduced MuSK-Ab titers, contrasting with a marginal decline in AChR-Ab. Longitudinal analysis further revealed a critical methodological distinction in interpreting antibody-clinical correlations. In AChR-MG, fluctuations in individual AChR-Ab titer were significantly associated with concurrent changes in symptom severity. This finding reconciles the long-standing clinical paradox: although AChR-Ab levels do not consistently reflect disease severity across different patients, their variation over time within the same individual does correlate with clinical status.38,39 Conversely, for MuSK-MG, the apparent cross-sectional correlation stemmed entirely from between-patient differences, with no significant within-person dynamic identified, consistent with the nonsignificant LMM finding. This suggests that MuSK-Ab titers are useful for stratifying disease severity across patients but are less reliable for tracking individual disease activity over time. Consequently, in the context of OFA treatment, clinical improvement with OFA may not synchronize perfectly with MuSK-Ab reduction, potentially depending more on factors such as IgG class switching or autoantibody affinity.4,40,41
The IgM/IgG ratio demonstrated a significant association with QMGS in both LMM and within-person analyses, suggesting it is a dynamic biomarker of within-person clinical status rather than merely reflecting cross-sectional severity. During OFA treatment, the declining ratio reflects a concerted immunologic shift: IgM decreases due to the depletion of OFA-sensitive naïve/unswitched B cells, 2 while IgG is maintained by depletion-resistant long-lived plasma cells. In MuSK-MG, this likely correlates with clinical improvement, as the depletion of MuSK-specific CD20+ memory cells indirectly reduces autoreactive plasmablasts. Thus, the ratio may serve as a surrogate marker for effective B-cell subset depletion and the resultant shift in the humoral immune milieu, aligning with—though not necessarily causing—symptom relief. Interestingly, the positive correlation in AChR-MG may involve distinct mechanisms, such as IgM-mediated complement activation. 42 Kappa and Lambda chains exhibited patterns similar to IgG.
Following OFA treatment, distinct immunological patterns emerged between subtypes: synchronized declines of BAFF and APRIL in MuSK-MG, versus transient BAFF elevation with sustained APRIL in AChR-MG. This mirrors their divergent immunopathology: MuSK-MG is a B cell/plasmablast-driven disease (IgG4), while AChR-MG is a T cell-dependent, germinal center-driven disease (IgG1/IgG3) sustained by long-lived plasma cells.2,3 We hypothesize that in MuSK-MG, effective depletion of the central pathogenic B-lineage cells could disrupt the inflammatory loop, reducing cytokine demand. In AChR-MG, the persistent long-lived plasma cell pool (resistant to OFA) might continue to consume APRIL via B-cell maturation antigen for survival, while the removal of mature B cells could trigger a homeostatic BAFF rebound that may reseed the autoreactive B-cell pool.43,44 If validated, this model suggests MuSK-MG may respond adequately to B-cell depletion alone, whereas AChR-MG could potentially require a dual-targeting strategy that combines B-cell depletion with inhibition of the BAFF/APRIL axis to target both upstream reservoirs and downstream antibody-producing cells. The partial BAFF inhibition by corticosteroids may partly explain the accelerated MSE attainment observed with combination therapy in our cohort. 45
Moreover, sCD40L, generated by CD40L shedding, promotes B cell proliferation, plasmacytoid differentiation, and IgG secretion.46,47 Previous research has found elevated sCD40L levels both in MuSK-MG and refractory AChR-MG,48,49 which can decrease significantly after tacrolimus treatment. 48 In our cohort, OFA consistently reduced sCD40L levels over time without intergroup differences, indicating effective pathway suppression independent of autoantibody status.
However, unlike the aforementioned cytokines primarily targeting downstream pathways, MIF, secreted by diverse cell types, promotes B-cell survival and proliferation via CD74 signaling.14,15 Although MIF is abundantly expressed in early-onset MG thymus and correlates with disease activity in treatment-naïve or conventionally immunosuppressed patients, we observed no such association with QMGS in OFA-treated individuals.14,15 This discrepancy may stem from several factors: the inclusion of AChR-MG with comorbid autoimmune diseases or prior thymectomy; OFA-mediated depletion of CD74-expressing effector cells, which may decouple MIF levels from clinical symptoms; and the rapid rebound of MIF within 31–90 days since last treatment in AChR-MG, highlighting dynamics that cross-sectional analysis cannot capture. Interestingly, the trend of higher MIF levels in AChR-MG patients throughout the treatment course, though not statistically significant, aligns with the hypothesis of a more prominent T-cell-driven autoimmunity in this subtype. Given MIF’s established role in T/B-cell activation cascades, 14 its sustained elevation may reflect a resilient T-cell network that is less completely suppressed by B-cell depletion alone, offering a compelling insight for future research.
Our study has limitations. First, its retrospective design and small sample size from a rare disease limit statistical power and may introduce selection bias. Critically, the absence of a control group (e.g., healthy individuals or refractory MG patients on other therapies) prevents definitive attribution of observed immunological changes specifically to ofatumumab, as opposed to natural fluctuation or concomitant therapies. Methodologically, the use of different assays (ELISA for AChR-Ab; RIA for MuSK-Ab) may affect the comparability of antibody titer trajectories between groups, as assays differ in sensitivity and dynamic range. Cell-based assays are now recommended. Furthermore, the observed clinical efficacy should be interpreted within the context of our proactive, biomarker-driven dosing intensity, affecting comparisons with studies using different thresholds or fixed intervals. Finally, the absence of long-term follow-up precludes assessment of sustained efficacy and long-term safety, a key gap given OFA’s novelty in MG. Future prospective studies with larger cohorts, controlled designs, and extended follow-up are warranted to validate the retreatment threshold, establish long-term outcomes, and clarify the specific immunological effects of OFA.
Conclusion
Ofatumumab demonstrates greater clinical efficacy in MuSK-MG than in AChR-MG, facilitating earlier MSE in patients with shorter disease duration and concomitant corticosteroid use. Mechanistically, OFA induces significantly greater reductions in BAFF and APRIL levels in MuSK-MG. Within-person increases in BAFF levels correlate with higher QMGS in MuSK-MG, while IgM/IgG is associated with QMGS in both subtypes.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-docx-1-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-docx-2-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-docx-3-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-10-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-10-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-11-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-11-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-12-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-12-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-4-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-4-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-5-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-5-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-6-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-6-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-7-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-7-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-8-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-8-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-9-tan-10.1177_17562864261421717 – Supplemental material for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study
Supplemental material, sj-tif-9-tan-10.1177_17562864261421717 for Differential response to ofatumumab in anti-AChR and anti-MuSK positive myasthenia gravis patients: a single-center retrospective study by Qian Zhou, Yuzhen Ouyang, Fei Jiang, Qiuming Zeng, Ting He, Zeyi Wen, Guanzhong Shi, Xiaohua Dong, Haobing Cai, Wei Xiang, Miao Su, Kailin Li and Huan Yang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
