Abstract
Background:
Neuromyelitis optica spectrum disorder (NMOSD) may be triggered by environmental risk factors.
Objectives:
We aimed to explore and integrate the recent research advances in this field. Here we describe relevant studies and summarize current knowledge on non-genetic factors that influence the onset of the disease.
Design:
Systematic review.
Methods:
We performed a systematic review up to May 21, 2024, following preferred reporting items for systematic reviews and meta-analyses guidelines. Two independent reviewers evaluated the quality of the included studies using the Joanna Briggs Institute checklist for risk of bias assessment.
Data sources:
MEDLINE, EMBASE, Scopus, and Web of Science databases.
Results:
A total of 15,869 articles were evaluated. Of those 50 studies met the eligibility criteria. A total of 21,410 NMOSD patients were included in the studies; 17,080 patients were females. Totally, 14 risk factors, including vitamin D deficiency, vaccination, virus infections, lifestyle, and dietary factors, were assessed. A total of 37% of the included articles were conducted in East Asia, mainly focusing on the effects of infection and vitamin D deficiency. These studies suggested vitamin D deficiency as a possible NMOSD risk factor. A total of 25% of the studies included Caucasian populations from Western countries. They showed that smoking decreased the odds of NMOSD, in contrast to observations from Eastern studies. Few cases reported NMOSD onset after COVID-19 vaccination. Antibodies against Epstein-Barr virus, Mycobacterium paratuberculosis, and Helicobacter pylori were observed to be more frequently positive in the serum of NMOSD patients. Lower protein and fat and higher carbohydrate intakes were correlated with NMOSD development.
Conclusion:
Vitamin D deficiency, cigarette smoking, Mycobacterium avium subspecies paratuberculosis infection, and diet were reported as environmental risk factors for NMOSD. The difference in the onset of NMOSD between Asian and Caucasian populations could be affected by smoking and vitamin D deficiency. Knowledge of modifiable risk factors for NMOSD may be beneficial in preventing and improving disease outcomes.
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an inflammatory autoimmune disease of the central nervous system (CNS). 1 Serum immunoglobulin G autoantibodies (NMO-IgG) are found in most NMOSD patients. 2 The target antigen of NMO-IgG is the astrocyte water channel aquaporin-4 (AQP4) in the CNS.3,4 AQP4-IgG is a distinct biomarker for NMOSD and mediates pathogenesis by binding to the autoantigen AQP4 on CNS astrocytes, leading to complement-dependent astrocyte injury, inflammation, subsequent demyelination, and neuronal damage.5,6 NMOSD is clinically characterized by predominant involvement of the optic nerves and the spinal cord.
The prevalence of NMOSD is variable geographically and ethnically. 7 NMOSD is prevalent in populations that are not Caucasian, especially in Asians, 8 and epidemiological studies indicate that the prevalence of NMOSD in East Asian countries (Japanese, Chinese, and Koreans) is ~3.5 per 100,000.9,10 In blacks, the prevalence is higher compared to whites, as some studies report it as ~1.8 per 100,000. 8 The prevalence among Caucasians is reported to be ~1 per 100,000. Additionally, the age of onset, disease course, and outcome differ between races. 11 Non-Caucasians patients experience a younger age of onset, severe motor and visual disabilities and more frequent attacks on the brainstem. 12 Although the exact etiology of NMOSD remains under investigation, emerging evidence suggests that a combination of genetic and environmental factors contributes to various prevalences in different regions. Genetic studies confirm that HLA-DRB1*08:02 and HLA-DRB1*16:02 are risk alleles for NMOSD, while HLA-DRB1*09:01 is associated with a lower risk of NMOSD onset. 13 The coexistence of NMOSD with other autoimmune diseases is well recognized, and the genetic autoimmune dependency of NMOSD is partly illuminated. 14 Recently, a report that MHC class I-related biological processes and the interferon-gamma-mediated signaling pathway may be involved in the pathogenesis of NMOSD coexisting with systemic lupus erythematosus, Sjögren’s syndrome, and autoimmune thyroid disease. 15
As to the effect of environmental factors, previous literature indicates that infections, smoking, and dietary habits increase the risk of CNS inflammatory disorders, including Multiple Sclerosis.16–19 However, recent studies have yielded conflicting results regarding the effects of these factors on NMOSD. 20 Epstein-Barr virus (EBV) infection is considered as one of the main MS risk factors, but several studies observed no significant increase in humoral response against EBV in NMOSD patients compared to healthy individuals. 21 It has long been considered that latitude significantly influences the prevalence of MS, whereas no study has confirmed this effect on the onset of NMOSD. Some studies have reported that vitamin D deficiency, habitual risk factors such as smoking, and infections with atypical agents, such as Mycobacterium avium, are possible risk factors for NMOSD.22–24 Also, diet may influence NMOSD occurrence; a high-carbohydrate and low-dairy diet has been reported to increase the risk of NMOSD.11,25
Identifying environmental and genetic risk factors is crucial for a deeper understanding of this disease, which in turn aids in establishing prevention strategies, assessing disease risk, and enhancing clinical efficacy. To the best of the authors’ knowledge, no comprehensive systematic review has been published on the environmental risk factors of NMOSD development. This systematic scoping review aims to explore the non-genetic risk factors of NMOSD, focusing on the causes of regional disparity.
Materials and methods
Search strategy
A systematic literature search of MEDLINE, EMBASE, Scopus, and Web of Science was conducted to evaluate non-genetic and environmental risk factors of NMOSD using the keywords neuromyelitis optica (NMO), environmental, risk factor, and each risk factor name, covering from 1999 to May 21, 2024. Furthermore, a manual search of the references and related reviews was performed. Database-appropriate syntax was used to optimize searching. The exact queries used in each database are available in the Supplemental Material.
Data were reviewed from human population studies, including cross-sectional, case-control, and cohort studies written in English. Two of the authors (M.M.L. and E.B.) separately searched, screened the records, and reviewed the titles and abstracts of identified records. A manual hand search of the reference lists of primary identified articles and relevant reviews to discover additional studies and congress abstracts. The two authors (M.M.L. and E.B.) read the full text of potentially eligible records and checked for the following inclusion criteria: (1) human population-based studies and (2) data on the environmental risk factors of NMOSD. Review articles, letters, editorials, and genetic studies were excluded (Table 1). Two experts (M.M.L. and E.B.) independently reviewed the articles. Disagreements were resolved by consensus. The preferred reporting items for systematic reviews and meta-analyses flow chart demonstrated the study selection (Figure 1).26–28 Initially, 15,871 records from PubMed (1387), Scopus (2795), Embase (9311), and Web of Science (2378) were included. Before the screening, 9522 duplicate records were removed. Further, 6041 records were excluded due to irrelevant titles and abstracts, and from the remaining 307 articles, 261 were not related, 34 were conference abstracts, and 12 were review articles. Fifty articles were included in the current study.
Eligibility considerations in study selection.

NMOSD, neuromyelitis optica spectrum disorder.
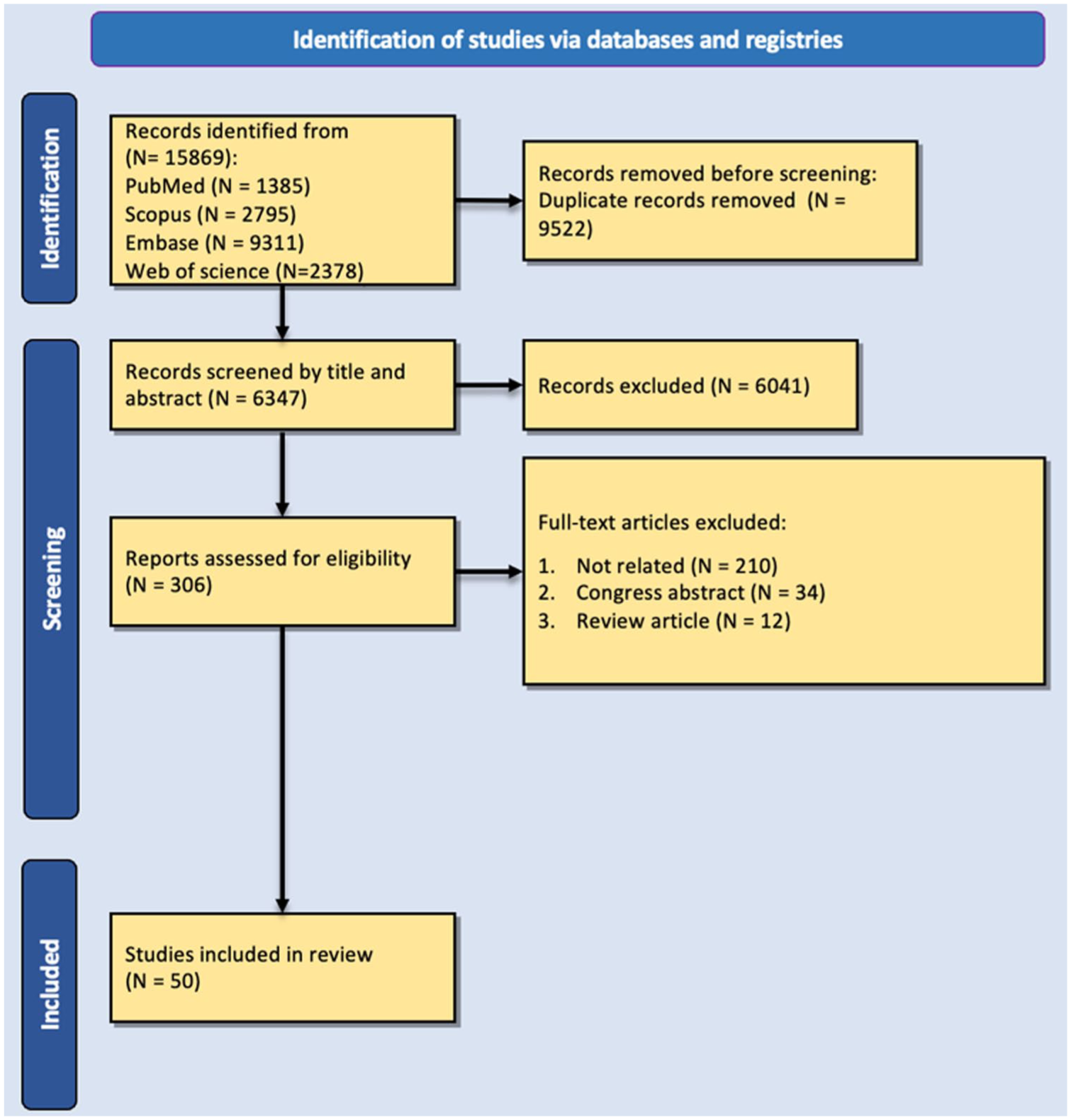
PRISMA chart of the study.
Data extraction and quality assessment
Data extraction and quality assessment were performed by two independent researchers (M.M.L., E.B.). Quality assessment was evaluated based on the Newcastle-Ottawa tool, which was designed by the Cochrane Handbook for the assessment of observational studies. 29 Repeated publications of the same data were excluded. The following information was collected: author and year, study type, risk factor(s), demographic data, sample size, and outcome. Measures of effect (e.g., odds ratio (OR), relative risk, confidence interval (CI), and hazard ratio (HR)) from each eligible study were extracted and then summarized in Tables 2–8.
The effect of patients’ demographics on NMOSD development.
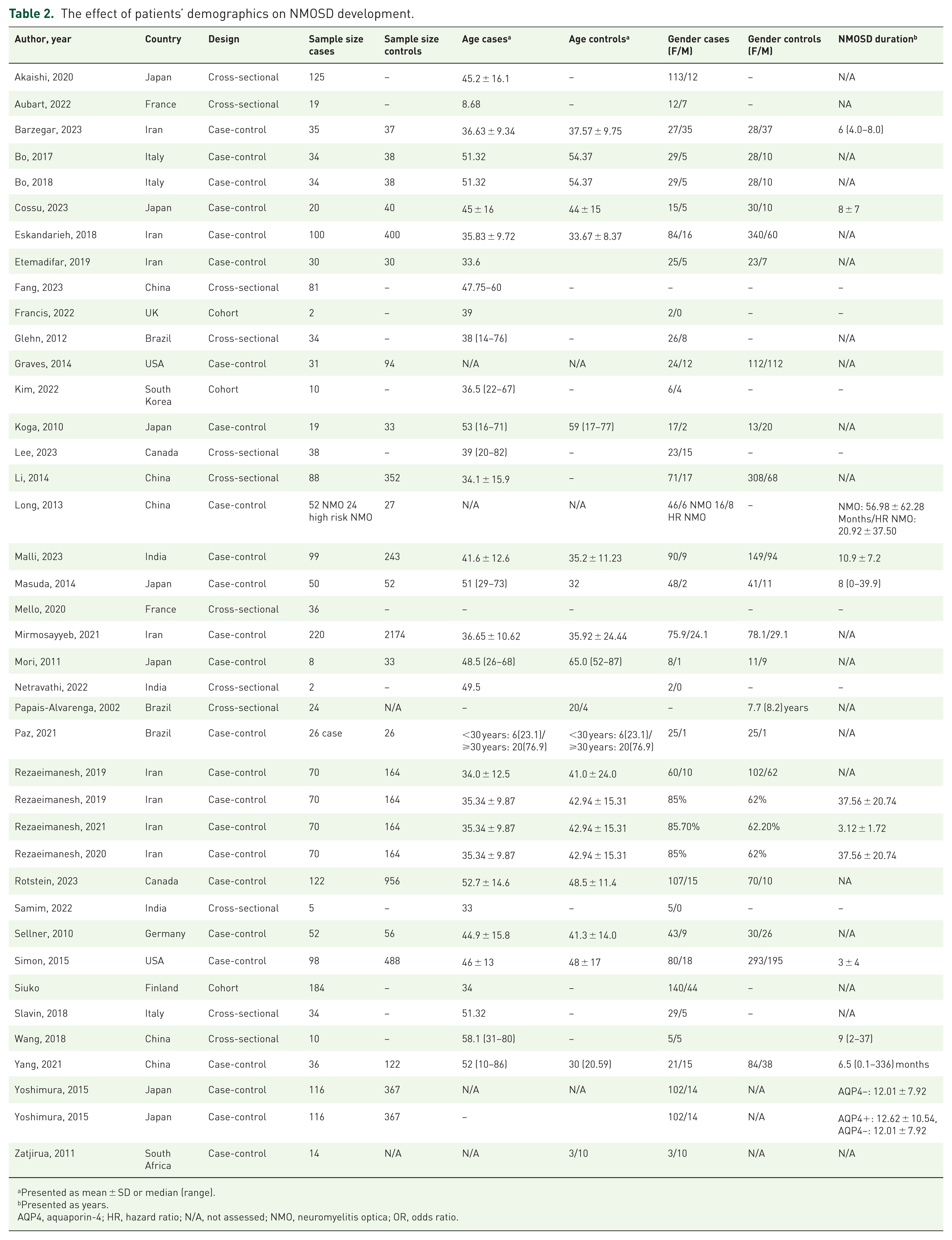
Presented as mean ± SD or median (range).
Presented as years.
AQP4, aquaporin-4; HR, hazard ratio; N/A, not assessed; NMO, neuromyelitis optica; OR, odds ratio.
The effect of patients’ lifestyle on NMOSD development.

CI, confidence interval; MNA, mini-nutritional assessment score; N/A, not assessed; NMOSD, neuromyelitis optica spectrum disorder; OR, odds ratio; TAC, total antioxidant capacity.
The effect of infection on NMOSD development.
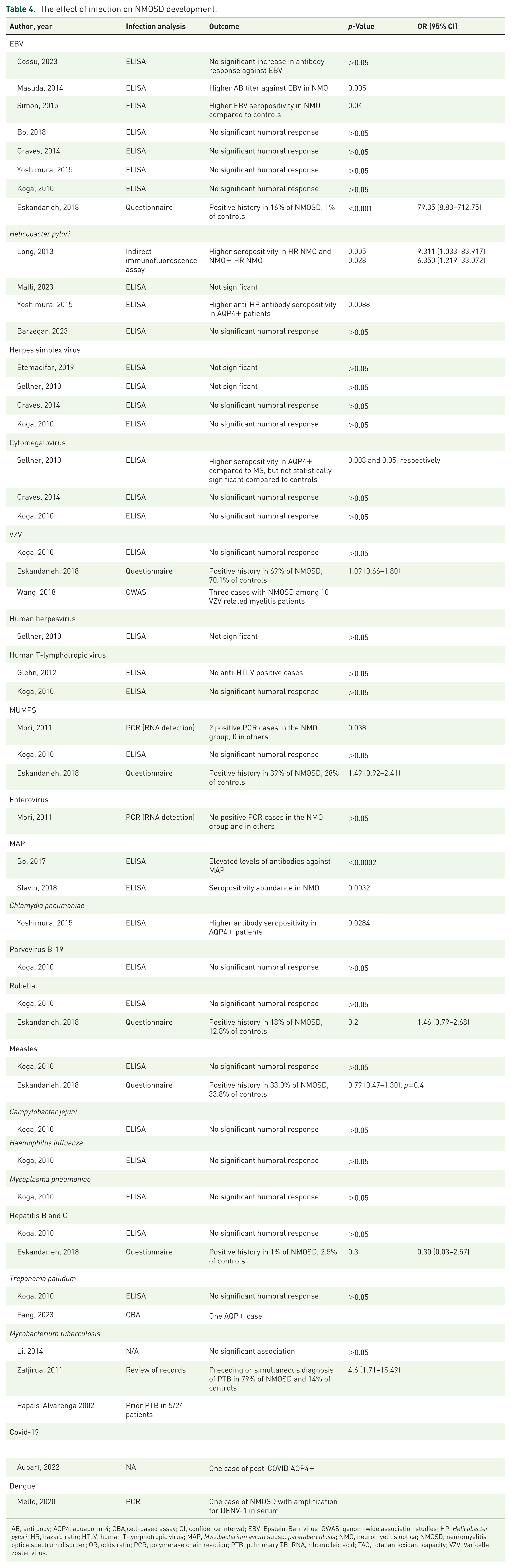
AB, anti body; AQP4, aquaporin-4; CBA,cell-based assay; CI, confidence interval; EBV, Epstein-Barr virus; GWAS, genom-wide association studies; HP, Helicobacter pylori; HR, hazard ratio; HTLV, human T-lymphotropic virus; MAP, Mycobacterium avium subsp. paratuberculosis; NMO, neuromyelitis optica; NMOSD, neuromyelitis optica spectrum disorder; OR, odds ratio; PCR, polymerase chain reaction; PTB, pulmonary TB; RNA, ribonucleic acid; TAC, total antioxidant capacity; VZV, Varicella zoster virus.
The effect of vaccination on NMOSD development.
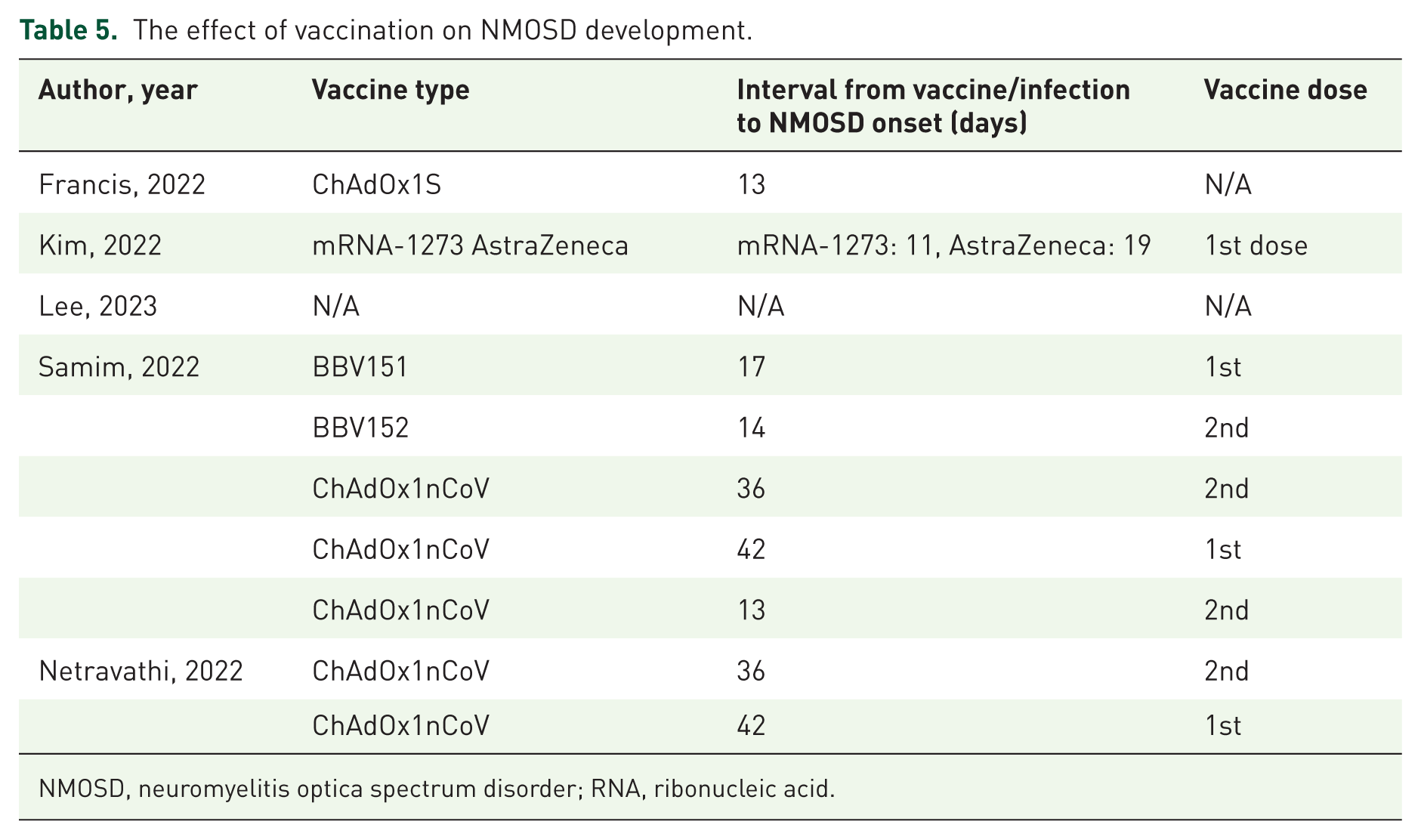
NMOSD, neuromyelitis optica spectrum disorder; RNA, ribonucleic acid.
The effect of hormonal factors on NMOSD development.

CI, confidence interval; IUI, intrauterine insemination; IVF, in vitro fertilization; NMOSD, neuromyelitis optica spectrum disorder; OCP, oral contraceptive pills; OR, odds ratio.
The effect of regional factors on NMOSD development.

CI, confidence interval; NMOSD, neuromyelitis optica spectrum disorder; OR, odds ratio.
The effect of vitamin D deficiency on NMOSD development.

N/A, not assessed; NMOSD, neuromyelitis optica spectrum disorder; N/S, Not significant; OR, odds ratio.
Results
As shown in Figure 2, the search resulted in 50 articles after the removal of duplicates and irrelevant studies from the following countries: China: 10, Iran: 10, Japan: 6, Brazil: 3, India: 3, Canada: 2, France: 2, Italy: 2, USA: 2, Australia: 1, Finland: 1, Germany: 1, Indonesia: 1, Korea: 1, South Africa: 1, South Korea: 1, Sri Lanka: 1, Thailand: 1, UK: 1. The remaining records were all written in English, and the method of the studies were case-control or cross-sectional. Except for one study, 30 all included studies had moderate to high qualities based on the NOS system. In total, 21,410 NMOSD patients were included in the studies, of which 17,080 were females.
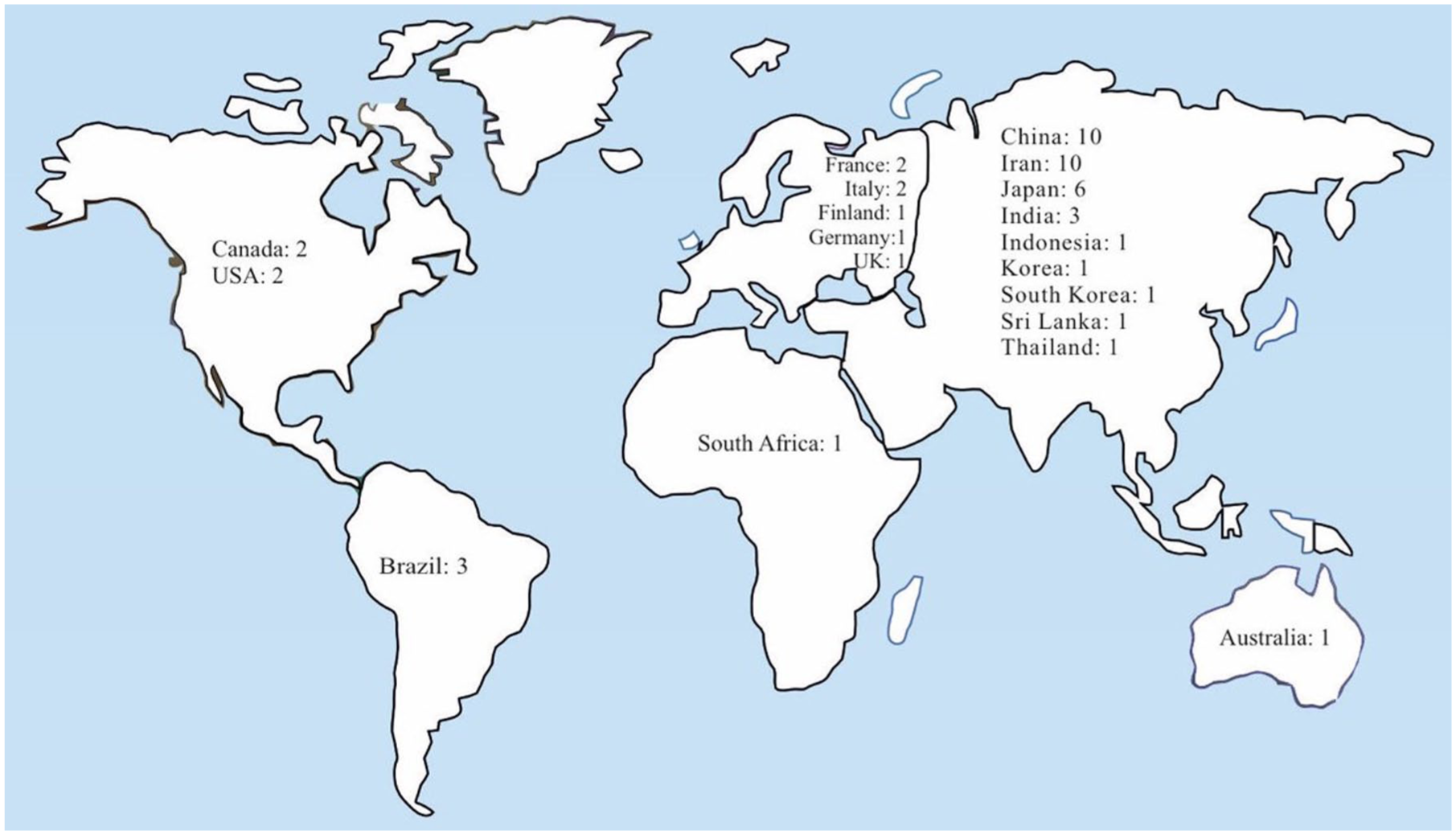
Distribution of the studies on environmental risk factors of NMOSD.
Eligible articles assessed 14 factors such as vitamin D deficiency, smoking, substance and alcohol abuse, vaccination, infection, diet, hormonal factors, seasonality, month of birth, latitude, sun exposure and physical activity and weight, stressful life events, and socioeconomic status (SES). A total of 37% of the included articles were conducted in East Asia, mainly focusing on the effect of infection and vitamin D deficiency.22,31–46 Thirty-one percent of the Asian articles included in this review were published from non-east-Asian countries,9,11,47–59 62% of which were from Iran.9,11,47–54,59 Iranian studies mainly focused on different environmental risk factors, such as diet, smoking, hormonal factors, and SES, history of oral contraceptive pills use (OCPs), month of birth, sun exposure and physical activity, and socioeconomic factors were evaluated as risk factors of NMOSD.9,11,47–51,59 Twenty-five percent of the studies included in the present review are from countries with mostly Caucasian populations.23,24,25,60–68 The main focus of the studies was smoking and infections.23–25,61,64–68.
We summarize the articles in Tables 2–8.
Demographics
Table 2 summarizes the effect of demographics on NMOSD onset. In the study of Rafiee et al., 47 marriage showed a negative impact on the odds of NMOSD development (OR = 0.10; 95% CI: 0.03–0.35). There was no relationship between NMOSD and the SES of patients and parental educational level. 47 In contrast, mothers’ education was significantly associated with NMOSD risk in the study by Graves et al., more than high school but less than college: OR = 0.46, 95% CI: 0.10–2.05, college and higher OR = 0.16, 95% CI: 0.03–0.79. The authors also evaluated daycare (OR = 0.33, 95% CI: 0.14–0.78) and age at the start of daycare (<1 year old: OR = 0.39, 95% CI: 0.13–1.15 and >1 years old: OR = 0.28, 95% CI: 0.09–0.87). 25 Akaishi et al. 32 observed a spring-summer predominance in the onset of AQP4+NMOSD. In another study, Siuko et al. 63 reported that the clinical onset of NMOSD in April–June was two times more prevalent than October–December (p = 0.03, OR = 2.0 (95% CI: 1.09–3.68)). Mirmosayyeb et al. 51 observed that the birth rate was reduced in September–October in the NMOSD group compared to controls.
Lifestyle
Table 3 shows possible lifestyle-related risk factors for NMOSD.
Smoking, substance abuse, and alcohol
Regarding the impact of smoking, a significant disparity was observed between studies on Asians and Caucasians. In two studies from Canada and the USA, patients with a history of smoking had a significantly lower risk of NMOSD. Rotstein et al. 23 reported a decreased risk of NMOSD in ever smokers in the crude (OR = 0.4, 95% CI: 0.3–0.7) and adjusted (OR: 0.5, 95% CI: 0.4–0.9) analyses. However, no correlation was observed in the analysis limited to Ontario residents (OR = 1.1, 95% CI: 0.5–2.2). In the study of Simon et al., 64 patients were less likely to report a history of smoking (OR = 0.5, 95% CI: 0.3–0.8). In contrast, smoking did not affect the risk of NMOSD in Asian studies, and in a study from Iran, passive smoking even increased the susceptibility. 11 Yang et al. 33 observed no statistical significance between active and passive smoking and the risk of NMOSD occurrence. Similarly, the study by Graves et al. 25 did not support tobacco exposure in early childhood as an environmental risk factor (p = 0.25, OR = 0.50, 95% CI: 0.16–1.5). Eskandarieh et al. found that being a passive smoker, passive smoking for more than 20 years and a positive history of smoking waterpipe showed a significant correlation with NMOSD risk (OR = 1.79, 95% CI: 1.12–2.85, OR = 2.46, 1.37–4.40 and OR = 2.39, 95% CI: 1.33–4.29, respectively). However, current smoking and smoking for more than 10 years did not correlate with the development of NMOSD. 11
Substance abuse of opioids, cannabis, stimulants, and hallucinogens did not increase the risk of NMOSD. Alcohol consumption at least once for 6 months led to an OR of 2.32 (95% CI: 1.21–4.43) for wine and 2.11 (95% CI: 1.08–4.11) for whiskey, and beer consumption did not differ between the two groups. 11
Diet
Table 3 summarizes the possible association between diet and NMOSD risk. Rezaeimanesh et al. 48 determined the odds of developing NMOSD with a focus on the impact of dietary total antioxidant capacity (TAC). Compared to the first quartile of TAC, the risk of NMOSD reduced across the quartiles (p-trend <0.01). Adjusted for age and gender, reduced risk of NMOSD was observed in second, third, and fourth quartiles with the following odds: 76% (OR: 0.24; 95% CI: 0.10–0.54) for second, 87% (OR: 0.13; 95% CI: 0.05–0.34) for third, and 97% (OR: 0.03; 95% CI: 0.00–0.12) for the fourth quartile. 50 In another study, they concluded that lower protein and fat and higher carbohydrate intakes might be associated with an increased risk of developing NMOSD. Adjusting for age, sex, BMI, and energy intake, a higher score of a low carbohydrate diet resulted in a significantly lower OR for NMOSD for the fifth (OR: 0.22; 95% CI: 0.05–0.86) and sixth (OR: 0.24; 95% CI: 0.06–0.93) decile (p-trend = 0.11). 49 In line with the findings mentioned above, lower sugar intake was associated with decreased odds of NMOSD in another study by Rezaeimanesh et al. 49 Per every 10 g increase in total sugar intake, the authors reported a 1.72-fold increase in odds of NMOSD (CI: 1.43–2.03; p-for-trend: 0.00). Moreover, various types of dietary sugar showed the following odds: glucose: 0.02 (CI: 0.00–0.08; p-for-trend: 0.00), fructose: 0.02 (CI: 0.00–0.10; p-for-trend: 0.00), galactose: 0.23 (CI: 0.08–0.61; p-for-trend: 0.00), lactose: 0.19 (CI: 0.06–0.58; p-for-trend: 0.00), and sucrose: 0.16 (CI: 0.05–0.51; p-for-trend: 0.00). In another study, Rezaeimanesh et al. reported a 90% (95% CI: 0.02–0.39) and 92% (95% CI: 0.01–0.52) reduced odds of NMOSD in the third and fourth quartiles of whole grains intake. In addition, in the third and fourth quartiles of legume intake, an 81% (95% CI: 0.05–0.71) and 95% (95% CI: 0.01–0.27) reduction in the odds of NMOSD was observed.48–50
In the study of Eskandarieh et al., the following dietary habits during adolescence showed a significant association with NMOSD risk: low dairy consumption, weekly (OR = 18.09; 95% CI = 6.91–47.37), low seafood intake (OR = 13.91; 95% CI: 6.13–31.57), low red meat consumption (OR = 3.07, 95% CI: 1.68–5.62), low egg consumption (OR = 9.39, 95% CI: 4.03–21.86), low chicken consumption (OR = 4.05, 95% CI: 1.59–10.35), low-fat consumption (OR = 3.01, 95% CI: 1.64–5.49), and low fruit and vegetable consumption (OR = 6.23; 95% CI: 3.07–12.62). Moreover, Eskandarieh et al. reported that monthly consumption of multivitamins (OR = 5.49; 95% CI: 1.90–15.85), iron (OR = 7.64; 95% CI: 2.98–16.61), vitamin B12 (OR = 2.78, 95% CI: 1.05–7.36), and vitamin C (OR = 3.80, 95% CI: 1.63–8.87) decreases the risk of NMOSD. However, calcium, folic acid, and vitamin D consumption did not differ in the chance of NMOSD onset (p > 0.05). 11
Paz et al. 30 reported a higher risk of developing NMOSD when the dietary inflammatory index was ⩾4.41 (OR = 30.25; 95% CI: 6.70–136.47). In the study of Huang et al. 69 using the mini-nutritional assessment score, the nutritional score of NMOSD patients was significantly lower than healthy controls.
Other lifestyle factors
Physical activity was assessed in two articles. In the study of Eskandarieh et al., low and moderate amounts of light physical activity at 13–19 years of old age were associated with being assigned to the NMOSD group (OR = 6.32, 95% CI: 3.08–12.95 and OR = 3.95, 95% CI: 1.89–8.28). Patients more frequently reported low and moderate amounts of heavy physical activity at 13–19 years old age, compared to controls (OR = 16.11, 95% CI: 7.03–36.91 and OR = 2.60, 95% CI: 1.06–6.35, respectively). 11
In the study of Paz et al., 30 physical activity and weight did not differ between cases and controls. A study by Rafiee et al. 47 showed the association of NMOSD and first-degree relatives (OR = 5.10; 95% CI (1.78–14.61), homelessness periods (OR = 4.35; 95% CIs (1.18–14.74), joblessness (OR = 4.24; 95% CI (1.91–5.15)), family disruption (OR = 12.68; 95% CI: 3.53–45.46), and divorce (OR = 14.18; 95% CI (1.91–23.15), university entrance exam (OR = 0.31; 95% CI (0.11–0.88)) showed a negative association with NMOSD risk (Table 3).
Infection
The association of a single infectious agent with autoimmune diseases is not conclusively established; patients with autoimmune diseases often experience relapses associated with seasonal infections during winter, suggesting an influence of environmental factors in addition to well-established genetic predispositions such as polymorphism in the HLA system. 70
Table 4 summarizes the possible association between the history of infection and the risk of NMOSD development.
Epstein–Barr virus
The history of EBV infection was evaluated in eight studies, three of which reported it as a risk factor. According to Masuda et al., serum anti-early antigen (anti-EA IgG) was more frequently positive in NMOSD patients, showing a more active viral replication than the controls. Furthermore, higher antibody titers of anti-EA IgM and anti-EA IgG were detected in NMOSD patients compared to controls (p < 0.001 and p = 0.005, respectively). 38 In the study of Simon et al., EBV seropositivity was more common in NMOSD patients than the controls, and they seemed likely to have a history of infectious mononucleosis (OR = 1.4, 95% CI: 0.8–2.7). However, an antibody against Epstein-Barr nuclear antigen (Anti-EBNA1 IgG Ab) was not associated with the risk of NMOSD. 64 Eskandarieh et al. 11 revealed a higher frequency of mononucleosis in NMOSD cases than in healthy controls (OR = 79.35, 95% CI: 8.83–712.75). Other studies did not observe an association.11,35,40,46
Helicobacter pylori
Four studies compared the rate of H. pylori (HP) seropositivity in NMOSD patients and healthy controls. Seropositivity against HP in NMOSD patients was only observed in East Asian studies. In the study of Long et al., 34 higher seropositivity was observed in high-risk NMOSD patients (OR = 9.311, 95% CI = 1.033–83.917) and NMO + high-risk NMO (OR = 6.350, 95% CI: 1.219–33.072). Yoshimura et al. 35 reported a higher anti-HP antibody seropositivity in AQP4+ patients (p = 0.0088). The other two studies did not report any significance. 35
Cytomegalovirus
Among three studies that evaluated the seroprevalence rate of Cytomegalovirus IgG in NMOSD patients, a trend was observed in the study of Sellner et al. 65 in AQP4+ patients (p = 0.05), while other studies did not show any significance.25,43
Mumps
Mori et al. 39 reported that polymerase chain reaction results of mumps virus Ribonucleic acid (RNA) were more frequently positive in cerebrospinal fluid of NMOSD patients compared to controls. The other two studies did not support the correlation (p > 0.05).11,43
Mycobacterium avium
The correlation between NMOSD onset and M. avium ssp. paratuberculosis (MAP) infection was strongly supported in the study of Bo et al. 24 and Slavin et al. 66 The sera of NMOSD patients showed a higher antibody level against peptides derived from MAP, compared to the healthy control group (p < 0.0002), in the study of Salvin et al., 66 the greater humoral response against two virulence factors of MAP, protein tyrosine phosphatase A, and protein kinase G detected in NMOSD patients (p < 0.0001 and p = 0.0032, respectively).
Chlamydia pneumoniae
Yoshimura et al. 35 reported a higher Chlamydia pneumoniae antibody seropositivity in AQP4+ patients than controls.
Mycobacterium tuberculosis
Tuberculosis infection was evaluated in three studies, one of which reported it as a risk factor (OR: 4.6 (95% CI: 1.71–15.49)).42,71,72 Papais-Alvarenga et al. 72 reported a history of pulmonary TB in 5 out of 24 evaluated NMOSD patients. One study did not report any significance.
Treponema pallidum
Fang et al. 41 reported one AQP4+ case among 81 patients with neurosyphilis. Koga et al. 43 did not report any significance.
Covid-19
Aubart et al. 67 reported a case of AQP4 seropositive among 19 children who had a history of COVID-19 infection and manifested a CNS inflammatory disease.
Dengue
Mello et al. 68 reported one case of NMOSD with amplification for DENV-1 in serum among 36 patients.
Varicella zoster virus
Wang et al. 73 reported 3 cases of NMOSD among 10 varicella zoster virus related myelitis patients.
Other infections
Studies did not evaluate the following microorganisms as possible risk factors of NMOSD development: parvovirus B-19, rubella, measles, Campylobacter jejuni, Haemophilus influenzae, Mycoplasma pneumoniae, hepatitis B and C, and enterovirus.
Human herpesvirus, human T-lymphotropic virus, and Herpes simplex virus.11,39,43,54,65,73
Vaccination
Table 5 summarizes the studies that report NMOSD onset after COVID-19 vaccination.41,52,57,58,71 Francis et al. 60 reported two patients who developed AQP4+ NMOSD 13 days after ChAdOx1S vaccination. Kim et al. 44 also reported two patients who developed NMOSD after the first dose of the vaccine. The interval from vaccine to clinical onset ranged from 11 days in the mRNA-1273 vaccine to 19 days in AstraZeneca. Lee et al. 61 also reported an AQP4+ patient after COVID-19 vaccination; however, vaccine type and the interval from vaccination to NMOSD onset were not specified in this study. In another study, Samim et al. 55 observed five cases of post-vaccination NMOSD, which occurred between 13 and 36 days after ChAdOx1nCoV and BBV152 vaccines. Netravathi et al. 74 reported two female patients who developed NMOSD after the first and second doses of ChAdOx1nCoV.
Hormonal factors
Table 6 demonstrates the association between hormonal factors and the risk of NMOSD. In the study of Eskandarieh et al., the history of Intrauterine Insemination or In Vitro Fertilization did not relate to NMOSD onset. Regarding the use of contraceptives, they observed that NMOSD patients report a more frequent use of OCP (OR = 2.11, 95% CI: 1.21–3.66). However, the duration of OCP use did not alter the risk. 11 Graves et al. reported that breastfeeding is associated with lower odds of having NMOSD (OR = 0.42, 95% CI: 0.18–0.99), cesarean delivery was not associated with increased odds of NMO compared with vaginal delivery (p > 0.05).
Regional factors
Table 7 shows the possible impact of regional factors on NMOSD onset.
Latitude
No association was found regarding the effect of latitude on NMOSD onset.31,46,62
Sun exposure
In the study of Eskandarieh et al., low sun exposure at 13–19 years old was associated with NMOSD onset (OR = 17.97, 95% CI: 7.11–45.40, p < 0.001). Low and moderate sun exposure at >20 years old also increased the risk of onset (OR = 10.83, 95% CI: 4.40–25.47, p < 0.001 and OR = 5.21, 95% CI: 2.27–11.94, p < 0.001, respectively). 11
Vitamin D
Table 8 indicates the possible association between vitamin D deficiency and NMOSD development. All seven studies were conducted in Asia.22,31,33,53–55 While East Asian studies evaluated vitamin D deficiency as a risk factor,22,36,37 another study from Thailand also reported significance, 57 and three other studies did not observe any significance. The significant results are as follows: Min et al. 22 reported a significant difference in the frequency of vitamin D deficiency in NMOSD cases versus controls. Puangsricharoen et al. 57 and Shan et al. 37 also observed lower vitamin D levels in NMOSD patients.
Levels of 25(OH)D, 25(OH)D2, and 25(OH)D3 were reported to be lower in the study of Gao et al. 36
In two other studies, no significant difference was observed between the vitamin D levels of NMOSD patients and healthy controls.56,58 Ashtari et al. 59 observed no difference between vitamin D levels in NMOSD and MS patients.
Discussion
The current systematic review summarizes the environmental factors associated with NMOSD onset. Even though the exact pathogenesis of the disease is yet to be understood, addressing triggers might help reach a prompt and accurate diagnosis. Besides individual factors such as age, gender, education, and ethnicity, studies indicate an environmental involvement in the onset of NMOSD. Environmental factors with seasonal variation influence the development of neurological conditions related to NMOSD. Spring-summer predominance in NMOSD onset was observed in the studies, which might be related to a higher prevalence of viruses like adenoviruses or varicella zoster and lower vitamin D levels during winter; moreover, changes in electrolytes due to dehydration could also affect disease onset.7,75 Environmental risk factors in this study have mainly focused on vitamin D deficiency, smoking, infection, and diet.
Effect of ethnicity on NMOSD risk
According to the literature, the prevalence of NMO is 0.5–4 per 100,000; however, in certain racial groups it may reach 10 per 100,000. 76
East Asians
NMOSD prevalence in China, Japan, and Korea has been reported as ~3.5 per 100,000, which is higher compared to other racial groups. A nationwide survey reported a prevalence of 3.42/100,000 in Japan, reaching 4.1/100,000 in other Japanese studies. The prevalence among Chinese is estimated as 3.1/100,000, in Korea, it has been recorded as 3.56 per 100,000. 8 Genetic studies indicate that Chinese and Japanese share HLA-DPB1*05:01 and HLA-DRB1*16:02, which increases NMOSD risk. 77 A third (37%) of the included articles was conducted in East Asia, mainly focusing on the effect of infection and vitamin D deficiency. Studies support that low vitamin D is significantly associated with higher odds of MS onset, and vitamin D supplements are proven to lower the relapses and activity measured by MRI in MS cases. 78 Vitamin D response elements form when the active form of vitamin D binds to intracellular vitamin D receptors, and directly influence the regulation of innate and adaptive immunity. Vitamin D may play its role in the pathogenesis of NMOSD by directly affecting AQP4-IgG as a part of humoral immunity, as it induces B cell apoptosis, which leads to a decrease in the production of antibodies.79,80 Moreover, vitamin D enhances an anti-inflammatory environment by inducing IL-10 and suppressing IL-6 and TNF production. 81
Vitamin D deficiency is more prevalent in Asia than in other regions. 82 When vitamin D deficiency is defined as less than 50 nmol/L, the prevalence is estimated as 18%—30% in the USA, 40% in Europe, 34% in Africa, and 54% in Asia. Several reasons are mentioned to be responsible for this issue. Asians tend to wear clothes that cover larger areas of the skin, leading to a reduction in ultraviolet B to penetrate the skin and vitamin D synthesis. Moreover, developing countries in Asia lack access to proper vitamin D supplements, unlike those in America and Europe. 82 All seven studies that evaluated vitamin D deficiency as a possible risk factor were conducted in Asia. East Asian studies evaluated vitamin D deficiency as a risk factor; another study from Thailand reported significance, and three other studies did not observe any significance.22,31,34,53–55 Consequently, countries in East Asia have relatively lower levels of vitamin D. Based on a meta-analysis by Jiang, the mean level of 25(OH)D concentration in Korea and China is 49.3 and 52.6 nmol/L, while in Indonesia and Sri Lanka, the mean level reaches 65.8 and 69.6 nmol/L. Different prevalence of vitamin D deficiency and a small sample size might explain the disparity. 82
The other main focus of East Asian studies was infections. Several case reports have reported the onset and worsening of NMOSD after bacterial and viral infection; however, we only included studies with a population of more than three cases.83,84 Seropositivity against HP in NMOSD patients was only assessed in East Asian studies.34,35 It is important to note that the highest prevalence of HP among the normal population is found in African countries, followed by Asia, and the lowest in Oceania. Among Asian countries, East Asians such as China and Korea show the highest prevalence, while Indonesia and Sri Lanka have the lowest. 85
Studies indicate a significant association between MAP and NMOSD onset; however, controversial results have been reported regarding other infections such as EBV and mumps, although they are considered triggers of MS. 86 Viruses and bacteria can, for example, trigger NMOSD onset through molecular mimicry and increased blood-brain barrier permeability, thus allowing the entry of antibodies into the CNS.
Even though tuberculosis is more common in Asian populations compared to Europe and North America, the strength of the evidence is relatively weak regarding the impact of this infection in provoking disease onset. Out of three studies evaluating TB in NMOSD, two report frequent prior pulmonary tuberculosis in NMOSD patients (79% and 21% positive history). The studies above evaluated small populations and did not report the AQP4-IgG status of the patients, leaving this as an open question. A possible effect of TB might be related to the fact that aquaporin extracellular loops in the Mycobacterium share homologous epitopes with human AQP4, so molecular mimicry may create autoantibodies that lead to astrocyte damage. 87 Interestingly, a prospective study showed that in patients with steroid-refractory NMOSD, anti-TB treatments (a four-drug regimen consisting of isoniazid, rifampicin, pyrazinamide, and streptomycin and three three-drug regimens consisting of isoniazid, rifampicin, and pyrazinamide) improve neurological deficits. 88
It is important to note that the abovementioned factors, such as vitamin D deficiency and some infections, are reportedly more frequent in Africans; however, due to a limited number of studies, environmental risk factors of NMOSD have not been properly assessed in the African region.
Caucasians/Whites
The prevalence of NMOSD among Caucasians ranges from 0.55/100,000 in Australia and New Zealand to 1.04/100,000 in Sweden. In Denmark, the prevalence is reported to be 1.09/100,000; however, recent studies indicate that by including the total clinical phenotype of NMOSD (AQP4-negative cases and myelin oligodendrocyte glycoprotein antibody-positive cases), the prevalence reaches 4.4 per 100,000. The prevalence of NMOSD in Caucasians is remarkably less than the rates observed in Asia; however, in Hungarians, it reaches 1.91, which may be due to the admixture of Asian genes. 8 In northern Europe, the HLA-types HLA-DRB1*03 and HLA-DRB1*01:01 have been observed to increase the susceptibility of NMOSD. 77 The main focus of the studies in Caucasian populations was smoking and infections.
Regarding the impact of smoking, a significant disparity was observed between studies on Asians and Caucasians. In two studies from Canada and the USA, respectively, patients with a history of smoking had a significantly lower risk of NMOSD.23,64 In contrast, smoking did not impact the risk of NMOSD in Asian studies, and in a study from Iran, passive smoking even increased the susceptibility.11,33 It has been established that smoking can affect several both innate and adaptive immune responses, and the effect of smoking in the onset of MS has been previously established. 17 The function of toll-like receptors, dendritic cells, and natural killer cells is altered by smoking, and the number and activity of CD8+ cells increase in smokers. 86 Smoking also leads to an increase in memory B cells and a decrease in regulatory B cells. 89 Moreover, studies indicate that smoking can damage astrocytes, which may consequently lead to NMOSD development. 90
Nevertheless, some studies report the anti-inflammatory effect of nicotine, which may help explain the paradox. Nicotine can decrease the proliferation rate of lymphocytes and leukocyte infiltration into the CNS. Jiang et al. showed that in a model of MS, the infiltration of proinflammatory monocytes and neutrophils is inhibited by nicotine through its impact on nicotinic acetylcholine receptors. Moreover, in nicotine-treated animal models, CCL2 and CXCL2 are reportedly reduced. Such chemokines involve proinflammatory cell chemotaxis and might affect disease onset. 91 More studies are needed further to evaluate the effect of smoking habits on NMOSD occurrence.
Diet
Studies show that high carbohydrate and sugar intake increases the risk of NMOSD. A possible explanation for the effect of sugar on the nervous system might be dysbiosis of gut microbiota and the pro-inflammatory effect. By increasing pro-inflammatory factors like CRP, sugar consumption can release reactive oxygen species. 92 Sugar increases intestinal permeability and dysbiosis in gut microflora, leading to the stimulation of proinflammatory factors, such as TNF-α and IL-6, that can cause systemic inflammation. 93 By the Brain-Gut axis hypothesis, modifying sugar intake can improve the microflora profile and reduce inflammation, which helps decrease the odds of NMOSD. 94 A high-energy and high-carbohydrate diet causes excess weight, which some studies indicate is a risk factor for neurodegenerative disease. 95 Adipose tissue is also a significant factor in autoimmune diseases through its impact on the proliferation of TH17 cells. 96
Socioeconomic status
Regarding SES, the included study did not show any significance, and according to the literature, MS follows the same pattern. In a systematic review by Goulden et al. 97 in 2015, no association was observed between SES and NMOSD risk. Another cohort in Denmark found the same result. 98
Hormonal factors
It was observed in the studies that, in contrast to OCP use, breastfeeding prevents NMOSD onset. The same result was reported when assessing MS cases. In a 2016 case-control study, it was observed that a history of OCP use is related to a 50% increase in the risk of MS. 99 It is believed that breastfeeding alters the risk of MS onset, which might be due to ovarian suppression that leads to a reduction in inflammatory activity. 100 Moreover, being breastfed as an infant also lowers the risk of MS onset and delays it, as it contains IL-10 and transforming growth factor-beta, which have anti-inflammatory properties. 101
The development of NMOSD might be due to the same reasons as well.
Limitations
Like any other systematic review, a limitation of this study is publication bias and heterogeneity in studies that make conclusions difficult for the readers; moreover, limited studies address the environmental risk factors of NMOSD, especially in African populations.
Conclusion
The review of 50 environmental risk factors in the current study showed that vitamin D deficiency, cigarette smoking, MAP infection, and diet had a significant association with NMOSD. In East Asians, vitamin D deficiency contributed to increased risk of NMOSD. Controversial results were reported regarding the effect of smoking among Caucasians and other races. Further studies are required to elucidate the findings presented in this review study.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251363293 – Supplemental material for Environmental risk factors of neuromyelitis optica spectrum disorder: a systematic review
Supplemental material, sj-docx-1-tan-10.1177_17562864251363293 for Environmental risk factors of neuromyelitis optica spectrum disorder: a systematic review by Mahsa Mohammadi Lapevandani, Elham Bazmi, Shima Jahani, Nasrin Asgari and Mohammad Ali Sahraian in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
