Abstract
Background:
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is the most frequently observed autoimmune neuropathy in patients with diabetes mellitus (DM). While intravenous immune globulin (IVIG) is a well-established treatment for CIDP, its efficacy in diabetic patients remains uncertain due to their exclusion from prior randomized trials, largely because of concerns about confounding diabetic axonal neuropathy.
Objectives:
To evaluate the effectiveness of IVIG therapy in CIDP patients with diabetes mellitus (CIDP-DM) compared to those without diabetes (CIDP).
Design:
Multi-center, prospective, observational study after at least 3 monthly infusions of IVIG therapy.
Methods:
Thirty-six patients meeting diagnostic criteria for CIDP were enrolled and stratified into CIDP or CIDP-DM. All patients were followed for a minimum of 3 months after initiating IVIG therapy. Clinical outcomes were assessed at baseline (visit #1) and after 3 monthly IVIG infusions (visit #4) using the adjusted Inflammatory Neuropathy Cause and Treatment Disability Score, the Rasch-built Overall Disability Scale, and the Chronic Acquired Polyneuropathy Patient-reported Index, measured at baseline and at the point of maximal improvement.
Results:
No significant differences were observed in clinical outcomes, treatment-related adverse events, or tolerance between CIDP and CIDP-DM groups, indicating comparable effectiveness of IVIG therapy. However, subgroup analyses revealed that longer duration of diabetes and elevated HbA1c levels were associated with delayed response to IVIG, likely due to cumulative axonal degeneration.
Conclusion:
Despite the small number of enrolled patients, IVIG appears equally effective in CIDP patients with and without diabetes. Earlier initiation of IVIG treatment should be considered in CIDP-DM patients to mitigate potential delays in therapeutic response associated with a possibly chronic diabetic neuropathy-related component.
Keywords
Introduction
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is one of the most common autoimmune inflammatory neuropathies in patients with diabetes mellitus (DM) with the occurrence of CIDP up to 11 times higher in DM patients1,2 and prevalence increasing with age. 2 Because 16.9% of patients with diabetic neuropathies meet the electrophysiologic criteria of CIDP3–6 there have been concerns, dating back to over 40 years ago, 7 as to whether the association is coincidental, if one cause is contributing to the other or even whether some clinical, neurophysiologic, and pathologic features are indistinguishable between the two during the disease course or even if any pathogenetic mechanisms may have similarities.
Although intravenous immune globulin (IVIG) has been proven to be a safe and effective treatment for CIDP in randomized controlled trials, 8 the optimal treatment benefits of CIDP in patients with DM remain uncertain.9–12 This is primarily because patients with DM have been excluded from most randomized CIDP trials due to concerns about the confounding effects of diabetic neuropathy. 5 As a result, it remains unclear whether IVIG therapy in CIDP-DM patients should be initiated earlier, whether the response is suboptimal possibly due to underlying axonal degeneration caused by DM, or whether such patients may actually have an enhanced response to IVIG given the potential autoimmune basis of DM itself. 12 Therefore, increased awareness of CIDP associated with DM is essential to promote earlier diagnosis and timely initiation of IVIG treatment considering that among all neuropathies seen in DM, only CIDP is treatable.
The purpose of this study is to: (a) determine the efficacy of IVIG in the treatment of CIDP patients with DM compared to CIDP patients without DM, by measuring changes in scores from baseline to maximal improvement; (b) assess whether differences in treatment response justify earlier initiation of therapy in DM patients with neuropathy meeting CIDP criteria; and (c) explore whether broader inclusion of CIDP-DM patients is warranted in future CIDP trials to better reflect real-world clinical populations and end their systematic exclusion from immunotherapeutic research studies.
Methods
This is an observational study designed to prospectively and systematically analyze collected research data, including validated investigator/patient assessments, demographics, treatment responses, disease-related clinical features such as proximal muscle weakness, or effects of DM among patients with CIDP associated with DM or without DM treated with IVIG at the standard therapeutic regimen of 2 g/kg/month or maintenance dose of 1 g/kg/month. Data of enrolled subjects were collected into a central repository from each IVIG infusion and from validated assessments performed by investigators, study staff, and infusion nursing staff.
Study groups
This observational study was conducted at the following five sites: Eisenhower Health (Palm Springs, CA, USA); Los Angeles Neuromuscular Center (Los Angeles, CA, USA); Mass General Brigham (Boston, MA, USA); Massachusetts General Hospital (Boston, MA, USA); Thomas Jefferson University (Philadelphia, PA, USA); and Vagelos College of Physicians and Surgeons (New York, NY, USA). Prospective clinical data related to patients’ responsiveness to IVIG were collected from patients with confirmed diagnosis of CIDP with DM and analyzed against a comparator group of CIDP patients without DM. Candidate patients were invited to participate after they have been prescribed IVIG from a participating study investigator and had signed informed consent after assessed for eligibility. Those who met the eligibility requirements were enrolled in the study and placed in the appropriate observational study arm Diabetic (A) or Non-Diabetic (B).
The Diabetic arm (A) included participants with CIDP and DM who have been receiving IVIG therapy for ⩾3 months (current), while the Non-Diabetic (Arm B), the comparator group, included CIDP patients without DM who have been receiving IVIG therapy for ⩾3 months (current comparator).
Key data assessments
These included: (a) demographics, sex, race, geographic location; (b) clinical data obtained from the patients’ charts including diagnostic criteria, electromyographic parameters fulfilling CIDP criteria, comorbidities, and concomitant medications; (c) laboratory assessments as standard of care; (d) treatment history; and (e) IVIG treatment information: dosing/dose frequency, rate, therapy adjustments/changes in therapy, adverse reactions/tolerability.
Exclusion criteria
CIDP patients were not eligible for inclusion if: (a) did not fulfill the CIDP criteria and there were uncertainties as to whether the neuropathy was more related to diabetes rather than CIDP; (b) discontinuation of IVIG was planned within 3 months of screening; (c) lack of records related to onset of DM and/or symptoms and treatment; and (d) patients were taking systemic corticosteroids at prednisone-equivalent doses of ⩾10 mg/day.
Evaluations and assessments
Subjects were evaluated using the Inflammatory Neuropathy Cause and Treatment (INCAT) disability score as well as the Rasch-built Overall Disability Scale (R-ODS), Chronic Acquired Polyneuropathy Patient-Reported Index (CAP-PRI), and Medical Research Council (MRC) sum score, which were collected at baseline (visit #1) and after 3 monthly infusions (visit #4) to assess the maximal improvement following IVIG therapy.13,14 The mean change in grip strength, measured with a Martin Vigorimeter from baseline to visit #4 was also assessed. 15 Patients were followed for 3 additional months (6 months overall) to capture safety data and perform routine assessments. Key data were collected and analyzed for comparison of demographics, treatment, dosing, and outcomes between subjects with and without DM receiving treatment. The overall duration of the study was approximately 24 months, considering the slow recruitment of patients fulfilling the study criteria, with each subject’s participation duration of approximately 6 months.
Objectives
The primary objective was responsiveness to IVIG from baseline to maximal improvement after 3 monthly infusions on adjusted INCAT disability score. Improvement of a >1 point increase in adjusted INCAT disability score was considered treatment-responsive.
Secondary objectives were assessed from baseline to maximal improvement after 3 monthly IVIG infusions based on: (a) mean change in R-ODS; (b) mean change in CAP-PRI; (c) MRC sum score; and (d) mean change in grip strength, measured with a Martin Vigorimeter (if these data were available).
Statistics
Statistical tests were employed to evaluate the differences in outcomes at the baseline to at least after 3 monthly infusions based on: MRC (commonly used to assess muscle strength from 0 (no muscle contraction) to 5 (normal strength)), INCAT, the R-ODS, and the CIDP Activity and Prednisone-Induced (CAP-PRI) score, which represent pain relief or improvement between the groups. Scores between the diabetic (Group A) and non-diabetic (Group B) measuring the sum score were compared using independent t tests to compare the means at the second study visit between the groups.
Results
Demographics
The A group consisted of 16 patients and the B group of 20. The age distribution across both groups demonstrates variability, with group A displaying a slight skew toward older individuals as compared to Group B (Table 1). In terms of body mass index, individuals in Group A consistently exhibit higher values relative to those in Group B, aligning with the summarized data (Figure 1 and Table 1). The distribution of HbA1C levels, which serve as indicators of blood glucose control, was generally at higher levels in Group A, classified as diabetic, than in Group B, identified as non-diabetic.
Demographic summary.
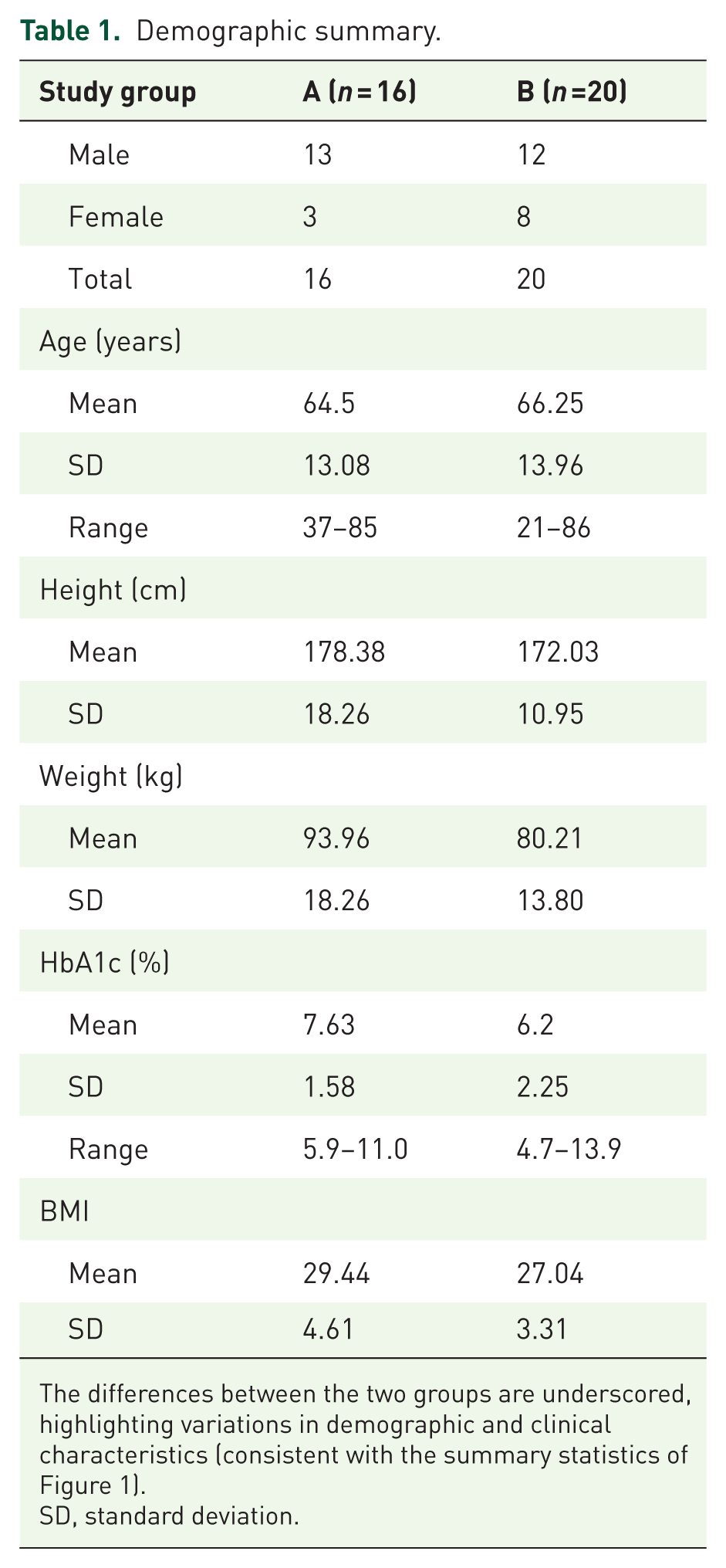
The differences between the two groups are underscored, highlighting variations in demographic and clinical characteristics (consistent with the summary statistics of Figure 1).
SD, standard deviation.

Descriptive statistics visuals for diabetic versus non-diabetic groups. The analysis of demographic and clinical characteristics between the two groups reveals distinct patterns in age, BMI, HbA1C levels, gender distribution, and diabetic status. Specifically, the age distribution across both groups demonstrates variability, with the Diabetic group displaying a slight skew toward older individuals as compared to Non-Diabetic group. In terms of BMI, individuals in Diabetic group consistently exhibit higher values relative to those in Non-Diabetic group. Furthermore, the distribution of HbA1C levels, which serve as indicators of blood glucose control, presents a broader range and generally higher levels in the Diabetic group than in the Non-Diabetic group.
Outcome measures
As shown on Table 2, the MRC analysis suggests a slight but not statistically significant improvement in muscle strength for the diabetic group (Group A) from baseline to the follow-up study visit, compared to the non-diabetic group (Group B) which showed a slight decrease in muscle strength on average (Table 2 and Figure 2); no statistically significant difference was, however, noted between the diabetic and non-diabetic groups in terms of the efficacy of IVIG treatment (p = 0.343).
Mean baseline MRC sum score versus maximum improvement.

A slightly higher mean MRC score (41.63) is noted in group A (CIDP-DM) compared to group B (40.50), suggesting a marginal but not statistically significant difference in muscle strength outcomes between the two groups (p-value = 0.694) confirming similar efficacy of IVIG between diabetic and non-diabetic CIDP patients.
CIDP, chronic inflammatory demyelinating polyradiculoneuropathy; DM, diabetes mellitus; IVIG, intravenous immune globulin; MRC, Medical Research Council.
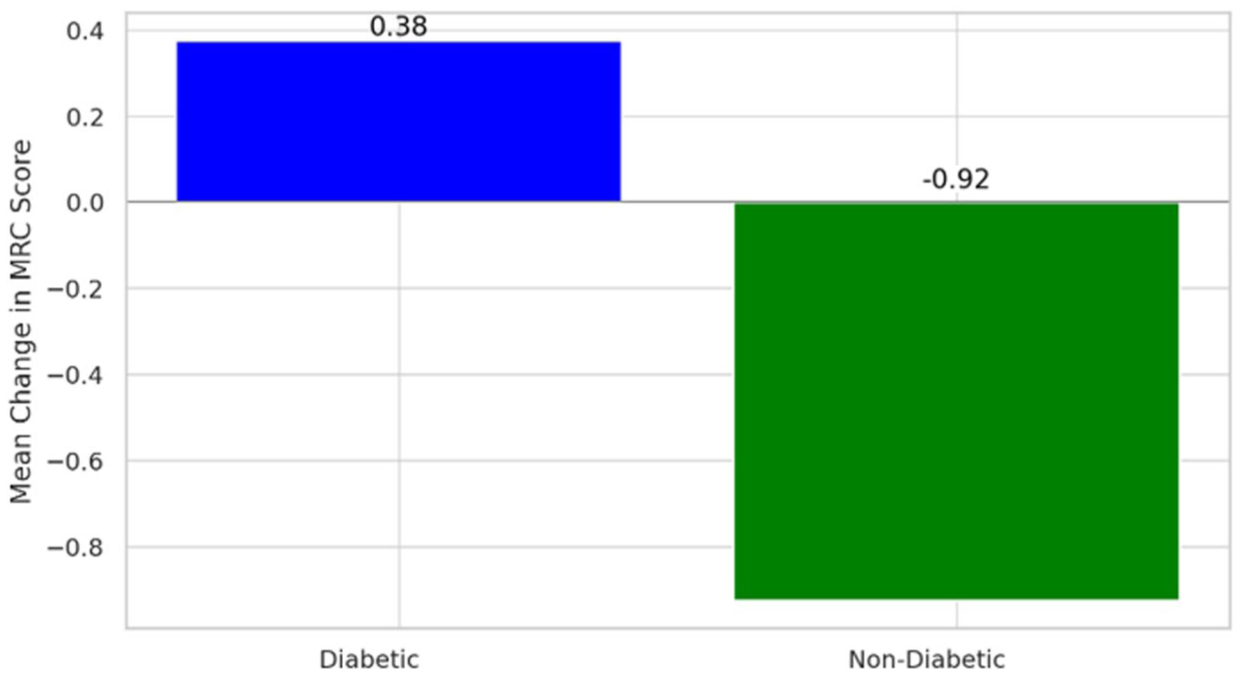
Average MRC score change from baseline to second visit. A slight improvement in muscle strength for the Diabetic group (Group A) from baseline to the second study visit, as indicated by the positive change in the MRC sum score. Conversely, the Non-Diabetic group (Group B) shows a slight decrease in muscle strength on average.
Analysis of the INCAT, R-ODS, and CAP-PRI scores across the observations in all 36 patients also reveals nuanced shifts in patient outcomes, among group A and group B, as shown in Figure 3. For INCAT scores, a noted mean change of −0.33 suggests a marginal average decline, with the data exhibiting variability as indicated by the standard deviation of 1.07. Notably, the range of change spans from a significant decrease (minimum change of −3) to an improvement (maximum change of 2), with inter-quartile analyses revealing that 25% of subjects experienced a decrease of 1 point or more, and 75% showed no change or improvement, highlighting the distribution’s skew toward stability or positive adjustment. Similarly, the R-ODS scores demonstrated a mean decrease of −0.72, with a broader variability (standard deviation of 1.34) among subjects. This measure’s distribution spans from a substantial decrease (minimum change of −5) to a slight improvement (maximum change of 2), with the median change at 0, indicating no change for half of the subjects, underscoring a mixed response in recovery or progression. For CAP-PRI scores, the analysis presents a mean decrease of −1.06, signifying an overall decline, accompanied by considerable variability (standard deviation of 1.90) in changes among the subjects. The range of change extends from a significant decrease (minimum change of −8) to an improvement (maximum change of 2), with the median change of −1, illustrating a predominantly negative shift with some instances of recovery or improvement. Overall, these statistics offer a comprehensive view of the changes in clinical measures over the course of the study and highlight the average trends and range of individual responses. The slight decreases observed in the mean changes for INCAT, R-ODS, and CAP-PRI scores indicate no meaningful differences from the first to the last follow-up visit between the two groups despite the variably observed minor score differences in outcome measures among the subjects, reflecting a mixture of improvements, stability, or decline in scores.

Comparison of INCAT, R-ODS, and CAP-PRI scores at the second visit between groups. INCAT, R-ODS, and CAP-PRI scores between the diabetic (in blue) and non-diabetic (in green) groups. Each bar represents the mean score for the respective clinical measure within each group. INCAT: Shows a slight difference in mean scores between the groups, with the diabetic group having a slightly higher mean score than the non-diabetic group. However, this difference is not statistically significant. R-ODS: The non-diabetic group has a higher mean score compared to the diabetic group, indicating better outcomes in terms of disability score. Again, this difference is not statistically significant. CAP-PRI: The diabetic group shows a higher mean score compared to the non-diabetic group, suggesting a difference in pain relief or improvement. This difference is also not statistically significant.
MRC scores sub-analysis of CIDP-DM in reference to HbA1C (Group A)
A slight, but not significant, improvement of HbA1c levels was noted after IVIG from baseline (Figure 4). Most notable—even if not statistically significant—improvements or worsenings in this subset of 16 patients measured by MRC scores were: (a) Right Plantar Flexors improved from a mean of 4.27–4.67 (+0.40); (b) Left Foot Extensors improved from 3.73 to 3.93 (+0.20) and (c) Left Tibialis Anterior and Right Quadriceps Femoris showed an improvement of +0.13. Several muscle groups, including Right Iliopsoas, Left Wrist Extensors, and Left Iliopsoas, maintained their mean scores without any improvement or decline. These minor changes in muscle strength had a negative correlation with HbA1C levels (−0.242896) with correlation coefficients ranging from −0.004051 to −0.355198, indicating varying degrees of weak to moderate associations, but overall suggesting that higher HbA1C levels might be associated with less improvement or even deterioration in muscle strength across all measured groups.
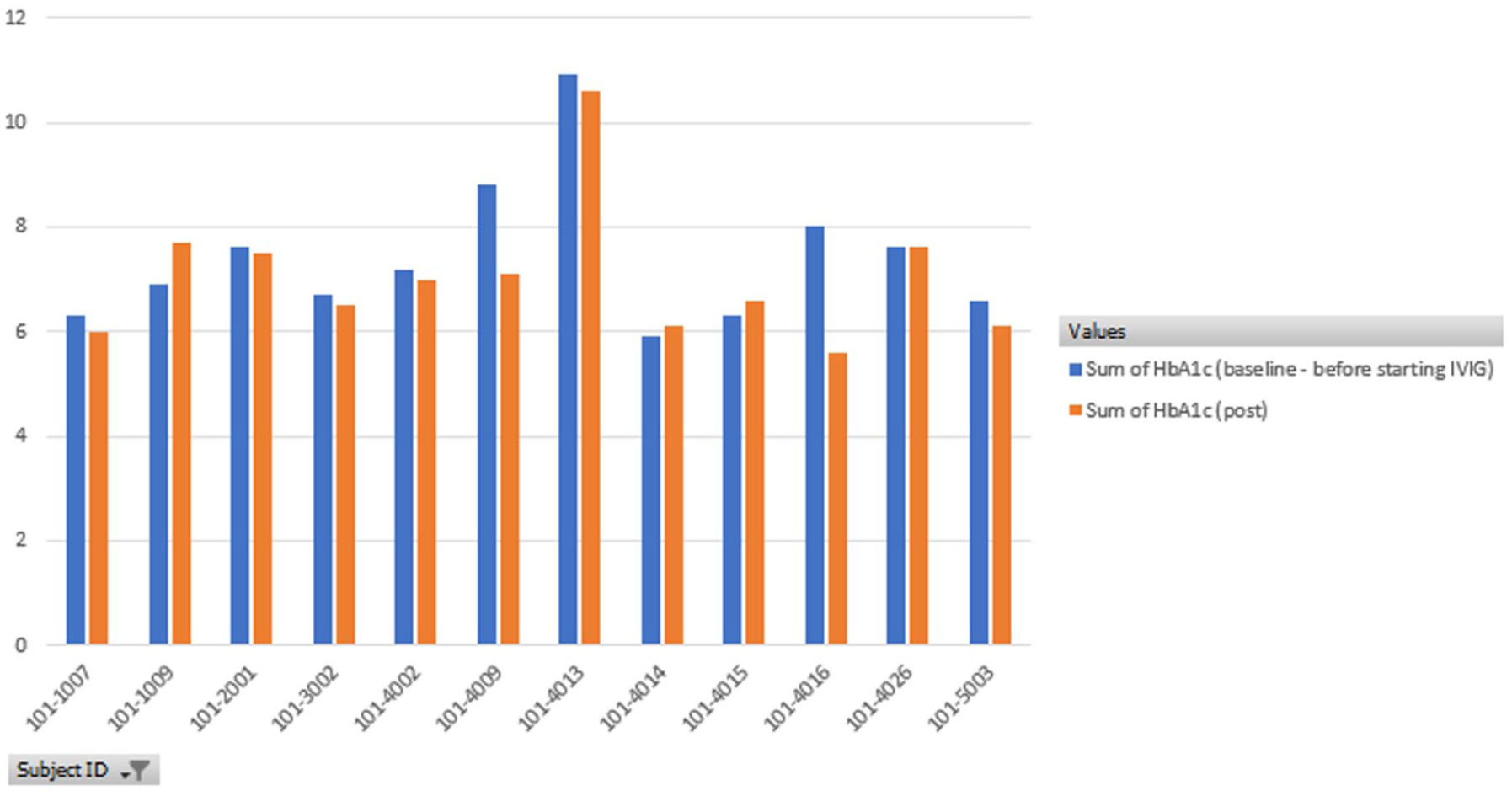
Hemoglobin A1C at baseline (blue) and post study (red). A slight improvement on average was noted in HbA1c levels after IVIG from baseline, but the difference was not significant.
Safety findings
Most patients reported not having any treatment-related significant adverse events, regardless of diabetes. Three patients had mild headache; one reported unplanned hospitalization (unrelated to IVIG); and another a visit to the emergency department (unrelated to IVIG).
Discussion
The study provides useful insights into the treatment of CIDP in patients with coexisting DM, suggesting that IVIG therapy yields comparable clinical benefits in both diabetic and non-diabetic patients based on the MRC, adjusted INCAT, R-ODS, and CAP-PRI scores. The results challenge the long-standing concern that diabetic neuropathy may confound the treatment response in CIDP and support the inclusion of diabetic patients in future therapeutic studies. The lack of statistically significant differences in outcome measures among the IVIG-responders in the two groups reinforces, despite the small sample size, the view that IVIG remains effective despite the possibility of any diabetes-related neuropathic changes. Accordingly, when CIDP is appropriately diagnosed based on the updated criteria, 15 IVIG can be a valuable treatment option regardless of diabetes, especially if DM is under reasonably good control.
The findings also highlight the clinical value of early immunotherapy initiation because patients with prolonged or poorly controlled diabetes, based on elevated HbA1c levels, experience a seemingly delayed or diminished therapeutic response, underscoring the need for timely recognition and initiation of IVIG in CIDP patients with DM before advanced glycemic neuropathic complications obscure the clinical picture or limit treatment efficacy. Interestingly, IVIG administration in patients with stiff person syndrome with diabetes in a controlled study had previously shown slight improvement of diabetes in some patients, 16 likely due to IVIG immunomodulatory effects as described. 17 Furthermore, DM did not appear to increase the risk of adverse events associated with IVIG, collectively concluding that IVIG is as safe and well-tolerated in CIDP patients with or without diabetes. The information is especially useful because in CIDP with DM corticosteroids are not used6,10,18,19 although they are effective based on controlled studies 20 and CIDP was called from the outset as a “steroid-responsive polyneuropathy.”7,9
The use of multiple validated outcome measures with detailed visual analytics allowed for a nuanced appreciation of both group-level trends and individual variability in treatment response. This approach strengthens the reliability of our findings but also provides a valuable framework for future studies aiming to personalize treatment strategies in complex patient populations. Despite the strengths of our prospective, multi-centered study design, several limitations should be acknowledged. The small sample size, while adequate for exploratory analysis, may limit the generalizability of the findings, and the absence of statistically significant differences may simply reflect insufficient power rather than a genuine absence of differences. Additionally, the observational nature of the study without blinding controls precludes definitive conclusions about any diabetic effects on CIDP causality or neuropathy progression. Future randomized controlled trials that include diabetic patients are warranted to confirm these results and explore whether glycemic control influences long-term outcomes in CIDP.
Conclusion
In conclusion, despite the noted limitations, our study supports the use of IVIG in patients with CIDP and coexisting diabetes, with outcomes comparable to those without diabetes, and advocates for earlier treatment initiation in diabetic patients fulfilling CIDP criteria and broader inclusion of such patients in future CIDP trials to better reflect real-world clinical populations.
Footnotes
Acknowledgements
We extend our deepest gratitude to Dr King Engel and Dr Valerie Askanas and for their lifelong dedication to the field of neuromuscular research, and initial guidance on the study concept. We are also grateful for the support provided by KabaFusion, whose team delivered infusion services to patients participating in this study.
