Abstract
Background:
Migraine is a disabling neurovascular disorder characterized by recurrent attacks that lead to extracranial and visual involvement. Several studies have investigated the retinal vasculature features in individuals with migraine, but there have been conflicting results.
Objective:
To evaluate retinal structure in migraine patients before (T0) and after 6-month therapy (T1) with anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs), using optical coherence tomography (OCT) imaging.
Design:
A case-control and longitudinal study was conducted between January 2021 and December 2023, including 20 eyes from 10 healthy controls (HCs) and 32 eyes of 16 patients with migraine and treated with anti-CGRP mAbs according to AIFA criteria.
Methods:
Patients underwent OCT angiography (OCT-A) to assess retinal vessel density (VD) and spectral-domain OCT (SD-OCT) to evaluate central retinal thickness, macular structure, and peripapillary retinal nerve fiber layer thickness. These parameters were assessed in both groups at T0 and again after 6 months (T1) as part of routine clinical care.
Results:
All migraineurs exhibited a significant reduction in disease disability at T1, as assessed by clinical parameters. OCT data analysis revealed that individuals with migraine showed a significant increase in temporal retinal nerve fiber layer (RNFL) thickness and a reduction in nasal RNFL thickness compared to HCs. No differences in retinal circulation were observed between the groups at baseline. At T1, RNFL thickness remained sustained in the superior temporal sector, while the percentage VD of the superficial capillary plexus and radial peripapillary capillary significantly increased in the nasal perifoveal, inferior temporal, and hemi-inferior subregions.
Conclusion:
Our study suggests that specific retinal structural changes could precede vascular dysfunction in migraine and can be detected early by combining SD-OCT and OCT-A acquisitions. Short-term treatment with anti-CGRP mAbs may exert neuroprotective effects, potentially preventing permanent ocular damage.
Trial registration:
EyeHEAD Study (Trial registration number AIFA July/2024: IT 1735, www.aifa.gov.it/registro-studi-osservazionali).
Plain language summary
Migraine patients show significant quadrant-specific structural RNFL changes compared to healthy controls, in the presence of still-preserved vascular networks; After short-term treatment with anti-CGRP mAbs, vascular perfusion increases in the superficial and radial peripapillary capillary, possibly helping to prevent irreversible ocular injury; OCT imaging may represent a rapid, useful, and non-invasive tool for monitoring migraine course and treatment outcome.
Introduction
Migraine is a global medical condition afflicting millions of people worldwide, and its prevalence is on the rise. 1 The recent Global Burden of Diseases report identified migraine as one of the top 10 primary contributors to nervous system health deterioration, especially among individuals under 50 years old. 2 Headache attacks can last for hours or even days, causing severe pain along with hypersensitivity to light, sound, smell, and touch. Patients often also experience nausea and vomiting, and about a quarter of them report visual disturbances known as an aura. 3
Although its pathophysiology has yet to be fully elucidated, 4 migraine is considered a complex neurovascular disorder with an increased risk of ischemic events that extend beyond the brain. 5 Other intracranial and extracranial vascular complications, such as stroke, angina, and myocardial infarction, are closely associated with long-term migraine. 6 Moreover, perfusion deficits occurring during visual aura or recurrent pain attacks can lead to ischemia and structural damage in the retina and optic disc, making migraineurs more vulnerable to several vascular ocular diseases (i.e., retinal artery occlusion, ischemic optic neuropathy, and glaucoma). 7 Interestingly, the retinal blood supply derives from the internal carotid artery and is regulated by vasoactive neuropeptides, such as calcitonin gene-related peptide (CGRP), which also plays a key role in headache pathogenesis and therapy. 8 Furthermore, CGRP and its receptors are widely expressed in the retinal nerve fiber layers; however, their function within the ocular system has not been fully established. 9
In the past 5 years, there has been a major breakthrough in managing high-frequency episodic migraine (HFEM) and chronic migraine (CM) with the introduction of monoclonal antibodies (mAbs) targeting CGRP (fremanezumab, 10 galcanezumab,11,12 and eptinezumab) or its receptor (erenumab). 13
These new medications have demonstrated high effectiveness in real-life settings, not only by reducing the frequency of pain attacks but also by alleviating other associated bothersome symptoms, such as visual disturbances like photophobia and aura.14–16 However, how these drugs act within the ocular system remains unknown.
Similar to other neurological diseases,17,18 some authors have suggested that the use of noninvasive, rapid spectral-domain optical coherence tomography (SD-OCT) and optical coherence tomography angiography (OCT-A) could serve as a potential clinical biomarker for the early detection and follow-up of retinal neuronal and microcirculatory changes in migraineurs.17,18 Unfortunately, many studies have yielded heterogeneous results, often assessing either retinal structural thickness or the eye’s vascular characteristics in isolation.19–21 Moreover, there is a lack of reports focused on monitoring these ophthalmological disturbances in patients treated with CGRP antagonists.
Hence, our study aimed to assess the retina and optic disc structure, and the microvasculature, in migraine sufferers and healthy controls (HCs), by combining SD-OCT and OCT-A imaging. Additionally, we evaluated these findings after 6 months of treatment with anti-CGRP mAbs to determine whether this new targeted therapy could be useful in reducing or preventing ocular changes in patients with HFEM or CM.
Materials and methods
Study population
This mixed-design study, combining a case-control design at baseline with a longitudinal observational approach, enrolled patients admitted to the Headache Center at Tor Vergata University Hospital of Rome between January 2021 and December 2023. Participants were required to be over 18 years of age and diagnosed with either HFEM or CM, according to the ICHD-3 criteria. 3 The study only included migraine patients who were prescribed specific anti-CGRP medications (erenumab 70–140 mg monthly; galcanezumab 240 mg for the first dose and 120 mg monthly; fremanezumab 225 mg monthly; or eptinezumab 100 mg–300 mg quarterly), and had completed at least 6 months of treatment according to local medical guidelines (AIFA). To ensure that the results were not biased by other treatments, the use of other migraine preventive drugs (e.g., antiseizure medications, tricyclic antidepressants, beta-blockers) was limited.
Healthy volunteers (HCs), matched for age, sex, socioeconomic status, and educational level, and screened for headache symptoms to avoid overlap with the migraineur group, were recruited from hospital staff, friends, and colleagues to serve as the control group.
Exclusion criteria for all participants included: best corrected visual acuity worse than 0.1 LogMAR, refractive error ⩾ ±3 diopters, intraocular pressure > 21 mmHg, previous eye surgery, and major ocular disorders, such as cataracts, ocular surface disease, uveitis, maculopathy or retinopathy, glaucoma, or other optic neuropathies. Additionally, patients with systemic diseases affecting the vascular system, such as diabetes, Sjögren’s syndrome, systemic hypertension, and/or other neurological disorders, were also excluded.
The sample size was calculated based on the existing literature 20 and our prior experience with similar study designs. 22
This study was conducted in accordance with the STROBE guidelines (Supplemental Material) for observational studies, 23 and the Helsinki Declaration. All subjects signed written consent before inclusion.
All the following data and examinations were collected as part of routine clinical care.
Neurological examination
At baseline (T0), a headache specialist neurologist gathered a detailed medical history from each patient through an in-person interview, using semi-structured scales. This included questions about sociodemographic factors, clinical migraine features and associated symptoms, previous and current migraine therapies, other concomitant medications, and any other health conditions.
During the course of the mAb treatment, patients were required to keep a monthly paper headache diary, recording the number of monthly migraine days (MMDs), the number of monthly headache days (MHDs), and the total number of painkillers used per month (MPNs).
To evaluate the level of migraine burden, patients were also asked to complete the Migraine Disability Assessment (MIDAS) Scale 24 and the Headache Impact Test-6 (HIT-6) Questionnaire 25 on a quarterly basis, as recommended in clinical practice.
Adverse events were reported for all participants.
Ophthalmological evaluation
At baseline (T0) and after 6 months (T1), all HCs and migraine patients underwent a complete ophthalmological examination, including visual acuity assessment with ETDRS charts, Goldmann applanation tonometry, and slit lamp biomicroscopy of the anterior and posterior segments after instillation of 1% tropicamide for pharmacological mydriasis, according to routine clinical care. For migraineurs, these ocular tests were performed during an attack-free period, with at least 3 days from the last pain episode or acute medication intake.
Retinal morphology was assessed using SD-OCT (Spectralis; Heidelberg Engineering, Heidelberg, Germany) with the “Posterior Pole-H” (horizontal) program of the Glaucoma Premium Module for the central retina and “ONH” scan for the optic nerve head, as part of routine clinical practice. 26 Automated segmentation was used to obtain the ganglion cell layer and peripapillary retinal nerve fiber layer (pRNFL; 3.5 mm) thicknesses (Figure 1).

Spectral-domain OCT representing total retinal thickness in the macular and ETDRS area. On the right side, thickness changes between two scans at different time points are shown.
Retinal and peripapillary vasculatures were evaluated using OCT-A (RTVue XR Avanti with AngioVue (Optovue, Inc., Fremont, CA, USA) with 6 × 6 mm2 HD Angio Retina scans (Figures 2-3). Specifically, the percentage vessel density (VD) of the retinal superficial capillary plexus (SCP) and deep capillary plexus (DCP), as well as the size of the foveal avascular zone (FAZ), were evaluated and measured. HD Angio disc 4.5 scans were performed to evaluate optic disc vascularization (Figure 4).

OCT-angiography scan representing vessel density in the superficial and deep retinal vascular plexi (a). Retinal flow is superimposed in red on the axial scans (b). Colorimetric maps of vessel density and retinal thickness in the ETDRS area (c).

En face representation of the superficial retinal vessel density in the ETDRS area (a). Confocal scanning laser ophthalmoscopy of the ETDRS area (b). Vessel density and OCT Thickness values of the different ETDRS sectors (c). Retinal flow is superimposed in red on the axial scans (d). Colorimetric maps of vessel density and retinal thickness in the ETDRS area (e).

Confocal scanning laser ophthalmoscopy of the optic nerve head and peripapillary area (a). Vessel density of the radial peripapillary capillary plexus (b). Colorimetric maps of optic nerve head and peripapillary area thickness (c) and vessel density (d).
Statistical analysis
All data were initially entered into an Excel spreadsheet (Microsoft, Redmond, WA, USA), and subsequently analyzed using Graphpad Prism, version 8.01 (Boston, MA, USA). After confirmation through histograms and the Kolmogorov–Smirnov test, descriptive statistical analysis was performed, with normally distributed parameters expressed as the mean ± the standard deviation and non-Gaussian parameters expressed as the median and range (minimum; maximum).
A two-sample t test was used to compare clinical and optical coherence tomography (OCT) parameters between migraine patients and HCs at T0 and T1. A paired t test was used to compare the mean change in clinical and OCT parameters between T0 and T1 within the migraine and the HCs group. Subsequently, a repeated-measures ANOVA was performed within the migraine group, with a decrease in MMDs as a covariate, specifically for those OCT parameters that showed significant changes in the paired t test analysis. This approach allowed us to investigate the effect of changes in MMDs on the changes in OCT parameters over time. A p-value of <0.05 was considered statistically significant.
All graphs were created using Excel (Microsoft).
Results
Cohort features
Overall, 38 patients were treated with anti-CGRP mAbs between January 2021 and December 2023 (those with the potential for a 12-month follow-up). Ten patients were excluded from the study either because they were lost to follow-up or did not receive treatment for at least 6 months. An additional 12 patients were excluded due to incomplete data regarding ophthalmological assessments or the presence of concomitant ocular pathologies.
The final sample included 32 eyes of 16 subjects with migraine (mean age: 46.1 ± 8.6 years, M/F = 2/14) and 20 eyes of 10 age- and sex-matched HCs (mean age: 49.9 ± 8.5 years, p = 0.270; M/F = 3/7, p = 0.271). Among the 16 migraine patients included in the study, 9 had CM (56.2%) and 3 experienced visual aura at baseline (18.7%).
Migraine features
Table 1 presents the main demographic and clinical features of each migraine patient at baseline (T0) and after 6 months of anti-CGRP mAb treatment (T1).
Migraineurs’ demographic and clinical features at baseline (T0) and after 6 months of treatment with anti-CGRP mAbs (T1).

Psychiatric comorbidities.
Gastrointestinal comorbidities.
Endocrine comorbidities.
CGRP, calcitonin gene-related peptide; E, erenumab; F, female; Fr, fremanezumab; G, galcanezumab; HIT-6, Headache Impact Test-6; M, male; MA, migraine with aura; mAb, monoclonal antibody; MHDs, monthly headache days; MIDAS, Migraine Disability Assessment; MMDs, monthly migraine days; MPNs, number of painkillers used per month; MwA, migraine without aura; N, no; NA, not available; SD, standard deviation; T0, baseline; T1, evaluation performed at 6 months; Y, yes.
Regarding the specific anti-CGRP mAb, 50% of patients were taking fremanezumab, 25% were taking galcanezumab, and 25% were on erenumab. None of the migraine sufferers received eptinezumab, as it was not available in Italy during the patient enrollment period.
At baseline, all migraine patients exhibited a high disease burden (MMDs: 15.6 ± 3.8, MHDs: 2.3 ± 1.9, MPNs: 17 ± 7.8, HIT-6: 68.6 ± 3.9, and MIDAS score: 65.6 ± 49.6). After 6 months of anti-CGRP mAbs, all these parameters showed a significant decrease (p < 0.0001, p = 0.002, p < 0.0001, p < 0.0001, and p < 0.0001, respectively) (see Table 1).
Ophthalmological features
Regarding retinal structural morphology, SD-OCT revealed that migraine patients had a significantly increased thickness of the pRNFL in the temporal area compared to HCs (p = 0.04). On the contrary, the thickness of the pRNFL in its nasal area was significantly lower in the former group (p = 0.01). Both of these parameters tended to increase in migraine patients after treatment with anti-CGRP mAbs, reaching statistical significance only in the superior temporal quadrant (p = 0.02). GLC thickness was similar across all groups. No differences in key ophthalmological parameters were found between T0 and T1 in the HCs group.
Table 2 summarizes the main SD-OCT features in HCs and in migraine patients at both T0 and T1.
Comparison of interictal structural retinal parameters across groups by using statistical analysis.
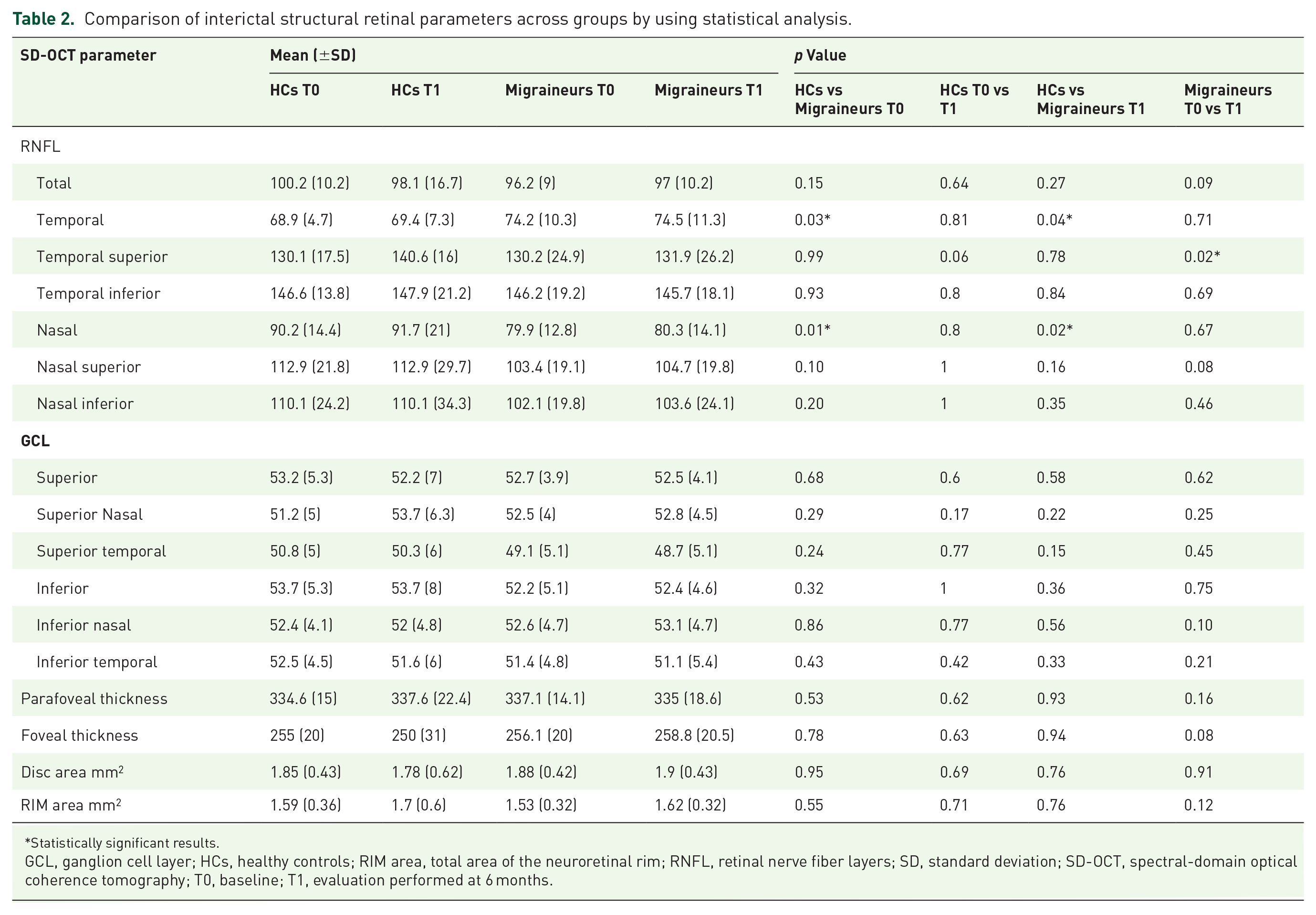
Statistically significant results.
GCL, ganglion cell layer; HCs, healthy controls; RIM area, total area of the neuroretinal rim; RNFL, retinal nerve fiber layers; SD, standard deviation; SD-OCT, spectral-domain optical coherence tomography; T0, baseline; T1, evaluation performed at 6 months.
The OCT-A data analysis showed no statistically significant differences in retinal circulation status between migraineurs and controls. After 6 months of mAb treatment, the percentage VD significantly increased in the nasal perifoveal SCP and the inferior temporal and hemi-inferior subregions of the radial peripapillary capillary (RPC), respectively (p = 0.02, p = 0.02, and p = 0.03) (see Table 3).
Comparison of interictal retinal vascular parameters across groups by using statistical analysis.

Statistically significant results.
FAZ, foveal avascular zone; HCs, healthy controls; OCT-A, optical coherence tomography angiography; RPC, radial peripapillary capillary; SD, standard deviation; T0, baseline; T1, evaluation performed at 6 months; VD, vessel density.
The percentage VD of the retinal DCP and the size of the FAZ were similar across all groups (see Table 3).
There were no differences between groups in the VD of the other foveal, perifoveal, and parafoveal parameters (see Table 3).
Finally, the repeated-measures ANOVA, with the decrease in MMDs as a covariate, revealed a significant effect of MMDs only on temporal superior retinal nerve fiber layer (RNFL) thickness after 6 months of mAb therapy within the migraine group (see Table 4). No significant differences were found in OCT-A parameters between T0 and T1 following the improvement in migraine days (see Table 4).
Comparison of interictal OCT parameters within the migraine group across different time points using repeated-measures ANOVA, with the decrease in MMDs included as a covariate.

Statistically significant results.
MMDs, monthly migraine days; OCT, optical coherence tomography; OCT-A, optical coherence tomography angiography; RNFL, retinal nerve fiber layers; RPC, radial peripapillary capillary; SD, standard deviation; SD-OCT, spectral-domain optical coherence tomography; T0, baseline; T1, evaluation performed at 6 months; VD, vessel density.
Discussion
Our retrospective observational study identified a differential retinal status between migraine patients and control eyes through combined structural and vascular OCT scanning. Migraineurs presented with a thinner nasal and thicker temporal RNFL compared to HCs. These alterations did not correspond to a baseline deficit in retinal circulation on OCT-A, suggesting that axonal damage might occur—or at least be detectable—in the presence of still-preserved vascular networks.
Of interest, after 6 months of treatment with anti-CGRP mAbs, vascular perfusion increased in the superficial plexus and RPC, while the temporal retinal thickness remained sustained, possibly leading to an improvement of neural structure and vasculature and helping prevent the development of permanent, irreversible ocular injury.
OCT and OCT-A are noninvasive, low-cost techniques that allow quantitative and qualitative assessment of retinal status and have been widely used in various medical conditions. Mounting evidence points to both RNFL thickness and retinal vasculature as promising biomarkers for early diagnosis, monitoring clinical progression, and assessing treatment response in several neurological conditions, especially in multiple sclerosis and other neurodegenerative disorders.27,28 Nonetheless, in migraine sufferers, although ocular manifestations such as photophobia and visual aura are commonly associated with headache, 29 OCT data evaluating the specific ophthalmological profile during and between attacks are inconsistent across studies, most probably due to the high variability of patient cohorts and the different technical applications of either OCT or OCT-A alone.19–21,30
Some authors have reported no differences or an increased RNFL thickness between patients with migraine and controls.19–21 Conversely, a recent meta-analysis found a significant lowering of the average pRNFL thickness in migraineurs, and these abnormalities appeared to be prominent in those with aura and/or CM. 21 They postulated that abnormal vasospasm occurring during repeated migraine attacks could affect retrobulbar circulation, leading to optic nerve head hypoperfusion and retinal ganglion cell death. 21
In this case-control and observational study, we first identified significant quadrant-specific RNFL thinning in migraine patients’ eyes, localized selectively in the nasal region. This suggests a preferential structural impairment of those retinal ganglion cell axons that are mostly unmyelinated and require more energy supply, making them more susceptible to local perimetric blood changes. 31 It is noteworthy that these nerve fibers project to the magnocellular pathways localized outside the macula and therefore do not specifically contribute to visual acuity. These findings, along with the structural integrity of the outer macular layers assessed by means of SD-OCT, explain why visual acuity is preserved in migraineurs and suggest that these changes could represent an early indicator of subclinical visual dysfunction.
We also noted RNFL thickening in the temporal sector, where the nerve fibers are directly connected to the highly vulnerable papillo-macular bundle. Interestingly, this structural OCT change mirrors patterns found in various ocular disorders that commonly complicate migraine, such as glaucoma, due to a dynamic fluid shift from the nearby vascular plexus and disruption in axoplasmic flow. 32 Although the pathophysiology underlying these alterations remains unclear, we propose that the increased temporal RNFL thickness in our sample may represent a compensation mechanism that occurs as the privileged optic nerve attempts to maintain adequate function.
Regarding OCT-A imaging, some authors have shown that migraine patients tend to have larger FAZ areas and lower superficial and deep foveal vascular density compared to control groups.20,21,30 Here, we found no significant differences at baseline in retinal vasculature between the eyes of migraine patients and controls, reinforcing our hypothesis that the primary insult in the pathogenesis of ocular impairment in migraine is more likely to occur directly in the structural nerve layers rather than in the blood vessel networks.
In patients treated with anti-CGRP mAbs for 6 months, we observed that RNFL thickness remained sustained in its superior temporal sector, whereas the vascular density increased in the surrounding SCP and RPC, possibly helping to protect neural conduction along the papillo-macular bundle and potentially preventing axonal retrograde degeneration. Hence, the optic nerve head could be more resistant to ischemic, hypoxic, and inflammatory processes compared to the remaining retinal regions.
Different hypotheses might explain our OCT-A results.
First, since migraine patients often experience a significant reduction in headache days and migraine severity with these new targeted drugs, they may also have reduced vasospasm. This could lead to improved extracranial perfusion and vascular morphology. However, in our sample, only changes in temporal superior RNFL thickness—rather than OCT-A parameters—were found to be significantly associated with the reduction in migraine days following mAb therapy.
On the other hand, functional CGRP receptors have been identified both in the retinal nerve fiber layers and on the surface of inner blood vessels. CGRP is thought to contribute to retinal and visual physiopathology through its role in promoting neurogenic inflammation.33,34 Therefore, the ocular disturbances observed in our study may be closely linked to CGRP activity, which not only plays a central role in migraine pathophysiology but may also exert a direct effect on ocular tissues. 9
In animal models, intracerebroventricular injection of CGRP has been shown to induce significant photophobia, while this response was prevented by the concomitant use of the human CGRP receptor antagonist olcegepant. 35 These effects were associated with the regulation of synaptic signaling and sensory perception, modulation of the trigeminovascular system, and activation of ocular neuronal sites. 35
Studies have also reported that individuals receiving CGRP infusions can develop photophobia. In healthy volunteers, severe light sensitivity occurred within 30 min of the infusion, followed by the onset of acute migraine approximately 3 h later. 36
Furthermore, in migraine patients, Schiano di Cola et al. recently demonstrated a significant improvement in ictal photophobia beginning 1 month after initiating treatment with the anti-CGRP mAb galcanezumab. This improvement occurred regardless of the subsequent change in headache frequency. 16
Taken together, these findings suggest that the activation of endogenous CGRP and its receptors may play a role in the ocular changes seen in migraine patients, particularly those with more severe and disabling migraines.
Thus, targeting the CGRP pathway may help alleviate neuronal injury and maladaptation associated with migraine-related ocular impairment, potentially providing both therapeutic and neuroprotective benefits.
Elucidating the pathophysiologic mechanisms behind such visual dysfunctions and identifying their clinical phenotypes in the future is crucial.
Our study had several limitations: the relatively small cohort size, the absence of evaluations during migraine attacks and after a longer treatment period, and the lack of differentiation based on the specific mechanism of action of anti-CGRP mAbs (targeting either the ligand or its receptor). Additionally, there were no considerations regarding previous failed preventive antimigraine drugs, nor was there any analysis of the degree and timing of improvement, worsening, or stability of ictal ocular symptoms.
Conclusion
Our data indicate that retinal and neural structural changes could serve as early indicators of subclinical ocular disturbances in migraine, potentially marking the beginning of prevention or early intervention in the disease progression.
The effects of anti-CGRP mAb therapy may be monitored sequentially using OCT in a noninvasive way and may be beneficial in the short-term, potentially reducing the development of irreversible vascular damage.
Further studies are warranted to thoroughly assess the effectiveness of anti-CGRP mAbs for the treatment of migraine complications, including the potential role of migraine attack therapies such as triptans, and to elucidate their mechanism of action in the ocular system.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251347277 – Supplemental material for Evaluating the impact of anti-CGRP monoclonal antibodies on retinal features in migraine patients: a retrospective optical coherence tomography study
Supplemental material, sj-docx-1-tan-10.1177_17562864251347277 for Evaluating the impact of anti-CGRP monoclonal antibodies on retinal features in migraine patients: a retrospective optical coherence tomography study by Massimo Cesareo, Alessio Martucci, Roberta Bovenzi, Marco Lombardo, Francesca Pistoia, Vittoria Carla D’Agostino, Alessandro Stefani, Carlo Nucci, Nicola Biagio Mercuri and Maria Albanese in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Dr. Roberto Sorge for his valuable assistance in revising the statistical analysis and contributing to the preparation of the tables.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
