Abstract
Background:
Amyotrophic lateral sclerosis (ALS) is a rapidly progressing and rare neurodegenerative disease. Therefore, evaluating the risk factors affecting the survival of patients with ALS is crucial. Constipation, a common but overlooked symptom of ALS, can be effectively managed. It is currently unknown whether constipation contributes to the progression and survival of ALS.
Objectives:
This study aimed to investigate the association between constipation and ALS development and survival using a novel overlap-weighted (OW) method to enhance the robustness and reliability of results.
Design:
This prospective matching nested case-control (NCC) study was conducted within an ongoing ALS cohort at the Second Hospital of Hebei Medical University. Baseline data were collected from patients meeting the inclusion and exclusion criteria, with constipation as the exposure factor. A 9-month follow-up was conducted, with death as the endpoint event.
Methods:
We primarily used the OW method in NCC studies to examine the association between constipation and ALS development and survival. Weighted Cox proportional hazards model was used to assess risk factors associated with overall survival. Survival differences between the two groups were analyzed using Kaplan-Meier’s plots and log-rank tests. Finally, the bioinformatic analysis explored common pathways between ALS and constipation.
Results:
Among the 190 patients included, the prevalence of constipation was 50%. Patients with ALS constipation exhibited faster disease progression (p < 0.001), with a positive correlation between constipation severity and progression rate (r = 0.356, p < 0.001). The constipation group had poorer survival before and after OW (log-rank test, p < 0.0001). In the Cox proportional hazards model of 114 patients, constipation was a risk factor for ALS both before (hazard ratio (HR) = 5.840, 95% confidence interval (CI) = 1.504–22.675, p = 0.011) and after (HR = 5.271, 95% CI = 1.241–22.379, p = 0.024) OW.
Conclusion:
Constipation in individuals with ALS is associated with faster disease progression and reduced survival rates, potentially through the peroxisome proliferator-activated receptor pathway.
Introduction
Amyotrophic lateral sclerosis (ALS), a motor neuron disease, is characterized by the degeneration of both upper and lower motor neurons, which leads to muscle weakness and atrophy.1,2 There is growing evidence that ALS is a multisystem disease, with 80% of patients reporting at least one non-motor symptom (NMS), 3 such as constipation, sleep disturbance, cognitive impairment, or salivation. Death occurs within 2–5 years of the onset of ALS motor symptoms, often as a result of respiratory failure, and there is currently no effective treatment. 4
Chronic constipation, which affects human health and daily life, is a functional gastrointestinal disease, and the older population is more likely to experience it than the younger population.5,6 The pathophysiology of constipation remains unclear and is generally believed to be a result of multiple factors, including age, dietary factors (such as insufficient fiber intake or low fluid intake), lifestyle factors (lack of mobility or sedentary lifestyle), intestinal neurodegeneration, microbiota disorders, drug stimulation and abuse, and psychological factors (anxiety/depression and sleep disorders).7–10 It is widely acknowledged that constipation can have a significant negative impact on patients, including psychological and physiological harm such as impaction, even perforation and death.11,12
Although there is currently limited literature on constipation symptoms in ALS, constipation is a common NMS in patients, with an incidence rate between 46.0% and 68.3%,13–16 which is much higher than that in the general population (14.0%). 17 Patients with ALS exhibit significant gut dysfunction, including delayed gastric and colonic motility.13,14,18 Previous articles have discussed some possible causes of constipation in patients with ALS. 19 In addition, patients with ALS have a high proportion of NMSs, including anxiety/depression, sleep disorders, and pain. Constipation is a common side effect of anxiolytics, antidepressants, sedatives, and analgesic drugs. Concurrently, NMSs and constipation are mutually causal,9,10 which can aggravate the incidence rate and disease severity of constipation in ALS. All of these factors are risk factors for constipation. As ALS progresses, the proportion of patients with constipation gradually increases. However, there is no research on the correlation between constipation and disease progression in ALS, which is a matter of significant concern.
Cytoplasmic accumulation of phosphorylated TAR DNA-binding protein 43 (pTDP-43) aggregates is a unifying proteinopathy in sporadic ALS.20,21 Studies have demonstrated pTDP-43 aggregates outside the central nervous system, as observed in muscle22,23 and peripheral nerve biopsies 24 in patients with ALS. Several TDP-43 animal models express pTDP-43 aggregates in cells of the myenteric plexus and exhibit intestinal abnormalities; some die suddenly from intestinal obstruction before the onset of neurological abnormalities.25–27 Furthermore, in the SOD1-G93A mouse model, we observed the aggregation of pathological SOD1 proteins in myenteric neurons accompanied by abnormal intestinal structure, myenteric neuron loss, and an excessively activated pro-inflammatory reaction in the intestine. 28 These findings support the hypothesis that gastrointestinal pathology occurs before motor symptoms in ALS, similar to other neurodegenerative disorders. 29 More importantly, it has been demonstrated that ALS and constipation share some pathogenesis that including oxidative stress, inflammation, and changes in metabolism and microbiota.30–34 Consequently, it is paramount to gain a more profound understanding of the interrelationship between the two.
Constipation is often ignored by individuals and their general practitioners as per epidemiological survey data of the general population. According to a 2016 YouGov survey of 2352 adults, 35% of patients with constipation indicated that they would wait for their constipation symptoms to resolve before seeking medical attention; only half of those surveyed thought it was not worth seeking medical help, and 39% considered it a minor health issue. 35 There is a similar dilemma exists in patients with ALS; clinicians and patients themselves attach low importance to it,36,37 and most patients use laxatives to relieve recurrent constipation symptoms. However, long-term use of laxatives can result in microbiota imbalance. Certain stimulant laxatives can damage intestinal nerves and reduce the sensitivity of the intestine to food residue stimulation, which can aggravate constipation, 38 accelerate the deterioration of ALS, and reduce the quality of life. According to Canadian management guidelines and the latest published European Academy of Neurology guidelines, improvements in NMSs can effectively improve patients’ quality of life.39,40 Constipation is a preventable and manageable illness that requires our attention.
As previously stated, constipation is influenced by many factors that are often associated with anxiety/depression and sleep disorders, and simultaneous treatment of all three symptoms is more effective than single-symptom treatment.41,42 However, there is currently insufficient research to determine whether it impacts disease progression and survival of patients with ALS. Therefore, the primary objective of this study was to examine the association between constipation symptoms and disease progression and survival outcomes in patients with ALS. We performed a nested case-control (NCC) study using the overlap-weighted (OW) propensity score (PS) proposed by Li et al. 43 to control for confounding factors and reduce bias. We used bioinformatics methods to identify common pathways between the two, ensuring an objective and precise description of the research findings.
Methods
Study design and participants
We conducted a population-based study using both cohort and case-control designs in parallel to preserve the advantages and complement the limitations of previous studies44,45; therefore, this research was designed as a prospective NCC study involving 491 participants from the “Hebei Province ALS cohort” launched in September 2019. In this cohort, the electronically recorded data contained information on patients with ALS attending The Second Hospital of Hebei Medical University, who were diagnosed with possible, probable, probable laboratory-supported, or definite ALS according to the revised El Escorial criteria (International Classification of Diseases, Tenth Revision (ICD-10): G12.2). The study period was from September 2019 to September 2023, and we followed the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
Inclusion criteria for patients with ALS were as follows: age 18–80 years old; forced vital capacity (FVC) >85%; no cognitive impairment (normal Mini-Mental State Examination and Montreal Cognitive Assessment scores). Exclusion criteria included patients with ALS-related gene mutations, such as those in SOD1, C90RF72, TDP-43, and FUS, as well as patients with frontotemporal dementia spectrum. Meanwhile, to ensure that patients with ALS maintain basic nutrition and water intake, we used the modified barium swallow to evaluate whether there it exists unsafe swallowing, if it exists, we consider percutaneous endoscopic gastrostomy, or nasogastric tube feeding if no other procedure was possible.39,46 In the event that the patient experienced difficulty swallowing and had not undergone surgical intervention, this was included in the exclusion diagnosis.
The sample size was calculated using the log-rank test using the PASS 15.0 software. 47 Given the lack of existing literature to inform the calculation, a preliminary experiment was conducted, resulting in a hazard ratio (HR) of 5.27. With power set at 0.8, alpha at 0.5, a follow-up period of 9 months, and a 5% dropout rate, the sample size for the case and control groups were determined to be 45 and 77, respectively, resulting in a total sample size of 122.
Finally, 301 patients were excluded, and 190 patients were included in the study. Concurrently, we gathered age- and sex-matched healthy volunteers (Table 1).
Demographic and clinical characteristics comparison between the normal group and ALS group, meanwhile between the non-constipation group and the constipation group.

p < 0.05. **p < 0.01. ***p < 0.001.
ALS, amyotrophic lateral sclerosis; ALSFRS-R, Amyotrophic Lateral Sclerosis functional rating scale; BMI, body mass index; FVC, forced vital capacity; KESS, Knowles-Eccersley-Scott Symptom Questionnaire; MiTos staging, Milano-Torino staging; PSQI, Pittsburgh Sleep Quality Index.
Follow-up
Follow-up was conducted from the date of inclusion in the cohort for 9 months or until the endpoint event, and the main outcome measure was survival. The endpoint was death (tracheotomy or tracheal intubation, equivalent to death). During the informed consent process, 40 patients declined further follow-up, while 150 consented to participate and continued with the follow-up assessments. Finally, 38 patients (25.3%) died, and 112 patients (74.7%) were still alive at the last follow-up.
Case-control selection
During the follow-up period after enrollment in the study, the ALS patients who had reached the endpoint event were designated as the case group. Each confirmed case was matched at a ratio of 1:2 in the control group based on age, sex, and body mass index (BMI). The control group comprises ALS patients who have not yet reached the endpoint event at the conclusion of the follow-up period. Finally, a total of 114 cases were collected, including 38 cases in the case group and 76 cases in the control group (Figure 1).

Flowchart of the establishment of the cohort and NCC populations.
Exposure
Constipation was the exposure of interest. It is defined in accordance with the diagnostic criteria set forth in Rome III criteria, 5 and no administration of opioids or antihypertensive agents and those not accompanied by organic diseases, including systemic diseases (such as hypothyroidism), or local pathology in the colon (such as colon cancer or diverticular stricture).
Data collection
The essential patient data included: (1) Age, sex, BMI, site of onset, FVC, 9 year compulsory education, and living environment. (2) The prevalence and severity of constipation were assessed using the Knowles-Eccersley-Scott Symptom Questionnaire (KESS). 48 ALS severity was assessed using the Amyotrophic Lateral Sclerosis functional rating scale (ALSFRS-R). The rate of disease progression (ΔFS) was calculated as the average monthly reduction in ALSFRS-R scores prior to enrollment in the experiment ΔFS = (48−ALSFRS-R)/symptom duration (months).49,50 Regarding functional deterioration, patients were categorized as “slow” (ΔFS <0.47/month), “moderate” (ΔFS 0.47–1.1/month), and “fast” (ΔFS >1.1/month). 50 Anxiety and depressive symptoms were assessed by using the Hospital Anxiety and Depression Scale (HADS). A score of >7 on the HADS anxiety or depression subscales indicated the presence of anxiety or depressive states, respectively, while scores ⩽7 were considered normal. 51 The Pittsburgh Sleep Quality Index (PSQI) was used to assess the sleep status of patients. 52 Scores ⩽5 were considered normal. To ascertain the survival stage of ALS, we employed the Milano-Torino staging (MiToS staging) method. Briefly, stage 0 is defined as no loss of function in any domain; stages 1–4 are defined as the loss of function in one to four domains; and stage 5 is defined as death. 53
Statistical analysis
All clinical data were statistically analyzed using SPSS v26.0 (IBM Corp., Armonk, NY, USA) and R 4.2.0 (R Core Team). Multiple imputations were used to impute the missing baseline covariates. Descriptive statistics were reported as counts and percentages, medians and interquartile ranges, or means and standard deviations.
We analyzed the full cohort data obtained from the matching NCC study. Among the 114 patients in the full ALS cohort, 38 died. The exposure of interest is constipation grouping, which is either “constipation group” or “non-constipation group.” 54 Then we balanced the baseline data between the constipation group and non-constipation group using the OW method. OW is a PS method that attempts to mimic the important attributes of randomized clinical trials, 55 and can generate an exact balance of the mean of every measured covariate when the PS is estimated using logistic regression to obtain more precise estimates with narrower confidence intervals (CIs) and greater statistical power. Specifically, a logistic regression was used to calculate the PS, with constipation (Y) as the dependent variable and age, sex, BMI, FVC, site of onset, ΔFS grouping, sleep grouping, anxiety grouping, depression grouping, 9-year compulsory education, living environment, and MiTos staging covariates. Where p (Y = 1|X) represents the probability of event Y occurring under the presence of covariates X1, . . ., Xk. Then we calculate the weights, for the treatment group, the weight is 1 minus the PS; for the control group, this weight is the PS. The impact of constipation on survival time was assessed through an observational study, wherein confounding arises when multiple covariates influencing the outcome also affect the exposure of interest, that is, constipation. Accordingly, an OW Cox proportional hazards model was employed to address the issue of multiple measured confounders. In contrast to the inverse probability-weighted Cox model, the OW Cox model may serve to minimize the potential exposure bias and improve the estimation precision of the HR. 44
Microarray profiles associated with ALS and constipation were obtained from the GEO database (https://www.ncbi.nlm.nih.gov/geo/). The GSE40438 series (GPL570 platform) contains gene expression data of lumbar motor neurons from four patients and oculomotor nerves from four patients, 56 and the GSE36701 dataset (GPL570 platform) contains gene expression data from rectal biopsies from donors with irritable bowel syndrome (n = 45) compared to controls (n = 21). 57 The “limma” package in R software was used to determine the differentially expressed genes (DEGs) between the case and control groups. Significant DEGs were thresholded at adj. p value < 0.05, p < 0.05, and |log2FoldChange| >1 or 2. A protein-protein interaction (PPI) network was constructed based on the STRING database (https://cn.string-db.org/). Cytoscape (version 3.7.2, Cytoscape Consortium, USA) was used to visualize the PPI network. The Cytoscape plug-in molecular complex detection (MCODE) was applied to the PPI network to explore key functional modules using the following selection criteria: K-core = 2, degree cutoff = 2, max depth = 100, and node score cutoff = 0.2. The CytoHubba plug-in of Cytoscape was used to identify hub genes from the PPI network, and five algorithms (Degree, MNC, MCC, EPC, and DMNC) were used to confirm the hub genes (top 15). Finally, we performed gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses to investigate the biological functional categories and underlying mechanisms of the common gene set (CGS) and hub genes.
All data plots were constructed using the R software. The “Weightlt” R package was used to calculate the standardized mean difference of variables before and after PS weighting. The “GGally” and “ggplot” R packages are used for Spearman correlation analysis, volcano plot, and barplot. The “Survival” R package was used for the Kaplan-Meier survival curve of all queues, and the “ggpubr” and “survminer” R packages were used for forest plots. The “VennDiagram” R package was used for Venn diagrams.
Results
Demographics and clinical characteristics
According to our inclusion and exclusion criteria, 190 patients with ALS (121 males and 69 females) were included, including 136 cases of limb-onset ALS and 54 cases of bulbar-onset ALS. Demographic characteristics are presented in Table 1. Statistically significant differences were observed in the PSQI, KESS, depression, and anxiety scores between the ALS group and the healthy group (p = 0.002, p < 0.001, p < 0.001, and p < 0.001, respectively). Additionally, compared with the non-constipation group, the constipation group was older (p = 0.001) and had a longer disease duration (p = 0.001). Moreover, the constipation group showed significantly higher scores in PSQI, KESS, depression, and anxiety compared to the non-constipation group (p < 0.001). Furthermore, significant statistical differences were observed in the ALSFRS-R score and ΔFS between the two groups (p < 0.001 and p < 0.001, respectively). These findings suggest that patients with ALS with constipation tend to have lower functional scores and experience more rapid disease progression, compared with patients with ALS without constipation.
Association of constipation symptoms with disease progression
We performed Spearman correlation analysis on the KESS, PSQI, depression and anxiety-related scores, ALSFRS-R scores, and ΔFS of the 190 patients, as shown in Figure 2. The results are summarized in Table 2. ALSFRS-R scores were negatively correlated with KESS (r = −0.585, p < 0.001), PSQI (r = −0.291, p < 0.001), anxiety (r = −0.251, p < 0.001), and depression (r = −0.366, p < 0.001) scores. Among these correlations, ALSFRS-R scores showed a particularly strong negative correlation with KESS scores. Conversely, ΔFS demonstrated a positive correlation with the KESS (r = 0.356, p < 0.001) and PSQI (r = 0.143, p = 0.049) scores, but no significant correlations were found with anxiety (r = −0.028, p = 0.701) and depression (r = 0.106, p = 0.145) scores.

Spearman correlation analysis between various scores.
Spearman correlation analysis between functional score and survival rate of ALS patients and various scale scores.

ALS, amyotrophic lateral sclerosis; ALSFRS-R, Amyotrophic Lateral Sclerosis functional rating scale; KESS, Knowles-Eccersley-Scott Symptom Questionnaire; PSQI, Pittsburgh Sleep Quality Index.
Furthermore, we found a significant positive correlation between the KESS scores and sleep (r = 0.506, p < 0.001), anxiety (r = 0.412, p < 0.001), and depression scores (r = 0.507, p < 0.001) (Figure 2).
Association of constipation symptoms with survival
We used Kaplan-Meier survival curves to examine the survival outcomes among the 114 patients of ALS and investigate the influence of potential risk factors on patient survival. Our results showed significant statistical differences (log-rank p < 0.0001) between the constipation group and the non-constipation group before and after OW (Figure 3(a) and (b)). Remarkably, constipation was highly prevalent (92.1%) in the 38 deceased patients with ALS. On the other hand, the presence of a moderate group (log-rank p < 0.0001), fast group (log-rank p < 0.0001), sleep group (log-rank p = 0.033), and depression group (log-rank p = 0.013) had an impact on patient survival, excluding anxiety (log-rank p = 0.58), as shown in Figure 3(c) to (f).

Kaplan-Meier survival curves for the primary endpoint were compared using the log-rank test. (a and b) There was a statistically significant difference between the curves in the constipation grouping before (p < 0.0001) and after (p < 0.0001) OW. (c–f) In ΔFS grouping, the sleep and depression grouping showed the same results (p < 0.0001, p = 0.033, p = 0.013), but not in the anxiety grouping (p = 0.58).
Constipation is considered a risk factor for the prognosis of ALS
Our analysis, which included single-factor, correlation, and Kaplan-Meier survival curves, revealed that constipation had a significant impact on patients with ALS. We first balanced the baseline data affecting constipation using OW to investigate whether constipation affects the prognosis of ALS and to exclude the effects of potential confounding factors. The results are presented in Table 3 and Figure 4. Simultaneously, we used a Cox proportional risk model to obtain specific HRs before and after OW (Figure 5). Besides the ΔFS grouping (moderate group before OW: HR = 32.513, 95% CI = 3.459–305.565, p = 0.002, moderate group after OW: HR = 39.670, 95% CI = 5.043–312.047, p < 0.001; fast group before OW: HR = 53.213, 95% CI = 5.642–501.877, p < 0.001, fast group after OW: HR = 54.641, 95% CI = 6.085–490.628, p < 0.001), site of onset (bulbar before OW: HR = 4.141, 95% CI = 1.666–10.292, p = 0.002, bulbar after OW: HR = 7.617, 95% CI = 1.534–37.815, p = 0.013), and MiToS staging (stage 5 before OW: HR = 12.481, 95% CI = 1.712–90.974, p = 0.012, stage 5 after OW: HR = 14.595, 95% CI = 1.029–206.941, p = 0.048), constipation was still identified as a risk factor for ALS, both before (HR = 5.840, 95% CI = 1.504–22.675, p = 0.011) and after (HR = 5.271, 95% CI = 1.241–22.379, p = 0.024) OW based on the findings.
Clinical characteristics of patients with ALS before and after propensity matching weighting in the constipation group and the non-constipation group.
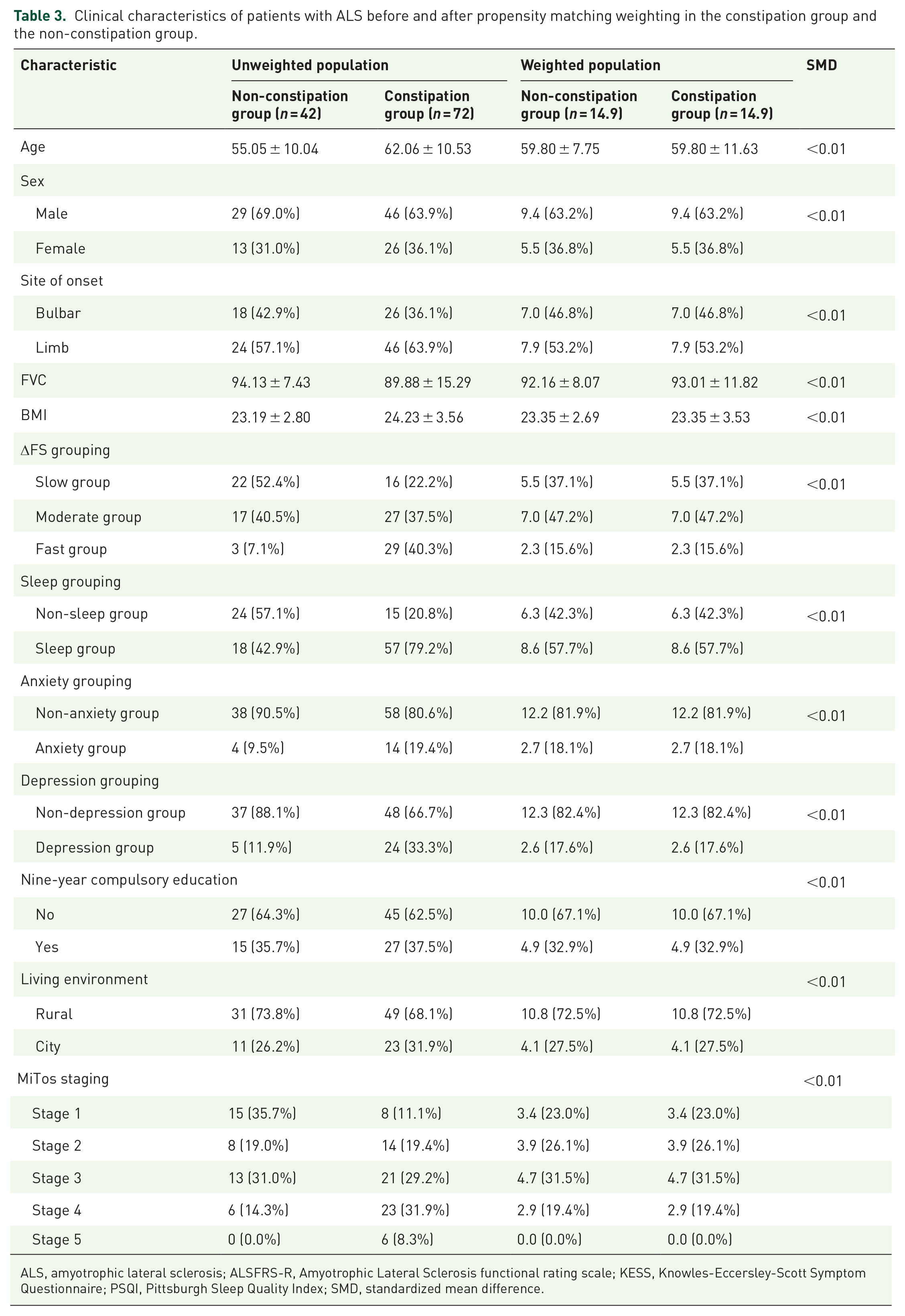
ALS, amyotrophic lateral sclerosis; ALSFRS-R, Amyotrophic Lateral Sclerosis functional rating scale; KESS, Knowles-Eccersley-Scott Symptom Questionnaire; PSQI, Pittsburgh Sleep Quality Index; SMD, standardized mean difference.

SMD of variables before and after propensity score weighting.

Forest plot of overall survival HRs of major subgroups in the cohort before (a) and after (b) OW.
Peroxisome proliferator-activated receptor pathway may be involved in ALS and constipation
We performed bioinformatics analysis to identify common regulatory genes and pathways between ALS and constipation and reveal the hub molecules and mechanisms involved in the co-occurrence of the two conditions. The GSE40438 (1779 DEGs) and GSE36701 (1719 DEGs) datasets were filtered from the GEO database (Supplemental Material 1(a) and (b)). The crosstalk genes between the two datasets were intersected using Venn diagrams, and 26 upregulated and 25 downregulated genes were identified (Supplemental Material 2(a) and (b)), suggesting that the pathogenesis of ALS and constipation might be similar. Then, GO enrichment and KEGG pathway analyses were carried out to understand further the CGS functional categories (Supplemental Material 2(c) and (d)). The results indicated that the negative regulation of proteolysis, neuronal dense core vesicles, enzyme inhibitor activity, and the peroxisome proliferator-activated receptor (PPAR) signaling pathway play vital roles in the progression of ALS and constipation.
Complex PPI networks are helpful in disease gene identification, gene function prediction, and drug treatment identification. 58 CGS was uploaded to STRING for PPI network construction with interaction scores >0.400, followed by visualization using the Cytoscape software (Supplemental Material 1(c)). The key cluster was identified using the MCODE algorithm based on the calculated scores (Supplemental Material 1(d)). Subsequently, the hub genes (top 15) were obtained using the CytoHubba plug-in based on degree algorithms (Supplemental Material 3), and 15 genes (FGF3, MMP1, CCR5, SERPIND1, SELP, KRT78, EGR2, CYP8B1, NPY, IFIT1, SERPINB5, ACADL, TGM1, PRDX5, and RPUSD2) were identified as hub genes (Supplemental Material 2(e)). Finally, GO enrichment and KEGG pathway analyses were performed on the hub genes, and the PPAR pathway was considered a key pathway (Supplemental Material 2(f) and (g)).
Discussion
Identifying modifiable risk factors for ALS is a promising approach to reduce the burden of this disease. To the best of our knowledge, this prospective NCC study represents the first attempt to investigate the relationship between constipation and mortality risk in patients with ALS. Constipation is a common NMS in patients with ALS and has received limited attention from researchers. Our study found that the incidence of constipation in patients is high at 50% (95/150), which is much higher than that in the general population (14.0%). 17 Indeed, the KESS score is based on a scale designed based on the More Stringent Rome III criteria; the use of the Roman IV standard will result in an even higher proportion of constipation cases. A more pessimistic finding was that only 24.7% (47/190) of patients were aware of their constitution, whereas <10.5% (20/190) of patients prioritized treatment for constitutional symptoms in our study. Similar to the general population, both healthcare providers and patients frequently overlook constipation; only one-third of individuals experiencing constipation seek formal medical consultation, and less than half take medications. 59 These findings suggest that constipation is not recognized and treated seriously despite occasional grave consequences.
Numerous investigations have indicated that individuals with constipation experience higher levels of anxiety, depression, and sleeplessness than those without the condition.60,61 It can be argued that anxiety/depression and sleep disorders predispose individuals to constipation and vice versa. Our study revealed that constipation, anxiety/depression, and sleep disorders are interconnected, implying that constipation may exacerbate anxiety/depression and sleep disorders and adversely affect the survival of patients with ALS. This interconnectedness of constipation, anxiety/depression, and sleep disorders is consistent with the results reported in previous studies. Therefore, it is important to note that NMSs are interdependent in ALS and that combined coping strategies are needed to achieve better outcomes.
As previously mentioned, various factors may cause constipation. Consequently, we employed a novel OW method that balances baseline data between the constipation group and the non-constipation group prior to regression. Our results are now more solid and reliable due to numerous simulation studies and illustrative data analyses in the medical literature that have compared the effectiveness of these weighted approaches with traditional epidemiological designs.62–64 Furthermore, our study showed that the OW results provided a more accurate description of the potential negative impacts of constipation on the prognosis and survival of patients with ALS. Importantly, the matching and weighting procedures lessened confounding factor bias, producing robust and comparable results. The results have attracted significant attention and highlight the potential distinctive impacts of constipation on the prognosis and progression of ALS patients.
The findings of our study indicate that site of onset, constipation, ΔFS grouping, and MiToS staging represent risk factors that influence the survival of ALS in Cox proportional risk model; other risk factors besides constipation have been reported in the literature. 65 After balancing the baseline variables that influence constipation using the OW method, we performed the Cox proportional risk model once more. The results confirmed that constipation is one of the risk factors for ALS, along with ΔFS grouping, site of onset, and MiToS staging.
Current research indicates that reduced physical activity is a cause of constipation in older adults, and reports suggest that active individuals are less likely to experience constipation than those with a sedentary lifestyle. 66 However, it may be challenging to increase the level of physical activity in patients with ALS, as exercising may be difficult due to muscle weakness and atrophy. Moreover, our results showed that patients with ALS and constipation exhibited lower ALSFRS-R scores than those without constipation. The ALSFRS-R score measures physical function in activities of daily living in patients with ALS. It is commonly used in research to measure the severity of the disease and track its advancement. 67 ΔFS that indicates a change in ALSFRS-R slope between disease onset and baseline. Similarly, the MiToS staging also reflects the progression of the disease. 53 We speculate patients with ALS and severe muscle weakness have a higher risk of constipation, which correspondingly affects their survival. Conversely, patients with constipation have poor intestinal function 68 and a poor intestinal microenvironment, 69 which promote chronic inflammation, oxidative stress, and other mechanisms,30–34 leading to faster disease progression, more severe muscle weakness, and shorter survival times. Additionally, ensuring adequate water intake is also necessary for normal bowel movements. 70 Scagnelli et al. 71 found that patients with bulbar-onset ALS are more likely to have inadequate total body water intake and water turnover than are those with limb-onset ALS. It can be reasonably deduced that both limb and bulbar onset have the potential to induce symptoms of constipation. In accordance with the findings of our previous research, 19 patients with limb onset may be more susceptible to constipation. Nevertheless, it can be confirmed that the onset of bulbar is a risk factor for the survival of patients with ALS. 72 In summary, our findings indicated that ΔFS grouping, MiToS staging, constipation, and onset of bulbar are risk factors for survival in patients with ALS.
Our study also found a correlation between constipation and ALS, with an increased risk of mortality in patients with ALS. However, a causal relationship between these two factors remains unclear. It is important to note that constipation can affect the progression of ALS and vice versa, creating a vicious cycle. In summary, our results showed that constipation is an independent risk factor for the survival of patients with ALS, and early detection of constipation symptoms may benefit these patients, as suggested by our previous dynamic prediction model of ALS constipation. 19 The role of the microbiota in ALS has been elucidated, 61 and it also affects the occurrence of constipation. 73 Therefore, the administration of probiotics to patients with high-risk ALS and early-stage constipation has shown benefits not only for constipation but also for ALS.74–76
Finally, as mentioned earlier, ALS shares some common pathogenic features with constipation.31–36 So we screened for some CGS and hub genes between ALS and constipation to find a common pathway between the two and make our results more reliable. GO enrichment and KEGG pathway analyses indicated that the PPAR pathway may be a common pathway between the two conditions. PPARs are ligand-inducible transcription factors that belong to the hormone nuclear receptor superfamily. 77 They are involved in various cellular processes, including cellular differentiation, apoptosis, inflammatory modulation, and attenuation of acute and chronic neurological damage in vivo and in vitro. 78 Some studies also indicate that ALS and constipation progression involve the PPAR pathway; PPARγ can be activated by lipid peroxidation metabolites in ALS motor neurons, and this can prompt the expression of antioxidant enzymes that exert a direct protective effect in ALS motor neurons, 79 and xylooligosaccharides can alleviate constipation symptoms through the PPAR pathway. 80 A PPARγ agonist not only protected transgenic mice carrying the ALS-related SOD1-G93A mutation from motor neuron neurodegeneration, improved muscle strength and body weight, and delayed disease onset, 80 but also improved the intestinal barrier, downregulated pro-inflammatory cytokines, and reduced free radical production through antioxidant mechanisms. 81 These findings support our conclusions.
Our study represents the first attempt to investigate the effect of constipation as a NMS on the prognosis and survival of patients with ALS, which is a significant strength. Additionally, we used a prospective matching NCC study design, multivariate analysis, and the OW method to control potential confounding variables, thereby increasing the validity and reliability of our results.
However, our study has some limitations that should be considered. First, the study was conducted at a single center and the sample size was relatively small. The conclusions derived from a single-center study are subject to referral bias. Future studies should include larger and more diverse groups of patients from different institutions to confirm our results and expand on our findings. Second, the dropout rate of patients was relatively high, and the disease progression rate was rapid. Third, the limited follow-up period and the low proportion of endpoint events (<50%) preclude the calculation of a median survival time. Fourth, while the OW method serves to balance some of the known confounding factors, it is unable to correct for unknown confounding factors. Finally, as the irritable bowel syndrome database (GSE36701) includes two types of constipation and diarrhea, but patients with ALS are mostly constipated, more accurate databases may be required for further verification.
Conclusion
In this prospective matching NCC study of a population-based cohort, we used a novel OW method and found that constipation, as an NMS in patients with ALS, had a detrimental effect on the rate of disease progression and overall survival. Therefore, objective assessment of these symptoms is critical for effective disease management. Additionally, implementing early intestinal management strategies could alleviate constipation symptoms and potentially enhance patients’ quality of life. Finally, we discovered that the PPAR pathway may be involved in constipation observed in patients with ALS.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241309811 – Supplemental material for An overlap-weighted analysis on the association of constipation symptoms with disease progression and survival in amyotrophic lateral sclerosis: a nested case-control study
Supplemental material, sj-docx-1-tan-10.1177_17562864241309811 for An overlap-weighted analysis on the association of constipation symptoms with disease progression and survival in amyotrophic lateral sclerosis: a nested case-control study by Tongyang Niu, Peize Wang, Xiaomeng Zhou, Tingting Liu, Qi Liu, Rui Li, Haitao Yang, Hui Dong and Yaling Liu in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
