Abstract
Orthostatic hypotension (OH) is a common, disabling manifestation of Parkinson’s disease (PD) and a substantial driver of discomfort and functional disability. Clinicians caring for people with PD benefit from a working knowledge of many different treatment options for PD with OH, including medications and nonpharmacological treatments. This review provides clinicians a working understanding of PD OH management strategies that may help them in clinical practice. This includes a summary of clinical features, pathophysiological considerations, pharmacological and nonpharmacological treatments, and a proposed integrated approach to the PD patient with OH.
Introduction
Symptoms of Orthostatic Hypotension (OH) are a highly disabling feature of Parkinson’s disease (PD). The most common of these symptoms—seen in association with a corresponding drop in blood pressure (BP)—include lightheadedness or dizziness when adjusting posture from a supine or seated position to a standing position. In PD, OH contributes to poor quality of life, increased fall risk, and may be an independent marker for more aggressive disease progression as well. Encouragingly, several symptomatic treatment approaches, many of which have been developed outside of the field of PD, can alleviate bothersome symptoms in affected individuals. In this clinically-focused review intended for medical providers caring for people with PD (PwPD) and OH, we aim to summarize the signs and symptoms of OH, pathophysiological models of OH in PD relevant to different treatment rationales, and pharmacological and nonpharmacological treatment approaches—many of which are off-label considerations—that can improve OH symptoms in affected individuals with PD.
Symptoms and diagnosis
Orthostatic hypotension (OH) affects about 30% of PwPD 1 and can be identified either by the presence of orthostatic symptoms suggesting OH or by measuring BP and pulse in associated positions. Common symptoms of OH in PD include feeling faint, lightheaded, or dizzy when changing from a lying or seated to a standing position. Many people with OH and PD also experience postprandial hypotension (PP) which manifests with similar symptoms that come on within 2 hours after eating. 2
Although OH is typically thought of as a nonmotor feature of PD, it has a profound deleterious impact on PD motor disability. PD with OH associates with greater activity of daily living (ADL) impairment, slower gait speed, greater physical frailty indices, and worse motor scoring on the Unified Parkinson’s Disease Rating Scale. 3 These features may be caused directly through OH-linked perfusion deficits to various organs. They may also be linked indirectly through either shared causal pathology or compensatory behavioral changes brought about by the uncomfortable symptomatic experience of chronic OH when superimposed upon PD. OH also associates with poor prognosis in PD and with the presence of other symptoms that drive morbidity and mortality. These include an increased risk of falls and poor quality of life, 4 cognitive decline, 5 and dementia. 6
OH in PD has a host of other associated symptoms that can be seen in association with postural changes (i.e., standing) and can be relieved by sitting or lying down (Table 1). These include generalized weakness, fatigue, pain in the neck and shoulders sometimes referred to as “coat hanger syndrome,” breathing difficulties, and headache. 7 A significant fraction of people with profound OH, with or without PD, present with atypical symptoms including backache and headache, lower extremity discomfort, shakiness and/or tremor, warmth and tingling, and upper extremity discomfort; 33% of patients can also be asymptomatic. 8 In a cohort of 210 PwPD, 50% had OH by classic criteria and 69% of these were asymptomatic. 9 OH symptoms are a common—though not universal—feature of prodromal PD.10,11 When present without manifest parkinsonism, this can be a feature of pure autonomic failure (PAF), a synucleinopathy with a high risk of conversion to manifest PD or Dementia with Lewy Bodies (DLB). A meta-analysis has shown that OH symptoms are increasingly common with advancing disease duration. 12 How much of this increased prevalence is due to progression of underlying synucleinopathy burden versus both the combination of advancing age and/or iatrogenic effects caused by additional medications is less clear.
Symptoms of OH in PD.

Please note that this list is not an exhaustive list of all possible symptoms of OH in PD.
OH, orthostatic hypotension; PD, Parkinson’s disease.
Postprandial OH (PP OH) is seen in several neurological conditions with autonomic dysfunction including PD. PP OH manifests with BP drops and associated OH-type symptoms within 2 hours of eating a meal—often one rich in carbohydrates. PP OH is triggered in part by pooling of blood in splanchnic blood vessels perfusing the enteric nervous system but also involves multiple putative “second hits” including the absence of effective compensatory sympathetic vascular or cardiac activity in the minutes following eating. 13
OH can be measured by checking BP changes with position alterations. The generally accepted method in PD involves checking systolic (SBP) and diastolic blood pressure (DBP) and pulse in the supine position after at least 5 min of rest. If lying recumbent in a supine position is not possible, a seated position could be substituted. The participant is next instructed to stand and remain standing for at least 60 s but no longer than 3 min before the same set of vital signs is rechecked again. A drop of ⩾20 mmHg in SBP or ⩾10 mmHg in DBP when assessed during the standing position is consistent with the diagnosis of OH. 14 Ideally, multiple BP measurements should be taken during the 60 second to 3-minute period in question to ensure that the fall in BP is sustained and not self-resolving or due to a spurious measurement. Heart rate typically rises in the setting of OH because a reduction in venous return to the heart leads to a reduction in cardiac stroke volume, thereby giving rise to a compensatory rise in heart rate. This phenomenon is sometimes called sympathetic or sympathotonic OH and is the form of OH most closely associated with PD. This should be differentiated causally from vasovagal or reflex syncope, one of the most common causes of syncope in adults, which is due to visceral afferent activation, triggering subsequent bradycardia and vasodilation.
Like conventional OH, PP OH can be measured analogously by detecting a drop in BP by ⩾20 mmHg in SBP when measured within 2 hours following the start of meal ingestion or when the SBP following meal ingestion decreases to 90 mmHg if the pre-meal SBP was greater than 100 mmHg. 15
Pathophysiology of OH in PD
There are multiple hypothesized explanations for why OH occurs in PD. None of these theoretical models are mutually exclusive. It is likely that several of these causal factors coexist in PD patients with OH. A dominant model isn’t likely to emerge until several key clinical research roadblocks are overcome including a widely accessible method for quantifying orthostatic BP shifts in a continual fashion in ambulatory settings, detailed circuit mapping of the human central and peripheral autonomic systems, and deeper pathophysiological understanding of the comparative vulnerability of the various neuronal cell types involved in autonomic tone modulation to synuclein pathology. For the ease of conceptual review, we have divided potential causes into two distinct categories: primary neurodegenerative causes and nonneurodegenerative causes.
Primary neurodegenerative causes
Orthostasis can be an early or even prodromal feature in synucleinopathies. Recent human subject data reveals that some of the earliest detectable preganglionic changes in PD and other synucleinopathies include the misfolding of synuclein within cholinergic neurons outside of the central nervous system. These include (1) cholinergic neurons in the gastric mucosa feeding into the myenteric plexus and dorsal motor nucleus of the vagus, (2) sudomotor cholinergic nerve fibers feeding into the sympathetic ganglia, and (3) cranial nerve 7 afferents innervating the submandibular ganglia.16,17 Collectively these regional peripheral pathologies associate with vasoconstrictive impairments, reduced peripheral vascular resistance when standing, greater pooling of blood in the periphery, and a relative reduction in cardiac output when shifting to a standing position. Cardiac sympathetic denervation is also seen in PD, DLB, and PAF (but not in Multiple System Atrophy where preganglionic autonomic denervation is more common) and may be a staging marker in PD where it has been shown to advance by about 4% every year. 18 Dysautonomia may also be seen in atypical parkinsonian conditions such as progressive supranuclear palsy. 19 Although the preganglionic pathologies may associate with OH in PD, postganglionic disorders are thought to mediate a substantial component of vasomotor dysfunction seen in PD OH. 20 Lewy bodies are known to accrue in autonomic postganglionic adrenergic neurons. 21 Such pathology not only leads to reductions in the resting plasma levels of epinephrine and norepinephrine, it also impairs neurons ability to secrete these catecholamines in times of normal physiologic need. 22 The end result is both a primary dysfunction of vasomotor tone in the body and secondary compensatory changes to other elements of peripheral vascular system as well. About two-thirds of PwPD and some measure of daytime dysautonomia will also have nocturnal supine hypertension, sometimes called “reverse dipping.” 23 The pathophysiology of this nocturnal supine hypertension is not well understood but may involve compensatory cardiac hypertrophy and diastolic dysfunction. 24
Synuclein pathology and Lewy body deposition is not thought to affect the hypothalamic nuclei until after the onset of PD motor dysfunction in Braak Stage 4. 25 Nevertheless, central autonomic regulation is coordinated (1) rostrally to the basal forebrain, (2) caudally to the brainstem and spinal cord nuclei, and (3) systemically through the neuroendocrine system regulating the hypothalamic-pituitary-adrenocortical axis by key hypothalamic nuclei including the paraventricular and supraoptic nuclei. 26 Different hypothalamic relay nuclei are also involved in other associated non-motor features including sleep regulation, thermoregulation, and weight changes—perhaps explaining the high rate of overlap of OH features with each of these individual symptom complexes in PD. Motor subtyping research in PD has also suggested that the akinetic-rigid subtype (compared to the tremor-predominant or mixed subtypes) may have a higher prevalence of OH. 27
Regional in vivo biomarker studies have also suggested insular and limbic dysfunction associates with OH in PD. This may be mediated through a common network involving hypothalamic Lewy Bodies or may be a separate network providing innervation to brainstem reticular formation and then to peripheral autonomic nuclei.28,29
Nonneurodegenerative causes
Levodopa is the most used medication to treat PD and can contribute to OH symptoms through multiple overlapping mechanisms. These include (1) the vasodilatory effects of levodopa’s active metabolite (dopamine) on peripheral and visceral vasculature, (2) direct effects on cardiac sympathetic function, and (3) effects on the neuroendocrinological renal-aldosterone axis. 30 In affected individuals with PD and OH induced by levodopa, standing BP shows reductions for about 2 hours after drug ingestion but starts to rise again after that point. 31 In a randomized controlled trial (RCT) involving levodopa-on and levodopa-off orthostatic evaluations in a cohort of PD participants with and without OH, levodopa use correlated with lower aggregate SBP values measured both when supine and standing in PD with and without OH, but did not associate with a statistically significant within-participant levodopa-induced change in the difference between supine and standing BP. 32 Interestingly, a recent PD study showed that risk for levodopa-induced OH is higher in PwPD and an abnormal response to the Valsalva maneuver, 33 suggesting that peripheral autonomic dysfunction may be an important ingredient in this pathological clinical OH response to levodopa. Other similar dopaminergic medications, including dopamine agonists, can have similar or even more profound effects on OH through analogous mechanisms.
PD and atherosclerosis are both disorders of aging and commonly coexist in the same person. Antihypertensive medications are used for the primary and secondary prevention of cardiovascular disease and can often be well tolerated without side effects in early PD or in relatively younger people. As PD advances, however, so too does time and the effects of aging on the elasticity of large and small arteries, thereby reducing their buffering capacity for antihypertensive drugs that iatrogenically alter the link between neuronal control of BP and the arteries that ensure time-sensitive dilation or contraction in response.
This latter situation can be a particular problem in individuals with the 3-part combination of PD, OH, and coexisting supine hypertension. Supine hypertension in PD affects about 28% of PwPD and is even more prevalent in those with PD OH compared to PD without OH. 34 The underlying biological causes of coexisting supine hypertension in PD OH is not completely understood. Some combination of sustained peripheral vascular resistance and neuroendocrinological dysfunction likely each plays additive roles. 35 In clinical setting such patients often also have direct or indirect contributions from non-PD pathologies including coexisting diabetes mellitus, refractory hypertension often predating PD with OH, and microvascular end-organ damage impairing vascular reserve through the body. All of these factors reduce the narrow therapeutic window for drugs that aim to decrease BP.
Clinical approach to PwPD and OH
We recommend screening for OH using an assessment of orthostatic vital signs in PwPD presenting with classic and atypical symptoms of OH, unexplained falls, or syncope. Screening methods anchored only on common symptoms may underestimate the number of PwPD who have OH, especially if office-based BP assessments are only available in seated-to-standing contexts, so home monitoring obtained first thing in the morning after lying for 5 minutes then standing 3 minutes or as symptoms develop during the day may be helpful. A minority of patients with autonomic dysfunction have delayed OH (drops occurring after 3 minutes), so continuous ambulatory BP monitoring or autonomic testing could be considered if suspicion is high and other testing is negative.36,37 For patients with unrevealing orthostatic vital signs assessments in the setting of disabling orthostatic symptoms, referral for a tilt table test can help increase the sensitivity of a diagnostic work-up.
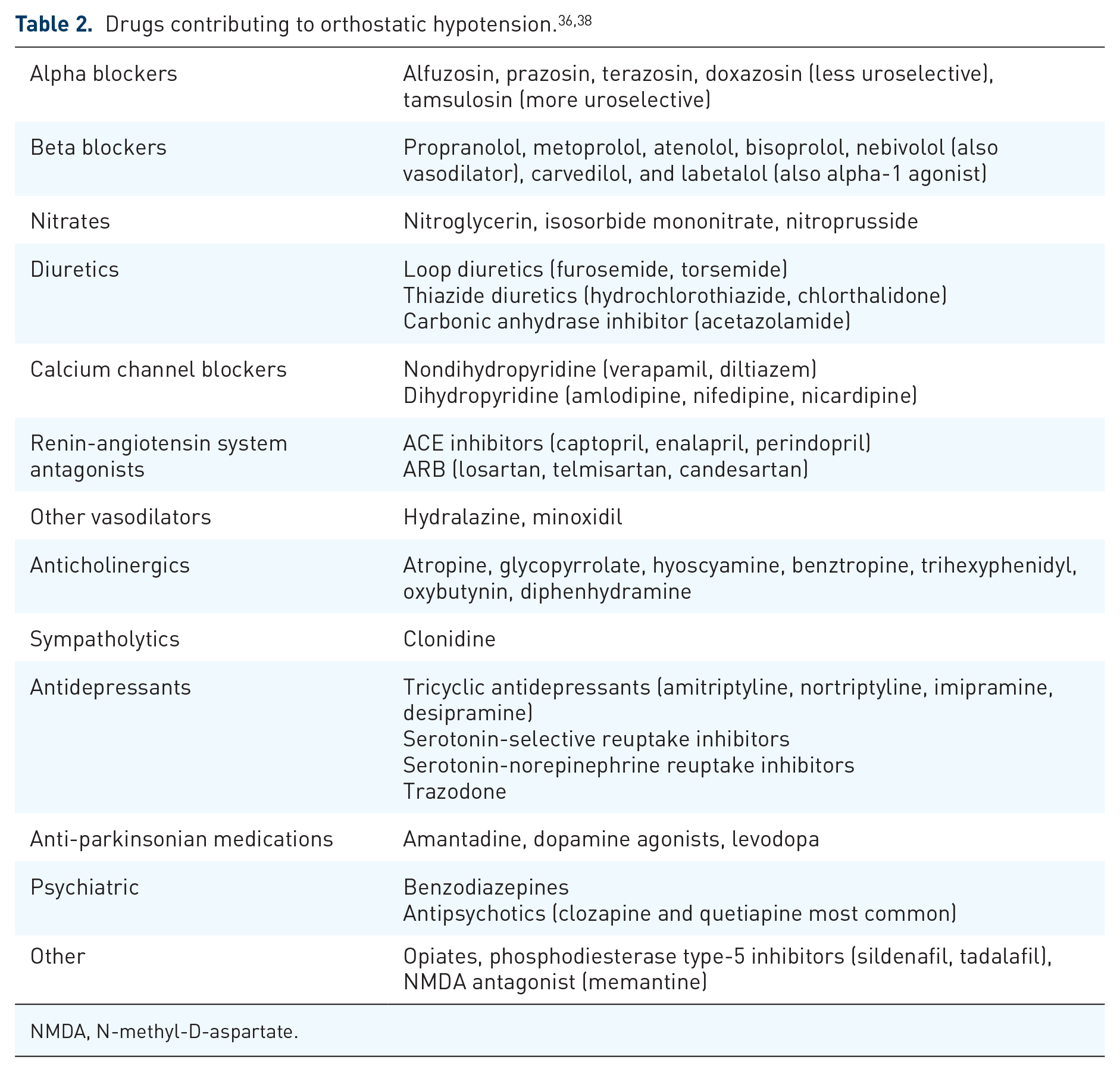
When OH is identified, an important early step is to evaluate for an alternate explanation, including cardiovascular disease, vasodilatory effects of subacute infections, and iatrogenic causes. Primary cardiac etiologies and infections are the most emergent and a full evaluation should be coordinated with the patient’s specialty providers. At the same time, a close review of medications should be undertaken. In addition to antihypertensive medications, it is important to recognize the hypotensive effects of alpha blocking agents commonly prescribed for benign prostatic hypertrophy as well as psychoactive medications, specifically tricyclic antidepressants and antipsychotics commonly used in patients with PD psychosis (Table 2). Patients with OH with a history of syncope with prolonged alteration of consciousness, myoclonus with syncope (i.e., convulsive syncope), or even, in more severe cases, post-hypoxic brain injury resulting in post-hypoxic myoclonus may present for evaluation after initiation of anticonvulsant drugs. A careful review of the underlying indication of such medications is an important step toward limiting polypharmacy, particularly in older patients.
NMDA, N-methyl-D-aspartate.
While dopaminergic medications can also contribute to OH, these medications may be needed to maintain mobility and independence. As such, adjustments should only be made after all other nonpharmacological and first-line pharmacologic approaches have been undertaken. If needed, we suggest first removing dopamine agonists with high affinity for the dopamine type-1 (D1) receptor (apomorphine, rotigotine) due to their intrinsic vasodilatory effects. After this, consider discontinuing adjunctive therapies (catechol-O-methyltransferase inhibitors: entacapone, tolcapone, opicapone), monoamine oxidase B inhibitors (rasagiline, selegiline, safinamide), amantadine, trihexyphenidyl, istradefylline, followed by non-ergot dopamine agonists (pramipexole, ropinirole), and lastly carbidopa-levodopa.
Nonpharmacological treatments of OH
PwPD and OH should be counseled about nonpharmacologic therapies to minimize BP drops, even early in the symptomatic course while an evaluation for secondary causes is being pursued. Important OH lifestyle modifications include increasing daily water intake, supplementing daily salt intake, avoiding potential triggers, changing behaviors to rise more slowly from a lying or seated position, learning and employing counter maneuvers, and considering compression garments. Physical exercise, as tolerated, may also help to improve muscle bulk, euglycemia, and cardiac output—all of which may improve the trajectory OH symptoms in PD.
The amount of water intake needed to replete daily losses is 64 ounces (oz; 2 L), so experts recommend patients with OH consume over 100 oz (3 L) per day. 36 This recommendation may be challenging for patients to operationalize. Drinking a bolus of water (16 oz in 5 min or less) can increase SBP by 30 mmHg within 5 min and last up to 1 h for patients with PP and post-exercise hypotension.36,37,39
Additionally, experts recommend increasing salt by 1–2 teaspoons (2.3–4.6 g) per day. 36 Aggressive intake of dietary salt can obviate the need for certain pharmacological approaches, namely fludrocortisone. The risks of worsening heart failure, supine hypertension, and peripheral edema should be carefully weighed against the potential benefits.
Patients with OH should also be encouraged to eat small, frequent meals to avoid blood pooling in the splanchnic vessels after eating, avoid alcohol as this can inhibit peripheral vasoconstriction, and avoid hot tub baths, showers, saunas, or excessive high-intensity exercise as this leads to peripheral vasodilation and possible dehydration exacerbating symptoms. 40
Physical countermeasures (bending forward, squatting, abdominal compression, leg crossing when standing) may improve OH by stabilizing BP through increasing peripheral vasoconstriction in patients with autonomic dysfunction and/or failure.40,41 For similar reasons, daily exercise focusing on lower body strengthening should be encouraged. Compression garments, specifically abdominal binders (exerting 20–40 mmHg) provide additional benefit by reducing venous pooling in the splanchnic vessels. Compression stockings (knee or thigh high) have less evidence 40 but are recommended by expert consensus. 36
Small studies suggest that tilting the head of the bed up 12° at night in combination with fludrocortisone will decrease nocturnal fluid and sodium excretion which can exacerbate morning OH.42,43 These studies were completed in patients with neurogenic OH, and not PD specifically, but we expect that this intervention would be effective for PwPD. Other small studies showed head-up tilt sleeping without the addition of fludrocortisone was also beneficial, 44 and the Heads-Up Trial is assessing this intervention in PwPD and parkinsonism. 45 For the combination of systolic hypertension and PD OH, a pilot trial of a topical heating pad placed under the torso overnight yielded a reduction in nocturnal hypertension and a decrease in urinary volume. 46 These changes are most likely due to cutaneous and/or splanchnic vasodilation in response to heat which then secondarily influences cardiac output.
Pharmacological treatments of OH
Patients with PD have peripheral noradrenergic degeneration and low plasma norepinephrine, so drugs that mimic elements of sympathetic tone—midodrine and droxidopa—are the most effective (Table 3).
Pharmacological therapies for OH in Parkinson’s disease.

PwPD, people with Parkinson’s disease.
Midodrine is a prodrug metabolized to a peripherally acting alpha-1 agonist which increases peripheral vascular resistance to decrease venous pooling in the legs and splanchnic vessels. While this drug has not been tested in PwPD, there are three RCTs documenting improvement in orthostatic BPs in patients with autonomic failure47–49 and improved orthostatic tolerance. 50
Droxidopa is a synthetic prodrug of norepinephrine that is converted to norepinephrine by aromatic L-amino acid decarboxylase, leading to vasoconstriction of peripheral vasculature. Four short RCTs were completed with mixed results,51–56 but suggest improvement in symptoms, ability to complete ADLs, and possibly decrease falls. There is only one long-term safety study that was limited by a high drop-out rate, 57 so this medication is only FDA approved for short-term use in PD patients with OH. Droxidopa has been reported to have less risk of causing supine hypertension compared to midodrine and fludrocortisone, but it should still not be given within 3–5 hours of lying supine. In addition, this medication was not tested in combination with midodrine, so close monitoring is recommended if using these drugs in combination.
Fludrocortisone is a synthetic mineralocorticoid that increases intravascular volume through increasing water and sodium reabsorption at the kidney. Evidence is weak for benefit in PwPD,58,59 but there is documented benefit in other populations and could be considered as an add-on therapy to midodrine. This medication is typically given at lower doses which allow for sensitization of alpha receptors and improve response to midodrine; 60 higher doses may increase risk of hypothalamic-pituitary-adrenal axis suppression with minimal additional benefit for OH so should be avoided.
Pyridostigmine is a cholinesterase inhibitor that prevents metabolism of acetylcholine. Atomoxetine is a norepinephrine reuptake inhibitor. Both medications can have a pressor-like effect in patients with intact peripheral sympathetic fibers.61–63 A post hoc analysis of a large 24-week rivastigmine PD dementia trial showed that compared to placebo, rivastigmine (a cholinesterase inhibitor) results in less OH and modestly improved cognitive outcomes as well, although the underlying trial may not have been adequately powered to detect a treatment difference in these outcomes. 64
Finally, a trial reduction in levodopa or other dopaminergic drugs should be considered in PwPD presenting with disabling orthostatic symptoms. Such reductions can run the risk of exacerbating motor impairments but in our experience, patients are often willing to take this trade-off if they experience a corresponding improvement in bothersome OH symptoms.
We recommend using midodrine as the first-line therapy for treatment of OH in PwPD based upon the strength of evidence (Figure 1). After maximizing the dose, if symptoms persist and supine hypertension is not limiting then add low-dose fludrocortisone to enhance the effect. If not tolerated or symptoms remain problematic, then we would recommend adding droxidopa. Midodrine can be continued or gradually tapered off in this setting, depending on the strength of clinical response to the droxidopa addition.

Management approach for OH in Parkinson’s disease.
Special considerations
For patients with PP who have failed the above therapies, octreotide could be considered. This medication is a somatostatin analog that blocks the release of gastrointestinal vasodilating peptides, leading to splanchnic vasoconstriction.65,66 Acarbose, an anti-diabetic medication that slows glucose absorption, may also improve PP. 67 Additionally, patients could consider increasing caffeine intake,68,69 and should be counseled to eat small frequent meals and avoid simple carbohydrates.
As patients with PD develop significant supine hypertension, due to neurogenic OH or as the result of pharmacological therapies, it is important to remember the goal of treatment is to maintain standing BP at levels that will prevent falls. This short-term benefit commonly outweighs the long-term risk reduction of cardiovascular complications from treatment of hypertension. In PD OH patients with nocturnal supine hypertension and a particularly high risk for cardiovascular morbidity and mortality due to stroke or heart disease, nocturnal dosing of a short-acting antihypertensive medication could be considered. Consensus guidelines recommend the following medications and doses: Captopril 25 mg at bedtime, clonidine 0.2 mg with evening meal, hydralazine 10–25 mg at bedtime, losartan 50 mg at bedtime, and nitroglycerine patch 0.1 mg at bedtime. 36 Monitoring of nocturnal supine hypertension is an important management step to identifying the indications and potential efficacy of any of these specific medication changes.
Conclusion
OH treatments in PD touch many different therapeutic targets including those related to vascular physiology and neuroendocrinologic factors. A care coordination discussion between PD specialists, patients, care partners, and/or other clinicians is often needed to optimize the sequence of OH treatment preferences in PwPD, particularly in those with coexisting comorbidities.
Footnotes
Appendix
Acknowledgements
None.
Declarations
Disclosures
K. J. W.: Dr Wyant receives royalties from UpToDate and MedMastery, research funding from the Michael J. Fox Foundation and PhotoPharmics. V. K.: Dr Kotagal receives payment and a travel stipend from the Movement Disorders Society for his role as an Associate Editor on the journal Movement Disorders, research grant funding from the US National Institutes of Health (NIH) and the US Department of Veterans Affairs (VA), and funding from the Lewy Body Dementia Association.