Abstract
In acetylcholine receptor (AChR) antibody-positive generalized myasthenia gravis (gMG), neonatal Fc-receptor (FcRn) inhibition has broadened the therapeutic spectrum. Myasthenic crisis (MC), heralded by an impending myasthenic crisis (iMC), is a critical condition requiring treatments with rapid onset and sustained efficacy. Currently treatments used for iMC, including intravenous immunoglobulins and plasma exchange/immunoadsorption, have limitations, such as delayed onset of action and potential side effects. So far, there is limited data on the use of FcRn inhibitors in the management of impending or manifest MC (mMC). Here, we present a case of AChR antibody-positive gMG with iMC, where subcutaneous administration of the FcRn inhibitor efgartigimod resulted in rapid clinical remission. Within 24 h of administration, the patient exhibited significant improvement in respiratory and bulbar muscle function, preventing progression to manifest MC and the need for mechanical ventilation. This rapid response was accompanied by a marked reduction in AChR antibody level by 89.8% within 4 weeks. This case supports the potential of efgartigimod as a fast-acting and effective treatment option for managing iMC, offering an alternative to existing therapies.
Introduction
Generalized myasthenia gravis (gMG) is an autoimmune disorder affecting neuromuscular transmission due to autoantibodies and is characterized by fluctuating exertional weakness of voluntary muscles. 1 In the majority (85%–90%) of seropositive gMG patients, the postsynaptic nicotinic acetylcholine receptors (AChR) represent the target antigen, less-frequent targets are the muscle-specific tyrosine kinase (MuSK) and lipoprotein-related protein (LRP4). 2
In the pathophysiology of antibody-mediated disorders, as in myasthenia gravis (MG), the neonatal Fc-receptor (FcRn) plays a crucial role. It is a widely expressed major histocompatibility complex class I-like receptor capable of binding endogenous immunoglobulin G (IgG) and protecting them from lysosomal degradation resulting in an extension of IgG half-life. 3
The FcRn antagonist efgartigimod and monoclonal antibodies against the FcRn (i.e., rozanolixizumab, nipocalimab) are engineered to bind to FcRn; thus, reduce IgG recycling process, and lead to reduction of IgG levels, including pathogenic IgG antibodies in case of antibody-mediated diseases. 3
Few years ago, the approval of the FcRn inhibitor efgartigimod as an add-on therapy for AChR-antibody positive gMG has broadened the therapeutic spectrum. 4
Efgartigimod is a humanized IgG1 Fc-fragment with high affinity to FcRn and inhibits its function. In a phase II study, 5 all patients with AChR-antibody positive gMG treated with efgartigimod intravenously showed a rapid decrease in total IgG and AChR-antibody levels within 2 weeks. Three of four patients showed a prompt and sustained clinical improvement (i.e., reduction in Myasthenia Gravis Activities of Daily Living (MG-ADL) score ⩾2 points/Quantitative Myasthenia Gravis (QMG) ⩾ 3 points, significant reduction in Myasthenia Gravis Quality of Life scale revised (MG-QoL15r)) compared to 25% in the placebo group. Adverse effects were rare and mostly mild. 5
Safety and efficacy data have been confirmed in the phase III study ADAPT 6 that enrolled 129 AChR-antibody positive study participants with rapid clinical response (reduction of MG-ADL ⩾ 2, sustained for ⩾ 4 weeks) in 68% compared to 30% in the placebo group after the first dose and clinical response in 87% of study participants after further doses of efgartigimod. 6
Of note, in the phase II study, 5 patients with severe bulbar symptoms (MGFA, Myasthenia Gravis Foundation of America classification system, IVb), and in the phase II and III study5,6 patients with myasthenic crisis (MGFA V), as well as patients with prior changes in standard-of-care medication were excluded.
The ADAPT-SC trial, and ADAPT-SC+, an open-label extension study, showed noninferiority between subcutaneous (SC) efgartigimod 1000 mg and efgartigimod intravenously (IV) 10 mg/kg, as well as long-term safety, tolerability, and efficacy of SC efgartigimod for the treatment of gMG. 7
Myasthenic crisis (MC) is the life-threatening, maximal manifestation of MG, which affects up to 20%–25% of patients at least once during their lifetime and is characterized by rapidly progressive weakness.8,9 A manifest myasthenic crisis (mMC) is characterized by rapidly progressive weakness of the respiratory and bulbar muscles, culminating in aspiration and respiratory insufficiency necessitating intensive care treatment and (non) invasive ventilation.8,10 An impending myasthenic crisis (iMC) is defined by a rapid clinical worsening of MG that, in the opinion of the treating physician, could lead to MC in the short term (days to weeks). 10
Currently recommended immunotherapies in MC are plasma exchange (PE)/immunoadsorption (IA), intravenous immunoglobulins (IVIG), and glucocorticosteroids (GCS). 11 However, a number of patients does not respond sufficiently,3,12,13 within an acceptable span of time or with adequate tolerance. 13
So far, there have been only few reports on the use of intravenous (IV) efgartigimod in mMC und iMC with seven cases as an add-on (to PE/IA and/or IVIG) treatment14–19 and ten cases as first-line therapy, one case in iMC.14,15,20
Here, we present a case of AChR-antibody positive gMG experiencing iMC with prompt and sustained clinical response to SC efgartigimod as primary rescue therapy within 24 h, with a rapid decrease of IgG- and AChR-antibody levels, first measured after 1 week.
Methods
The case report is based on patient documentation during inpatient and outpatient visits at the University Hospital Augsburg, Germany.
To identify reports on the use of efgartigimod in MC, a systematic literature search was conducted in PubMed, September 1, 2024, with the search terms: ((“myasthenia gravis” [MeSH]) AND “efgartigimod”) AND (“refractory” OR “crisis”)).
Case report
A 54-year-old female with AChR-antibody positive gMG was admitted to our emergency department with clinical worsening, that is, exertional dyspnea, neck flexor weakness, and ptosis in August 2024.
In 2009, AChR-antibody positive gMG had been diagnosed with predominantly bulbar symptoms (AChR-antibody titer 18.6 nmol/L). In 2013, she underwent surgery for a malignant thymoma (WHO type B1, Masaoka stage lll, pericardial infiltration). Following radiation, there has been no recurrence until July 2024.
Since diagnosis, she had continuously been treated with prednisolone, with a dose ranging from 5 up to 40 mg daily (depending on her clinical condition), and pyridostigmine up to 180 mg daily, resulting in minor fluctuations with remissions after transient dose increases.
Within the last 9 months, myasthenic symptoms had gradually worsened, with intermittent double vision and right-sided ptosis, masticatory weakness, and exercise-induced dyspnea. For that, azathioprine had been started 3 months before admission at a daily dose of 150 mg by the outpatient neurologist; furthermore, prednisolone had been increased to 20 mg and pyridostigmine to 240 mg daily 2 weeks prior to hospital admission, without relevant effect.
At admission in August 2024, the patient was awake, exhibited exertional dyspnea, mild dysarthrophonia, neck flexor paresis, and ptosis. The patient was admitted to our monitoring unit. Pyridostigmine was continued intravenously, and prednisolone increased to 30 mg per day. Further up-titration of pyridostigmine did not lead to clinical improvement, but higher doses of pyridostigmine (up to 12 mg per day IV) were not tolerated due to gastrointestinal side effects. Despite treatment intensification, within 3 days after admission, the patient experienced significant myasthenic exacerbation, including dyspnea at rest. Vital capacity was 0.7 L (previously recorded highest value 1.83 L, June 2024), and the single count breath test (SCBT) yielded a score of 20 (23, at admission). Furthermore, she developed dysphagia with high aspiration risk necessitating oral withholding and dysarthrophonia (MGFA lVb, QMG 18 points, MG-ADL 10 points, MG-QoL 43 points). Immunotherapies for iMC, that is, IVIG and PE/IA, were comprehensively discussed with the patient; additionally, experiences with the fast and positive effect of FcRn and complement inhibition in severe gMG. PE/IA and IVIG were declined by the patient for invasiveness of PE / IA and treatment duration of several days regarding both treatment options, despite her poor clinical condition. After thorough education of the patient about the fact that there are only case-based experiences with its use in similar situations, treatment with efgartigimod was decided. After obtaining informed consent, efgartigimod was administered at a dose of 1000 mg SC.
Within 1 day, dysphagia, dysarthrophonia, and dyspnea markedly improved. The patient was allowed to resume eating, vital capacity increased to 1.7 L, and the SCBT yielded a score of 30. After 1 week, dysarthrophonia, ptosis, and neck flexor paresis had improved. Treatment was tolerated without side effects. The patient received the second dose of efgartigmod after 7 days and was discharged in nearly complete remission (MGFA llb, QMG 3 points, MG-ADL 3 points, MG-QoL 16 points, vital capacity 2.0 l) 9 days after the initial efgartigimod administration, with pyridostigmine 240 mg, prednisolone 30 mg, and azathioprine 150 mg daily. Remission was sustained, when the patient returned for the third and fourth efgartigimod injection 2 and 3 weeks after treatment start and 1 week thereafter (Figure 1(a)).
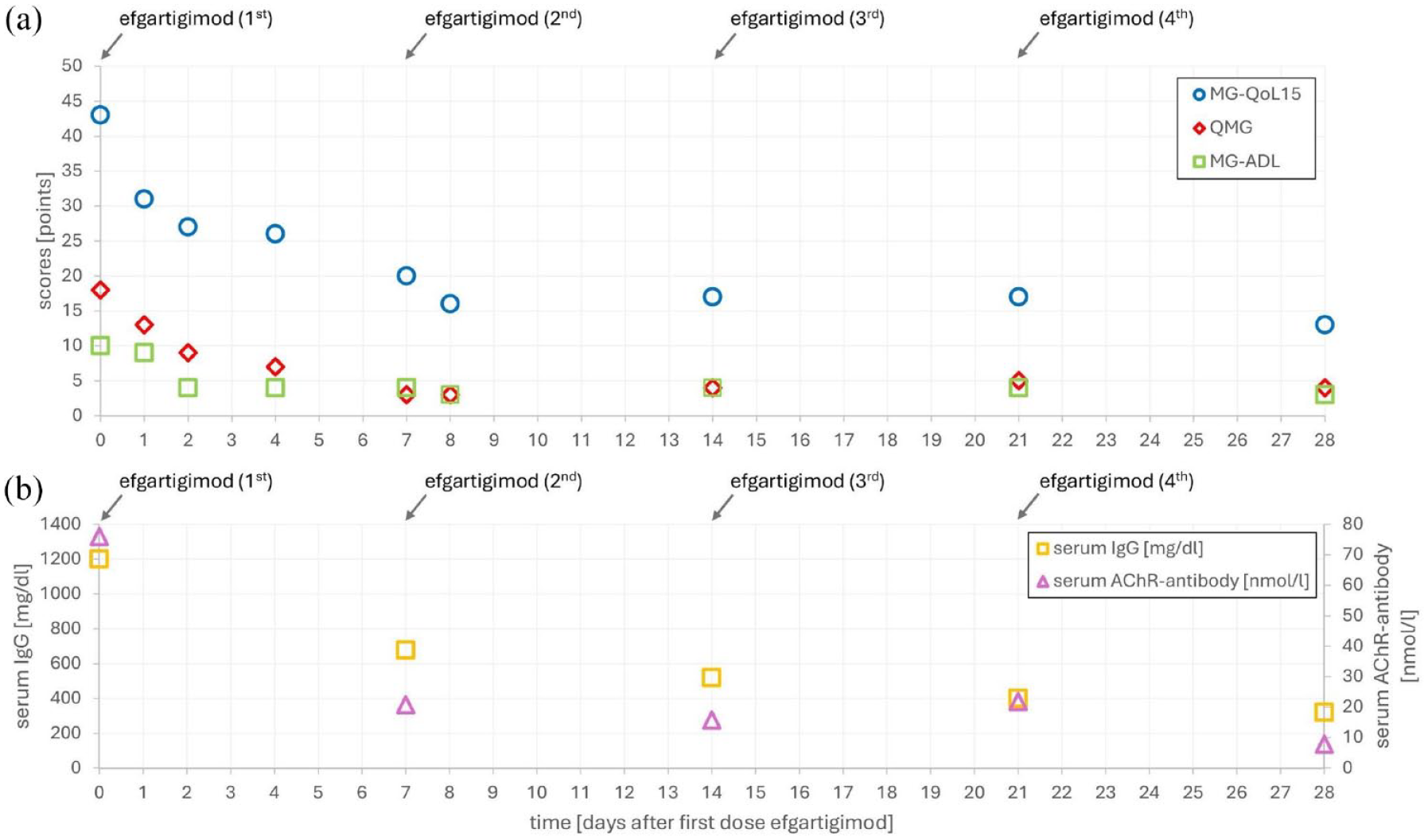
Time course after efgartigimod subcutaneous administration. (a) Clinical course: myasthenia Gravis-Quality of Life 15, MG-QoL15; quantitative myasthenia gravis score, QMG; myasthenia gravis-specific activities of daily living scale, MG-ADL and (b) Serum levels of acetylcholine receptor (AChR)-antibodies and immunoglobuline G (IgG).
As expected, 6 serum IgG levels decreased from 1200 mg/dL (reference 700–1600 mg/dL) to 678 mg/dL 7 days, and to 320 mg/dL 28 days after the first efgartigimod injection (Figure 1(b)). In parallel, AChR-antibody levels dropped from 76.0 nmol/L to 20.8 nmol/L 7 days, and to 7.8 nmol/L 28 days after the first efgartigimod injection (Figure 1(b)).
Between 1 day and 5 weeks after first efgartigimod SC injection, total lymphocytes increased from 1.5 to 3.7/nL, CD19+ B-cells (7.2% vs 7.4%), CD3+ T-cells (83.4% vs 87.4%), CD4+ T-cells (23.5% vs 26.4%), and CD8+ T-cells (59.8% vs 59.3%) remained stable, CD3-/CD56+/CD16+ natural killer (NK) – cells decreased (9.6% vs 4.4%).
In the follow-up, azathioprine was reduced from 150 to 100 mg daily due to liver enzyme increase diagnosed 7 weeks (normal at week 6) after first efgartigimod administration (initially, and during follow-up, ALT and AST <2-fold, gamma-glutamyl transferase <5-fold of upper limit of normal, bilirubin normal) with stable values thereafter. For further stabilization, and after azathioprine had been reduced, she received a second cycle efgartigimod consisting of four weekly injections à 1000 mg SC, starting 50 days after the first injection. With this regimen, she remained mainly stable (MGFA llb, QMG 7 points, MG-ADL 1 point, MG-QoL 20 points, when receiving the last efgartigimod injection). Prednisolone was reduced to 17.5 mg.
Discussion
PE/IA, IVIG, and GCS, alongside symptomatic and supportive therapy, are the basic principles of iMC and mMC treatment. 10 In recent years, complement inhibition as well as inhibiting the FcRn have broadened the therapeutic spectrum in gMG. Efgartigimod and the recently approved rozanolixizumab, bind to the FcRn, resulting in a rapid reduction of circulating IgG antibodies, including pathogenic autoantibodies in MG and a prompt clinical improvement within 1–2 weeks.6,21,22
In the present case, myasthenic symptoms markedly improved within 1 day after starting efgartigimod SC without prior treatment with IVIG or PE/IA, preventing mMC. Although the patient had been diagnosed with thymoma-associated myasthenia gravis, typically more treatment-resistant than nonthymoma-related MG, 23 she greatly benefited from treatment.
In the literature, there are 17 reports on the treatment of iMC (n = 2) and mMC (n = 15) by efgartigimod IV in AChR-antibody positive gMG (Table 1). In 7 of 17 cases, patients had been treated with either IVIG or PE/IA or both before.
Effect of efgartigimod in iMC and mMC.

Improvement refers to MG-ADL/QMG change from last reported scores before efgartigimod administration and first scores thereafter.
AChR-antibody follow-up: first and last reported value.
Nine AChR-antibody positive and one anti-muscle-specific kinase positive patients.
AChR, acetylcholine receptor; iMC, impending myasthenic crisis; MG-ADL, myasthenia gravis activities of daily living; mMC, manifest myasthenic crisis, QMG, quantitative myasthenia gravis.
In the cases published so far, variable intervals for a meaningful clinical improvement have been reported, ranging from 2 days to 3 weeks (Table 1). Two patients receiving efgartigimod as a first-line rescue therapy, demonstrated a swift clinical response, allowing them to be successfully weaned off bilevel positive airway pressure ventilation within 5 days and oral intubation within 14 days. 14
GCS, often also used for treatment intensification, typically exert effects within 2–4 weeks in approximately 75% of patients. 1 How fast IVIG unfold effectiveness, is a subject of an ongoing debate. Some studies suggest a brief duration, typically around 4–5 days 13 or 7–10 days, 24 while other reports indicate that the peak effect may be observed after approximately 20 days.19,25 For PE, a treatment response as soon as after 7 days has been reported. 26 Besides, PE may be followed by a rebound of anti-AChR antibodies that may result in a secondary clinical deterioration. 27 Of interest, a multicentre study in MC revealed that weaning failure occurred more often in patients treated with first-line IVIG (50%), and less often first-line PE/IA (29%) showing that current first-line treatments for MC are not sufficiently effective in a number of cases. 28
This is the first report indicating that efgartigimod, in our case given SC, may have a very rapid onset of action even within 1 day. It has been shown that after efgartigimod administration in humans, IgG1 levels decreased from 1 or 2 days depending on the dose given, thus enabling its rapid effect. 29 It may be speculated that the fast effect of efgartigimod in our case within 1 day compared to other published cases may be related to the variable number of tandem repeat (VNTR) polymorphism of the FCGRT gene. The FcRn is encoded by FCGRT, a gene located on chromosome 19. Five different alleles with VNTR polymorphisms in the FCGRT gene result in different expression levels of FcRn mRNA and protein levels. The most common allele, VNTR3, is associated with an increase in promoter activity raising FcRn level in homozygous VNTR3/3 individuals. VNTR3 homozygous patients with normal circulating IgG levels have a lower monoclonal antibody distribution clearance compared to other polymorphisms. Based on that, it has been considered if efgartigimod results in a greater reduction in AChR antibody titers and has a better efficacy in VNTR3 homozygous patients. 3 However, the polymorphism has not been investigated in our case.
Efgartigimod is generally well tolerated, with common side effects being mild including infusion-related reactions and transient infections. 30 In the present case, no side effects occurred confirming the good tolerability in iMC or mMC with manageable side effects like dyspnoea, 15 headache,14,20 upper respiratory infection (n = 1), and urinary tract infection (n = 2). 14
High-dose IVIG and PE/IA may also have potential side effects, and for PE/IA a central venous catheter is often necessary, making treatment more invasive and burdensome for the patient.
In comparison to IVIG, efgartigimod is associated with a lower risk of thrombosis and hemolysis. 4 Regarding application, efgartigimod SC is easy to administer within only 90 s.
Song et al. reported that serum IgG levels (n = 8) were reduced by 50.9% and AChR-antibody levels (n = 6) by 42.0%. However, two patients exhibited increasing AChR-antibody levels despite a decrease or stabilization of IgG following efgartigimod treatment. 14 In another case report decreasing AChR-antibody titers after efgartigimod initiation were described with a slight increase 2 weeks later. 16
In the present case, clinical improvement was accompanied by a reduction of serum IgG and AChR-antibodies by 73.3% and 89.8% after 28 days, respectively. This reduction exceeds findings described in most other reports (Table 1).
Concerning economic aspects, IVIG have limited availability due to dependence on plasma donors resulting in increasing costs. 31 In Germany, the net price for efgartigimod 1000 mg for SC use is 14,402.34 Euros (in August 2024, according to the manufacturer), making it a comparably costly treatment. However, this must be weighed against the total costs of IVIG and PE/IA treatment, also considering the length of hospital stay mainly in intensive care or monitoring units.
In some reports, not consistent effects on T-cell subsets and CD19+ B-cells have been reported.14,15 In the study by Zhang et al., a gradual decrease in total T-cells and CD4+ T-cells from the second week was observed; furthermore, after a gradual decrease of CD19+ B-cells from the first week after treatment, an increase of CD19+ B-cells 69 days after the last injection. 18
In our case, total lymphocytes increased between 1 day and 5 weeks after the first efgartigimod SC injection, but percentages of lymphocyte subsets remained stable. NK-cells showed a decrease, its significance however remains unclear. Although inhibition of FcRn can also reduce T-cell activation, 32 these findings at the cellular level are too preliminary and in part not consistent to draw meaningful conclusions.
Of interest, besides efgartigimod, complement inhibition has been discussed as a potential rescue therapy in MC as well. So far, there is limited data with few reports indicating rapid improvement after administration of eculizumab33–36 or ravulizumab, 37 with eculizumab being effective within approximately 1 week34,36 and ravulizumab resulting in hospital discharge after about 2 weeks. 37 Based on these reports, complement inhibition may be another fast-acting option in MC.
Limitations of this case report include the sample size of only one patient and the absence of previous iMC or mMC episodes in the patient, resulting in lacking information on the potential effects of IVIG or PE/IA.
In our case, clinical improvement was observed very early. However, due to currently very few published reports on the rapid onset of clinical improvement after efgartigimod, this observation is premature and must be interpreted with caution.
Conclusion
Efgartigimod, with its rapid onset of action, combined with an acceptable safety profile, and easy application, may evolve as a promising option for the management of therapy-refractory MC and iMC. Therefore, inhibiting the FcRn may enrich the therapeutic armamentarium in the management of severe gMG, encompassing a range of benefits over the currently available treatment options.
