Abstract
Background:
There is currently no established standard of care for recurrent glioblastoma (GBM). Re-irradiation (re-RT) and Bevacizumab (BEV) are both used in salvage treatment, but their combined efficacy remains uncertain.
Objectives:
To evaluate whether combining re-irradiation with BEV improves survival outcomes compared to BEV alone in patients with recurrent high-grade gliomas (rHGG).
Design:
Systematic review and meta-analysis of two-arm clinical trials.
Data sources and methods:
A comprehensive literature search was conducted in Scopus, PubMed, Web of Science, and the Cochrane Library up to April 2024. Two independent reviewers assessed studies for eligibility and extracted data. Study quality was evaluated using the ROBINS-I and ROBINS-II tools. The primary outcome was overall survival (OS); secondary outcomes included progression-free survival (PFS), toxicity, and prognostic factors.
Results:
The meta-analysis demonstrated a significant improvement in OS with combined BEV and re-irradiation compared to BEV alone (hazard ratio (HR) 0.69, 95% confidence interval (CI: 0.56–0.85); p = 0.0005), corresponding to a 31% reduction in the risk of death. PFS also improved significantly (HR 0.64, 95% CI (0.45–0.90); p = 0.01). No significant increase in grade 3 toxicities was observed with the combination therapy. Subgroup analyses indicated that younger age and female gender were statistically associated with better OS, though the effect of age was modest and male gender was linked to poorer survival. Karnofsky performance status significantly influenced survival. Pulsed versus non-pulsed re-irradiation showed no differential effect on outcomes.
Conclusion:
The combination of re-irradiation and BEV significantly improves both OS and PFS in patients with rHGG, without increasing severe toxicity. These findings support the safety and efficacy of the combined approach. Prospective trials are warranted to guide standardized treatment protocols.
Trial registration:
This review was prospectively registered with PROSPERO (CRD42023463183).
Plain language summary
Recurrent glioblastoma (GBM) is a highly aggressive form of brain cancer, and there is no universally accepted treatment once the tumor returns. This study examined whether combining two therapies—Bevacizumab (BEV), a medication that targets tumor blood vessels, and re-irradiation (a second round of radiation therapy)—is more effective than using BEV alone.
By analyzing data from several clinical trials, researchers found that patients who received the combination therapy lived longer overall (overall survival, OS) and went longer without disease worsening (progression-free survival, PFS) compared to those who received BEV alone. Importantly, adding re-irradiation did not lead to more serious side effects.
The study also found that patients with better overall health (measured by Karnofsky Performance Status) had improved survival. While younger patients tended to do slightly better, the benefit was small. Male patients, however, had worse survival outcomes compared to females. Additionally, using a specific radiation method called pulsed re-irradiation did
These results suggest that combining BEV with re-irradiation is a safe and effective strategy for treating recurrent GBM, helping patients live longer without increasing side effects. More studies are needed to confirm these findings and to guide treatment decisions more precisely.
Introduction
Glioblastoma (GBM) is the most common and aggressive primary brain tumor in adults, accounting for approximately 60% of high-grade gliomas and 50% of all malignant brain tumors. Despite aggressive treatment, including surgical resection, temozolomide chemotherapy, and external beam radiotherapy, the prognosis remains poor. The recurrence of GBM is nearly inevitable, and post-recurrence survival is limited, averaging just 6 months for GBM and 10 months for anaplastic astrocytoma.1–3 The latest WHO classification of gliomas highlights the importance of genetic markers, such as IDH mutations and 1p/19q co-deletions, in aiding diagnosis and guiding treatment strategies. 4
In 2009, the FDA-approved Bevacizumab (BEV), a monoclonal antibody targeting vascular endothelial growth factor (VEGF), for use in recurrent GBM (rGBM). BEV has been shown to improve progression-free survival (PFS) in recurrent cases; however, it does not significantly extend overall survival (OS). 5 Radiation therapy remains a cornerstone for OS enhancement in newly diagnosed GBM, but the recurrence of high-grade gliomas presents unique challenges. Treatment-related toxicities, such as radiation-induced necrosis, often complicate management. In recurrent cases, the goal is to balance local tumor control with minimizing additional treatment-induced morbidities. 6
Re-irradiation (re-RT) has increasingly been considered for patients with localized recurrence of GBM, although its use is limited by the risk of radiation-induced brain necrosis, especially when higher doses or larger volumes of tissue are involved.7,8 Despite careful patient selection, the overall benefit of re-RT remains uncertain, and its application must be approached with caution. 9
Combining re-RT with BEV offers the potential for synergistic benefits, as BEV’s antiangiogenic effects may target radioresistant glioma stem cells by disrupting their vascular niches, potentially improving tumor control while minimizing radiation-induced damage.10,11 This approach could overcome some of the limitations of re-RT alone, providing a promising treatment strategy for rGBM.
This meta-analysis systematically evaluates the impact of combining BEV with re-RT on both OS and PFS in rGBM patients, compared to BEV alone. In addition to efficacy, we also explore the safety profile of this combined approach to provide a comprehensive assessment of its potential clinical application. The findings from this study aim to inform clinical decision-making and optimize patient outcomes by better understanding the balance between efficacy and safety in rGBM treatment.
Methods
This review was registered prospectively on PROSPERO CRD42023463183. We adhered to PRISMA guidelines when reporting this systematic review and meta-analysis.
Eligibility criteria
We included studies meeting the following criteria:
Population: Studies on patients with recurrent high-grade gliomas (rHGG).
Intervention: Studies where the exposed group received re-RT combined with BEV.
Comparator: Studies where the control group received BEV alone.
Outcomes: Studies reporting on efficacy outcomes (such as OS and PFS) and safety outcomes (such as the incidence of grade 3 or higher toxicities).
Study design: Comparative designs, including randomized controlled trials (RCTs) and observational studies comparing the outcomes of re-RT + BEV versus BEV alone.
We excluded conference abstracts, studies in languages other than English, and studies that did not directly compare re-RT + BEV to BEV alone.
Data sources and searches
A comprehensive search of PubMed, Ovid, Scopus, Web of Science, and the Cochrane Central Register of Controlled Trials was conducted from inception to April 2024. The search query included terms like “High Grade Glioma,” “Glioblastoma,” “Bevacizumab,” and “re-irradiation” to capture relevant studies.
Selection process and data extraction
Studies were screened and selected independently by two reviewers. Data were extracted into a standardized sheet, including (1) study characteristics, (2) population demographics, (3) risk of bias, and (4) outcome measures, including both efficacy outcomes (such as OS and PFS) and safety outcomes (such as the incidence of grade 3 or higher toxicities).
Risk of bias assessment
Two authors independently assessed the risk of bias using the Cochrane Risk of Bias tool (ROB 2.0). ROB2 is a standardized tool developed for assessing the risk of bias in randomized trials. It was created by researchers affiliated with the Cochrane Collaboration for RCTs and the ROBINS-I tool for non-randomized studies.
Data synthesis and analysis
Statistical analyses were performed using RevMan version 5.4. The Cochrane Collaboration, 2020. London, United Kingdom.Pooled hazard ratios (HR) with 95% confidence intervals (CIs) were calculated for dichotomous outcomes. The significance threshold was set at p < 0.05. Heterogeneity was assessed using the I2 statistic. For an I2 > 50% or a p-value < 0.05, a random-effects model was applied, and a sensitivity analysis was conducted to identify sources of heterogeneity.
To address the heterogeneity, the PythonMeta tool was applied, alongside subgroup analyses to identify and manage sources of variability. 12
Results
Study selection
A total of 355 studies were identified through database searches (PubMed: 112, Scopus: 145, Web of Science: 98). After removing duplicates, 210 studies were screened, with 140 excluded due to irrelevance or study design. Full-text screening for eligibility was conducted on 30 studies. After excluding 23 studies for reasons such as insufficient outcome data or wrong interventions, 7 studies were ultimately included in the final analysis (Figure 1).
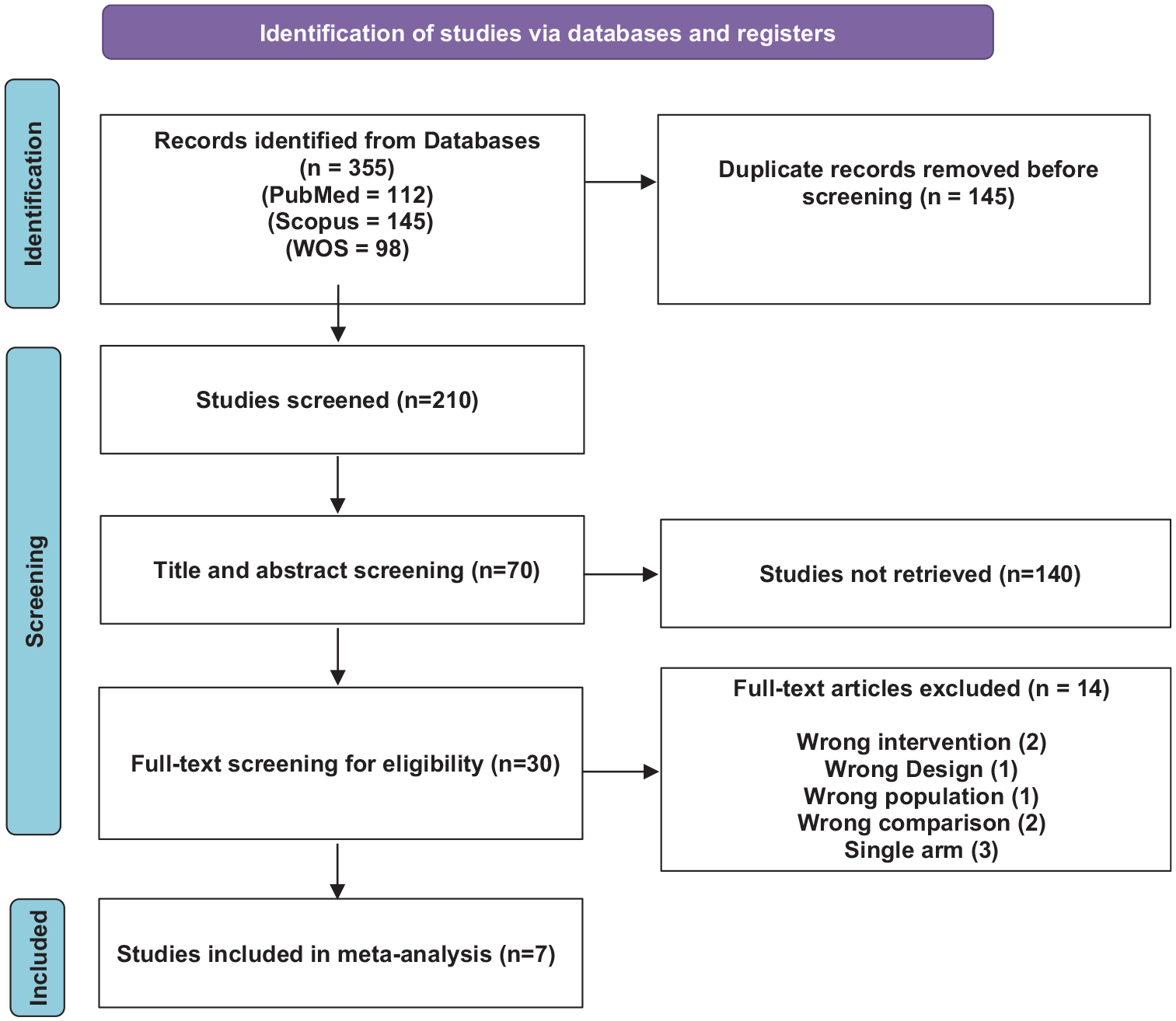
PRISMA flow diagram of the included studies.
Study characteristics
Study and treatment characteristics are outlined in Table 1. The analysis included a total of 1023 participants. Two studies were RCTs, while 5 studies were non-randomized studies. Of the seven studies included, the majority of patients were diagnosed with WHO grade 4 GBM, comprising approximately 70%–90% of the population across studies. WHO grade 3 gliomas, including astrocytomas and oligodendrogliomas, accounted for the remaining 10%–30% of participants. Data on genetic markers, specifically IDH mutations and 1p/19q co-deletion, were inconsistently reported. IDH mutation status was available in three studies, showing 10%–20% of patients with IDH mutations, while information on 1p/19q co-deletion was largely absent. For more comprehensive insights into the study and treatment specifics, refer to Table 1.
Summary of included studies.

BEV, bevacizumab; FSRS, fractionated stereotactic radiosurgery; N/A, not available; pRDR, pulsed reduced dose rate radiotherapy; RCT, randomized controlled trials.
Risk of bias within studies
The RoB assessment is demonstrated in Figures 2 and 3. The two RCTs were of low RoB. Of the five observational studies, four were of moderate RoB and one was of low.

Risk of bias assessment—ROBINS 2—for RCT studies.

Risk of bias assessment—ROBINS 1—for non-RCT studies.
Outcomes
Overall survival
A meta-analysis of 7 studies involving a total of 1023 patients assessed the efficacy of both treatment methods on OS. The pooled HR indicated that concurrent BEV and re-irradiation were significantly associated with improved OS (HR 0.69, 95% CI (0.56, 0.85), p = 0.0005; Figure 4).

Forest plot summary for OS: The forest plot summarizes the log HR of the study outcome for OS. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also highlights the p for χ2 (evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for overall effect), and p for the significance of the overall effect.
Progression-free survival
A total of five studies, involving a combined total of 587 patients, reported on the efficacy of both methods in terms of PFS. The pooled HR indicated that concurrent BEV and re-irradiation were significantly associated with improved PFS (HR 0.64, 95% CI (0.45, 0.90), p = 0.01; Figure 5).

Forest plot summary for PFS: The forest plot summarizes the log HR of the study outcome for PFS. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also highlights the p for χ2 (evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for overall effect), and p for the significance of the overall effect.
To address heterogeneity, a subgroup analysis was conducted based on radiotherapy fractionation (>5 fractions vs ⩽5 fractions).
The HR for the >5 fractions subgroup was 0.65 (p = 0.00015), while for the ⩽5 fractions subgroup, the HR was 0.64 (p = 0.00001). The test for heterogeneity between subgroups yielded an I2 value of 0%, with a p-value of 0.98, indicating no significant variability in effect sizes across subgroups.
These results suggest that the benefit of concurrent BEV and re-irradiation in improving PFS is independent of the number of radiotherapy fractions administered (Figure 6).
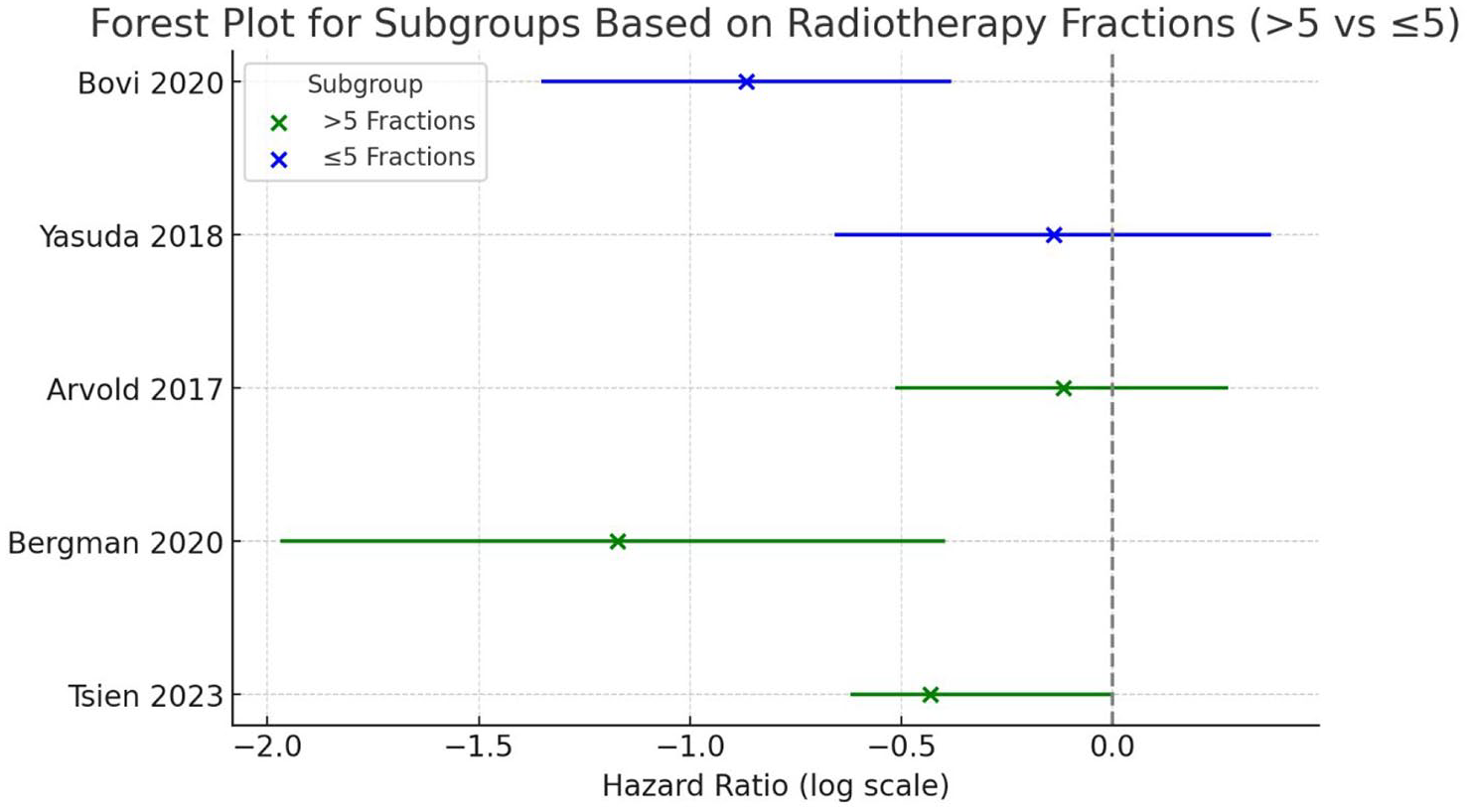
Forest plot summary for subgroup analysis based on radiotherapy fractionation protocols for PFS.
Relationship between Karnofsky performance status and OS
The results of the meta-analysis revealed an association between Karnofsky performance status (KPS) and OS in two types of treatment: concurrent BEV with re-irradiation, or BEV alone (HR 0.97, 95% CI (0.96, 0.99), p = 0.005; Figure 7).
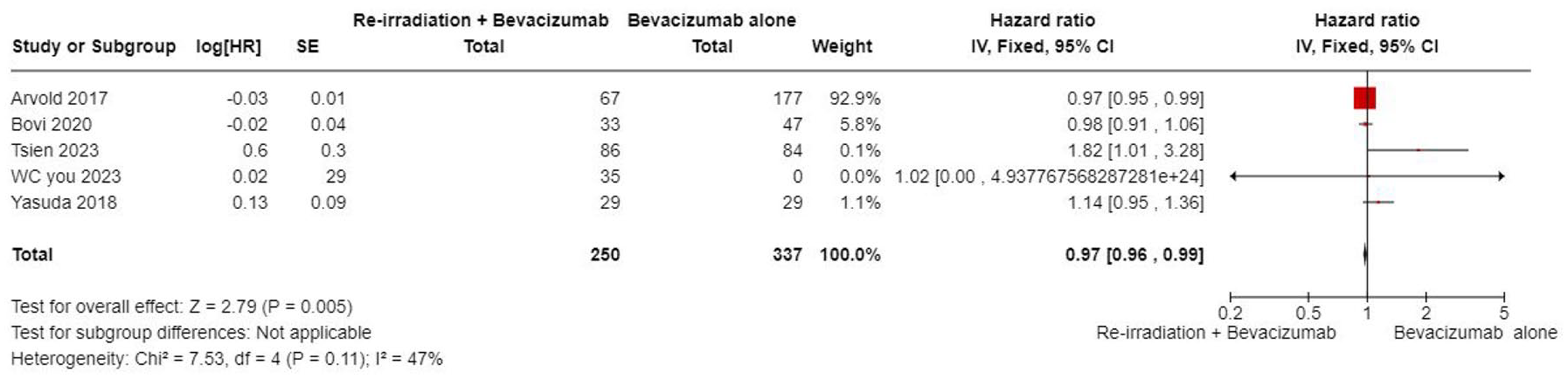
Forest plot summary for the relationship between KPS and OS. The forest plot summarizes the log HR of the relationship between KPS and OS. Key metrics presented include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also highlights the p for χ2 (indicating evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for the overall effect), and the p for the significance of the overall effect.
Grade 3 toxicities as an adverse effect of treatments
Five studies, comprising a total of 879 patients, reported significant grade 3 toxicities as an adverse effect of the treatments. The meta-analysis results indicated no significant difference in the occurrence of grade 3 toxicities between the two methods (HR 1.25, 95% CI (0.80, 1.95), p = 0.33; Figure 8).

Forest plot summary for significant grade 3 toxicities as an adverse effect of treatments. The forest plot summarizes the log HR for significant grade 3 toxicities as an adverse effect of the treatments. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also details the p for χ2 (indicating evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for the overall effect), and the p for the significance of the overall effect.
Subgroup meta-analysis based on the type of radiotherapy applied
The subgroup meta-analysis, based on the type of radiotherapy applied in group 1 (re-irradiation with BEV), revealed no significant difference in OS between the two treatment methods (χ2 0.06, df = 1, p = 0.81; Figure 9).

Forest plot summary for subgroup meta-analysis based on the type of radiotherapy applied. The forest plot summarizes the log HR for the subgroup analysis focusing on significant grade 3 toxicities as an adverse effect, based on the type of radiotherapy applied. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also provides the p for χ2 (indicating evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for the overall effect), and the p for the significance of the overall effect.
Subgroup meta-analysis on age as a prognostic factor for OS
The subgroup meta-analysis identified age as a statistically significant prognostic factor for overall survival (OS). The hazard ratio (HR) was 1.01 (95% CI: 1.00–1.02, p = 0.03), indicating that older age was associated with slightly worse OS. However, the effect size was minimal, suggesting no clinically meaningful difference in survival outcomes based on age alone (p = 0.03; Figure 10).
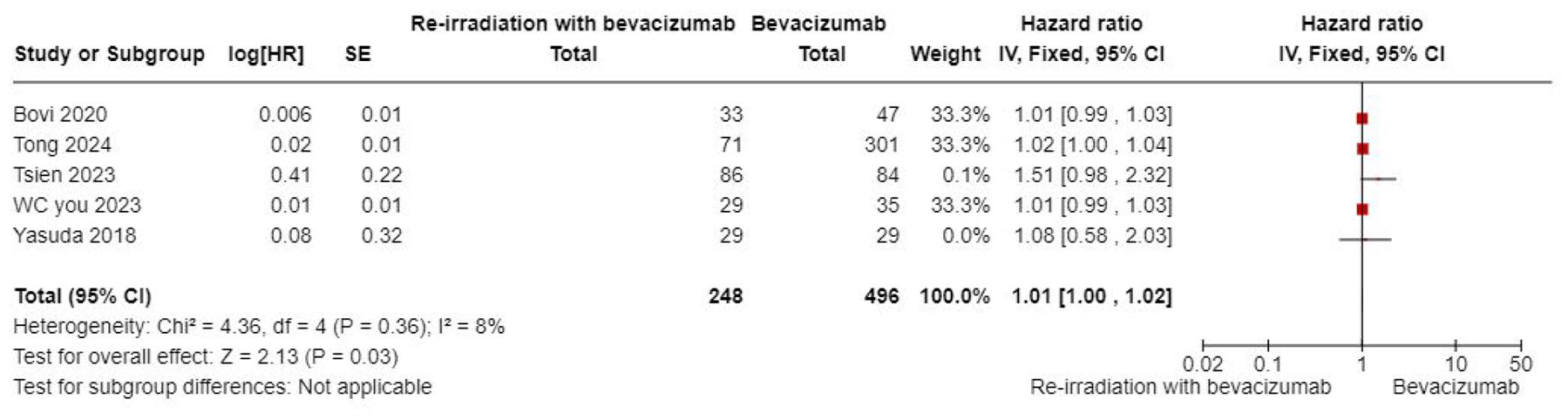
Forest plot summary for subgroup meta-analysis on age as a prognostic factor for OS. The forest plot summarizes the log HR for the subgroup analysis examining age as a prognostic factor for OS. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also presents the p for χ2 (indicating evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for the overall effect), and the p for the significance of the overall effect.
Subgroup meta-analysis on gender as a prognostic factor for OS
The meta-analysis identified gender as a statistically significant prognostic factor for overall survival. The pooled hazard ratio (HR) was 1.15 with a 95% confidence interval of 1.09 to 1.22, indicating that male patients had worse overall survival compared to female patients. Although statistically significant (p < 0.00001), the effect size indicates only a modest clinical impact on OS (p < 0.00001; Figure 11).
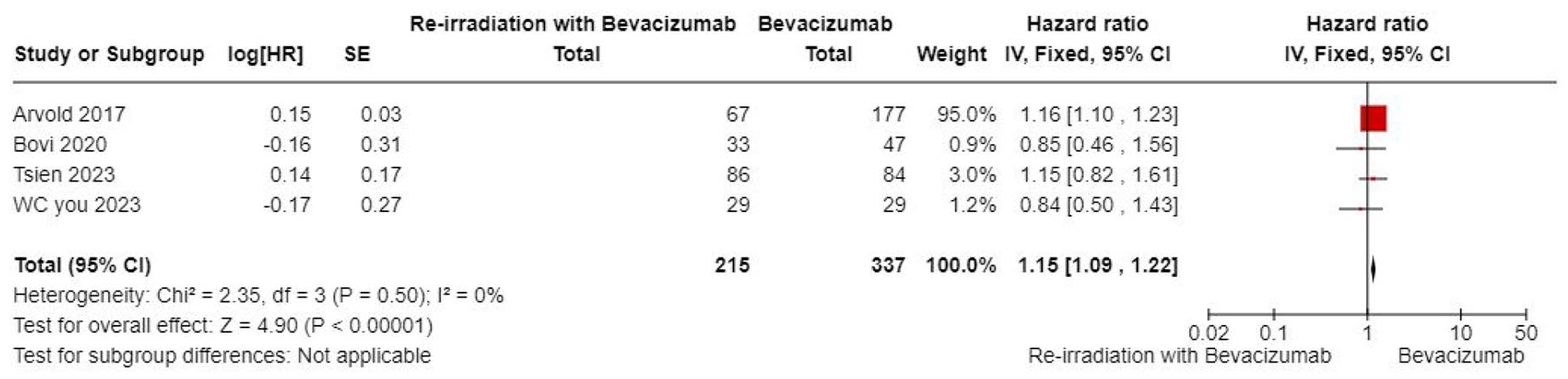
Forest plot summary for subgroup meta-analysis on gender as a prognostic factor for OS. The forest plot summarizes the log HR for the subgroup analysis investigating gender as a prognostic factor for OS. Key metrics include the HR, SE, p, CI, df, and χ2 (statistical test for heterogeneity). The plot also includes the p for χ2 (indicating evidence of heterogeneity in intervention effects), I2 (degree of heterogeneity between studies), Z-statistic (test for the overall effect), and the p for the significance of the overall effect.
Discussion
Summary of the results
This meta-analysis evaluated the efficacy of combining bevacizumab (BEV) with re-irradiation (re-RT) compared to BEV alone in patients with recurrent glioblastoma (rGBM). The results demonstrated significant improvements in both overall survival (OS) and progression-free survival (PFS) with the combination therapy. Specifically, the combination was associated with a 31% reduction in the risk of death (HR: 0.69; 95% CI: 0.56–0.85; p = 0.0005) and a 36% reduction in the risk of disease progression (HR: 0.64; 95% CI: 0.45–0.90; p = 0.01). Improved OS was also associated with higher Karnofsky Performance Status (KPS), younger age (albeit with minimal clinical impact), and female sex. Importantly, toxicity analysis revealed no significant increase in grade 3 or higher adverse events, underscoring the safety and tolerability of the combined treatment approach.
Efficacy in context of existing literature
The findings align with and expand upon Marwah et al.’s 19 prior meta-analysis, which reported improved OS (HR 0.42) and PFS (HR 0.46) with BEV + re-RT, although with lower certainty and a broader patient population. Our analysis incorporates three additional studies and focuses exclusively on comparisons between BEV + re-RT and BEV alone, offering stronger and more targeted evidence of efficacy.
GBM remains one of the most aggressive and treatment-resistant tumors. While re-resection and re-RT have shown potential benefits in rHGG, their effectiveness is often limited by tumor location and patient condition. 7 BEV, as an anti-VEGF agent, complements re-RT by improving tumor oxygenation and enhancing radiosensitivity.20–22 This synergistic effect may explain the improved survival outcomes observed in our analysis.
Supporting this, Vordermark et al. 23 found a median OS of 7.9 months for rGBM patients undergoing re-RT alone. In contrast, Kreisl et al. 5 reported a 43% reduction in the risk of death (HR 0.57, 95% CI (0.38–0.86)) with concurrent BEV and re-RT. These survival outcomes exceed those typically observed with re-irradiation alone, reinforcing the added benefit of BEV.
Further comparison with systemic therapies reveals that re-RT + BEV performs better in terms of OS and PFS. Marwah et al. 20 showed an OS HR of 0.57 for re-RT + BEV versus 0.73 for re-RT + systemic therapy, and a PFS HR of 0.64 versus 0.57, respectively. This suggests that BEV, through its antiangiogenic and immunomodulatory effects, may offer a more potent complement to radiation than conventional systemic agents.19,22
Safety profile and tolerability
Toxicity remains a critical consideration in re-RT for brain tumors, particularly with overlapping symptoms between tumor progression and treatment-induced radionecrosis. However, our analysis found no significant difference in the rate of grade ⩾3 adverse events between the BEV + re-RT group and the BEV-only group (HR 1.04, 95% CI (0.78, 1.38), p = 0.54).
This finding aligns with previous studies. Kazmi et al. 24 and Vordermark et al. 23 observed manageable toxicity levels in patients treated with this combination, and Levin et al. 10 showed that BEV may actively reduce radiation necrosis. BEV’s ability to normalize vasculature likely contributes to this safety advantage by mitigating radiation-induced vascular permeability and edema.19,22
In addition, our subgroup analyses explored whether treatment protocols, such as the use of pulsed versus fractionated re-irradiation, influence safety and efficacy outcomes. However, no statistically significant difference in OS was observed between these approaches (p = 0.81), suggesting that the type of re-irradiation technique may not substantially impact survival outcomes.
Factors influencing treatment outcomes
The exact mechanism behind the observed improvements remains unclear in the literature, but the synergistic effects of re-irradiation and BEV likely improve local tumor control while maintaining anti-angiogenic properties, leading to better outcomes.
The type of radiation therapy—whether pulsed or fractionated—may also affect survival rates. A meta-analysis of fractionated radiation therapy found that the incidence of brain radionecrosis was 5% at biologically effective doses of 120 Gy and 10% at 150 Gy for fraction sizes smaller than 2.5 Gy. 23 Despite the promise shown by pulsed reduced dose rate re-irradiation for recurrent grade 4 gliomas, its exact impact remains unclear, though its potential to reduce CNS toxicity is significant.23–28 Subgroup analysis in our study found no significant difference in OS between different radiotherapy approaches (pulsed vs fractionated), indicating that the type of radiotherapy used alongside BEV may not substantially influence survival outcomes.
Interestingly, KPS influenced OS in our meta-analysis, which may be explained by favorable patient characteristics and the KPS distribution in studies such as Tsien’s. This finding suggests that KPS may play a crucial role in determining outcomes between concurrent BEV with re-irradiation and BEV alone. 9
Other patient characteristics, such as age and gender, play an important role in predicting survival and prognosis. Ironside et al. 25 found that age is a significant prognostic factor for GBM survival, with patients over 65 years having lower OS compared to younger patients.25,28 Our findings identified age as a statistically significant but clinically minimal prognostic factor, with younger age being associated with slightly better OS. In addition, the role of gender in GBM survival has been debated. Lun et al. 29 demonstrated no significant difference in OS between males and females. However, our meta-analysis confirmed that female sex is a significant factor associated with better OS in GBM patients.
Limitations
While this meta-analysis highlights the potential benefits of combining BEV with re-irradiation for rGBM, important limitations must be acknowledged. The variability in study design, patient selection, and treatment protocols may impact the consistency of the results. Differences in re-irradiation methods, radiation doses, and BEV administration timing were inconsistently reported, affecting the overall applicability. The predominance of retrospective or observational studies introduces bias, weakening the reliability of the conclusions. In addition, the limited long-term follow-up in some studies complicates the evaluation of treatment durability and late toxicities. Differentiating outcomes between WHO grade 4 GBM and WHO grade 3 gliomas further emphasizes the need for a more nuanced interpretation.
Conclusion
The combination of BEV with re-irradiation showed a significant improvement in both OS and PFS compared to BEV alone, without a corresponding increase in grade 3 toxicities. This demonstrates that the concurrent use of BEV and re-irradiation is not only effective but also safe for managing rGBM. Nevertheless, further clinical trials are essential to better understand the long-term effects and optimize the use of this combined treatment strategy in clinical practice. Comprehensive trials will also help refine treatment protocols and provide clearer guidance for improving patient outcomes.
Supplemental Material
sj-doc-1-tan-10.1177_17562864251343574 – Supplemental material for Efficacy and safety of combining re-irradiation with bevacizumab compared to bevacizumab alone in the management of recurrent high-grade gliomas: a meta-analysis and systematic review
Supplemental material, sj-doc-1-tan-10.1177_17562864251343574 for Efficacy and safety of combining re-irradiation with bevacizumab compared to bevacizumab alone in the management of recurrent high-grade gliomas: a meta-analysis and systematic review by Ali Hammed, Almonzer Al-Qiami, Ali Hasan, Gregor Richter, Asmaa Zakria Alnajjar, Josef Rosenbauer, Karel Kostev, Omar Ismail, Veit Braun and Christian Tanislav in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We would like to express our gratitude to Hamza Alsalhi, Mostafa Naguib, and Hosny Elkhawaga for their contributions to this meta-analysis. Although their involvement was limited and some tasks were not fully completed, their early efforts in data collection and preliminary analysis were valuable to the overall project.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
