Abstract
Background:
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease with limited treatment options and significant variability in care. Racial and ethnic disparities in ALS management and outcomes have been reported, but findings remain inconsistent.
Objectives:
This study aimed to evaluate racial and ethnic disparities in ALS care, specifically differences in healthcare utilization, treatment patterns, and survival, within a large healthcare system.
Design:
This retrospective cohort study analyzed electronic health records from a large healthcare system in Texas for patients diagnosed with ALS between 2013 and 2023, examining racial and ethnic differences in treatment and outcomes.
Methods:
Patients were identified using International Classification of Diseases (ICD) codes. Baseline characteristics, including race/ethnicity and socioeconomic factors, were collected. Primary outcomes included the use of noninvasive ventilation (NIV), tracheostomy, gastrostomy, mobility aids, and ALS medications; secondary outcomes included time to diagnosis and survival. Racial and ethnic disparities were assessed using generalized linear regression and Cox proportional hazards models, adjusting for demographic and socioeconomic factors.
Results:
A total of 636 patients were included (74.5% Non-Hispanic White, 5.3% Non-Hispanic Black, 7.4% Hispanic, and 12.7% Other). Non-Hispanic Black patients had significantly higher tracheostomy rates than Non-Hispanic White patients (35.3% vs 8.7%; adjusted odds ratio (OR), 6.20; 95% confidence interval (CI), 2.43–15.84). Hispanic patients had lower odds of receiving riluzole (42.6% vs 61.8%; adjusted OR, 0.36; 95% CI, 0.18–0.71) and higher rates of emergency department visits (adjusted OR, 2.00; 95% CI, 1.09–3.65) and hospitalizations (adjusted OR, 2.57; 95% CI, 1.37–4.81). No significant racial or ethnic differences were observed in time to diagnosis or survival after adjustment.
Conclusion:
Significant racial and ethnic disparities exist in ALS care, particularly in tracheostomy utilization, medication prescribing, and healthcare access. These findings underscore the need for targeted interventions to promote equitable ALS management, including provider education and improved healthcare accessibility.
Plain language summary
Amyotrophic lateral sclerosis (ALS) is a progressive and fatal neurodegenerative disease with no cure. While treatments can help manage symptoms, access to ALS care and treatment may vary by race and ethnicity. Previous studies have reported racial and ethnic disparities in ALS management, but findings have been inconsistent. Understanding these differences is essential for ensuring equitable healthcare access and improving patient outcomes.
The research team analyzed electronic health records from a large healthcare system in Texas to examine racial and ethnic differences in ALS care. They studied 636 patients diagnosed with ALS between 2013 and 2023, assessing healthcare utilization, treatment patterns, and survival. Statistical models were used to account for demographic and socioeconomic differences.
Non-Hispanic Black patients were more likely than Non-Hispanic White patients to undergo tracheostomy (35.3% vs 8.7%). Hispanic patients were less likely to receive riluzole, a medication that may slow ALS progression (42.6% vs 61.8%). Hispanic patients also had more emergency room visits and hospitalizations. However, there were no significant racial or ethnic differences in time to ALS diagnosis or overall survival after adjusting for socioeconomic factors.
The study highlights disparities in ALS care, particularly in access to key treatments and healthcare services. Addressing these gaps requires targeted efforts, such as improving healthcare access, reducing barriers to receiving medications, and educating providers on disparities to ensure equitable ALS care for all patients.
Introduction
Amyotrophic lateral sclerosis (ALS), also referred to as Lou Gehrig’s disease, is a progressive neurodegenerative disease characterized by gradual muscle weakness, atrophy, and ultimately paralysis. 1 The average life expectancy following the onset of symptoms ranges from 2 to 5 years, with many patients ultimately succumbing to respiratory failure. 2 While no cure exists, recent years have seen the introduction of novel therapies and an expanding clinical pipeline, signaling progress in the search for ALS treatments.
ALS is a rare disease, with a global incidence of 1.59 per 100,000 person-years and a prevalence of 4.42 per 100,000 individuals. 3 In the United States (US), rates are slightly higher at 1.6 per 100,000 person-years and 6.6 per 100,000 individuals.4,5 The global burden of ALS is projected to increase substantially in coming decades, largely driven by an aging population.6,7 While disease onset can occur at any age, incidence peaks between 60 and 79 years of age. 6 Late-onset cases often progress more aggressively, and while ALS is more common in men, this sex disparity diminishes with age.8,9 Most ALS cases are sporadic, while familial ALS, which is often associated with genetic mutations, has an earlier onset. 10 The disease typically manifests as either spinal-onset ALS (70% of cases), which affects limb muscles, or bulbar-onset ALS, which impairs speech and swallowing. 11
Advanced age, male sex, and genetic predisposition are the most consistently reported risk factors for ALS.11 Environmental exposures such as toxins and smoking have been suggested, but evidence remains mixed.12,13 Additional factors including head trauma, electrical burns, and high levels of physical activity have also been identified as potential risk factors.14 –17 Moreover, studies have suggested that patients with lower body mass index (BMI) are at a higher risk of developing ALS and have worse survival outcomes.18,19
The U.S. Food and Drug Administration has approved several pharmacologic agents for the treatment of ALS, including riluzole, edaravone, sodium phenylbutyrate/taurursodiol, dextromethorphan/quinidine, and tofersen. In April 2024, sodium phenylbutyrate/taurursodiol, which had received prior approval based on a small Phase 2 trial influenced by patient advocacy groups and the Accelerating Access to Critical Therapies for ALS Act, was withdrawn from the market after the Phase 3 PHOENIX trial failed to show efficacy over placebo.20,21 Currently, over 100 experimental therapies, including gene-specific therapies, monoclonal antibodies, and stem cell-based treatments, are in clinical trials, instilling optimism for future breakthroughs. 22
Beyond pharmacological treatment, physical therapy has demonstrated effectiveness in reducing motor deterioration, and mobility aids are frequently utilized to reduce fall risk. 23 As the disease progresses, dysphagia becomes a major concern, often necessitating the placement of a percutaneous endoscopic gastrostomy tube for nutrition and hydration. Noninvasive ventilation (NIV) is also used for respiratory management, with evidence indicating prolonged survival, while tracheostomy may be necessary in cases of severe respiratory failure.24,25
Racial and ethnic disparities in ALS diagnosis and care have been reported. Some studies report that Black patients in the US experience longer diagnostic delays and receive fewer interventions, though findings are inconsistent.26 –30 In terms of survival outcomes, findings in the literature remain inconsistent as well.26,27,30 –34 Due to ALS being more prevalent in White populations, studies examining disease burden and outcomes in other racial and ethnic groups remain limited. Research has largely focused on Black–White comparisons, leaving gaps in understanding disparities across broader populations. This study aims to address these gaps by analyzing a diverse patient cohort while accounting for socioeconomic factors to better inform equitable treatment strategies.
Methods
Study design
This retrospective longitudinal cohort study utilized electronic health record (EHR) data from Baylor Scott & White Health (BSWH), a large, integrated healthcare system that serves both urban and rural regions across north and central Texas. The BSWH system includes specialized ALS multidisciplinary clinics in addition to general neurology and primary care practices, ensuring comprehensive capture of ALS care within the region.
The EHR system documents inpatient and outpatient encounters, procedures, prescriptions, and laboratory results. Data from external healthcare facilities connected via the Care Everywhere health information exchange network were also incorporated when available. Data spanning January 1, 2013, to December 31, 2023, were extracted from the EHR using Epic Caboodle, the enterprise data warehouse platform for the BSWH system.
The study was prepared following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines to ensure comprehensive and transparent reporting of cohort study methods and findings. 35
Patient sample
All patients with a recorded diagnosis of ALS, identified by International Classification of Diseases (ICD; ICD-9-CM: 335.20; ICD-10-CM: G12.21) codes, between January 1, 2013, and June 30, 2023, were extracted. Manual chart review was performed for all identified cases, confirming ALS diagnoses and excluding patients with other motor neuron diseases. As this was a retrospective cohort study utilizing an existing EHR dataset, no formal sample size calculation was performed a priori.
Eligible patients were followed from their date of diagnosis until their last recorded interaction with the BSWH system or the end of the study period (December 31, 2023), whichever occurred first. A minimum follow-up duration was not required, allowing for the inclusion of patients regardless of the length of observation. The median follow-up duration was 17.4 months (interquartile range (IQR): 7.6–36.0 months), with 82.7% of patients having at least 6 months of documented follow-up.
Baseline characteristics and outcomes
Baseline characteristics included race/ethnicity, age at disease onset, age at diagnosis, sex, site of symptom onset, primary insurance type, median household income, smoking status, marital status, and BMI at diagnosis. Race/ethnicity was derived from self-reported data and categorized as Non-Hispanic White, Non-Hispanic Black, Hispanic, or Other. Patients with missing race or ethnicity were classified as Other. Age at disease onset was defined as the earliest documented ALS symptom recorded in the EHR (e.g., patient self-reported disease onset). Age at diagnosis was determined by the date of the first ALS diagnosis recorded in the EHR, irrespective of classification (definite, probable, possible, or suspected). Median household income was estimated using zip code-level data. 36 BMI at diagnosis was based on the most recent value recorded within 365 days prior to ALS diagnosis.
Primary outcomes:
Use of NIV, gastrostomy tube placement, tracheostomy, and mobility equipment.
Prescribing of riluzole, edaravone, sodium phenylbutyrate/taurursodiol, and dextromethorphan/quinidine.
Hospital utilization rates per patient-month: Emergency department (ED) visits. Outpatient visits. Inpatient admissions. Inpatient hospital length of stay (LOS).
Secondary outcomes:
Time from disease onset to ALS diagnosis (months).
Time from disease onset to death (months).
Date of death was obtained from the EHR. If unavailable, publicly accessible obituaries were reviewed. Patients without a confirmed death date were censored at the time of their last recorded healthcare interaction.
Statistical analysis
Descriptive statistics were used to summarize baseline characteristics, including sample size (n), proportions (%), means, standard deviations (SDs), medians, and IQRs. Differences across racial and ethnic groups were assessed using Chi-square tests for categorical variables and Kruskal–Wallis tests for continuous variables. These methods were also applied in unadjusted analyses of healthcare resource utilization (HCRU).
For adjusted analyses, generalized linear models (GLMs) were used to control for baseline covariates, including age at disease onset, age at diagnosis, sex, site of symptom onset, primary insurance type, median household income, smoking status, marital status, and BMI at diagnosis. Covariates were selected based on prior literature indicating their association with HCRU in ALS populations.
Model selection was informed by the distributional properties of each outcome. Binary outcomes (e.g., intervention use) were analyzed using logistic regression with a binomial distribution and logit link. Count-based outcomes (e.g., visit rates) were modeled using negative binomial (NB) regression with a log link. Continuous, positively skewed outcomes (e.g., inpatient hospital LOS) were modeled using gamma-distributed GLMs with a log link.
Although a zero-inflated NB model was initially considered for outcomes with excess zero counts (e.g., ED visits, inpatient admissions), model convergence was not achieved due to the small number of patients with at least one such event (see Supplemental Table 1). Consequently, standard NB models were applied to estimate incidence rate ratios for ED visits and inpatient admissions. NB regression was also applied to outpatient outpatient visits per patient-month, given its non-normal distribution. For inpatient hospital LOS, a gamma-distributed GLM was used to account for its right-skewed, non-negative distribution.
For survival analyses, Cox proportional hazards models were used to estimate hazard ratios (HRs), and Kaplan–Meier curves were constructed to visualize unadjusted time-to-event outcomes.
All data management and statistical analyses were conducted using SAS Enterprise Guide Version 8.2 (SAS Institute Inc., Cary, NC, USA) and Stata/BE 18.0 (StataCorp LLC, College Station, TX, USA). Statistical significance was defined as α = 0.05 for all comparisons.
Results
Baseline characteristics
A total of 636 patients were included in the study, with their baseline characteristics summarized in Table 1. Among them, 474 individuals (74.5%) were Non-Hispanic White and 328 (51.6%) were male. Hispanic patients were significantly younger at both disease onset (median 58 vs 65 years, p = 0.005) and diagnosis (median 59 vs 67 years, p = 0.002) compared to Non-Hispanic White patients. Additionally, the type of insurance coverage varied significantly, with a higher proportion of Hispanic patients classified under “Other” insurance types (19.2% vs 4.9%, p < 0.001) in contrast to Non-Hispanic White patients. The “Other” category predominantly included self-pay patients, often indicative of a lack of insurance coverage. Non-Hispanic Black patients had lower median household incomes (⩽$49,999: 23.5% vs 5.5%, p = 0.003), a lower likelihood of being married or partnered (41.2% vs 75.3%, p = 0.001), and a higher prevalence of underweight status (BMI < 18.5) at diagnosis (17.7% vs 2.5%, p = 0.002) compared to Non-Hispanic White patients.
Baseline characteristics of patients by race/ethnicity.
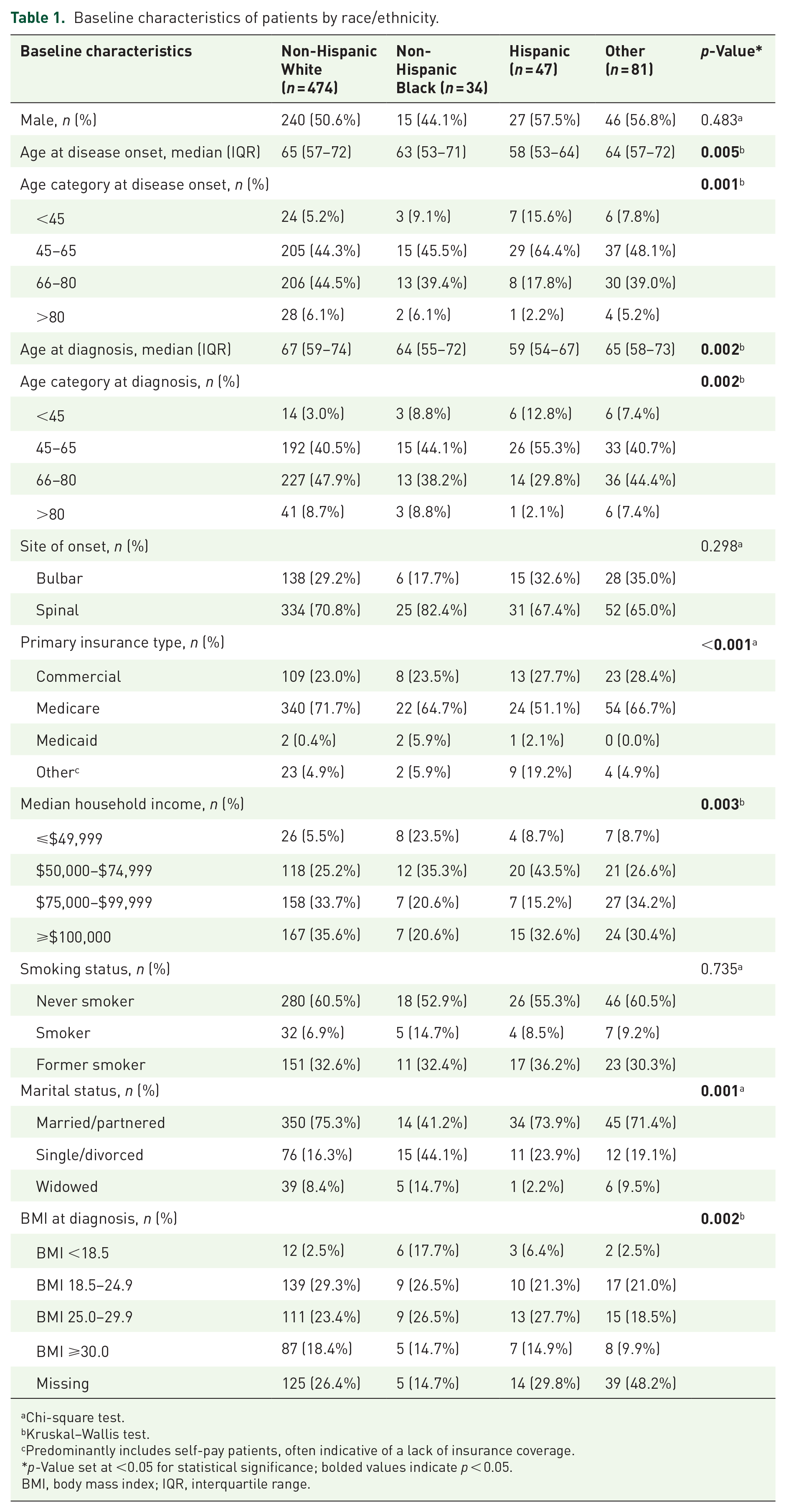
Chi-square test.
Kruskal–Wallis test.
Predominantly includes self-pay patients, often indicative of a lack of insurance coverage.
p-Value set at <0.05 for statistical significance; bolded values indicate p < 0.05.
BMI, body mass index; IQR, interquartile range.
HCRU and survival
Significant racial and ethnic disparities were observed in HCRU (Table 2). Tracheostomy was most frequently performed among Non-Hispanic Black patients (35.3%), followed by Hispanic (14.9%), Non-Hispanic White (8.7%), and Other (8.6%) patients. After adjusting for baseline characteristics, Non-Hispanic Black patients had significantly higher odds of undergoing tracheostomy compared to Non-Hispanic White patients (odds ratio (OR) = 6.20, 95% confidence interval (CI): 2.43–15.84; Table 3).
Unadjusted HCRU of patients by race/ethnicity.

Chi-square test.
Negative binomial regression, unadjusted.
Includes the 160 total patients with any inpatient admissions, calculated in days.
p-Value set at <0.05 for statistical significance; bolded values indicate p < 0.05.
ED, emergency department; HCRU, healthcare resource utilization; LOS, length of stay; NIV, noninvasive ventilator; SD, standard deviation.
Adjusted a HCRU for baseline characteristics of patients by race/ethnicity.
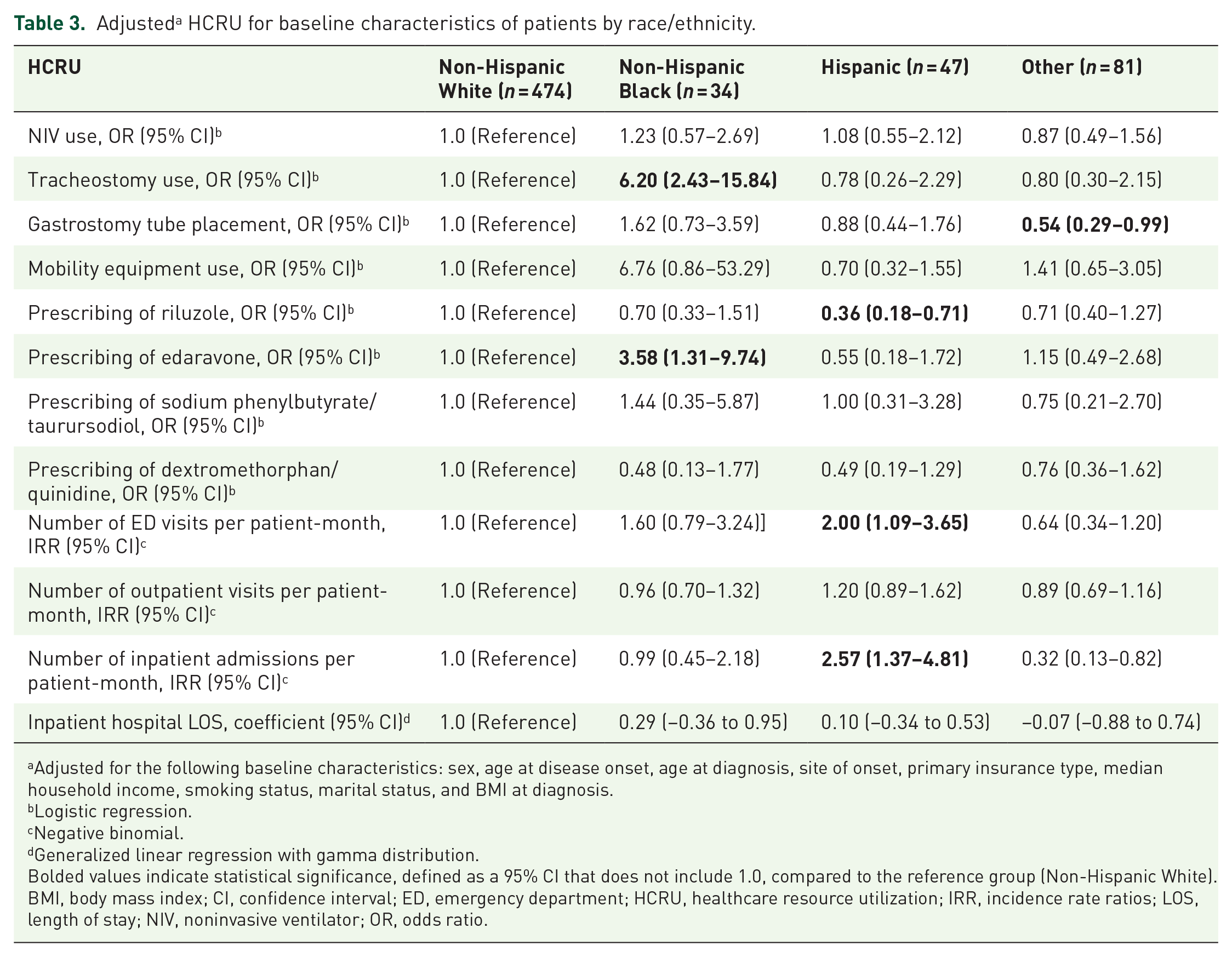
Adjusted for the following baseline characteristics: sex, age at disease onset, age at diagnosis, site of onset, primary insurance type, median household income, smoking status, marital status, and BMI at diagnosis.
Logistic regression.
Negative binomial.
Generalized linear regression with gamma distribution.
Bolded values indicate statistical significance, defined as a 95% CI that does not include 1.0, compared to the reference group (Non-Hispanic White).
BMI, body mass index; CI, confidence interval; ED, emergency department; HCRU, healthcare resource utilization; IRR, incidence rate ratios; LOS, length of stay; NIV, noninvasive ventilator; OR, odds ratio.
The use of mobility equipment was highest among Non-Hispanic Black patients (97.1%), followed by Non-Hispanic White (80.2%), Hispanic (76.6%), and Other (74.1%; Table 2). However, this association was not statistically significant in the adjusted analysis (OR = 6.76, 95% CI: 0.86–53.29; Table 3). While not statistically significant, it is notable that Non-Hispanic Black patients in our cohort had a higher prevalence of spinal-onset ALS compared to Non-Hispanic White patients, which may contribute to greater mobility equipment use. Although this difference in spinal onset was also not statistically significant, it may be a clinically plausible factor underlying the observed trend.
Regarding pharmacologic treatment, Hispanic patients were least likely to be prescribed riluzole (42.6%) compared to Non-Hispanic White patients (61.8%; Table 2). Adjusted analyses confirmed this disparity, with Hispanic patients demonstrating significantly lower odds of being prescribed riluzole compared to Non-Hispanic White patients (OR = 0.36, 95% CI: 0.18–0.71; Table 3). In contrast, Non-Hispanic Black patients had significantly higher odds of being prescribed edaravone in adjusted analysis (OR = 3.58, 95% CI: 1.31–9.74; Table 3) compared to Non-Hispanic White patients, although this difference was not significant in the unadjusted analysis (Table 2).
Differences were also observed in healthcare visit rates. Hispanic patients had a higher mean number of ED visits per patient-month compared to Non-Hispanic White patients (mean 0.14 (SD = 0.33) vs 0.05 (SD = 0.15); Table 2), a difference that persisted in the adjusted analysis (OR = 2.00, 95% CI: 1.09–3.65; Table 3). Similarly, the mean number of inpatient admissions per patient-month was significantly higher among Hispanic patients (mean 0.16 (SD = 0.63) vs 0.06 (SD = 0.03); Table 2), with the adjusted analysis confirming greater odds of hospitalization (OR = 2.57, 95% CI: 1.37–4.81; Table 3).
The median time from disease onset to ALS diagnosis was longest among Non-Hispanic Black patients (16.4 months, IQR: 8.0–33.9), followed by Non-Hispanic White (12.0 months, IQR: 6.6–23.2), Hispanic (10.5 months, IQR: 7.3–21.4), and Other (10.6 months, IQR: 8.3–21.8). However, race and ethnicity were not significantly associated with diagnostic delay in either unadjusted (log-rank p-value = 0.021; Figure 1) or adjusted models (Non-Hispanic Black vs Non-Hispanic White: HR = 1.01, 95% CI: 0.69–1.48; Table 4).
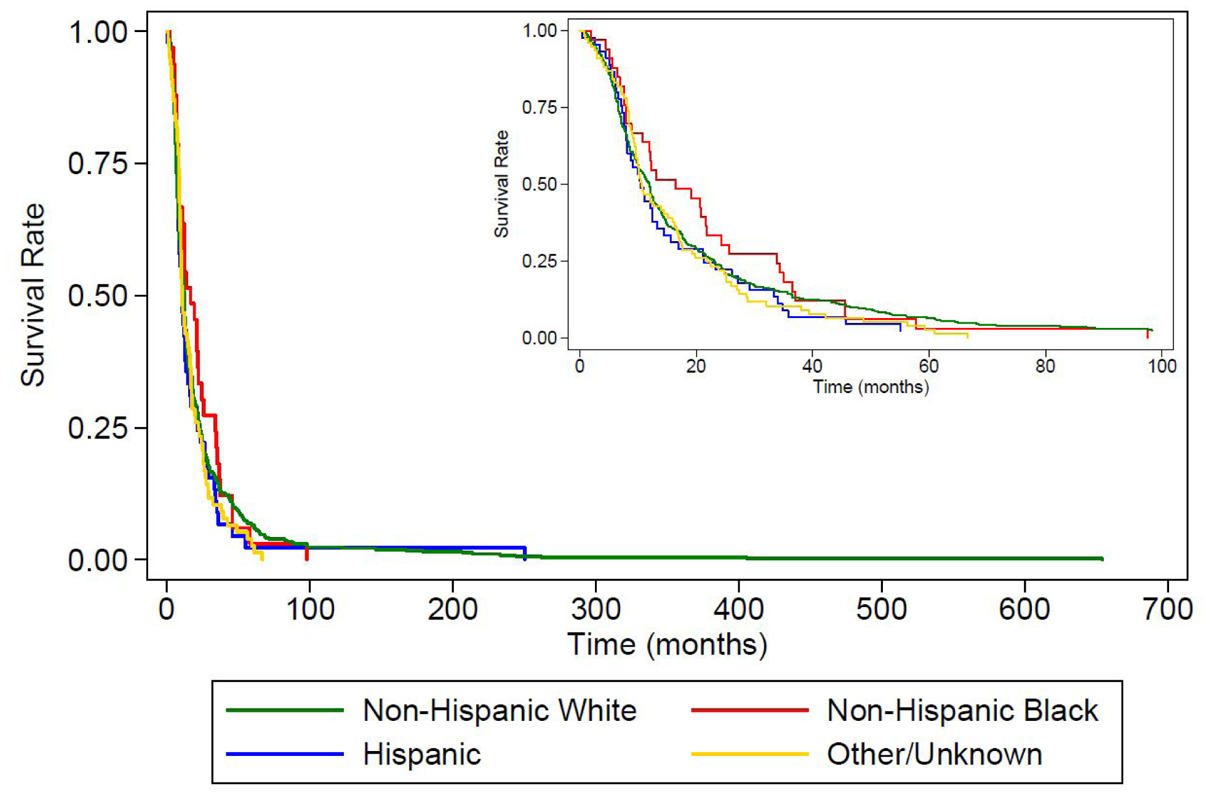
Kaplan–Meier curve of time from disease onset to diagnosis by racial/ethnic group.
Estimated HRs for time from disease onset to diagnosis of ALS. a
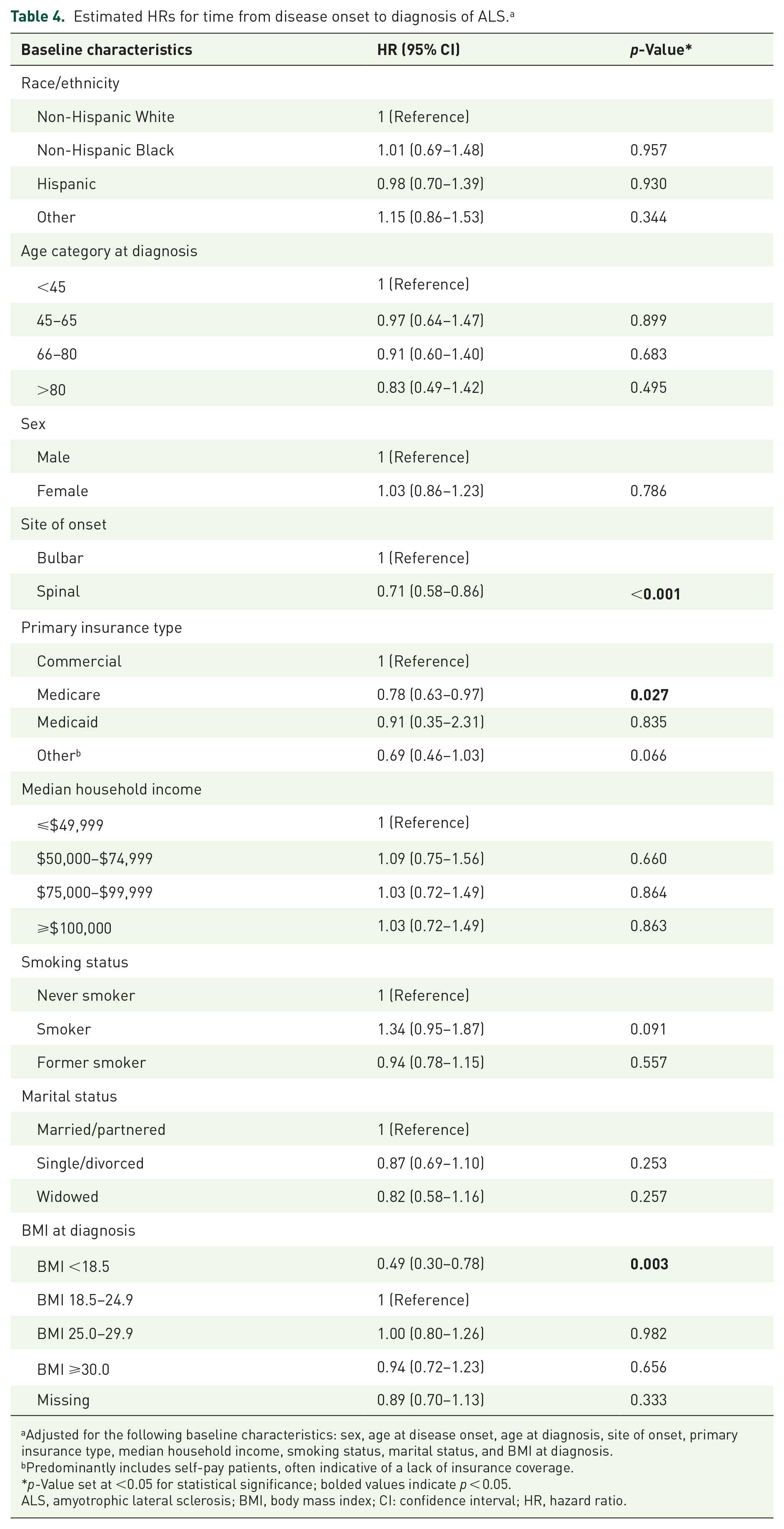
Adjusted for the following baseline characteristics: sex, age at disease onset, age at diagnosis, site of onset, primary insurance type, median household income, smoking status, marital status, and BMI at diagnosis.
Predominantly includes self-pay patients, often indicative of a lack of insurance coverage.
*p-Value set at <0.05 for statistical significance; bolded values indicate p < 0.05.
ALS, amyotrophic lateral sclerosis; BMI, body mass index; CI: confidence interval; HR, hazard ratio.
Regarding overall survival, Non-Hispanic Black patients had the longest median survival (51.4 months, IQR: 37.8–79.5), followed by Hispanic (38.1 months, IQR: 21.9–56.6), Non-Hispanic White (33.7 months, IQR: 21.4–56.7), and Other (30.0 months, IQR: 21.5–48.6). Although significant differences in survival across racial and ethnic groups were observed in the unadjusted Kaplan–Meier analysis (log-rank p < 0.0001; Figure 2), these associations were not statistically significant after adjustment for baseline characteristics (Non-Hispanic Black vs Non-Hispanic White: HR = 0.75, 95% CI: 0.48–1.19; Table 5).

Kaplan–Meier curve of time from disease onset to death by racial/ethnic group.
Estimated HRs for time from diagnosis of ALS to death. a
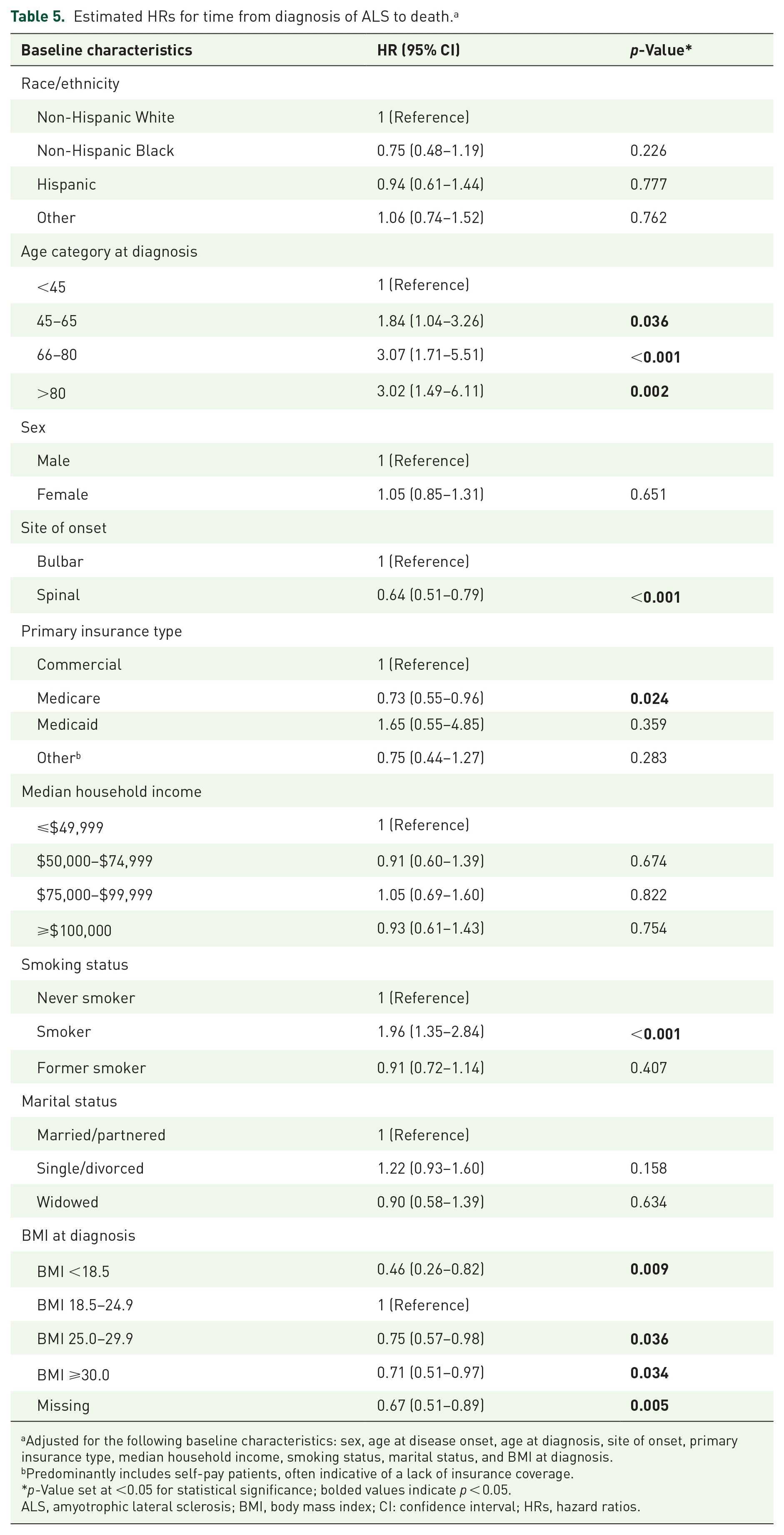
Adjusted for the following baseline characteristics: sex, age at disease onset, age at diagnosis, site of onset, primary insurance type, median household income, smoking status, marital status, and BMI at diagnosis.
Predominantly includes self-pay patients, often indicative of a lack of insurance coverage.
*p-Value set at <0.05 for statistical significance; bolded values indicate p < 0.05.
ALS, amyotrophic lateral sclerosis; BMI, body mass index; CI: confidence interval; HRs, hazard ratios.
Given the potential impact of tracheostomy on survival, an exploratory analysis was conducted stratifying patients by tracheostomy status. Median survival was longer in patients who underwent tracheostomy (55.7 months; IQR: 28.3–93.0) compared to those who did not (41.0 months; IQR: 24.0–79.9), although this difference was not statistically significant (HR = 0.84; 95% CI: 0.61–1.14; p = 0.264; Supplemental Figure 1). After adjusting for tracheostomy status along with baseline characteristics in survival models, Non-Hispanic Black patients continued to show a non-significant trend toward longer survival compared to Non-Hispanic White patients (HR = 0.76; 95% CI: 0.48–1.22; Supplemental Figures 2 and 3).
Discussion
This 10-year study (2013–2023) leveraging EHR data from a large, integrated healthcare system in Texas offers a comprehensive analysis of racial and ethnic disparities in HCRU and survival among patients with ALS. Our findings reveal substantial differences in care delivery and healthcare utilization patterns across racial and ethnic groups.
One of the most notable disparities observed was the higher rate of tracheostomy among Non-Hispanic Black patients, who had more than six times the odds of undergoing the procedure compared to Non-Hispanic White patients. This aligns with previous research reporting increased use of invasive ventilation among Black individuals.29,37,38 However, unlike earlier studies attributing this to delayed diagnosis and more advanced disease at presentation, our analysis found no significant difference in diagnostic timing across racial and ethnic groups.26,27 This challenges the assumption that clinical presentation alone accounts for higher tracheostomy rates.
Socioeconomic status (SES) has also been linked to higher tracheostomy utilization, with patients in lower income brackets more likely to undergo the procedure.39,40 In our study, Non-Hispanic Black patients were disproportionately represented in the lowest income group. Yet, the disparity persisted even after adjusting for SES and other baseline characteristics, suggesting that other factors beyond SES, such as differences in patient education, cultural perceptions of invasive life-sustaining treatments, implicit provider biases, and structural barriers to NIV, may be at play. Although tracheostomy has been shown to prolong survival in ALS, it is also associated with substantial risks, including infection, hemorrhage, increased caregiver burden, and reduced quality of life.39,41 –43 Further research is needed to determine whether higher tracheostomy rates reflect disparities in access to earlier interventions or differences in treatment preferences.
Disparities were also observed in the prescribing of riluzole, with Hispanic patients significantly less likely to receive the drug compared to Non-Hispanic White patients. Given that riluzole remains the only widely approved therapy shown to prolong survival in ALS, this disparity raises concerns about potential inequities in treatment access and clinician prescribing behavior.44,45 While insurance coverage may be a contributing factor, as nearly one in five Hispanic patients in our cohort lacked Medicare, Medicaid, or commercial insurance, this alone is unlikely to explain the disparity, as medication orders are typically recorded regardless of insurance approval.
Other possible explanations include provider-related factors (e.g., differences in prescribing practices, implicit bias) and patient-related factors (e.g., treatment refusal, side effect concerns, mistrust of the healthcare system). Prior studies have not consistently found racial differences in pharmacologic management of ALS, but most have largely focused on comparisons between Black and White patients, leaving a critical knowledge gap regarding Hispanic and other underrepresented groups.29,32
An interesting finding emerged in the prescribing patterns for edaravone, in which Non-Hispanic Black patients had more than three times the odds of being prescribed the medication compared to Non-Hispanic White patients. While the inclusion of edaravone in the Texas Medicaid drug formulary was initially considered a potential explanation, further investigation indicates it doesn’t fully account for this trend. Given the adjustments made in our model, other factors such as differential provider preferences, patient advocacy, and targeted outreach may be influencing this prescribing pattern.
Healthcare utilization patterns also revealed disparities in access to care. Hispanic patients had twice the odds of ED visits and inpatient admissions compared to Non-Hispanic White patients, a pattern consistent with prior studies. 46 ,47 This suggests that Hispanic patients with ALS may face barriers to timely primary and specialty care, leading to an increased reliance on acute care services. These findings highlight the need for healthcare systems to implement policies aimed at improving access to timely ALS care, such as expanding telemedicine services, increasing the availability of interpretation services, and strengthening community-based care coordination.
In terms of diagnostic delay, our findings also align with previous research showing that Black patients experience longer delays in ALS diagnosis compared to White individuals.26,27 In our cohort, Non-Hispanic Black patients had a median time to diagnosis of 16.4 months, compared to 12.0 months for Non-Hispanic White patients, though this difference did not reach statistical significance. Additionally, the median time from disease onset to death was longest among Non-Hispanic Black patients (51.4 months) and shortest among Non-Hispanic White (33.7 months) and Other (30.0 months). Although this trend mirrors findings from previous studies in which Black patients had longer median survival compared to White patients, the differences in survival in our study did not reach statistical significance.27,29 The median survival time for Non-Hispanic Black patients in our study exceeded previously reported ranges (32–38 months), suggesting the possibility of unmeasured protective factors that warrant further investigation.
Limitations
This study has several limitations. First, since our study cohort was drawn from a single healthcare system in Texas, the demographic and clinical characteristics of included patients may not fully reflect those of the broader national ALS population. This geographic and system-based constraint may limit the generalizability of our findings to other regions or healthcare settings across the US.
Second, as a rare disease, ALS presents inherent challenges related to sample size, particularly among racial and ethnic groups other than Non-Hispanic White patients. This may have limited statistical power to detect certain disparities or to assess nuanced differences across underrepresented populations.
Third, the absence of a continuous eligibility requirement may have affected data completeness. To promote inclusivity, especially for marginalized groups with potentially fragmented care, we included all patients with at least one outpatient visit within the healthcare system. While this approach enhances representation, it may have captured individuals with intermittent or out-of-network care, resulting in potential gaps in longitudinal data. Given the rapid progression of ALS, such care transitions and interruptions are common and complicate continuous tracking.
Fourth, although our EHR database includes data from Care Everywhere to capture some care delivered outside the system, it is possible that hospitalizations, ED visits, office encounters, and prescriptions occurring outside the healthcare system may not have been fully captured. This limitation may have led to an underestimation of healthcare utilization, particularly among patients with lower or inconsistent engagement with the BSWH system.
Fifth, our analysis of medication prescribing patterns was based solely on prescription orders documented within the EHR. Due to limitations in structured EHR data, we were unable to distinguish whether the absence of a documented prescription reflected a provider’s decision not to offer the medication, a patient’s decision to decline therapy, or other factors such as clinical ineligibility or insurance barriers. In addition, clinical measures such as forced vital capacity and ALS Functional Rating Scale-Revised scores, which are often required for insurance authorization of medications, were not consistently available in the dataset and therefore could not be incorporated into our analysis. As a result, the observed disparities in prescribing rates across racial and ethnic groups should be interpreted with caution.
Sixth, temporal changes in treatment practices over the 10-year study period were not explicitly modeled. Evolving clinical guidelines, approval and availability of new medications, and shifts in healthcare policy may have introduced time-dependent confounding.
Finally, median household income was estimated at the zip code level, reflecting median neighborhood income where the patients reside rather than individual income, which introduces the potential for ecological fallacy.
Conclusion
This study identifies significant racial and ethnic disparities in ALS care, with notable differences in tracheostomy utilization, medication prescribing, and healthcare visits. These findings point to systemic inequities in ALS care delivery and underscore the need for multi-level interventions. Efforts to address these disparities should include enhancing provider education to promote guideline-based, equitable care, implementing policies to expand healthcare access to underserved populations, and conducting targeted research to identify the underlying drivers of these disparities. By prioritizing health equity in ALS care, providers and policymakers can work toward ensuring that all individuals with ALS receive high-quality, evidence-based, and culturally competent care throughout the course of their disease.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251365001 – Supplemental material for Racial and ethnic disparities in ALS: a longitudinal electronic health records study
Supplemental material, sj-docx-1-tan-10.1177_17562864251365001 for Racial and ethnic disparities in ALS: a longitudinal electronic health records study by Tiffany Kuo, Timothy Reynolds, Linda Chen, Chanhyun Park, Karen Rascati and Paul Godley in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251365001 – Supplemental material for Racial and ethnic disparities in ALS: a longitudinal electronic health records study
Supplemental material, sj-docx-2-tan-10.1177_17562864251365001 for Racial and ethnic disparities in ALS: a longitudinal electronic health records study by Tiffany Kuo, Timothy Reynolds, Linda Chen, Chanhyun Park, Karen Rascati and Paul Godley in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
