Abstract
Background:
Cladribine (CLAD), an immune reconstitution therapy for active multiple sclerosis (MS), can reduce intrathecal antibody production.
Objectives:
In this study, we investigated the long-term impact of oral CLAD on protective antibody levels, essential for preventing infections and immune defense.
Design:
Observational long-term study including a cohort of 15 CLAD-treated MS patients.
Methods:
We longitudinally studied the humoral immunity to seven common pathogens (measles, mumps, varicella-zoster virus, diphtheria and tetanus toxin, rubella, hepatitis B virus (HBV)) and absolute immunoglobulin G (IgG) levels prior to CLAD treatment (baseline, BL; 12/2017–03/2020) and after an average of 73 months (long-term) follow-up to explore the impact on pre-existing IgG. At long-term, we assessed IgG response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to evaluate potential inhibitory effects on the formation of new immunity.
Results:
We found no CLAD associated loss of humoral immunity over up to 7 years. Pathogen-specific IgG antibodies were present in 60%–100% and 67%–100% of patients at BL and long-term, respectively. We found no decline in absolute IgG levels 73 months after starting CLAD treatment. Patients who received subsequent anti-CD20 treatment had significantly lower SARS-CoV-2 antibody levels (p = 0.011) compared to the rest of the cohort, which developed adequate anti-SARS-CoV-2 IgG. One patient had a clinically silent tick-borne encephalitis (TBE) infection mounting appropriate IgG and IgM. No severe COVID-19 cases occurred, and no new safety concerns were identified.
Conclusion:
These long-term data suggest that CLAD treatment does not impact preexisting humoral immunity or antibody production toward novel antigens. Our results support the positive long-term safety profile of the drug.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic, immune-mediated disease affecting approximately 2.8 million people worldwide. 1 The primary therapeutic approach involves immunotherapies, which aim to prevent inflammation in the central nervous system (CNS) and reduce frequency and severity of MS relapses. Due to the chronic nature of the disease and its early onset, MS patients are treated with disease-modifying therapies (DMTs) for many years, and even decades.
Cladribine (CLAD) is an oral immune reconstitution therapy (IRT) used to treat active MS. 2 It features a unique short-course dosing regimen, administered in two cycles approximately 1 year apart, leading to long-lasting efficacy in the majority of MS patients.2–5 CLAD is synthesized as a purine nucleoside analog, allowing it to cross the blood–brain barrier and target both peripheral and central inflammatory mechanisms. 6 CLAD modifies the peripheral immune system by reducing lymphocytes, particularly B and T cells up to 80% and 50%, respectively, thereby remodulating the detrimental proinflammatory regulation in MS.7,8 Following IRT with CLAD, repopulation rates vary among different B cell subsets. While absolute B cell counts largely recover by the end of the first and second treatment years, the memory B cell compartment remains markedly depleted, which may contribute to the sustained efficacy of CLAD.9–12 The profound impact on B cell lymphocytes, accompanied by reorganization of the B cell transcriptome, 13 may have long-term effects on the number and functionality of terminally differentiated, antibody-producing plasma cells.12,14 Indeed, reductions in cerebrospinal fluid (CSF)-specific oligoclonal bands (OCBs), a hallmark of MS, 15 have been reported during a 10-year treatment period with CLAD. 16 OCBs are isoelectrically focused immunoglobulin G (IgG) antibodies reflecting the polyspecific humoral immune response within the CNS. 17 Although the disappearance of OCBs may represent a favorable therapeutic outcome in MS, it raises the question of whether CLAD—given its higher concentrations in blood compared to CSF 6 —might also compromise peripheral humoral immunity in the long term. Impaired production of pathogen-specific antibodies could reduce the immune system’s capacity to respond effectively to infections and pose potential safety concerns.
While the effects of CLAD on cellular immunity have been extensively studied, its impact on humoral immune responses remains less explored.7,10,18 No decline in preexisting humoral immunity against frequent pathogens was identified after 24 months of CLAD treatment initiation. 19 Given that plasma cells have a lifespan of several years, potential long-term effects on humoral immune memory may however become evident only with extended follow-up periods.
We conducted a longitudinal study to explore whether CLAD induces long-term changes in the humoral immunologic memory. Serum antibodies against common pathogens were monitored both before CLAD start and after up to 7 years of follow-up. Additionally, we evaluated the impact of CLAD on the ability to generate novel humoral immune responses.
Methods
Study design and patient recruitment
This observational study included 15 patients with active relapsing MS who received their first CLAD cycle between December 2017 and March 2020 at the outpatient MS clinic of the Christian Doppler University Hospital in Salzburg, Austria.
BL demographic and clinical data, including age, sex, MS disease duration, previous DMTs, and Expanded Disability Status Scale (EDSS) score, were collected before treatment initiation.
All participants were regularly screened for signs of (opportunistic) infections, clinical deterioration, and MS relapses. Patients were recommended to perform annual MRI scans of brain and cervical spinal cord. Clinical data were collected through March 2025.
Oral CLAD was administered following the approved dosing regimen according to the manufacturer’s guidelines, consisting of CLAD tablets with a cumulative dose of 3.5 mg/kg body weight over 2 years, administered as 1.75 mg/kg/year. Each year’s treatment course is divided into two treatment weeks, one at the beginning of the first month and one at the beginning of the second month.
Patients were eligible for inclusion if they had a diagnosis of active relapsing MS based on the 2017 McDonald criteria, 15 were aged between 18 and 60 years, and treated with CLAD as part of routine clinical care at the Christian Doppler University Hospital between December 2017 and March 2020. Inclusion was further limited to individuals who had completed at least one treatment cycle of CLAD and for whom serum samples were available for both baseline and long-term follow-up antibody analysis. Patients were excluded if they had any concomitant autoimmune or immunodeficiency disorders, active infections at BL, or incomplete clinical or insufficient laboratory follow-up data. Furthermore, individuals with less than 24 months of follow-up were not considered eligible for the study.
Sample collection and antibody measurements
The study protocol involved collecting peripheral blood samples into 10 mL serum tubes (Becton Dickinson, Franklin Lakes, NJ, USA) from each patient at least two time points: At the initiation of CLAD treatment (BL) and during the last follow-up visit (long-term follow-up, LT). The blood collections at BL were conducted from December 2017 until July 2020. The final blood assessments (LT) were conducted between November 2024 and February 2025. Serum samples for LT analyses of immunoglobulins were sent to the in-house laboratory for immediate processing. For BL assessments, measurements were either performed immediately or, if unavailable, analyzed retrospectively using stored serum samples collected during the BL visits. The serum tube at BL was centrifuged for 10 min with 3000g at room temperature. Subsequently, serum aliquots were stored at −80°C for later batch processing. Antibody levels against measles, mumps, varicella-zoster virus (VZV), diphtheria toxin, tetanus toxin, rubella, and hepatitis B virus (HBV) were longitudinally measured. Additionally, we assessed levels of IgG antibodies against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2, anti-Spike IgG). For SARS-CoV-2, we used the cut-off of 264 binding antibody units/milliliter (BAU/mL), which, according to the literature, correlates with approximately 80% immunity against coronavirus disease 2019 (COVID-19). 20
Due to changes in laboratory measurement methods and/or reference values over the follow-up period, we focused on whether patients retained or lacked humoral immunity against these seven specific pathogens, and did not assess changes in pathogen-specific antibody levels. Enzyme-linked immunosorbent assay or chemiluminescent microparticle immunoassay techniques were used to measure antibody titers. The respective cut-off criteria for antibody titers were applied. Detailed information on instruments, assays, and reference values is provided in Supplemental Table 1. Total IgG concentrations were quantified using kinetic nephelometry on a Siemens BNII system (Siemens Healthineers, Erlangen, Germany) with corresponding reagents (N Antiserum against IgG; Siemens Healthineers). All laboratory analyses were performed using standard routine methods at the Department of Laboratory Medicine, which is accredited to ISO-9001 and ISO-15189 standards.
EDTA whole blood samples were analyzed immediately after collection. Absolute lymphocyte counts were determined using the Sysmex XN-Series hematology analyzer (Sysmex Corporation, Kobe, Hyogo Prefecture, Japan). For lymphocyte subset analysis, flow cytometry was performed to quantify T helper cells (Th, CD3+ CD4+), suppressor T cells (CD3+ CD8+), and B cells (CD19+) using the BD FACSLyric™ flow cytometer (Becton, Dickinson and Company).
Statistical analysis
Descriptive statistics were used to summarize demographic and clinical characteristics. Data are presented as mean ± standard deviation (SD) or median with interquartile range (IQR), unless otherwise specified. For IgG antibody counts, the Shapiro–Wilk test was used to assess normal distribution. As the data passed the normality test, paired t tests were subsequently used to determine statistical significance between BL and LT data. All reported tests were two-sided, and p-values <0.05 were considered statistically significant. To analyze changes in IgG antibody counts among the five patients who received additional anti-CD20 depleting therapies, a repeated measures one-way analysis of variance with Geisser–Greenhouse correction and Tukey’s multiple comparisons test was performed. To determine statistical significance in SARS-CoV-2 titers between patients treated with and without subsequent anti-CD20 depleting therapies, an unpaired t test with unequal variance was used. No imputation for missing data was performed. Statistical analysis was performed using GraphPad Prism (V10.2.3; GraphPad Software Inc., San Diego, CA, USA). A multivariable generalized binomial model with loss of humoral immunity as dependent variable and various baseline characteristics as predictors was conducted. Additionally, a backward variable selection algorithm was applied.
Due to the exploratory and observational nature of this long-term follow-up study, a formal sample size calculation was not conducted. The study cohort consisted of all eligible individuals who met the inclusion criteria and had complete longitudinal data available. The statistical analyses applied in this study were designed to detect robust effects on humoral immunity over time, while acknowledging the limitations imposed by the cohort size.
This study was conducted following the STROBE guidelines for reporting observational studies.
Results
Patients’ characteristics and disease activity
At the time of CLAD start (BL), the 15 patients included in this analysis had a mean age of 34 years (SD ±9), a median disease duration of 6 years (1–13 IQR), and a median EDSS score of 1.5 (IQR 0.0–2.5). The average follow-up period after CLAD initiation was 73 months, with a range from 58 to 84 months. The demographic characteristics of the cohort at the time of their final blood collection (LT) are summarized in Table 1. Twelve individuals (80%) had received DMTs prior to initiating CLAD, with an average interval of 1 month (range: 0–4 months) between discontinuation of the last DMT and the start of CLAD treatment. Overall, 12 patients underwent 2 cycles of CLAD, while 1 patient received only 1 cycle, and 2 patients completed 3 cycles of treatment. Five patients (33%) required subsequent B-cell-depleting therapies due to clinical or radiological disease reactivation. Of these, two patients were treated with ocrelizumab, two patients received ofatumumab, and one additional patient was switched from ocrelizumab to ofatumumab. Another patient started a siponimod treatment. The mean time from the first CLAD cycle to the start of subsequent anti-CD20 depleting therapy was 41 months (SD ±20). The duration from anti-CD20 therapy initiation to the LT follow-up blood draw averaged 34 months (SD ±18). Lymphocyte counts are presented in Supplemental Figure.
Characteristics of the study population at the time of the final blood collection for long-term assessment.
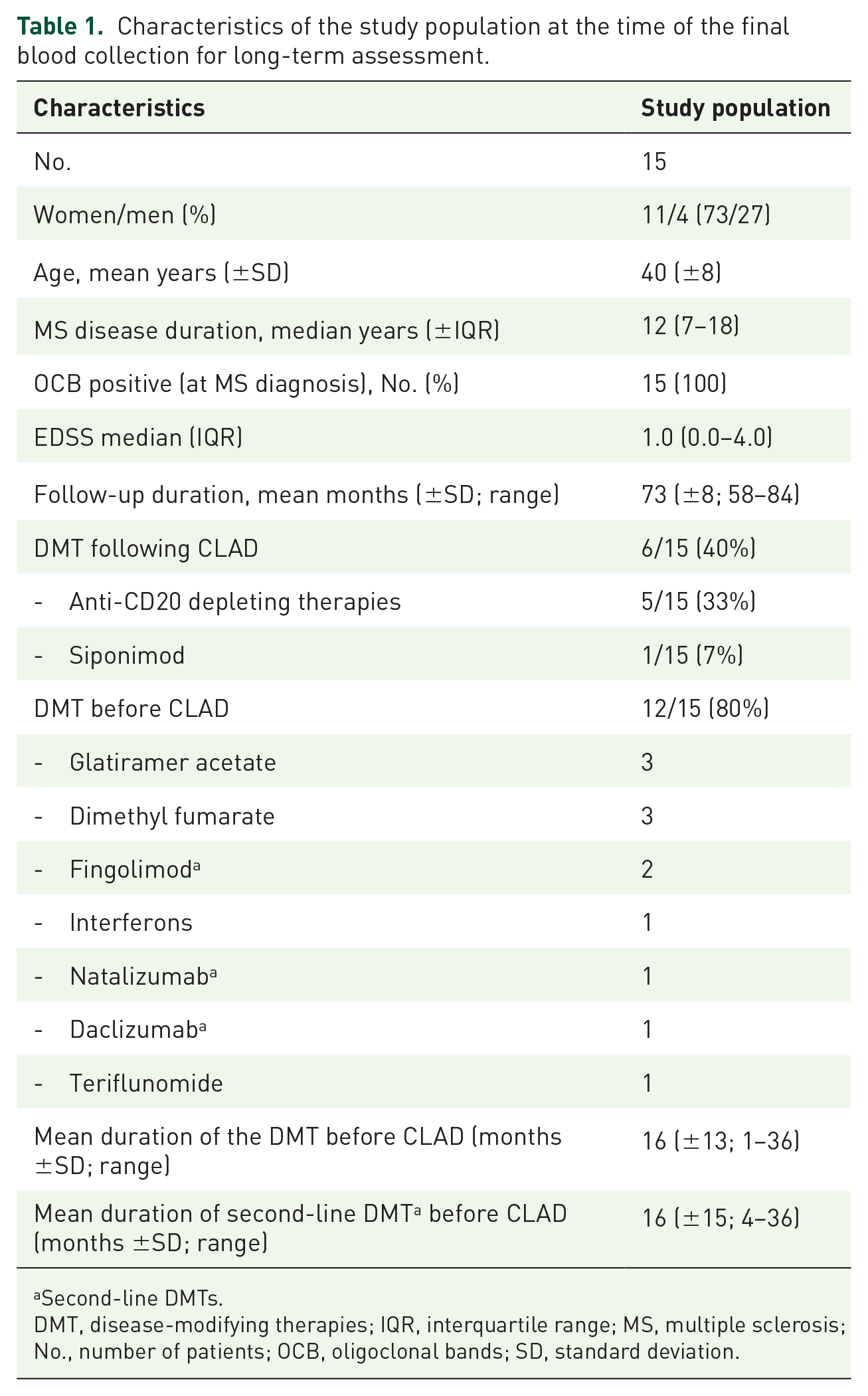
Second-line DMTs.
DMT, disease-modifying therapies; IQR, interquartile range; MS, multiple sclerosis; No., number of patients; OCB, oligoclonal bands; SD, standard deviation.
During the follow-up period, four patients (27%) experienced clinical relapses. Three patients had a single relapse, while one patient had three relapses. New lesions on MRI were detected in eight patients (53%) during the entire follow-up period. No relapse and new lesions on MRI were found in 6/15 (40%) patients throughout the LT follow-up.
Long-term impact of CLAD on preexisting pathogen-specific antibodies
During the follow-up period, four patients (27%) received a tick-borne encephalitis (TBE) vaccination, two (13%) were vaccinated against HBV and influenza, and one patient received a combined tetanus–diphtheria vaccine as well as a VZV vaccine.
Compared to the BL immunity status, we observed no relevant impact of CLAD on pathogen-specific humoral immunity at 73 months posttreatment initiation (Figure 1(a)). Among the seven different pathogen-specific antibodies analyzed in our cohort (n = 15), a total of six immunities were lost in five individuals (n = 2 against measles, n = 2 against mumps, n = 1 against rubella, and n = 1 against HBV). In one patient, humoral immunity against HBV was lost 2 months after initiation of CLAD therapy; this individual had a BL anti-HBs titer of 114.7 U/L, only marginally above the threshold of 100 U/L. In the remaining four patients, protective pathogen-specific antibody levels were maintained at their last intermediate IgG assessments prior to LT follow-up, conducted at 5, 20, 23, and 23 months after CLAD initiation, respectively. Thus, any loss of immunity has occurred between these time points and the final follow-up.

(a) Preexisting pathogen-specific IgG immunity to seven common pathogens was not altered by CLAD treatment after LT (mean 73 months) surveillance. (b) Pathogen-specific IgG levels to SARS-CoV-2 (anti-Spike IgG) assessed 73 months after CLAD initiation. Cohort divided into patients with (CLAD → subsequent anti-CD20 therapy; green dots) and without (CLAD therapy; blue dots) subsequent anti-CD20 treatment. Individuals with subsequent anti-CD20 depleting therapy (n = 5) mounted significantly fewer antibodies to SARS-CoV-2 (anti-Spike IgG). Dashed lines indicate cut-off values. Error bars represent 95% confidence intervals. (c) Absolute IgG blood levels remained stable despite immune reconstitution by CLAD. (d) Absolute IgG blood levels of five patients with consecutive anti-CD20 B cell depleting therapy remained stable during the follow-up period. Gray dotted lines represent the normal range of IgG levels. Error bars represent 95% confidence intervals with n = 15, for VZV n = 14.
On the other hand, five immunities were gained in five individuals (n = 2 against HBV, n = 1 against mumps, n = 1 against diphtheria, n = 1 against tetanus) during follow-up.
The proportion of patients with pathogen-specific immunity at BL varied by antigen, ranging from 60% to 100%. Immunity was universal (100%) for measles and VZV, whereas 60% of patients were immune to HBV. HBV also had the lowest (67%) immunity rate after LT follow-up, with immunity levels across all pathogens ranging from 67% to 100%. Complete immunity (100%) was observed against tetanus toxin (n = 15) and VZV (n = 14).
Two patients in our cohort were treated exclusively with CLAD, with no prior or subsequent DMT exposure. In one of these individuals, humoral immunity to mumps was lost between 5 months after CLAD initiation and LT follow-up. No other changes in pathogen-specific humoral immunity were observed in association with CLAD treatment among these two patients. We applied a multivariable generalized binomial model to identify potential predictors of long-term loss of humoral immunity. The variables included age (p = 0.98), gender (p = 0.96), use (p = 0.93) and number of DMTs prior to CLAD initiation (p = 0.93), IgG levels at BL (p = 0.92), EDSS at baseline (p = 0.95) and at long-term follow-up (p = 0.93), disease duration at BL (p = 0.94), and absolute B cell counts at BL (p = 0.93), at the end of year 1 (p = 0.92), and year 2 (p = 0.96). Additionally, the use of second-line DMTs before (p = 0.90) and after CLAD initiation (p = 0.91), as well as administration of a third CLAD cycle (p = 0.93), were not predictive. No significant associations were identified, even after applying a backward variable selection algorithm.
Impact of CLAD on antibody production to novel antigens
We assessed the SARS-CoV-2 antibodies at LT to investigate whether the formation of a humoral immunity against novel antigens was impaired following immune reconstitution with CLAD treatment. Since the individuals from our cohort had undergone CLAD treatment before the COVID-19 was declared as pandemic, their first exposure to SARS-CoV-2 antigens occurred only after immune reconstitution with CLAD. Among the 14 patients studied, 8 (57%) had been vaccinated against SARS-CoV-2, and 10 (71%) reported at least 1 COVID-19 infection. Overall, two patients neither reported vaccination nor infection.
At LT, 12 out of 14 patients (86%) tested positive for SARS-CoV-2 IgG antibodies (Figure 1(b)). Of the two patients without detectable antibodies, one had a history of COVID-19 infection but was unvaccinated, while the other had been vaccinated but reported no infection. Both patients had received anti-CD20 treatment after CLAD. Overall, patients with subsequent anti-CD20 treatment had significantly lower SARS-CoV-2 antibodies (p = 0.011) as compared to the remaining CLAD cohort.
Impact of CLAD on overall IgG levels
The absolute serum IgG levels at CLAD start were within the normal range (700–1600 mg/dL) in 12/15 patients (80%), while three individuals (20%) had reduced IgG levels (Figure 1(b)). We observed no significant reductions in absolute blood IgG levels associated with CLAD treatment during the follow-up period of up to 7 years (p = 0.1; Figure 1(c)). At LT, IgG levels remained reduced in two (13%) patients, while the remaining had either normal (80%) or increased (7%) IgG levels. We further analyzed IgG levels in the subgroup of five patients who received consecutive anti-CD20 B-cell depleting therapies. Even among these five patients, no significant reductions in absolute blood IgG levels were observed (p = 0.47; Figure 1(d)).
Infections and side effects
During the long-term follow-up after CLAD treatment, three patients (20%) reported bacterial infections and nine (60%) viral infections. Of the patients with bacterial adverse events, one had repeated episodes of bacterial pharyngitis and otitis media, none of which required hospitalization. Another patient (women with EDSS 6.0) suffered from recurrent urinary tract infections and was hospitalized for cholecystitis. The third patient experienced sinusitis. One individual suffered from recurrent herpes labialis. Of the eight patients with COVID-19, five experienced a single infection and three reported two episodes. However, none required hospitalization, and no severe COVID-19 courses occurred.
One patient (a 41-year-old woman, EDSS: 0, CLAD-only therapy) had a laboratory-confirmed TBE infection at the final blood assessment in December 2024. This patient mounted appropriate IgG and IgM antibodies. She remained asymptomatic throughout the last clinical follow-up in March 2025, indicating a clinically silent, incidental TBE infection. Beyond the reported infections, no moderate or severe side effects were observed during long-term follow-up.
Discussion
CLAD treatment was not associated with a loss of pathogen-specific humoral immunity in our long-term study. Over a follow-up period extending up to 7 years, the proportion of patients with protective systemic antibodies against seven common pathogens remained stable compared to pre-treatment levels, suggesting that CLAD does not significantly compromise preexisting protective IgG responses. A preserved capacity to protect from infections despite IRT with CLAD is supported by the clinical safety outcomes from pivotal trials and their extensions.21,22 In contrast to our findings regarding pathogen-specific antibodies in the peripheral blood, previous studies have reported effects of CLAD on intrathecal antibody production, with reductions in kappa free light chains and IgG index in CSF observed as early as 48 weeks. 14 Moreover, OCBs decreased after 10 years. 16 A potential explanation for its selectivity could involve CLAD-induced reductions in intrathecal C-X-C motif chemokine 13 (CXCL-13), a key B cell chemoattractant. 14 Additionally, plasma cells responsible for peripheral antibody production primarily reside in the bone marrow and may be more resistant to CLAD’s cytotoxic effects compared to their counterparts in the CNS. In fact, analyses of peripheral blood from MS patients showed minimal impact of CLAD treatment on antibody-producing cells.12,14
In our cohort, approximately half of the patients required no further treatment beyond the initial two cycles of CLAD throughout the 7-year follow-up. Moreover, the treatment was associated with a favorable long-term safety profile. No severe COVID-19 cases were reported, and a patient diagnosed with TBE remained clinically stable and mounted adequate antibodies, suggesting preserved immune competence.
In line with previous studies,14,23 we observed no significant effects on total peripheral IgG levels over time, even among patients who received consecutive anti-CD20 B cell treatment during the follow-up. This is noteworthy because long-term anti-CD20 B cell depletion is frequently associated with hypogammaglobulinemia, thereby possibly increasing the risk of infectious adverse events. 24 While preexisting humoral immunity remains largely unaffected also under anti-CD20 treatments,25,26 these therapies significantly impair the formation of a humoral immune response to novel antigens, as was evident during the COVID-19 pandemic.25,27 Our findings complement existing data from the literature, 27 showing that this is not the case with CLAD, even in the long-term. We demonstrate that antibody production to novel antigens, whether induced by vaccination or infection, is preserved years after immune reconstitution with CLAD. The patients in our cohort, treated with CLAD prior to the COVID-19 pandemic, mounted substantial antibody responses to SARS-CoV-2. An exception was the subgroup of patients who subsequently received anti-CD20 B cell therapy: among this cohort, antibody production to SARS-CoV-2 was significantly reduced.
The transient nature of CLAD-induced B cell depletion, 12 which differs from the sustained B cell depletion under anti-CD20 therapies, 28 may offer two key advantages: (a) the preserved ability to generate novel pathogen-specific antibodies and (b) maintenance of absolute IgG levels. In contrast, the impairment of humoral immunity associated with anti-CD20 therapies has been linked to the highest infection risk among MS DMTs, 29 as well as an increased likelihood of severe COVID-19 courses 30 —concerns not observed with CLAD. 31
The long-term stability of protective IgG antibodies under CLAD is particularly relevant for women of childbearing age, as neonatal immunity depends on the maternofetal transfer of IgG across the placenta. 32 Moreover, CLAD has been proposed as an exit strategy after high-efficacy DMTs such as natalizumab or fingolimod, often in older patients, to mitigate the risk of rebound disease activity. Given the effects of immunosenescence, our data support CLAD as a viable option even for elderly individuals with MS.
This study has several important limitations. First, the relatively small sample size of 15 patients limits the generalizability of our findings. Second, changes in antibody assessment methods over the study period prevented direct comparisons between BL and LT concentrations. However, antibody titers naturally fluctuate within individuals and decline over time—even in the absence of immunotherapies—making direct comparisons challenging to interpret.33,34 Additionally, the threshold for defining sufficient antibody levels for infection prevention remains uncertain. Therefore, our approach, which focuses on the loss or preservation of humoral immunity, is considered appropriate. An additional consideration is the heterogeneity in prior and subsequent DMT exposure within the cohort, with 40% of patients receiving further immunotherapies during the study follow-up. While this variability complicates attribution of effects solely to CLAD, it mirrors real-world clinical practice. Notably, despite these treatment dynamics, our key finding remains that CLAD-treated patients did not experience a significant loss of humoral immunity to the pathogens investigated, regardless of prior or subsequent DMT use.
Conclusion
Treatment with CLAD does not appear to have a significant impact on protective antibodies, neither in the short-term 19 nor in the long-term. This potential preservation of humoral immunity may contribute to its generally favorable safety profile. Our findings may have practical relevance, particularly for more vulnerable MS patient cohorts, such as the elderly and women considering pregnancy.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251357275 – Supplemental material for Long-term impact of oral cladribine on humoral immunity in multiple sclerosis
Supplemental material, sj-docx-1-tan-10.1177_17562864251357275 for Long-term impact of oral cladribine on humoral immunity in multiple sclerosis by Marc Messner, Michael Unterhofer, Jonas Strauss, Sylvia Mink, Janne Cadamuro, Hannes Oberkofler, Wolfgang Hitzl, Peter Wipfler, Eugen Trinka and Tobias Moser in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
