Abstract
Background:
Dysphagia is a common complication following intracerebral hemorrhage (ICH) and is associated with an increased risk of aspiration pneumonia and poor outcomes.
Objectives:
This study aimed to explore associated lesion patterns and contributing factors of post-ICH dysphagia, and predict dysphagia outcomes following ICH.
Design:
A multicenter, prospective study.
Methods:
Patients with ICH from two stroke centers within 72 h of symptom onset received baseline bedside swallowing evaluations. Dysphagia-related lesion patterns were identified using support-vector regression-based lesion-symptom mapping. Predictors of swallowing impairment on the 7th and 30th day, as well as stroke-associated pneumonia (SAP), were determined through multiple logistic regression analyses, and nomograms were developed.
Results:
A total of 153 patients were included in the final analysis. Of those, 28 had dysphagia. Dysphagia-related lesions predominantly affected bilateral subcortical and adjacent cortical regions. Stroke severity, hematoma expansion, and basal ganglia hemorrhage were significantly associated with initial dysphagia. Baseline aspiration risk and age were identified as independent predictors of impaired swallowing function on days 7 and 30, and SAP. Moreover, ICH volume was significantly correlated with swallowing impairment on day 7 and SAP occurrence. Midline shift and basal ganglia hematoma remained independent predictors of impaired swallowing on day 30. Predictive models for swallowing impairment on days 7 and 30, as well as SAP, demonstrated strong calibration and discriminatory ability, with C indices of 0.867, 0.895, and 0.773, respectively.
Conclusion:
Post-ICH dysphagia can be predicted based on stroke severity, hematoma expansion, and basal ganglia hemorrhage. Incorporating aspiration risk and imaging evaluation can further improve the identification of patients at high risk for swallowing impairment at both 1 week and 1 month after ICH.
Introduction
Spontaneous intracerebral hemorrhage (ICH) is the second most common and most lethal stroke subtype.1,2 Compared with ischemic stroke, higher rates of mortality and disability are often expected in patients with ICH.3,4 Dysphagia is a common complication following stroke and is associated with an increased risk of stroke-associated pneumonia (SAP) and leads to catastrophic infectious consequences.5,6 In the latest American Heart Association/American Stroke Association (AHA/ASA) Guideline for Spontaneous Intracerebral Hemorrhage, dysphagia screening is recommended before oral intake to reduce pulmonary infection. 7 Early detection of swallowing disorders allows for timely interventions, such as nasogastric tube placement and percutaneous endoscopic gastrostomy. 8 Previous studies showed that advanced age, stroke severity, larger hematoma volume, and strategic lesion location were associated with post-ICH dysphagia.9,10 Clinical presentation and prognosis of ICH differ based on the location of the hemorrhage. A hospital-based study demonstrated that patients with lobar hemorrhages had poorer outcomes compared to those with subcortical ICHs. 11 Models for post-stroke dysphagia based on clinical and radiological features showed reliable predictive values in ischemic stroke patients.12,13 Therefore, accurate prognostic techniques for swallowing disorders following ICH based on prospective data are still in need to guide clinicians’ decisions. The aims of this study were: (1) to investigate the incidence of post-ICH dysphagia and identify associated lesion locations and contributing factors; (2) to determine predictors of swallowing outcomes and SAP in ICH patients; and (3) to develop predictive models for post-ICH dysphagia and swallowing outcomes based on imaging and clinical characteristics.
Methods
Study population
The study cohort consisted of pooled data from two prospective cohorts of acute ICH patients. The first cohort was admitted to the First Affiliated Hospital of Chongqing Medical University between November 2021 and October 2022, and the second cohort was admitted to the Second Affiliated Hospital of Anhui Medical University between January and December 2023. The exclusion criteria were as follows: (1) secondary ICH resulting from the structural lesion, trauma, tumors, or hemorrhagic transformation; (2) lack of computed tomography (CT) scans within 72 h post-ictus; (3) inability to undergo baseline swallowing evaluation due to impaired consciousness, intubation, or tracheostomy; (4) preexisting dysphagia caused by other concomitant diseases; (5) occurrence of a recurrent stroke during the study; and (6) non-compliance with the evaluation procedure. This study has been reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies.
Dysphagia evaluation
Baseline swallowing evaluations were conducted within 48 h of admission for conscious patients capable of sitting upright. We utilized Any2 Scale to evaluate the risk of aspiration, a validated tool that can be easily applied by bedside and comprises the following features: dysphonia, dysarthria, abnormal gag reflex, abnormal volitional cough, cough after swallow, and voice change after swallow. 14 We scored swallowing ability according to the functional oral intake scale (FOIS), which ranges from level 1 (nothing by mouth) to level 7 (total diet with no restriction) based on the consistency and amount of food that a patient could bear. 15 We defined initial dysphagia as a FOIS of ⩽5 at baseline, indicating that these patients could not tolerate food of various consistencies.
Outcomes
Oral intake ability on the 7th and 30th days was evaluated using FOIS. Patients with a FOIS score of ⩽6 who did not recover to their premorbid state were considered to have impaired swallowing function. 16 SAP was defined as the use of antibiotics for newly diagnosed pneumonia within 7 days after admission, or as determined by the treating physician. 17 Functional outcomes were evaluated using the modified Rankin Scale (mRS).
Clinical variable collection
Clinical information on patients’ gender, age, vascular risk factors, and medication were prospectively collected. Stroke severity was assessed at each visit using the National Institutes of Health Stroke Scale (NIHSS).
Imaging analysis
Admission and follow-up CTs were performed with a slice thickness of 5 mm. Hydrocephalus was defined as either contralateral dilation in the temporal horns or unilateral dilation in the frontal or posterior horns from deep ICH, or as dilation in the bilateral lateral ventricles or the third ventricle caused by an obstructive clot in the ventricle or mass effect from deep ICH. 18 Midline shift was defined as dislocation over 4 mm of pineal gland, septum pellucidum, and cerebral falx. 19 Hematoma volume was calculated by MRICron software. Hematoma expansion (HE) was defined as an absolute hematoma growth >12.5 mL from baseline CT or an increase in hematoma volume >33%. 20
For voxel lesion-symptom mapping (VLSM) analyses, baseline CT scans were spatially normalized to match the Montreal Neurological Institute 152 standard space template by Clinical Toolbox embedded in Statistical Parametric Mapping 12 (https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) based on Matlab 2021a. Hematomas were delineated using MRICron software (https://www.nitrc.org/projects/mricron) to generate binary lesion masks.
Statistical analysis
The demographic, radiological, and clinical parameters were compared between patients with and without initial dysphagia. Continuous data were compared using Student’s t-test or the Mann–Whitney U test, while categorical variables were analyzed using the χ 2 test or Fisher’s exact test, as appropriate. To identify potential prognostic variables, univariate logistic regression analysis was performed for each covariate. Factors with a p < 0.2 were included in the multivariate analyses. We examined multicollinearity using variance inflation factors (VIFs), variables with a VIF > 5 were excluded from the multivariate model. Subsequently, nomograms predicting swallowing impairment on day 7 and 30, as well as SAP, were constructed based on the results from multivariate regression analyses. The concordance index was calculated using 1000 bootstrap samples to evaluate discriminatory ability. Calibration plots were generated to assess calibration ability. Statistical analyses utilized IBM SPSS Statistics version 25 (IBM SPSS Statistics; Chicago, IL, USA) and R version 4.2.0 (The R Foundation; Vienna, Austria).
Multivariate support-vector regression lesion-symptom mapping (SVR-LSM) analysis was performed utilizing SVR-LSM toolbox (version 2019-06-16).21,22 Compared to the traditional VLSM method, SVR-LSM is a multivariate analysis technique that makes use of the SVR function of Statistics Toolbox in Matlab, in which intervoxel correlation is taken into account and lesion volume is regressed out of both lesion location and behavioral score. Only voxels lesioned in at least 10% masks were included in the analysis to ensure generality. To control type II errors, extensive permutation testing (10,000 permutations) was used to generate voxel-wise distribution of SVR-β and then converted into p value map. Associations between voxel-wise lesion location and behavioral data were deemed significant at p < 0.05. Cluster-wise family-wise error (FWE) correction was then applied to control the false positive rate. To identify affected regions, the Automated Anatomical Labeling (AAL) and the Johns Hopkins University (JHU) white matter templates were employed. Clusters with fewer than 200 contiguous voxels were excluded from the final results.
Results
Baseline characteristics
A total of 153 individuals were included in the final analyses (Figure S1), and 13 patients were excluded from VLSM due to HE (n = 12), and poor image quality (n = 1). Among them, 28 (18.3%) were classified as initial dysphagia according to pre-specified criteria based on FOIS ⩽5. However, impaired swallowing ability was identified in 44 patients (28.8%) on the 7th day and in 18 patients (11.8%) on the 30th day according to our criteria. Median follow-up time was 30.6 days (interquartile range (IQR), 30.0–33.6). Clinical and radiological features are summarized in Table 1. The patients had a mean age of 60.1 years (standard deviation (SD), 13.3), and 30.7% were female. Initially, dysphagic patients had higher baseline NIHSS scores (p < 0.001) compared with non-dysphagic ones. Regarding imaging features, the median ICH volume for the entire cohort was 5.21 mL (IQR, 2.26–10.15), with no significant difference between the two groups (p = 0.319).
Patient characteristics.

FOIS, functional oral intake scale; ICH, intracerebral hemorrhage; IQR, interquartile range; IVH, intraventricular hemorrhage; mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; SAP, stroke-associated pneumonia; SD, standard deviation.
Hematoma location and variables associated with dysphagia at baseline
An overlap map of voxels present in at least 10% of ICH patients was generated during the SVR-LSM analysis (n = 105). As illustrated in Figure 1, the distribution of hematomas predominantly affected bilateral subcortical regions, with the majority of lesions localized within the basal ganglia and thalamus, extending to the adjacent insular cortex and corona radiata. SVR-LSM identified several clusters exhibiting potential associations with post-ICH dysphagia. Clusters located in the left insula (214 voxels), putamen (230 voxels), and external capsule (347 voxels) showed association with initial dysphagia, with mean Z scores of 1.9360, 1.9106, and 1.8879, respectively. These clusters also demonstrated a positive correlation with baseline Any2 scores (Table 2). However, the significance of these clusters did not persist after cluster-wise FWE correction.

Overlay of hemorrhagic lesions included in SVR-LSM (n = 105). The color bar indicates overlap frequency per voxel, with darker shades representing higher frequencies. The variable “N” denotes the number of overlaps. The regions included in the analyses are predominantly located in bilateral subcortical areas and adjacent cortical structures. SVR-LSM indicates support-vector regression-based lesion-symptom mapping.
Clusters significantly associated with swallowing dysfunction at different stages derived from SVR-LSM.
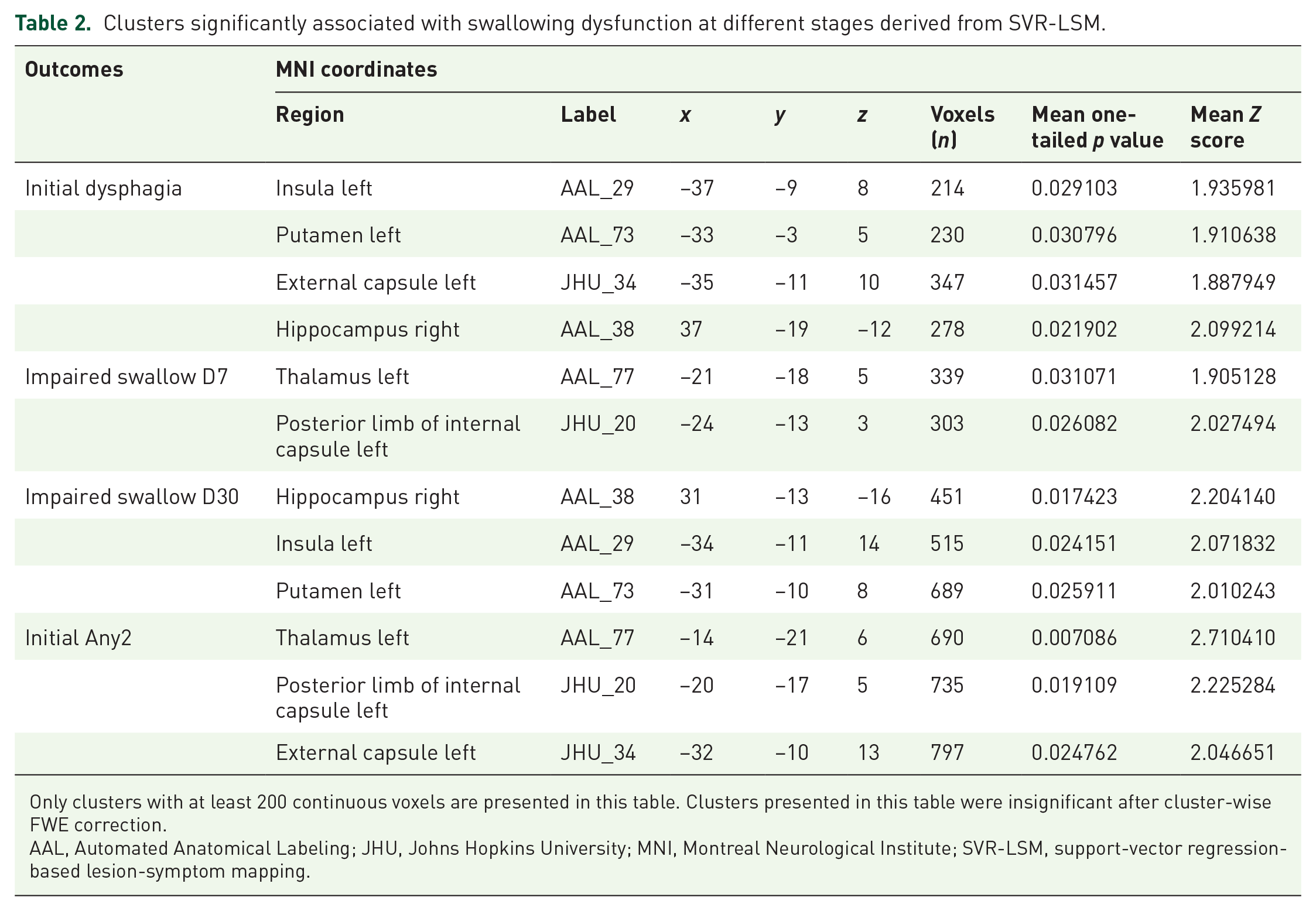
Only clusters with at least 200 continuous voxels are presented in this table. Clusters presented in this table were insignificant after cluster-wise FWE correction.
AAL, Automated Anatomical Labeling; JHU, Johns Hopkins University; MNI, Montreal Neurological Institute; SVR-LSM, support-vector regression-based lesion-symptom mapping.
To evaluate the influence of clinical and radiological factors on post-ICH dysphagia, we performed univariate logistic regression analyses (Table S1). In multivariate regression analyses, higher NIHSS score (odds ratio (OR), 1.26; 95% confidence interval (CI), 1.09–1.44; p = 0.001), hemorrhage in basal ganglia (OR, 5.12; 95% CI, 1.53–17.16; p = 0.008), HE (OR, 5.88; 95% CI, 1.04–33.43; p = 0.046) remained as independent predictors for initial dysphagia after adjusted for other covariates. Age (OR, 1.04; 95% CI, 1.00–1.09; p = 0.057) showed a marginal association with dysphagia during baseline evaluation (Figure 2(a)). Furthermore, putamen was strongly correlated with initial dysphagia after adjusting for covariates including age, NIHSS, and premorbid mRS (OR, 8.12; 95% CI, 1.42–46.44; p = 0.019; Table S1).

Forest plots based on the results from multivariate analyses of characteristics associated with initial dysphagia, impaired swallowing at day 7 and day 30, as well as SAP. (a) Initial dysphagia. (b) Impaired swallowing function on the seventh day. (c) Impaired swallowing function on the 30th day. (d) Stroke-associated pneumonia during follow-up.
Lesion pattern and multivariate analyses of prediction for swallowing outcomes
In the SVR-LSM analysis, clusters located in the left thalamus (339 voxels, Z score = 1.9051) and the left posterior limb of the internal capsule (303 voxels, Z score = 2.0275) exhibited a potential correlation with swallowing impairment on day 7 at a voxel-wise significance level of 0.05. A similar correlation was observed between the right hippocampus and impaired swallowing on both day 7 and 30 (Table 2).
In multivariate analyses, advanced age (OR, 1.05; 95% CI, 1.01–1.10; p = 0.015), elevated baseline Any2 score (OR, 3.48; 95% CI, 1.98–6.13; p < 0.001) and ICH volume (OR, 1.10; 95% CI, 1.02–1.19; p = 0.013) were significantly associated impaired swallow on the 7th day (Figure 2(b)). Notably, age (OR, 1.11; 95% CI, 1.04–1.18; p = 0.001) and the initial Any2 (OR, 4.58; 95% CI, 2.15–9.76; p<0.001) score were also independent predictors for impaired swallowing function at 1 month, as well as imaging characteristics including midline shift (OR, 8.20; 95% CI, 1.18–57.16; p = 0.034) and lesions in basal ganglia (OR, 5.24; 95% CI, 1.07–25.72; p = 0.041, Figure 2(c)). Moreover, age (OR, 1.06; 95% CI, 1.01–1.10; p = 0.012), ICH volume (OR, 1.09; 95% CI, 1.02–1.16; p = 0.007), and Any2 (OR, 1.53; 95% CI, 1.03–2.27; p = 0.036) score exhibited significant correlations with SAP (Figure 2(d)).
Nomogram construction and performance
Predictive models for impaired swallowing function on the 7th day and 30th day, as well as SAP were constructed based on results from multivariate regression analyses and presented as nomograms, with C indices of 0.867 (95% CI, 0.802–0.928), 0.895 (95% CI, 0.822–0.959), 0.773 (95% CI, 0.658–0.876), respectively (Figure 3). Points were assigned to various variables, enabling direct estimation of probability by summing all points. Calibration plots demonstrated consistent agreement between the nomogram predictions and observed incidents (Figure S2).

Nomograms for predicting swallowing outcomes. (a) Impaired swallowing function on the seventh day. (b) impaired swallowing function on the 30th day. (c) Stroke-associated pneumonia. Impaired swallowing function was defined as FOIS ⩽ 6 and failed to return to premorbid state. ICH volume was stratified into tertiles. A vertical line can be projected from each variable to the “points” scale, and these points are then summed to obtain a “total points” score, which corresponds to a specific “probability” on the nomogram. C indices for the models are 0.867 (95% CI, 0.802–0.928), 0.895 (95% CI, 0.822–0.959), and 0.773 (95% CI, 0.658–0.876), respectively.
Discussion
Our results indicate that dysphagia is a relatively common complication following acute ICH, occurring in 18.3% of conscious patients, thereby underscoring its significance as a post-ICH issue that requires careful monitoring and management, even in patients with relatively mild symptoms. We identified clinical and imaging features significantly associated with post-ICH dysphagia and its outcomes, and developed predictive models that demonstrated favorable calibration and discriminatory power in forecasting swallowing impairment at different time points during the rehabilitation phase and SAP. These models provide valuable tools for forecasting dysphagia risk in acute ICH patients, thereby assisting healthcare providers in delivering timely interventions and personalized care strategies to enhance patient outcomes.
Stroke severity and age are well-established clinical predictors of post-stroke dysphagia and pulmonary infection, consistently validated across various stroke cohorts.23,24 In our study, age emerged as an independent predictor across all outcomes in multivariate analyses, exhibiting a marginal association with initial dysphagia. Conversely, NIHSS scores demonstrated a strong correlation specifically with initial dysphagia (Figure 2). These findings suggest that the influence of age on post-ICH dysphagia predominantly emerges during the rehabilitation period, whereas initial dysphagia is more closely related to stroke severity and the specific location of the hemorrhage. Elderly individuals are more susceptible to difficulties with deglutition due to physical and physiological factors that reduce both the variety and quantity of oral intake. Despite these challenges, their nutritional requirements for essential nutrients like vitamins, proteins, and minerals tend to increase. Therefore, it is crucial to monitor the nutritional status of post-ICH patients, especially among the elderly. 25
Previous studies have shown that post-stroke complications can extend the length of hospital stay. 26 However, in our study, no significant differences in mortality or hospitalization duration were observed, which may be due to the fact that less severe cases were more likely to be recruited at baseline. Notably, hemorrhagic lesions in the basal ganglia remained independent predictors of initial dysphagia and swallowing impairment on the 30th day, suggesting that this brain region may contribute to acute dysphagia and potentially lead to slower long-term recovery. HE, ICH volume, and midline shift were identified as independent predictors for initial dysphagia, impaired swallowing on day 7, and impaired swallowing on day 30, respectively. These findings underscore the predictive value of visually inspected brain imaging features in forecasting swallowing function at various stages post-ICH.
Formal dysphagia screening is recommended for all acute stroke patients, as failure to pass such screening is associated with poor outcomes, including pneumonia, higher mortality rates, and increased disability.27,28 However, fewer ICH patients received formal swallowing screening compared to ischemic stroke patients according to a hospital-based study, likely due to higher admission rates to intensive care units and greater stroke severity. 29 In our study, we only included individuals who could sit up and complete the bedside screening procedure. The Any2 score was identified as the strongest predictor of follow-up swallowing impairment and pulmonary infection, this simple assessment can be performed by doctors and nurses at the bedside. We emphasize that utilizing this tool, in conjunction with timely brain imaging, can effectively predict subsequent swallowing function. The predictive models developed in this study demonstrated favorable calibration and discrimination, underscoring their potential utility in the clinical evaluation of swallowing function and prevention of SAP. These models could aid in dietary planning decisions for patients with acute ICH.
The effect of lesion lateralization on post-stroke dysphagia remains unclear.12,30 In our study, we found the majority of clusters associated with post-ICH dysphagia were located in the left hemisphere, suggesting its potential predominance in controlling deglutition. In the SVR-LSM analysis, none of the clusters exhibiting voxel-wise significance survived cluster-wise FWE correction, which may be attributed to insufficient sample size and the complex pathophysiology of ICH. However, the hemorrhagic lesion located in the putamen identified in the SVR-LSM analysis also showed a significant correlation with initial dysphagia in the multivariate regression model (Table S1). Meanwhile, even though clusters in the insula, external capsule, thalamus, and internal capsule did not demonstrate a significant association with swallowing impairment after multiple statistical comparisons, previous studies suggest that stroke lesions in these brain areas may elevate the risk of post-stroke dysphagia due to their critical roles in regulating deglutition movements.31 –33 Therefore, visual inspection of these regions could serve as a crucial indicator for identifying ICH patients at high risk of dysphagia following ICH.
The prevalence of dysphagia is higher in hemorrhagic strokes compared to ischemic strokes.34,35 Furthermore, ICH is characterized by complex pathophysiological processes and secondary injuries, including space-occupying effects, perihematomal edema, and inflammatory responses. 36 Consequently, it is crucial to identify hematoma locations associated with dysphagia in ICH populations. Our results revealed that only a hemorrhagic lesion in the left putamen was an independent predictor of acute dysphagia. Given the complex structure of these regions, we hypothesize that only damage to critical connectivity can lead to deglutition dysfunction. Implementing an automatic segmentation program on baseline brain imaging could help identify strategic hematoma locations that carry a high risk of dysphagia. These regions could then be integrated into predictive models for post-ICH dysphagia, guiding clinical decision-making for dietary plans and early interventions. Future research could explore post-ICH dysphagia of different mechanisms using advanced brain imaging techniques and identify optimal strategies for its prevention and management.
This study has several limitations. First, we only included patients who remained conscious and were able to sit upright, thus those with larger ICH volumes and those with brainstem hematoma were prone to be excluded, this selection criterion also contributed to the relatively small sample size in our study. Furthermore, hematomas affecting the brainstem, including midbrain, pons, and medulla, did not show a significant correlation with dysphagia, which may also be attributed to the small sample size. Future studies incorporating larger sample sizes are needed to further investigate the effects of hemorrhagic sites and affected tracts on the development of post-ICH dysphagia. Second, although predictive models in this study demonstrated favorable predictive value in internal validation, external validation within a larger cohort is required. Lastly, in VLSM, we primarily focused on the hematoma site, influence of ICH-related pathophysiologies on post-ICH dysphagia should be evaluated in further investigations.
Conclusion
Post-ICH dysphagia is associated with stroke severity, HE, and basal ganglia involvement, while baseline aspiration risk serves as an independent predictor of swallowing impairment at 1 week and 1 month. Integrating these features may improve the prediction of swallowing function following ICH, thereby enhancing the prevention and management strategies for post-ICH dysphagia.
Supplemental Material
sj-pdf-1-tan-10.1177_17562864241311130 – Supplemental material for Predictors and lesion patterns of dysphagia and swallowing outcomes after acute intracerebral hemorrhage
Supplemental material, sj-pdf-1-tan-10.1177_17562864241311130 for Predictors and lesion patterns of dysphagia and swallowing outcomes after acute intracerebral hemorrhage by Xiao Hu, Min Wang, Zijie Wang, Yanfang Xie, Mengqiu Zhang, Shanyu Zhang, Tiannan Yang, Chuanqin Fang, Libo Zhao, Yanghua Tian and Qi Li in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
