Abstract
Background:
Siponimod, a selective sphingosine-1-phosphate receptor modulator was approved for patients with Secondary progressive Multiple Sclerosis (SPMS) with ongoing disease activity. However, its real-world positioning and management after discontinuation remain challenging.
Objectives:
To evaluate the real-world use of siponimod in Italian real world setting, focusing on discontinuation rates, possible predictors, and post-treatment management.
Methods:
A retrospective, multicenter study on patients treated with siponimod across six Italian MS centers. Demographics, prior disease modifying therapy (DMTs), reasons for discontinuation, adverse events, and subsequent therapies were collected. Statistical analyses included propensity-adjusted Cox model.
Results:
A total cohort of 188 patients (63.8% female, median age 52 years) was enrolled, out of them 76(40.4%) discontinued siponimod, with a median treatment duration of 26 months. The main reason for discontinuation was safety concerns (72.4%), particularly persistent lymphopenia (43.6%) and recurrent infections (27.3%). Disease activity accounted for 27.6% of discontinuations. No significant demographic or clinical predictors of discontinuation were identified. After discontinuation, 49 patients (64%) started a new DMT, most commonly ocrelizumab (n = 22) or cladribine (n = 15), while 25 (32.9%) received no further therapy.
Conclusion:
High discontinuation rates, mainly due to safety, and frequent post-treatment gaps highlight the need for improved, individualized management strategies for SPMS after siponimod.
Plain language summary
Siponimod is a treatment for secondary progressive multiple sclerosis (SPMS), aimed at slowing disease progression. This study reviewed real-life data from six Italian MS centers, including 188 people treated with siponimod. About 40% stopped treatment after around two years, mostly due to side effects such as low white blood cells or infections. Some stopped because their MS became active again. After discontinuation, two-thirds started another therapy—most often ocrelizumab or cladribine—while one-third received no further treatment. No clear patient characteristics predicted who stopped therapy. These findings show that siponimod is often discontinued for safety reasons and that better strategies are needed to guide treatment choices and ensure ongoing care for people with SPMS.
Introduction
Siponimod, a selective sphingosine-1-phosphate (S1P) receptor modulator, was approved for the treatment of individuals with secondary progressive multiple sclerosis (SPMS) who continue to exhibit disease activity. 1 The transition from relapsing-remitting multiple sclerosis (RRMS) to SPMS remains a pivotal stage in the disease course and is associated with a reduction in the availability of disease-modifying therapies (DMTs).2–4 In this context, siponimod is the only treatment option specifically indicated for individuals with SPMS who exhibit clinical or radiological evidence of ongoing disease activity.
The introduction of siponimod has generated considerable debate regarding its optimal placement within the MS treatment algorithm. 2 Although its approval was initially welcomed as a much-needed option for patients with active SPMS for whom other therapies were unsuitable, the challenge of identifying the ideal patient profile has led to ongoing discussions, owing to the difficulty in clinically characterizing this patient population.5–7
In clinical practice, siponimod has been primarily considered for patients at the intersection between late RRMS and early, active SPMS—those who continue to show signs of inflammatory activity but are progressing despite prior treatments. This positioning reflects the complexity of the SPMS disease process and emphasizes the need for more individualized therapeutic strategies.8,9
Safety considerations have also played a central role in shaping the use of siponimod.5,10,11 The risk of adverse events (AEs), including cardiovascular effects, infections, and laboratory abnormalities such as lymphopenia, has necessitated careful patient selection and monitoring. 12 Furthermore, the management of therapy discontinuation—whether due to lack of efficacy, AEs, or patient preference—has emerged as a critical issue. The development of clear exit strategies is essential to minimize the risk of disease reactivation or progression following siponimod discontinuation and to ensure continuity of care. These strategies may involve transitioning to alternative DMTs, implementing close clinical and radiological monitoring, or, in some cases, temporarily interrupting treatment.
We have designed a multicenter Italian real-world study with the primary aim of evaluating the positioning and safety profile of siponimod in patients with SPMS in the routine clinical practice of specialized MS centers in Italy.
Methods
Study design and setting
This is a multicentric, retrospective, observational study on prospectively collected data conducted across six Italian MS centers. Data were extracted on April 30, 2025 from electronical medical records.
Study population
We included patients (a) aged >18 years, (b) who had a diagnosis of active SPMS according to 2017 McDonald criteria, 13 and (c) who initiated treatment with siponimod according to Italian prescription rules between January 2022 and October 2024 in the participating centers to ensure at least 6 months of follow-up.
We excluded data from patients who had incomplete baseline data or lost to follow-up.
Outcomes and covariates
The primary outcome of this study was the discontinuation of siponimod therapy, analyzed using Cox proportional hazards survival analysis adjusted by propensity scores (PS) to account for potential confounding factors. This approach allowed for the identification of predictors associated with therapy discontinuation.
Secondary outcomes included: the specific causes of siponimod discontinuation; the influence of prior DMTs on treatment persistence and outcomes; post-siponimod management strategies (“exit strategies”), including subsequent DMTs and related clinical outcomes.
Covariates considered in the analyses included demographic and clinical variables, pre-siponimod disease activity, and post-siponimod outcomes. Baseline characteristics comprised age at prescription, sex, comorbidities, disease duration, lag time, baseline Expanded Disability Status Scale (EDSS) and prior DMTs.
Evidence of disease activity was defined as the presence of relapses, confirmed disability progression (CDP), and active Magnetic Resonance Imaging (MRI) lesions (new or enlarged T2 lesions and/or contrast-enhancing lesions). 14 Relapses were defined as new or worsening neurological symptoms lasting at least 24 h, in the absence of concurrent illness or fever, and occurring at least 30 days after a previous relapse. CDP was defined as an increase in EDSS by ⩾1.5 points for those with a baseline EDSS of 0, by ⩾1 point for a baseline EDSS ⩽5.5, or by ⩾0.5 points for a baseline EDSS >5.5, sustained for at least 12 weeks. EDSS scores recorded within 30 days of a relapse onset were excluded. MRI activity was defined as the appearance of new T1-gadolinium enhancing brain lesions and/or new or enlarging T2 brain lesions.
AEs were classified according to the European Medicines Agency definitions. Lymphopenia was defined according to the Common Terminology Criteria for Adverse Events (CTCAE). 15
Our multicenter study adhered to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for reporting. 16
Statistical analysis
Descriptive statistics summarized baseline characteristics, treatment patterns, and outcomes. Categorical variables were reported as frequencies and percentages; continuous variables as means and standard deviations or medians and interquartile ranges (IQRs). Group comparisons used Chi-square or Fisher’s exact test for categorical variables and t test or Mann–Whitney U test for continuous variables.
We conducted a time-to-event analysis to evaluate predictors of siponimod discontinuation in a real-world cohort using a Cox proportional hazards model weighted by inverse probability of treatment weighting (IPTW) based on PS. The PS was estimated via logistic regression, modeling the probability of discontinuation as a function of baseline covariates: age at the time of siponimod prescription, sex, comorbidities, lag time, disease duration, EDSS at siponimod initiation, number of previous DMTs. The proportional hazards assumption was verified for all covariates using Schoenfeld residuals and graphical diagnostics. The potential impact of treatment center was evaluated by including the center variable as a categorical covariate in the multivariate model. A global Wald test was performed to assess the overall effect of center in the logistic regression model. The test yielded a Wald statistic of 2.38 with a p-value of 0.66, indicating no significant center effect, so it was not added to the analysis.
Variance inflation factor (VIF) was calculated to assess multicollinearity among predictor variables. A VIF value greater than 5 was considered indicative of moderate multicollinearity, while values above 10 were interpreted as evidence of significant multicollinearity.
All statistical tests were two-sided, and a p-value less than 0.05 was considered statistically significant.
All analyses were performed in Python 3.11 using the following packages: pandas 2.2.2, numpy 1.26.4, scikit-learn 1.4.2, lifelines 0.28.0, matplotlib 3.8.4, and seaborn 0.13.2.
Results
A total of 188 patients was enrolled, 63.8% were female, with a median age at siponimod prescription of 52 (44.8–58.0) years. Baseline characteristics are shown in Table 1.
Baseline demographic, clinical, and genetic characteristics of the SPMS cohort treated with siponimod.

Data are presented as n (%) for categorical variables, and as mean ± standard deviation and median (IQR) for continuous variables. CYP2C9 genotype distribution is also shown.
DMT, disease modifying therapy; EDSS, Expanded Disability Status Scale; IQR, interquartile range; SPMS, secondary progressive multiple sclerosis.
Of these patients, 76 (40.4%) discontinued therapy with siponimod, and the median time on the index drug was 26 (IQR 15–35.3) months.
The main reason for siponimod discontinuation was safety concerns, reported in 55 (72.4%) patients (Table 2). The most frequently reported AEs were persistent lymphopenia (grade 4 according to the CTCAE classification) in 24 (43.6%) patients, and recurrent infections (upper and lower respiratory tract and urinary infections) in 15 (27.3%). The second most frequent reason was disease activity, reported in 21 (27.6%) patients.
AEs leading to siponimod discontinuation.
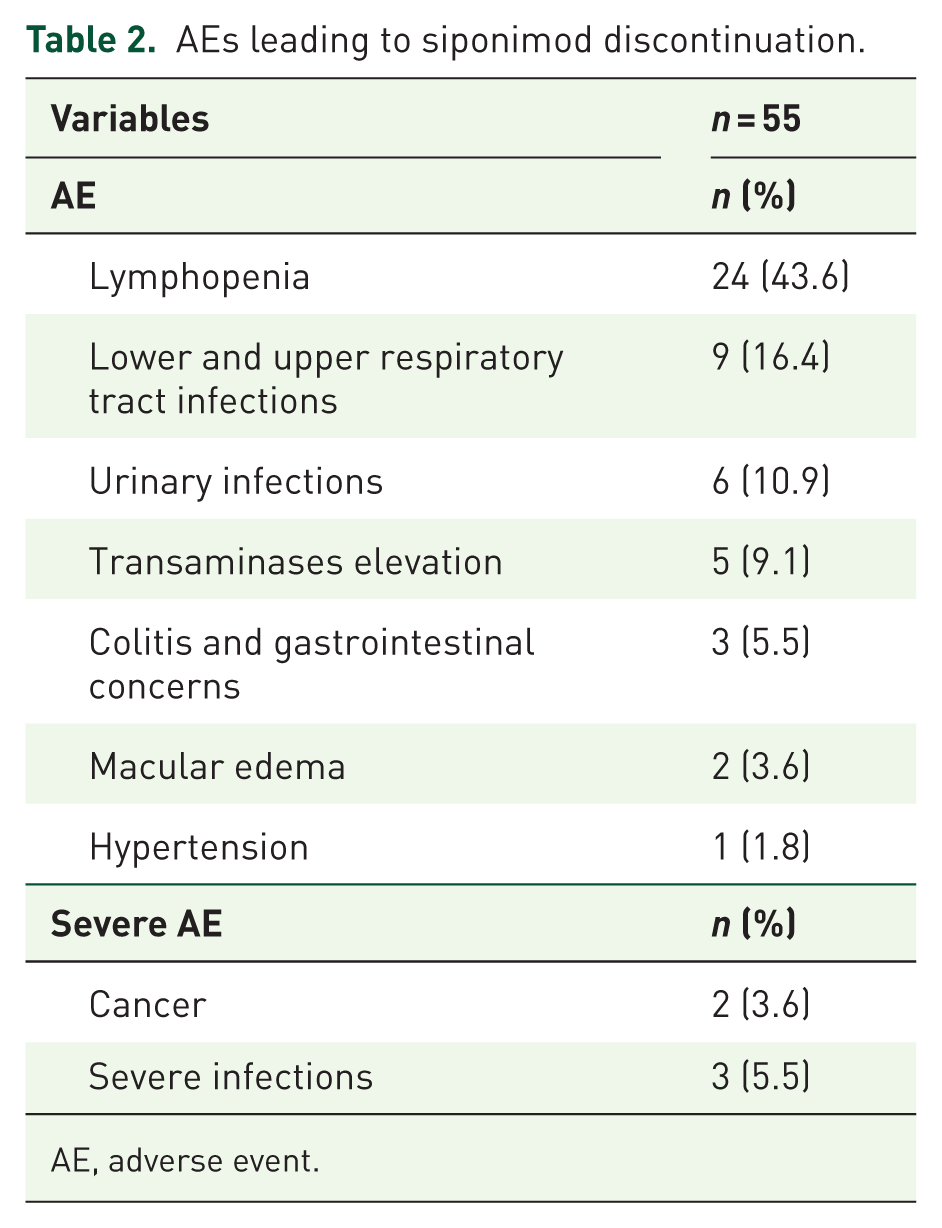
AE, adverse event.
The highest proportion of patients who discontinued siponimod were previously exposed to Ocrelizumab (70%; Figure 1).

Distribution of siponimod discontinuation rate according to DMT before its prescription.
Cox IPTW-PS regression model
The Cox IPTW-PS regression model assessing the outcome of DMT discontinuation did not identify any significant predictors among demographic or clinical characteristics (see Figure 2, Table 3). This result was confirmed after PS-IPTW adjustment (Figure 2, Table 3).

Cox model for siponimod discontinuation unadjusted and IPTW-PS adjusted plot.
Unadjusted and IPTW-PS adjusted coefficients from Cox regression model for siponimod discontinuation.

CI, confidence interval; DMT, disease modifying therapy; EDSS, Expanded Disability Status Scale; IPTW-PS, inverse probability treatment weighting propensity score.
Exit strategies post siponimod discontinuation
Following siponimod discontinuation, 49 patients (64%) initiated a new DMT. The most commonly prescribed DMTs were ocrelizumab (n = 21) and cladribine (n = 15; Figure 3).

DMTs prescribed post siponimod discontinuation.
Notably, 25 patients (32.9%) did not initiate any subsequent DMT following siponimod withdrawal. Among these patients, the median age was 56.0 years (Q1–Q3: 51–60), baseline EDSS was 6.5 (Q1–Q3: 4.0–6.5), disease duration was 22.0 years (Q1–Q3: 16–27), and the mean number of previous DMTs before siponimod was 2.4 ± 1.3. Of these 25 patients, 6 (24%) discontinued siponimod due to disease worsening, whereas 19 (76%) discontinued it for safety concerns.
During the available follow-up period (median 18 months, Q1–Q3 14–21), nine patients developed disease activity after discontinuing siponimod. Among them, only one was not receiving a new DMT. Among these patients, six had new MRI lesions, one experienced EDSS progression, one showed both MRI activity and a relapse, and one had a relapse only.
Discussion
This multicenter, real-world study provides an evaluation of siponimod utilization in Italian patients with SPMS, with an emphasis on patterns of treatment discontinuation and subsequent therapeutic strategies.
Among 188 patients, 40.4% discontinued siponimod, with a median treatment duration of 26 months. The principal reasons for discontinuation were safety concerns—most notably persistent lymphopenia and recurrent infections—followed by disease activity. Notably, no significant predictors of discontinuation were identified, even after adjustment for baseline characteristics, underscoring the multifactorial and unpredictable nature of treatment cessation in this population.
A finding that warrants further investigation is the increased risk of siponimod discontinuation among patients previously treated with ocrelizumab.17–19 The observed trend is clinically relevant and remains consistent with real-world clinical practice in Italy. These findings suggest the need to move beyond traditional phenotypic classifications and instead consider MS as a spectrum, with alternating active inflammatory and chronic phases leading to progressive disability. 6 In the setting of siponimod therapy, restrictive prescribing criteria further complicate management, increasing patient frustration and exposing them to cumulative risks—including immunosuppression and pharmacoeconomic burden.20,21
A considerable proportion of patients who discontinued siponimod subsequently resumed anti-CD20 therapies, such as ocrelizumab or rituximab, despite the well-documented risks of cumulative immunosuppression, including infections and hypogammaglobulinemia. 22 This pattern highlights the limited therapeutic alternatives for SPMS and the perceived efficacy of anti-CD20 agents, even in advanced disease stages. The decision to revert to anti-CD20 therapy, despite its risks, underscores the urgent need for more robust and diverse treatment options for this patient population.
Additionally, the emergence of an “orphan population”—patients who discontinue siponimod without initiating any subsequent DMT—represents a growing and under-recognized challenge in progressive MS care. Our subgroup analysis shows that these individuals tend to be older, with a median age of 56 years, have longstanding disease (median duration of 22 years), high levels of disability (baseline EDSS 6.5), and a history of multiple prior DMTs. Notably, the majority discontinued siponimod for safety concerns, further underscoring their clinical fragility.
These findings highlight the urgent need for structured, evidence-based therapeutic exit strategies for this complex patient population.23–27
The therapeutic landscape in Italy is evolving, particularly with the recent reimbursement approval of cladribine for progressive forms of MS. 28 This development introduces new possibilities for patients who have exhausted other options or are unsuitable for continued immunosuppression with anti-CD20 agents.29–33 Cladribine, with its distinct mechanism of action and intermittent dosing schedule, may represent a valuable alternative for patients with active SPMS, especially those at risk for AEs with other therapies. 34 Its approval constitutes a significant advancement in expanding the therapeutic armamentarium for progressive MS and may help address the unmet needs of the orphan population identified in this study.
Despite therapeutic advances, key gaps remain. There is no structured exit strategy from siponimod and limited guidance on how to manage patients left without treatment after discontinuation. A more proactive, individualized approach is needed to address the therapeutic void in late-stage MS.
The risks associated with ocrelizumab discontinuation, particularly in the context of sequential therapy, must be carefully weighed against its benefits. Addressing these gaps will be essential for improving outcomes and quality of life for individuals with progressive MS.
From a safety perspective, the predominance of lymphopenia and infections as reasons for discontinuation is consistent with previous clinical trial and real-world data. However, the absence of identifiable predictors in this cohort suggests that individual risk assessment remains challenging.
In a recent real-world study, grade 4 lymphopenia occurred early after siponimod initiation and was not predicted by any baseline demographic or laboratory characteristics. 10
Enhanced pharmacovigilance and the development of predictive biomarkers may facilitate improved patient selection and monitoring in the future.
Limitations of this study include its retrospective design, potential selection bias, and the limited duration of follow-up, which may affect the generalizability of the findings. Additionally, the relatively small sample size and the lack of standardized protocols across centers may have influenced the detection of predictors and outcomes. Importantly, data on siponimod dose modifications, temporary treatment interruptions, and the timing of grade 4 lymphopenia were not systematically collected. As a result, time-dependent factors potentially impacting discontinuation risk could not be evaluated. Additionally, cognitive outcomes were not assessed, and no direct comparisons across S1P receptor modulators were possible due to the retrospective design and dataset limitations.35–39
Future prospective, multicenter studies with larger cohorts, standardized data collection, and longer follow-up are needed to validate these results and to further elucidate optimal management strategies for SPMS patients discontinuing siponimod.
Conclusion
The management of SPMS following siponimod discontinuation is still complex and evolving. It is essential to carefully balance the risks of disease reactivation and the cumulative effects of immunosuppression when considering treatment discontinuation.
