Abstract
Background:
Accumulating evidence supports a role of the microbiota in health and disease, including in multiple sclerosis (MS). How MS drugs affect the microbiota and whether this is part of their mode of action is yet unknown.
Objectives:
To assess how dimethyl fumarate (DMF) affects the gut microbiota and whether the microbiota is associated with clinical response or adverse events (AEs) to DMF or diet.
Design:
An observational cohort study, in which the microbiota from 45 patients with relapsing-remitting MS pre-DMF initiation and following 6 months of DMF therapy, and from 47 matched healthy controls, were compared, and associations with clinical and dietary data assessed.
Data sources and methods:
Microbial DNA was sequenced and analyzed using MicrobiomeAnalyst. The clinical response was assessed after 1-year DMF therapy based upon evidence of disease activity (relapse, ΔEDSS increase >1, or MRI activity compared to pre-treatment). Dietary data were obtained by food questionnaires.
Results:
Alterations in relative abundance of several microbes were identified post 6-month DMF therapy compared to pre-treatment, including an increase in Firmicutes, Lachnospiraceae, and Ruminococcaceae, while reduction in Bacteroidetes and Proteobacteria. Patients who showed disease activity within 1 year from DMF initiation had pre-treatment higher abundance of Proteobacteria, Flavonifractor, and Acidaminococcaceae, while lower abundance of Firmicutes, Ruminococcaceae, Butyricicoccus, and Massiliprevotella massiliensis, compared to patients without disease activity. Patients who discontinued DMF therapy due to AEs had pre-treatment higher abundance of Proteobacteria, Bacteroidetes, Eggerthella, and Lachnoclostridium and lower abundance of Ruminococcaceae, Megamonas, and Holdemanella, among others. Differentially abundant microbes correlated with intake of several nutrients.
Conclusion:
DMF immunotherapy is associated with modifications of the microbiota. The microbiota may affect the severity of AEs and the clinical response to DMF, and is potentially modulated by diet. Microbiota-based, personalized treatment approach, integrating pharmacotherapy with dietary components, carries potential to improved clinical outcome.
Keywords
Introduction
Multiple sclerosis (MS) is an autoimmune, neurodegenerative disease, characterized by an aberrant immune response targeting the myelin sheet, causing demyelination, axonal, and neuronal loss. MS is a complex, multifactorial disease driven by interactions between genetic and environmental factors, such as smoking, obesity, Epstein–Barr virus infections, Vitamin D deficiency, etc. Growing evidence suggests that the gut microbiome plays a role in diseases such as MS, through the gut–brain axis of interacting substances and neural communication.1,2 The gut microbiome affects both the development and activity of the immune system, as well as gut permeability and function, production and release of immune-mediating substances such as microbial-associated molecular patterns, 3 short-chain fatty acids (SCFAs), and tryptophan/serotonin, 4 besides additional roles in energy and vitamin production. The microbiota composition of the individual develops during early childhood, influenced by host genetics and environment, but may undergo modifications later on in life, temporarily or stable, due to significant changes of lifestyle, diet, medications, age, or disease.2,5
In recent years, several publications have presented differences in the microbiota in patients with relapsing-remitting MS (PwMS) compared to healthy individuals,4,6–11 however with relatively low overlap between studies, not enabling the definition of a specific MS-microbiome signature. We have recently published a comparison of the microbiota between treatment-naïve PwMS and healthy individuals in the Israeli populations, where we found several microbes at different taxonomic levels to differ between cases and controls. 12 We also showed that the MS-associated microbiota seems in part to be population-specific, and that the MS-differential relative abundance of some microbes is associated with diet, specific nutrient intake, Vitamin D level, age, and gender, suggesting that the microbiota may serve a comprehensive and complex role in MS pathogenesis and pathology. In continuation to the published study, we investigate the effects of various MS disease-modifying therapies (DMTs) on the microbiota. Recent studies suggest that while DMTs seem not to affect the general microbiota composition, they may alter the abundance of specific microbes.9,13–16 Whether alteration of the microbiota is part of the beneficial immunomodulatory effects of some of the DMTs available for PwMS, or whether it is a byproduct, with either positive, neutral, or negative effects on the patient’s health, is unknown. The current observational, cohort, longitudinal study focused upon the effect of dimethyl fumarate (DMF) therapy on the microbiota, possible associations between microbiota alterations and clinical response, occurrence of adverse events (AEs) leading to drug discontinuation, and association with dietary data.
DMF is an orally administered fumaric acid ester, cleaved in the intestine by esterases to monomethyl fumarate (MMF), the main active metabolite. DMF acts through both neuroprotection, upregulating the nuclear factor (erythroid derived 2)-like 2 (Nrf2) anti-oxidative pathway, and through immunomodulation, by inhibition of the nuclear factor-kappa B pathway, which mediates pro-inflammatory signaling. 17 We and others have previously shown that DMF increases the proportions of anti-inflammatory and regulatory immune cells, while reduces inflammatory cells.18–21 Transient mild to moderate flushing and gastrointestinal (GI) AEs, such as nausea, vomiting, diarrhea, and abdominal pain, are commonly reported in PwMS treated with DMF, most frequently in the first month of treatment before declining.22,23 These DMF-associated AEs can be intolerable for some PwMS affecting the adherence to therapy and may lead to drug discontinuation. Some effects of DMF therapy on various microbes in PwMS have been described,13,16,24–27 however with very low repetition across studies.
Methods
Recruitment and sample collection
PwMS (N = 45) about to initiate DMF therapy, including 33 treatment-naïve PwMS, as well as 47 age- and gender-matched healthy controls (HCs), were recruited at an MS Center in Carmel Medical Center, Israel. The study was conducted under a protocol approved by the Institutional Ethical Review Board (0034-13-CMC), and all participants signed a written informed consent.
Inclusion criteria for PwMS were a diagnosis of relapsing remitting MS according to revised McDonald criteria,28,29 and being about to initiate DMF therapy. Healthy participants were recruited among PwMS’ escorts to the clinic (mainly unrelated spouses or acquaintances) and hospital’s visitors, who had no known relative with MS up to 2nd degree. Inclusion criteria for all participants were age 18–55 years, without antibiotics/probiotics/corticosteroids treatment within the last month, and no irritable bowel disease (IBD), other autoimmune disease, or history of gastric/bowel surgery. The cohorts included fecal samples from 30 PwMS and 43 HC, who were part of the cohorts examined in our recent published study comparing the microbiome of PwMS (N = 57) and healthy individuals (N = 43) in the Israeli populations. 12 PwMS initiated oral DMF treatment according to standard regime (starting dose of 120 mg twice a day for 7 days, then maintenance dose of 240 mg twice a day). Fecal samples were collected at recruitment and 6 months after DMF initiation, using a stool preservative tube (Norgen Biotek, Thorold, ON, Canada), frozen immidiately upon arrivel to the clinic and kept at −80° until DNA extraction. All participants completed a food frequency questionnaires (FFQ) and a Mediterranean Diet Score (MDS) questionnaire, both especially adapted to the Israeli population. The FFQs were analyzed at the Department of Public Health, Faculty of Health Sciences, Ben-Gurion University of the Negev, Israel, for daily intake of energy and 74 various nutrients.30–32 The MDS is a 17-item Mediterranean Diet adherence screener.33–35 Demographic and clinical data was obtained, including Vitamin D level at recruitment, body mass index (BMI), level of disability (Expanded Disability Status Scale, EDSS) pre-treatment initiation and during 1-year follow-up, annual relapse rate calculated as mean over the last 2 years prior to recruitment, relapses during 1-year follow-up since DMF initiation, evidence of disease activity on MRI after 1-year follow-up compared to MRI pre-DMF treatment and drug discontinuation due to flushing and GI AEs.
Clinical response definition
PwMS who discontinued DMF therapy within 1 year due to flushing and GI AEs were defined as patients with intolerable AEs (IAEs). PwMS who continued DMF therapy, despite any GI AEs, were defined as patients without IAE.
Clinical response was assessed after 1-year DMF therapy based upon any evidence of disease activity (relapse, MRI activity, and/or ΔEDSS increase >1). PwMS were defined as having no evidence of disease activity (PwNDA) if they had no relapse, no evidence of disease activity on MRI (no worsening compared to previous MRI, e.g., no new or enlarged lesions and no Gadolinium enhancement, according to the summary provided by a Radiologist), and no ΔEDSS increase >1. PwMS who had a relapse within 1 year of initiating DMF, any evidence of disease activity on MRI compared to the previous MRI (pre-DMF) or ΔEDSS increase >1 were defined as patients with disease activity (PwDA).
Microbial DNA extraction
Microbial DNA sequencing was performed as previously described. 12 DNA was extracted using the QIAamp® PowerFecal® Pro DNA kit (Qiagen, Tegelen, The Netherlands), according to manufacturer’s instructions. DNA 16s rRNA sequencing was performed at Hy Laboratories Ltd (Rehovot, Israel). The V3V4 region of the 16s rRNA gene was amplified using primers from The Earth Microbiome Project. Access Array primers for Illumina (Fluidigm, San Francisco, CA, USA) were used to add adaptor and index sequences and the reactions cleaned using Kapa Pure beads (Kapa, Roche, Basel, Switzerland). The concentration of each library was measured by Qubit (Life Technologies, Waltham, MA, USA) using the DeNovix dsDNA high sensitivity assay (DeNovix, Wilmington, DE, USA) and samples pooled. The pooled library was sequenced on Illumina Miseq using a Miseq v2 Kit (Illumina, Eindhoven, The Netherlands) to generate 2 × 250 PE reads, at a depth of 100,000 reads/sample. Reads were trimmed for adaptor sequences and quality, paired reads were merged and subjected to operational taxonomic units (OTUs) de novo picking against the SILVA database at >97% sequence similarity using the CLC-bio software (Qiagen); 5773 OTUs were identified.
Statistical analysis
Differences between groups in gender, age, ethnicity, MDS, BMI, proportion of smokers and vegetarians, and the average intake of 75 nutrients between groups were assessed by the Mann–Whitney U-test or Kruskal–Wallis test for continuous or ordinal variables and by Fischer’s exact test for nominal, categorical variables, using IBM SPSS statistics (v28) (Armonk, NY), at p-value < 0.05. Analysis of MDS in patients after 6 months of DMF therapy compared to pre-treatment was assessed by the Wilcoxon paired test or by the Friedman test (MDS groups). Differences in MDS or specific nutrient intake between PwDA and PwNDA or between patients with IAE and patients without IAE were assessed by the Mann–Whitney U-test, at p-value < 0.05.
Statistical analysis of microbiota data was performed with the MicrobiomeAnalyst web-tool version 2.0 (Xia Lab, McGill University, Montreal, QC, Canada), using the Marker Data Profiling (MDP) Module, according to published protocols.36–38 OTUs were filtered to include only features with ⩾4 counts in at least 10% of samples. Testing for differences in α-diversity across groups was assessed using the Shannon alpha index (Kruskal–Wallis test), comparing OTU richness and evenness, while β-diversity was calculated using the Bray–Curtis dissimilarity (PERMANOVA) comparing the similarity and distance between samples, on data normalized by total sum scaling (TSS). Differential relative abundance analysis of individual taxa between PwMS and HC was performed as previously published. 12 Differences in relative abundance of microbes between patients post 6-month DMF therapy compared to pre-treatment, was performed using multiple linear regression with adjustment for subject ID (paired analysis). As a complement, unpaired analyses were performed using DeSeq2 (normalization by Relative Log Expression), adjusted for multiple testing, at false discovery rate (FDR) < 0.1, and the Linear Discriminant Analysis Effective Size (LEfSe) tool (on TSS normalized data), which identify taxa that best discriminate between groups, at p-value < 0.05 and linear discriminant analysis (LDA) ⩾±2. The relative tolerant FDR reflects the exploratory nature of the study. Only microbes with significant difference in the paired analysis, or in both two unpaired analyses, were considered differential.
Differences in relative abundance of specific microbes (in pre-treatment samples or in samples after 6-month DMF therapy) between PwNDA and PwDA were calculated using two statistical tools: Linear Regression and DeSeq2, both adjusted for multiple testing at FDR < 0.1, as well as by LEfSe, at p < 0.05 and LDA ⩾ 2. Differences in relative abundance of specific microbes (in pre-treatment samples) between patients with IAE and patients without IAE were calculated by Linear Regression at FDR < 0.1, by DeSeq2 at FDR < 0.05 (to reduce the risk of false positive due to small number of IAE cases), and by LEfSe at p < 0.05 and LDA ⩾ 2.
Spearman correlations between the relative abundance of the differential microbes, identified in the various analyses, and the dietary data (MDS, Vitamin D, or daily nutrient intake) or disease severity (EDSS), were assessed using IBM SPSS statistics (v28), at p-value < 0.05 (data normalized by TSS).
Functional analysis
To predict metabolic functions and pathways affected by DMF therapy (from the identified altered microbiota) the Tax4Fun2 package was used, 39 under the MDP Module in the MicrobiomeAnalyst web tool. The tool was applied to all OTUs, filtered for ⩾4 reads in ⩾10% samples, scaled by TSS, and then functions predicted by Tax4Fun2 using the Kyoto Encyclopedia of Genes and Genomes (KEGG) orthologies (KOs). The KO abundance table was uploaded into the Shotgun Data Profiling module for differential pathway analysis, enrichment analysis, and metabolic network mapping. KOs were filtered for ⩾4 reads in ⩾10% samples and scaled by cumulative sum scaling. Functional metabolic pathways differing between PwMS post 6-month DMF therapy compared to baseline were identified using multiple linear regression adjusted for pairs, at FDR < 0.05, and the significant KOs mapped to enriched networks at FDR < 0.05. Similar differential pathway analysis between PwNDA compared to PwDA was performed at FDR < 0.05 and mapped for enrichment at p < 0.05. LEfSe was performed on KOs to detect metabolic functions that best distinguished between groups at FDR < 0.05 and LDA >±0.65.
Results
Demographic and clinical data of participants
Forty-five PwMS about to initiate DMF therapy, as well as 47 age- and gender-matched HC were recruited to this study (study design presented in Figure 1). Fecal samples were available from all participants at recruitment, and from 32 of the PwMS following 6 months of DMF therapy. Demographic and clinical data are presented in Table 1. PwMS included 33 treatment-naïve patients (1st line patients), and 12 2nd line patients, with a mean time since last DMT of 26 months. The mean disease duration was 2.4 months, reflecting the considerable majority of 1st line patients, recently diagnosed. There was no significant difference between PwMS and HC in age, gender, BMI, and frequency of smoking or of vegetarians. However, there was a significant difference in ethnicity between the cohorts, with more Jews and less Arabs in the HC cohort (p = 0.028), yet this was irrelevant for the longitudinal study of PwMS, following DMF initiation. There was no difference in MDS between the cohorts, also not between PwMS before and after 6-month DMF therapy. Five PwMS stopped DMF treatment during follow-up due to IAE, one PwMS due to an insufficient clinical response, and two PwMS were lost to follow-up. Clinical response was available following 1-year treatment for 38 PwMS, of these 27 PwMS (71%) showed no evidence of disease activity during follow-up (PwNDA). Eleven PwMS showed some evidence of disease activity (ΔEDSS increase >1, relapse, and/or evidence of active disease on MRI) (PwDA). Of these, one PwMS discontinued DMF and shifted DMT <1 year, four PwMS shifted DMT within 12–15 months, while six PwMS continued DMF therapy beyond at least 2 years from treatment initiation, indicating that despite the evidence, the clinical response was regarded sufficient at the time.

Study design.
Demographic and clinical data of participants.

Comparison of clinical and demographic data between PwMS (N = 45) pre-treatment, post 6-month and 1-year DMF therapy, and HC (N = 47). Statistical comparison of age, BMI, MDS was assessed using Mann–Whitney U-test; gender, frequency of smoking and vegetarian, ethnicity, MDS groups assessed by Fischer’s exact test.
Assessed by Wilcoxon’s paired test.
Assessed by Friedman test.
BMI, body mass index; EDSS, Expanded Disability Status Scale; HC, healthy controls; IAE, intolerable adverse events which lead to drug discontinuation; IFN, interferon; MDS, Mediterranean Diet Score; PwMS, patients with MS; PwNDA, PwMS with no evidence of disease activity.
Comparison of general microbiota composition
A comparison of the general microbiota composition showed no significant difference in Shannon alpha index diversity between HC, PwMS at baseline, and post 6-month DMF therapy (p = 0.21) (Figure 2(a)). A pairwise analysis showed also no significant difference between any of the groups (HC vs PwMS: p = 0.5; PwMS baseline vs 6-month DMF: p = 0.07 (trend)). There was a significant difference in Bray–Curtis beta-diversity between the three groups (p = 0.032) (Figure 2(b)). However, pairwise analysis revealed that this difference was only significant between HC and 6-month DMF patients, while not significant between patients pre- and post-DMF therapy (HC vs 6-month DMF: p = 0.006; HC vs PwMS: p = 0.5; PwMS baseline vs 6-month DMF: p = 0.08 (trend)).

General microbiome comparison. (a) Shannon alpha-diversity and (b) Bray–Curtis beta-diversity at the species level, between HC (healthy control), PwMS (baseline, pre-therapy), and following 6-month DMF treatment (6 m DMF).
Comparison of relative abundance of specific microbes
Effect of 6-month DMF treatment on the relative abundance of specific microbes
Microbes with significant alteration in relative abundance after 6-month DMF therapy compared to baseline (pre-treatment) were identified by Multiple Linear Regression adjusted as paired analysis, and complementarily by two unpaired analyses DeSeq2 and LEfSe. Twenty-five microbes with differential abundance post 6-month DMF therapy at different taxonomic levels were identified by the paired analysis, including four phyla, four classes, four orders, seven families, and six genera (presented in Table 2, Figure 3). In addition, another phylum, two families, seven genera, and one species were significantly different post DMF therapy in both unpaired analyses (Table 2, Figure S1 (LEfSe)). A multifactor subanalysis, adjusted both for subject ID (paired analysis) and for previous treatment (1st or 2nd line patients), is presented in Table S1, and confirmed the significantly differential abundance of the top five genera, seven families, four orders, five classes, and four phyla. This indicates that the effect of previous treatment in the 2nd line patients was minor, likely due to the relatively long period since previous DMT in most cases in our cohort. Microbes with increased abundance after 6-month DMF therapy all belonged to the Firmicutes phylum, the Clostridiales order, and were mostly from the Lachnospiraceae family, whereas some taxa belonging to the Selenomonadales or the Lactobacillales orders within the Firmicutes were reduced by DMF therapy. Other microbes reduced by DMF therapy mostly belonged to the Bacteroidetes, the Patescibacteria, or the Proteobacteria phyla.
Taxa with differential relative abundance post 6-month DMF therapy compared to pre-treatment.

Taxa with differential abundance after 6-month DMF therapy compared to baseline (pre-treatment). Multiple Linear Regression (paired analysis, N = 32 pairs), and unpaired analysis using DeSeq2, and LEfSe (N = 45 + 32 samples), at FDR < 0.1, corrected for multiple testing. Only taxa significant by the paired analysis or by both unpaired analyses are presented.
DMF, dimethyl fumarate; FC, fold change; FDR, false discovery rate; LDA, linear discriminant analysis; LEfSe, LDA effect size; m, months; ns, not significant.

Differential abundant taxa in PwMS following 6-month DMF therapy compared to pre-treatment. Representative graphs of taxa with significant difference in relative abundance, as determined by Multiple Linear Regression paired analysis at different taxonomic levels (FDR < 0.1).
Comparison of microbes differential post 6-month DMF therapy versus pre-treatment and microbes differential between PwMS (pre-treatment) and HC
As we have recently published data on the comparison of the gut microbiota between HC and treatment-naïve PwMS in the Israeli population, 12 on cohorts which in part overlap with the cohorts of the current study (as described in the section “Methods”), such comparison was not the aim of the current study. However a PwMS/HC analysis was performed (presented in Table S2) to enable a comparison with DMF-altered microbiota (Table 2), revealing only very few overlapping differentially abundant microbes. Some Lachnospiraceae genera had lower abundance in PwMS compared to HC, and DMF therapy enhanced several Lachnospiraceae family members. However, Prevotella 7 + 9 genera had lower abundance in PwMS compared to HC, and Prevotella 7 further decreased post DMF therapy. Likewise, the Tannerellaceae family was lower in PwMS than HC, and the abundance further reduced after 6-month DMF therapy.
Associations between microbe abundance (pre-treatment) and clinical response
In order to identify microbes which may be associated with disease activity, we compared the relative abundance of microbes between patients with or without disease activity during the 1 year follow-up (PwDA vs PwNDA), in baseline samples (Table 3, Figure 4), using three statistical methods. At baseline, two genera, two families, two orders, one class, and three phyla differed between PwNDA and PwDA, according to either DeSeq2 and/or Linear Regression, while LEfSe identified four species, nine genera, and three families that best discriminate between the groups of clinical response. Of these the abundance of the genus Solobacterium, a relatively rare bacteria, was significantly higher in PwDA by all three statistical methods, and the abundance of the family Ruminococcaceae was higher in PwNDA in two out of three methods, thus presenting the most robust results, while other microbes were only differential by one statistical method (Table 3, Figure 4).
Taxa with differential relative abundance between patients with or without evidence of disease activity during 1-year DMF therapy (pre-treatment samples).

Taxa with significance differential abundance at baseline (pre-DMF therapy) between PwNDA and PwDA (N = 38). Statistical analysis using DeSeq2, Linear Regression, corrected for multiple testing, at FDR < 0.1, and LEfSe, at p < 0.05, LDA > 2.
DMF, dimethyl fumarate; FC, fold change; FDR, false discovery rate; LDA, linear discriminant analysis; LEfSe, LDA effect size; ns, not significant; PwDA, PwMS with evidence of disease activity during 1-year follow-up from DMF initiation; PwNDA, PwMS with no evidence of disease activity during 1-year follow-up from DMF initiation.

Taxa with differential relative abundance between PwNDA and PwDA in baseline samples (pre-DMF therapy). (a) Representative graphs of taxa with significant difference in relative abundance between PwNDA and PwDA, as determined by Linear Regression, or * by DeSeq2 (FDR < 0.1). (b) LDA score of differential taxa as determined by LEfSe between PwNDA and PwDA (p < 0.05).
Associations between microbe abundance (post 6-month DMF treatment) and clinical response
Similar analysis of samples obtained after 6-month DMF therapy (Table 4, Figure 5) revealed three species and six genera with differential relative abundance between PwDA and PwNDA according to DeSeq2 (Linear Regression did not detect any significant results), while LEfSe identified three species, four genera, and two families that best discriminated between PwDA and PwNDA. Of these, the species Clostridium sp. K4410.MGS-306 and Bacteroides uniformis CL03T12C37, and the genus Megamonas were significant by both statistical methods, thus being the most robust results. There was no overlap between the microbes with differential abundance according to clinical response detected in baseline samples and those detected in samples after 6-month DMF therapy.
Taxa with differential relative abundance between patients with or without evidence of disease activity during 1-year DMF therapy (post 6-month DMF samples).

Taxa with significance differential abundance between PwNDA and PwDA in samples obtained after 6-month DMF therapy (N = 31). Statistical analysis performed using DeSeq2, corrected for multiple testing, at FDR < 0.1, and LEfSe, at p < 0.05, LDS > 2.
DMF, dimethyl fumarate; FC, fold change; FDR, false discovery rate; LDA, linear discriminant analysis; LEfSe, LDA effect size; ns, not significant; PwDA, PwMS with evidence of disease activity during 1-year follow-up from DMF initiation; PwNDA, PwMS with no evidence of disease activity during 1-year follow-up from DMF initiation.

Taxa with differential relative abundance between PwNDA and PwDA in 6-month DMF samples. (a) Representative graphs of taxa with significant difference in relative abundance between PwNDA and PwDA, as determined by DeSeq2 (FDR < 0.1). (b) LDA score of significant differential taxa as determined by LEfSe between PwNDA and PwDA (p < 0.05).
Associations between microbe abundance (pre-treatment) and IAE
Similarly, we compared the relative abundance of microbes in baseline samples from patients with IAE (N = 5) to patients without IAE (N = 39) (Tables 5 and S3, Figure 6). With the low number of patients with IAE, the risk of false positive is high, and results are considered with caution. The unspecified species uncultured rumen bacterium was significantly lower in patients with IAE by three out of three statistical methods, while two species were significantly higher in IAE patients by both Linear Regression and LEfSe. Five genera were significant by all three statistical methods, namely Lachnoclostridium (higher in IAE patients), Ruminococcaceae UCG-010, Ruminococcaceae NK4A214 group, Ruminococcaceae UCG-002, and Holdemanella (all lower in IAE patients), while another eight genera were significant by two out of three different methods. The family Clostridiales vadin BB60 group was significantly lower in IAE patients by all three methods, while Leuconostocaceae was higher in IAE patients by two out of three methods.
Taxa with differential relative abundance between patients who discontinue DMF therapy due to IAE and patients without IAE.
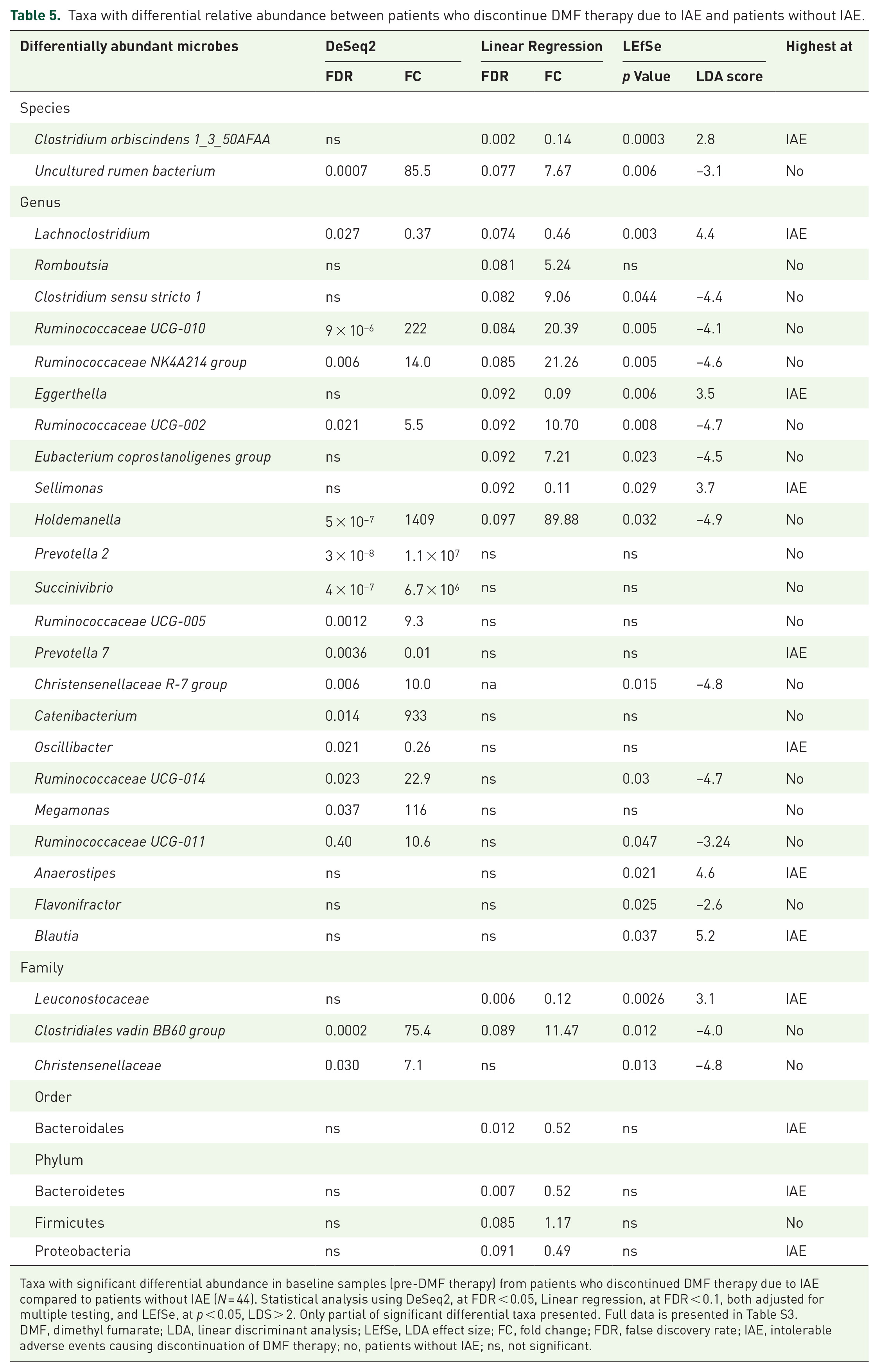
Taxa with significant differential abundance in baseline samples (pre-DMF therapy) from patients who discontinued DMF therapy due to IAE compared to patients without IAE (N = 44). Statistical analysis using DeSeq2, at FDR < 0.05, Linear regression, at FDR < 0.1, both adjusted for multiple testing, and LEfSe, at p < 0.05, LDS > 2. Only partial of significant differential taxa presented. Full data is presented in Table S3.
DMF, dimethyl fumarate; LDA, linear discriminant analysis; LEfSe, LDA effect size; FC, fold change; FDR, false discovery rate; IAE, intolerable adverse events causing discontinuation of DMF therapy; no, patients without IAE; ns, not significant.

Representative figures of taxa with differential relative abundance between patients with IAE and patients without IAE (at baseline). Only taxa significant by three methods are presented (DeSeq2, Linear regression, and LEfSe).
Associations between abundance of microbes altered by DMF therapy and MDS or clinical data
Only the Lachnospiraceae family, enhanced by DMF therapy, correlated positively with MDS (Rs = 0.3, p = 0.048). EDSS at baseline correlated negatively with the abundance of the Rikenellaceae family and the Alistipes genus (of the same family), the Barnesiellaceae and the Bacteroidaceae families, and the Bacteroides genus, all significantly reduced by DMF therapy (Rs = −0.4, p = 0.008; Rs = −0.43, p = 0.004; Rs = −0.3, p = 0.039; Rs = −0.3, p = 0.047, and Rs = −0.3, p = 0.047, respectively) (Figure 7(a)). Interestingly, the Barnesiellaceae and the Bacteroidaceae families and the Bacteroides genus correlated positively with Vitamin D levels in PwMS at baseline (Rs = 0.32, p = 0.03; Rs = 0.3, p = 0.048; and Rs = 0.3, p = 0.048, respectively).
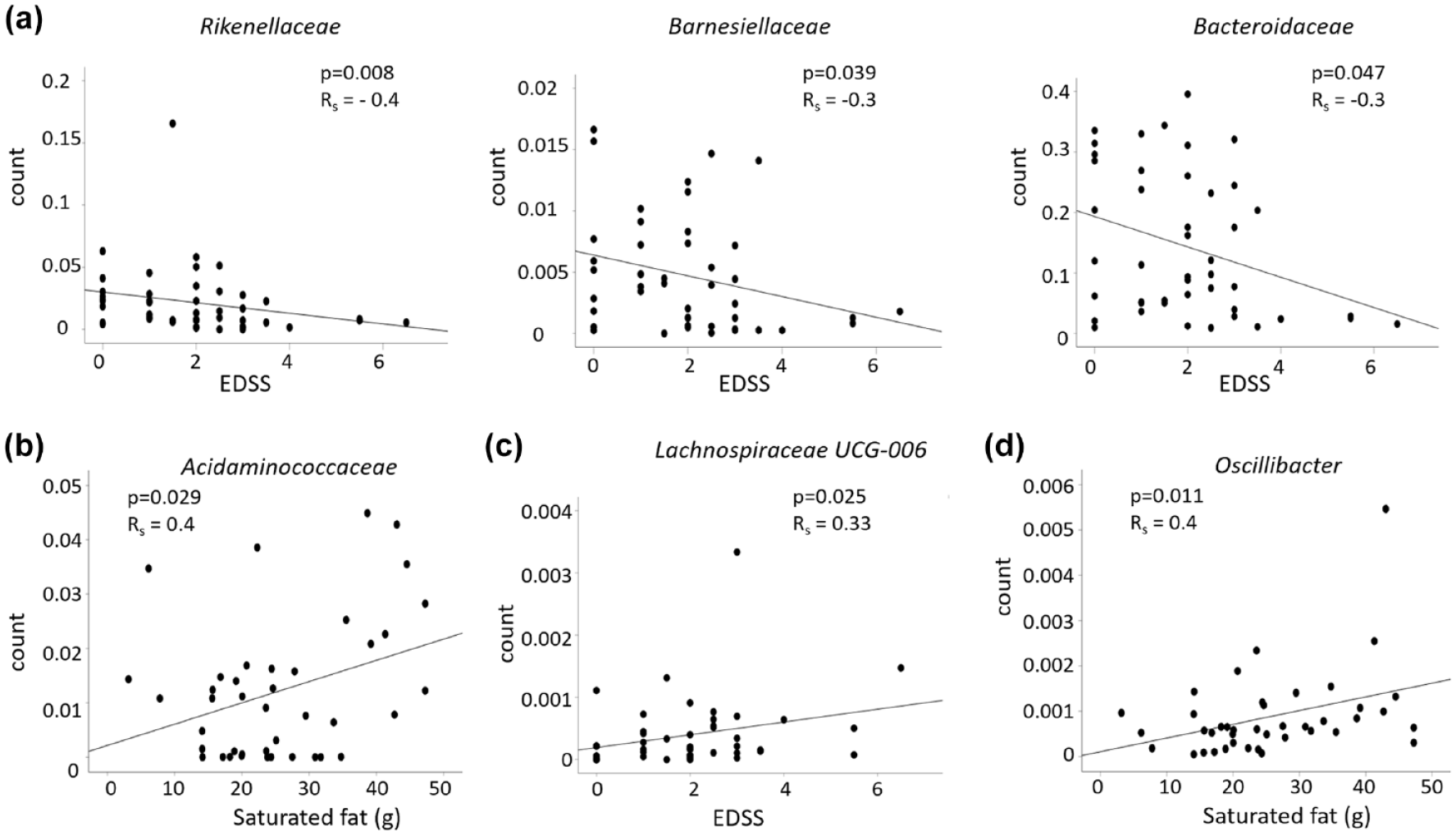
Correlations between EDSS or nutrients and abundance of differentially abundant taxa. (a) Differential taxa post 6-month DMF therapy compared to pre-treatment. (b and c) Differential taxa between PwNDA and PwDA. (d) Differential taxa between patients with or without IAE. Spearman correlation were calculated by SPSS on TSS normalized data.
Associations between abundance of disease-activity differential microbes and dietary or clinical data
There was no significant difference in MDS between patients with or without evidence of disease activity (7.7 ± 0.7 and 7.0 ± 0.4, respectively, p = 0.4). The mean alcohol intake was significantly lower in PwDA compared to PwNDA (0.17 gr ± 0.2 and 1.09 gr ± 0.5, respectively, p = 0.011), while no other differences in nutrient intake were detected between the two clinical response groups. Several significant correlations were identified between taxa with differential abundance between PwDA and PwNDA (from Table 3) and nutrient intake (from Table S4(A)). Of these, the family of Acidaminococcaceae, showing highest abundance in PwDA, was the only one that correlated positively with saturated fat and several fatty acids (Figure 7(b)). There were no significant correlations between baseline Vitamin D levels or MDS and abundance of the PwDA/PwNDA differential microbes, but the genus Lachnospiraceae UCG-006, highest in PwNDA, correlated positively with EDSS (p = 0.025, Rs = 0.33) (Figure 7(c)).
Associations between abundance of IAE-differential microbes and dietary or clinical data
There was no significant difference in MDS between patients with or without IAE (7.8 ± 1.3 and 7.3 ± 0.3, respectively, p = 1.0). There was a trend toward a higher intake of unsaturated fatty acids Parinaric and Decosapentaenoid (DPA, omega 3) in patients with IAE compared to patients without IAE (0.041 gr ± 0.01 and 0.025 gr ± 0.006, p = 0.065; 0.038 gr ± 0.007 and 0.027 gr ± 0.004, p = 0.053, respectively), while other nutrients did not differ between patients with/without IAE. Many significant correlations were identified between microbes with differential abundance between patients with/without IAE (from Table 5 and Table S3) and nutrient intake (from Table S4B). Of these, the Oscillibacter genus, highest in IAE patients, was the only one to correlate positively with saturated fat and several saturated fatty acids (Figure 7(d)). Eggerthella, highest in patients with IAE, correlated positively with various fatty acids, food energy, carbohydrates, total sugars, and unsaturated fat. Intake of Biotin (Vitamin B7), Vitamin B6, Thiamine (Vitamin B1), or Folate (Vitamin B9) correlated positively with some microbes with highest abundance in patients without IAE such as [Ruminococcus] gnavus group, Flavonifractor, or Ruminococcus gnavus CC55 001C. The abundance of the species Clostridium orbiscindens 1_3_50AFAA and the genus Eggerthella, both highest in IAE patients, correlated positively with MDS (p = 0.022, Rs = 0.34 and p = 0.004, Rs = 0.43, respectively), while the genera Megamonas and Succinivibrio, both lower in patients with IAE, correlated negatively with MDS (p = 0.028, Rs = −0.33 and p = 0.037, Rs = −0.31, respectively).
Enriched functional pathways
The Tax4Fun2 package, which predicts functional profiles based upon the KEGG database,37,39 was used to explore functional metabolic differences in DMF-treated patients compared to pre-treatment, or between PwNDA compared to PwDA, including functional diversity profiling and enrichment analysis. 1079 KO pathways significantly differed post-DMF therapy compared to pre-treatment (Table S5), and these mapped to 8 enriched pathways, at FDR < 0.05, presented in Table 6(a)). Top enrichment was found in pathways such as Citrate cycle (tricarboxylic acid (TCA) cycle), Lipopolysaccharide biosynthesis, Oxidative phosphorylation, and Biotin metabolism. Two KOs significantly differed between PwNDA and PwDA at FDR < 0.05, and 89 KOs were significant at p < 0.005 (Table S6), and these 89 KOs mapped to 3 enriched pathways, at p < 0.05 (Table 6(b)), but only included 1 hit each. Puzzling, this included the pathway of drug metabolism-cytochrome P450, although DMF has not been found to interact with hepatic cytochrome P450 (CYP) enzymes. 40
Enriched pathways from differential predicted functional profiles (KOs).

Enriched pathways mapped according to predicted functional profiles (KOs) that differ (a)—post 6-month DMF therapy compared to pre-treatment at FDR < 0.05, or (b)—in PwNDA compared to PwDA, at p < 0.05.
DMF, dimethyl fumarate; FDR, false discovery rate; KO, Encyclopedia of Genes and Genomes orthologs (KEGG) orthology; LPS, lipopolysaccharide; PwDA, PwMS with evidence of disease activity during 1-year follow-up from DMF initiation; PwMS, patients with multiple sclerosis; PwNDA, PwMS with no evidence of disease activity during 1-year follow-up from DMF initiation; TCA, tricarboxylic acid.
Discussion
With accumulating evidence of a role for the microbiome in human health and disease, including MS, revealing the interactions between microbiota, demographic and clinical factors, DMTs, and disease pathology is highly warranted. Studies from different geographic regions,4,6–10,41 including ours, 12 have found differential abundance between PwMS and healthy individuals of various microbes, suggestive of dysbiosis of the microbiota in MS. However, due to the complexity and many confounding factors affecting the microbiota, such as diet, gender, age, medications, disease type and state, comorbidities, geographic location, population, and lifestyle, the overlap between reports is limited, and a world-wide MS-microbiome signature is unavailable. MS drugs may have antimicrobial effects, and their efficacy and tolerability could in part be related to their effects on the microbiota. 25 Studies, mostly cross-sectional, comparing DMT-treated to untreated PwMS, found differences in the abundance of specific microbes, while the general microbiota composition, as determined by α- or β-diversity, seems not to be affected by treatment,7,9,13,15,16,25–27,41–44 similarly to our results. One could speculate that the significant difference in β-diversity found between HC and PwMS post DMF-therapy presents both DMF-induced and disease-related differences, while the trend difference between PwMS pre- and post DMF, presents only DMF-induced differences.
There are yet only a few publications on how a specific MS-DMT affects the microbiota, mostly cross-sectional, and with the many confounding factors that may affect the microbiota of an individual, these studies have a high risk of bias, compared to longitudinal studies, making comparison and validation across studies difficult. A recent large study by the iMSMS consortium found that microbial differences detected between untreated PwMS and their healthy house-hold members, were not replicated in treated PwMS, supporting that DMTs alter the microbiota composition. 25 Moreover, the authors found that several taxa altered by DMTs, did not differ between PwMS and HC, similarly to our results. This indicates that DMT-induced alterations do not necessary contribute to restore a healthy microbiota. Whether DMT-induced microbiota alterations are involved in the clinical response to treatment or drug tolerability is a pertinent question. Alterations could be favorable, promoting microbes that induce anti-inflammatory immune cells or intermediates, or could contribute to DMT-associated AEs, which may affect drug adherence. We addressed the effect of 6-month DMF therapy on the microbiota in PwMS in the present study, and investigated associations between microbiota, diet, treatment response, and AEs.
The function of the intestinal barrier, containing the gut mucosal immune system, is important for homeostasis and prevention of diseases, impeding penetrance of microbes and antigenic molecules from the gut. 45 Altered intestinal permeability has been demonstrated in PwMS. 46 Altered intestinal permeability was associated with disease severity in experimental autoimmune encephalomyelitis, and treatment with probiotics or administration of SCFAs improved barrier function, reduced inflammatory immune cells, and promoted anti-inflammatory cytokines.45,47 The gut barrier contains inter-epithelial tight junction molecules, such as occludins, claudins, junctional adhesion molecules, and zona occludens-1, as well as mucus, fluid, and innate and adaptive immune cells. 45 DMF was shown in vitro to increase the expression of zona occludens-1 in Experimental Colitis, 48 and to decrease intestinal permeability by strengthening tight junctions, through elevation of SCFA-producing bacteria in a mice model 49 ; thus, effects on gut permeability and microbiota could be part of the mechanism of action of DMF. Six recent studies have addressed the effect of DMF therapy on the microbiota composition in MS patients, of these three cross-sectional studies13,24,25 and three longitudinal studies,16,26,27 summarized in Table 7. Of these, Diebold et al. 27 also addressed whether DMF-associated changes in the microbiota were associated with lymphopenia, two studies addressed whether such changes were associated with flushing and GI AEs,16,26 while none have yet addressed associations with clinical response. We found in the present study 36 microbes at different taxonomic levels, with altered relative abundance after 6 months of DMF therapy compared to pre-treatment. We also identified several microbes, whose abundance differed according to evidence of disease activity following 1-year DMF therapy. Moreover, we identified microbes that differed pre-treatment between PwMS who eventually discontinued DMF therapy due to IAE compared to those who continued therapy, despite AEs (without IAE).
Summary of studies on effects of DMF therapy on the microbiome.

DMF, dimethyl fumarate; m, months; w, weeks.
One of the beneficial roles of the gut microbiota is the production of SCFAs, mainly butyrate, acetate, and propionate, from digestion of dietary fibers. SCFAs enhance the intestinal barrier integrity, mucus production, protect against gut inflammation, maintain intestinal homeostasis and function, and are also important for immunomodulation, blood brain barrier integrity, and neuroimmune homeostasis.2,45,50 Butyrate is a highly important SCFA, produced mainly by members of the phylum Firmicutes, such as the families Lachnospiraceae, Lactobacillaceae, and Ruminococcaceae, and serve a role in energy production, gut barrier integrity, and promotion of anti-inflammatory cytokines. In this study, DMF therapy increased the relative abundance of Firmicutes, the order Clostridiales, mainly in members of the Lachnospiraceae family, but also of the Ruminococcaceae family, while reduced the abundance of Bacteroidetes, an effect that could promote a beneficial increase in SCFA production. This is supported by the observation of higher abundance of Firmicutes, while lower abundance of Bacteroidetes in PwNDA, compared to PwDA. An increased abundance of Firmicutes and lower abundance of Bacteroidetes following DMF therapy was also reported after 12-weeks DMF treatment in another longitudinal study, 16 while the opposite was reported in a cross-sectional study. 15 Possible explanations for this contradiction include the cross-sectional versus longitudinal study design, technical differences, and variations related to different populations, which we have previously shown affect the microbiota. 12 The detected increased abundance of the Lachnospiraceae family and genera within the family following DMF therapy, included an increase in Blautia, a main SCFA producer associated with a healthy microbiota, and involved in controlling gut inflammation.44,51 Anaerostipes, another increased genus of this family, was also reported increased after 1-month DMF therapy. 26 Increased levels of various Lachnospiraceae taxa have been reported in several diseases, such as major depressive disorder, Diabetes Type 1 and Metabolic syndrome, while lower levels were found in persons with Crohn’s disease or ulcerative colitis, 51 thus emphasizing the complexity of determining disease-associated microbiota, and specific genera or species within a family may serve opposite roles. The Ruminococcaceae family, increased by DMF therapy, was highest pre-treatment in PwNDA, and so were members of this family such as Faecalibacterium, Butyricicoccus, and Ruminiclostridium. Faecalibacterium was previously reported to increase after 3-month DMF therapy, 16 and Butyricicoccus was reported to be less abundant in PwMS compared to healthy persons, 52 similarly to the current study. The reduction of Proteobacteria following 6-month DMF therapy could also be clinically beneficial, since the abundance of this microbe pre-treatment was highest in PwDA and in patients with IAE. A similar reduction in Proteobacteria after 1 and 3 months of DMF therapy was recently reported. 26 Proteobacteria include several known human pathogens, and dysbiosis of this phylum has been associated with promotion of pro-inflammation and inflammatory diseases, such as metabolic disorders and IBD,53,54 and is elevated in PwMS compared to healthy persons. 55 The genus Prevotella was reported in several studies to be lower in PwMS compared to healthy persons, including our previous and present study,6,10,12,56 and was further reduced here by DMF therapy, while not affected by DMF in a cross-sectional study. 15 Increased abundance of specific Prevotella species were reported in DMT-treated patients, such as Prevotella copri in Interferon-β-treated patients, 57 and Prevotella stercorea in interferon-β or teriflunomide-treated versus untreated patients. 41 Due to taxonomic resolution limitations of 16S sequencing used in the present study, 58 DMF-induced alterations at the species level may have been undetected.
Whether and how the microbiota is related to disease activity or to the clinical response to treatment is yet unknown. Differential microbiota composition was reported in patients with active versus remissive MS, 6 and we have previously found several specific microbes to correlate with disability (EDSS). 12 In pediatric MS, absence of Fusobacteria 59 and lower abundance of microbes involved in tryptophan metabolism 60 were associated with increased relapse risk. Moreover, higher abundance of Butyricicoccus, Lachnospiraceae NKA136 groups, Ruminococcaceae, and Odoribacter were associated with lower risk, while abundance of a member of Coriobacteriales was associated with higher risk of disease activity in pediatric MS. 61 Similarly, we found higher abundance of Butyricicoccus, Lachnospiraceae NKA136 groups, and Ruminococcaceae in pre-treatment samples of PwNDA. Moreover, the abundance of several genera from the Ruminococcaceae or Lachnospiraceae families (Firmicutes phylum), as well as of two orders from the Mollicutes class (Tenericutes phylum) was higher in PwNDA, while abundance of two species from the Bacteroidales order was higher in PwDA. Tenericutes was suggested to be associated with negative effects on inflammatory cytokines and protective in some autoimmune diseases, 62 and was elevated by probiotics in a murine model of MS. 63 Solobacterium, more abundant in pre-treatment samples of PwDA, has previously been associated with IBD, inflammation, and colorectal cancer. 64 Massiliprevotella massiliensis, a Prevotella species, was highest in PwNDA, supporting a beneficial abundance, as previously suggested for the Prevotella genus.6,10,12,56 Whether any of these microbes may predict disease activity or clinical response to treatment requires further validation in large studies. Moreover, the use of complementary assays for assessment of disease activity such as the MS Functional Composite could improve the detection of PwDA and the ability to address also the type and severity of activity. We also found microbes differentiating according to disease activity in samples obtained post 6-month DMF therapy. However, the observed alterations could be a consequence of lowered disease activity following treatment, and not present causality, since these microbes did not differ pre-treatment.
With the relatively common incidence of flushing and GI AEs after DMF initiation, which mostly decreases over time but, in some cases, can lead to drug discontinuation, it is pertinent to study whether specific microbes are associated with higher risk of such IAE. Two publications addressed this question assessing samples either pre-treatment or following DMF therapy.16,26 In this study, the analysis focused on pre-treatment samples from patients with AEs of a severity that caused discontinuation of DMF therapy. Other PwMS most likely may also have experienced flushing or GI AEs after DMF initiation, either at less severity, faster resolved, or persons may have had higher coping capability. We found that PwMS developing IAE had higher abundance of Bacteroidetes and Bacteroidales pre-treatment, in contrast to a former report. 16 Firmicutes and several genera from the Ruminococcaceae family were less abundant in IAE patients, while some genera belonging to the Lachnospiraceae family, such as Lachnoclostridium, Anaerostipes, and Blautia, as well as the Leuconostocaceae family were more abundant in IAE patients. Lower levels of Ruminococcaceae have been reported in people who develop antibiotic-associated diarrhea, 65 indicative that low abundance of members of this family may increase the likelihood of GI symptoms. Blautia was transiently increased in MS patients with GI symptoms after 3 months of DMF therapy, 26 and correlated positively with GI symptoms and diarrhea in the general population, 66 thus supporting our results. The genus Eggerthella, more abundant in IAE patients, was found to correlate positively with GI symptoms in children with autism spectrum disorder. 67 The genus Megamonas, less abundant in IAE patients, was found to be lower in patients with GI symptoms in people undergoing bariatric surgery. 68 The genus Holdemanella, less abundant in IAE patients, has been associated with a healthy state, 69 and the Holdemanella biformis species was reported to correlate negatively with symptoms of IBD. 70
Adherence to a Mediterranean diet did not differ between groups according to disease activity or IAE. The positive correlation between MDS and Eggerthella or C. orbiscindens, both higher in IAE patients, and the negative correlations between MDS and Megamonas or Succinivibrio, both lower in IAE patients, do not support that a Mediterranean diet may prevent IAE through alteration of these microbes. However, the Lachnospiraceae family, elevated by DMF therapy, correlated positively with MDS, supporting that a Mediterranean diet increases beneficial SCFA producers 71 such as Lachnospiraceae.
Several statistical correlations were found between microbes with differential abundance according to disease activity or IAE and the intake of various nutrients, indicating that diet and dietary recommendations could carry the potential to affect the clinical response or tolerance to DMF, through modification of microbe abundance. Interestingly, Acidaminococcaceae, enhanced in PwDA, and Oscillibacter, highest in patients with IAE, were the only identified differential microbes that correlated positively with saturated fat and saturated fatty acids. Dietary fibers, fructose, and Vitamin C correlated positively with Bacteroidetes, more abundant in patients with IAE, while correlated negatively with Firmicutes, lower in PwDA and IAE patients. While a high-dietary fiber diet intervention reduced the Firmicutes/Bacteroidetes ratio in persons with rheumatoid arthritis, 72 other studies found that dietary fibers increased the abundance of Firmicutes, 73 and such opposite effects appear to be caused by composition differences between various types of resistant starch in the dietary fiber, such as RS2, RS3, and RS4. 74 Carbohydrates and several unsaturated fatty acids correlated positively with Eggerthella, highest in patients with IAE, but also with Flavonifractor, lower in patients with IAE, thus not promoting dietary recommendations. Moreover, intake of several types of Vitamin B (Biotin, Thiamine, Folate, and B6) correlated positively with some microbes that showed higher abundance among patients without IAE, suggesting increased intake might potentially be beneficial in preventing IAE. Further investigation in larger cohorts are necessary to conclude on associations with diet and specific dietary recommendations; however, our observations propose that dietary interventions could have an intriguing potential to affect DMF response and tolerability, through modifications of abundance of specific microbes. Low Vitamin D levels have been associated with increased risk of disease activity in MS. 75 While still debatable whether Vitamin D supplement alters the microbiota,7,76 we only found a few associations between baseline Vitamin D levels and abundance of microbes modified under DMF therapy, while none with microbes that showed differential proportion according to disease activity or IAE.
The top enriched pathway of predicted functional profiles identified following 6-month DMF therapy was the Citrate cycle (TCA cycle). Predicted enrichment of TCA cycle intermediates following 3-month DMF therapy has also been reported elsewhere. 27 The active metabolite of DMF, MMF, is hydrolyzed within cells to fumaric acid, which is further metabolized through the TCA cycle, and DMF-treated patients have increased blood levels of TCA intermediates and metabolites, such as Succinate, Fumarate, and Malate. 77 Another enriched pathway of predicted functional profiles after DMF therapy was oxidative phosphorylation, interestingly, since DMF has been shown to affect oxidative phosphorylation and mitochondrial function.78,79 Biotin metabolism pathway was also enriched under DMF therapy. Biotin is a co-factor for four essential carboxylases expressed in oligodendrocytes and neurons, and supplement has therefore been suggested as a therapeutic strategy, although high-dose Biotin supplement in progressive PwMS was unsuccessful. 80 Additional longitudinal replication followed by experimental studies are necessary to assess biological relevance of these predicted functional findings.
A summary of the findings of this study (graphically presented in Figure 8) suggests that a relatively high abundance among PwMS initiating DMF therapy of: Firmicutes (highest in PwNDA, in patients without IAE, and increased by DMF therapy), Ruminococcaceae (highest in PwNDA and increased by DMF therapy), C. vadin BB60 group (highest in PwNDA and in patients without IAE), Megamonas, and Catenibacterium (highest in patients without IAE), is favorable for tolerability and clinical response. In contrast, high abundance of Proteobacteria (highest in PwDA, in patients with IAE, and decreased by DMF therapy), Bacteroidetes, and Bacteroidales (highest in patients with IAE and decreased by DMF therapy), as well as Leuconostocaceae (highest in patients with IAE and reduced by DMF therapy), may be unfavorable. The overlap between current publications on microbes altered by DMTs is small. However, there are accumulating indications that treated patients have a beneficial increase in SCFAs-producing microbes, such as members of Lachnospiraceae or Ruminococcaceae, as shown here following DMF therapy. The functional and clinical impact of therapeutic-induced changes in the abundance of specific microbe communities is for now unclear. Furthermore, while alterations of certain microbes under treatment of a specific DMT may be caused by the mechanism of action of the drug, alterations of microbes observed under the regime of different DMTs may reflect general improvement in disease activity, gut barrier function, and inflammatory state, due to immunomodulatory therapy. The therapeutic potential of these observations raises the question of whether targeting specific microbes can mimic or improve immunomodulatory effects of a DMT, and whether complementary treatment targeting the microbiota, may improve the clinical outcome for the patient, as well as drug tolerability.

Graphical summary: Interactions between DMF immunomodulation therapy and the gut microbiota in PwMS—associations with clinical response, drug tolerability, and diet. Graphical figure presenting a summary of results presented in this study.
The strength of the current study is the longitudinal study design addressing effects of one specific DMT and the inclusion of dietary data and clinical follow-up. The limitations include first the inclusion of both 1st line and 2nd line PwMS in the cohort (although a subanalysis showed this to have minimal effects on results), and, second, the limited sample size, especially of patients with IAE or PwDA. A third limitation is the self-report of dietary data, and the lack of scaling AE levels in patients. The taxonomic resolution limitations of 16S sequencing for identification of microbes below the genera level 58 is another limitation. Lastly, like most available publications in the domain of microbiome and autoimmunity, this study focused upon identifying disease-associated or therapy-induced compositionally effects on the microbiome, while the functional effects and biological relevance of such associations remain to be evaluated. Following further replication and establishment of robust findings across studies, future studies should proceed to functional experiments, targeting specific microbial communities to evaluate the causative effect on disease activity, drug response, or tolerability.
Conclusion
This study further clarifies the immunomodulatory role of DMTs, specifically DMF, on the gut microbiota composition, and the potential contribution of microbiota on clinical outcome and drug tolerability. Future goal would be to develop a microbiome-based, personalized treatment approach, integrating pharmacotherapy with dietary components, in synergy, toward improved clinical outcome.
Supplemental Material
sj-docx-2-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-docx-2-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-docx-3-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-4-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-docx-4-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-8-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-docx-8-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-1-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-tif-1-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-xlsx-5-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-xlsx-5-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-xlsx-6-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-xlsx-6-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-xlsx-7-tan-10.1177_17562864241306565 – Supplemental material for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy
Supplemental material, sj-xlsx-7-tan-10.1177_17562864241306565 for Immunotherapy-mediated modulation of the gut microbiota in multiple sclerosis and associations with diet and clinical response—the effect of dimethyl fumarate therapy by Elsebeth Staun-Ram, Anat Volkowich and Ariel Miller in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We gratefully thank Prof. Danit Shahar, the Department of Public Health Faculty of Health Sciences, Ben-Gurion University of the Negev, Israel for analysis of the FFQ; Dr Kathleen Abu-Saad and Dr Ofra Kalter-Leibovici, Gertner Institute for Epidemiology and Health Policy Research, Tel Hashomer, Israel for providing the MDS; Hy Laboratories Ltd for NGS service; Dr Ofra Barnett-Griness, Department of Community Medicine and Epidemiology, Lady Davis Carmel Medical Center, Israel for statistical consultancy; and Dr Neomi Gronich, Department of Community Medicine and Epidemiology, Lady Davis Carmel Medical Center, Israel for consultancy. We thank the MS patients for participation in this study, Sara Dishon, RN, M.PA., Natalie Zidon, RN, and Lea Glass-Marmor PhD, Carmel Medical Center, Haifa, Israel, for providing patient care and assistance in clinical data management.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
