Abstract
Background:
The inpatient Parkinson’s Disease Multimodal Complex Treatment (PD-MCT) is an important therapeutical approach to improving gait and activities of daily living (ADL) of people with PD (PwP). Wearable device-based parameters (DBP) are new options for specific gait analyses toward individualized treatments.
Objectives:
We sought to identify predictors of perceived ADL benefit taking clinical scores and DBP into account. Additionally, we analyzed DBP and clinical scores before and after PD-MCT.
Design:
Exploratory observational cohort study.
Methods:
Clinical scores and DBP of 56 PwP (mean age: 66.3 years, median Hoehn and Yahr (H&Y) stage: 2.5) were examined at the start and the end of a 14-day inpatient PD-MCT in a German University Medical Center. Participants performed four straight walking tasks under single- and dual-task conditions for gait analyses. Additionally, clinical scores of motor and nonmotor functions and quality of life (QoL) were assessed. Using dichotomized data of change in Movement Disorders Society Unified Parkinson’s Disease Rating Scale Part II (MDS-UPDRS II) as a dependent variable and clinical and DBP as independent variables, a binomial logistic regression model was implemented.
Results:
Young age, high perceived ADL impairment at baseline, high dexterity skills, and a steady gait were significant predictors of ADL benefit after PD-MCT. DBP like gait speed, number of steps, step time, stance time, and double limb support time were improved after PD-MCT. In addition, motor functions (e.g., MDS-UPDRS III and IV), QoL, perceived ADL (MDS-UPDRS II), and experience of nonmotor functions (MDS-UPDRS I) improved significantly.
Conclusion:
The logistic regression model identified a group of PwP who had the most probable perceived ADL benefit after PD-MCT. Additionally, gait improved toward a faster and more dynamic gait. Using wearable technology in context of PD-MCT is promising to offer more personalized therapeutical concepts.
Trial registration:
German Clinical Trial Register, https://drks.de; DRKS00020948 number, 30 March 2020, retrospectively registered.
Keywords
Introduction
Bradykinesia, rigidity, tremor, and postural instability are the well-known cardinal motor symptoms of Parkinson’s disease (PD). 1 But PD also causes nonmotor symptoms such as sleep disturbances, depression, pain, autonomous dysfunction, 2 and cognitive decline. 3 In addition, the gait of people with PD (PwP) often shows significant alterations in terms of a decreased gait speed, reduced stride and step length, increased stride time, double support time, and in general an increased variability and asymmetry of gait parameters.4,5
These symptoms have an important influence on the quality of life of PwP 6 through falls, activity limitations, restricted social participation, and loss of independence. 7
As the fastest-growing neurological disorder worldwide, 8 PD is seriously affecting our and future generations. 9 This causes a special need for individualized and multidisciplinary therapies.10,11
Multidisciplinary approaches in PD treatment combining medical adjustment with physical, occupational, and speech therapy are internationally recommended12,13 to offer a patient-centered and individualized treatment. 10 In Germany, the inpatient PD-Multimodal Complex Treatment (PD-MCT) is a widespread treatment approach for PwP that lasts between 7 and 21 days. 14 PD-MCT is offered at hospitals to about 23% of all inpatients in Germany. 14 Several observational studies already reported the positive effect of PD-MCT on motor symptoms, depression, and quality of life.15 –20
As an effect of the fast-developing technological opportunities, there is an increasing number of technology-based objective measures, 21 including parameters from wearable digital devices. These body-worn sensors provide objective and accurate metrics of gait, are quick and easy to use,22,23 and allow a specific, continuous, and more detailed quantitative assessment with a broader data collection 24 in contrast to established and validated clinical gait assessments. 25 Analyzing spatiotemporal aspects of gait can provide more individualized care. 21
Taking into account the activities of daily living (ADL) impairments caused by PD, the widespread application of PD-MCT, and the opportunities of wearable digital devices, there is a need to specify predictive factors for ADL benefit after PD-MCT to offer more patient-centered and individualized care. 26 Ziegler et al. 18 already identified age and baseline ADL impairment as important factors for ADL benefit after PD-MCT using clinical scores. We want to use the potential of device-based parameters (DBP) in combination with clinical assessments to identify promising outcome parameters for our analysis.
The implementation of DBP in research and clinical setting has already shown promising opportunities in previous studies.5,20,27 –30 Even if wearable technology is widely used in research, it still needs to be clarified which DBP are responsive to different types of interventions 31 before implementing them in clinical practice. In a previous analysis on PD-MCT, we already presented DBP and objective outcomes of gait and balance in a smaller sample size 20 and other groups presented preliminary data on wearable technology for evaluation of PD-MCT too. 32
In this exploratory observational study, we analyzed the most promising parameters for perceived ADL benefit after PD-MCT as a primary objective using device-based gait parameters as well as clinical scores.
As a secondary objective, we assessed clinical and device-based parameters before and after a PD-MCT using a larger sample size.
Methods
Study design
This exploratory subanalysis is part of the observational cohort study Park-Move. The study is listed in the German Clinical Trial Register (DRKS-ID: DRKS00020948) and is contributing data to the multicenter ComOn-Study 33 coordinated by UKSH University Hospital Kiel, Germany.
Participants
PwP undergoing an inpatient PD-MCT between September 2019 and August 2021 at the Department of Neurology at St. Josef-Hospital, Ruhr-University Bochum, Germany were screened for eligibility.
A PD diagnosis based on the criteria of the UK Brain Bank 34 and the Movement Disorder Society 35 was mandatory. Additional inclusion criteria were a minimum age of 18 years, written informed consent, and an adequate medical and mental condition to participate in the clinical and device-based assessments. Patients with secondary or atypical Parkinsonian syndromes 36 were excluded. Detailed exclusion criteria were described previously. 20
Setting and procedure
Participants were examined at the beginning (T1, days 1–2) and at the end (T2, days 13–14) of PD-MCT according to OPS 8-97d of the German Federal Institute for Drugs and Medical Devices. 37
Examinations included medical history, clinical examination, assessment of clinical scores, and device-based gait analyses (Figure 1). All assessments were performed during the ON state.

Study design and schedule of assessments.
Intervention
Participants underwent an inpatient PD-MCT for at least 14 days. While it is comparable to other forms of comprehensive rehabilitation available outside Germany, regulation and delivery of PD-MCT are specifically German. The treatment is part of a catalog defining conditions for reimbursement of inpatient healthcare services. Among these conditions are a minimum of 7.5 h of nonpharmacological therapies per week, weekly team meetings under the supervision of a board-certified neurologist and documentation of treatment goals and results. The costs are covered by statutory health insurances. While from a medical point of view, PD-MCT is a form of short-term rehabilitation, from a legal point of view its delivery is regulated separately from rehabilitation.
The study’s PD-MCT consisted of medication adjustment and nonpharmacological treatment elements which have been described in detail previously.15,16 The PD-MCT aims to improve the daily function, that is, the performance of physical, behavioral, and cognitive activities, 38 to support quality of life and focus on specific personal needs—a key element of personalized medicine. 10
A core element of the PD-MCT is nonpharmacological treatment in the form of individual sessions (physiotherapy, occupational, speech and language, and physical therapy). Endurance, safe walking, rising from a chair, or balance exercises were practiced. 39
Additionally, PwP attended weekly group therapy to manage PD in daily life. During the inpatient stay, they were accompanied by a multidisciplinary team, consisting of neurology specialists for movement disorders, psychiatrists, physiotherapists, occupational therapists, a PD nurse, and speech and language therapists. Every discipline assessed personal needs at the beginning. Corresponding therapeutic strategies, clinical changes, and individual therapy goals of every PwP were discussed and evaluated in weekly team meetings. According to the current guidelines of the German Society of Neurology (DGN), 40 medical treatment was adjusted to the needs and symptoms of the PwP.
Device-based gait assessment
For gait analyses, participants were equipped with three wearable digital devices of the CE-certified RehaGait® system (Hasomed, Magdeburg, Germany) which was placed at the ankles and the lower back (L5). These devices were connected to a tablet computer via Bluetooth during the examination. Under supervision, participants performed several gait tasks while the instructions were read by the investigator from the tablet computer to ensure that every patient received the same instructions and to avoid bias such as unintended additional motivation. Due to external interference, disease severity, or incorrect performance, repetitions up to two times were possible.
We chose the following gait tasks or walking test paradigms for this analysis:
1. Walking 20 m under single-task conditions in - a self-selected speed (single-task normal pace, STNP) - a fast-walking speed (single-task fast pace, STFP).
2. Walking 20 m in a fast-walking speed under dual-task conditions - motor-motor dual-task: checking boxes (dual-task checking boxes, DTCB) - motor-cognitive dual-task: serial subtraction of 7 (dual-task subtracting serial sevens, DTS7).
A distance of 20 m for device-based gait analyses has already been chosen and advised in other studies.29,41
Depending on the motor and cognitive condition, some patients only performed single-task examinations.
The wearable digital devices contained a three-axis accelerometer, gyroscope, and magnetometer to measure parameters as linear acceleration (±16 g), angular velocity (±2000°/s), and changes in magnetic field (±1,3 Gs) with a sampling rate of 500 Hz. 42 For data interpretation and comparability, data were processed into different parameters of gait using a validated algorithm. 43 Estimated gait parameters are explained in Table 1.31,44
Device-based gait parameters and domains.

Clinical and patient-reported assessments
The primary outcome was motor experience of daily living as assessed by the mean change in Movement Disorder Society Unified Parkinson’s Disease Rating Scale Part II (MDS-UPDRS II) score. 45 Secondary outcomes included motor symptoms and complications using the MDS-UPDRS Parts III and IV, 45 balance function using the Berg Balance Scale (BBS), 46 functional capacity of the lower extremities using the Short Physical Performance Battery (SPPB), 47 functional mobility using the Timed Up and Go (TUG) test, 48 and fine motor skills using the Perdue Pegboard Test (PPT).49,50 Nonmotor experience of daily living was measured with the MDS-UPDRS Part I, 45 and the Falls Efficacy Scale International (FES-I) 51 was used for assessing fear of falling.
Cognitive and neuropsychological aspects were assessed using the Montreal Cognitive Assessment (MoCA), 52 Frontal Assessment Battery (FAB), 53 and Trail Making Test (TMT). 54 Health-related quality of life was measured using the EuroQol 5-Dimensions 5-Level (EQ-5D-5L)55,56 and the Parkinson’s Disease Questionnaire (PDQ-39). 57
To evaluate disease severity, the modified Hoehn and Yahr (H&Y) scale58,59 was used. PD phenotypes were estimated from MDS-UPDRS according to Stebbins et al. 60 We also calculated the levodopa equivalent daily dose 61 for T1 and T2 and collected the exact amount of therapy hours.
The patient-reported questionnaires were self-reported by the participants. In case they needed further information for understanding and completing the questions, the details were explained by the examinator. In the vast majority of cases, the questionnaires were completed independently.
Statistical methods
Data were analyzed with IBM SPSS Statistics, Version 29 (IBM Inc., NY, U.S.). Using Shapiro–Wilk test, continuous data were tested for normality. Mean values (M) and standard deviations (SD) were calculated for all continuous clinical and device-based variables, whereas categorical data were described using median and interquartile range.
Device-based parameters were screened for correlating significantly and strongly (p < 0.05, ρ > 0.5) with gait speed using Spearman’s rank correlation coefficient. Parameters with significant and strong correlations were normalized for a velocity of 1 m/s. 20 To compare the parameters at T1 and T2, p values and Bayes factor (BF10) were calculated by using the Bayesian t test. P values <0.05 were considered significant. To avoid alpha error, Bonferroni correction for multiple testing was used to adjust the level of significance. P values considered significant after Bonferroni correction are emphasized by typographical notes.
To model PD-MCT benefit and adjust for confounders, binomial logistic regression was employed. The minimal clinically important difference (MCID) of MDS-UPDRS Part II improvement was chosen as a cutoff to generate a dependent variable (−3.05 points for improvement). 62 Data of the MDS-UPDRS II difference between T2 and T1 were dichotomized according to the MCID as “no benefit” and “benefit.” Independent variables were chosen by screening all variables with a simple logistic regression. The model was calculated with five predictors according to the number of cases (1 predictor per 10 participants as a rule of thumb). Predictors were also checked for multicollinearity through Spearman’s rank correlation coefficient. For evaluating the quality of the model and effect size, receiver operating characteristics (ROC) analysis, area under the curve (AUC) values, odds ratio, the overall percentage of accuracy, Hosmer–Lemeshow test, and Nagelkerkes R (R2 > 0.5) were assessed.
For this exploratory analysis, sample size calculation and power analysis were not performed. Missing values were not imputed. Variables of affected cases were excluded from the specific analysis.
Results
One hundred twelve PD-MCT patients were screened for eligibility and 56 met the inclusion criteria (Figure 2). Main reasons for exclusion were atypical Parkinsonian syndromes or overall health status making participation in the walking tasks impossible.
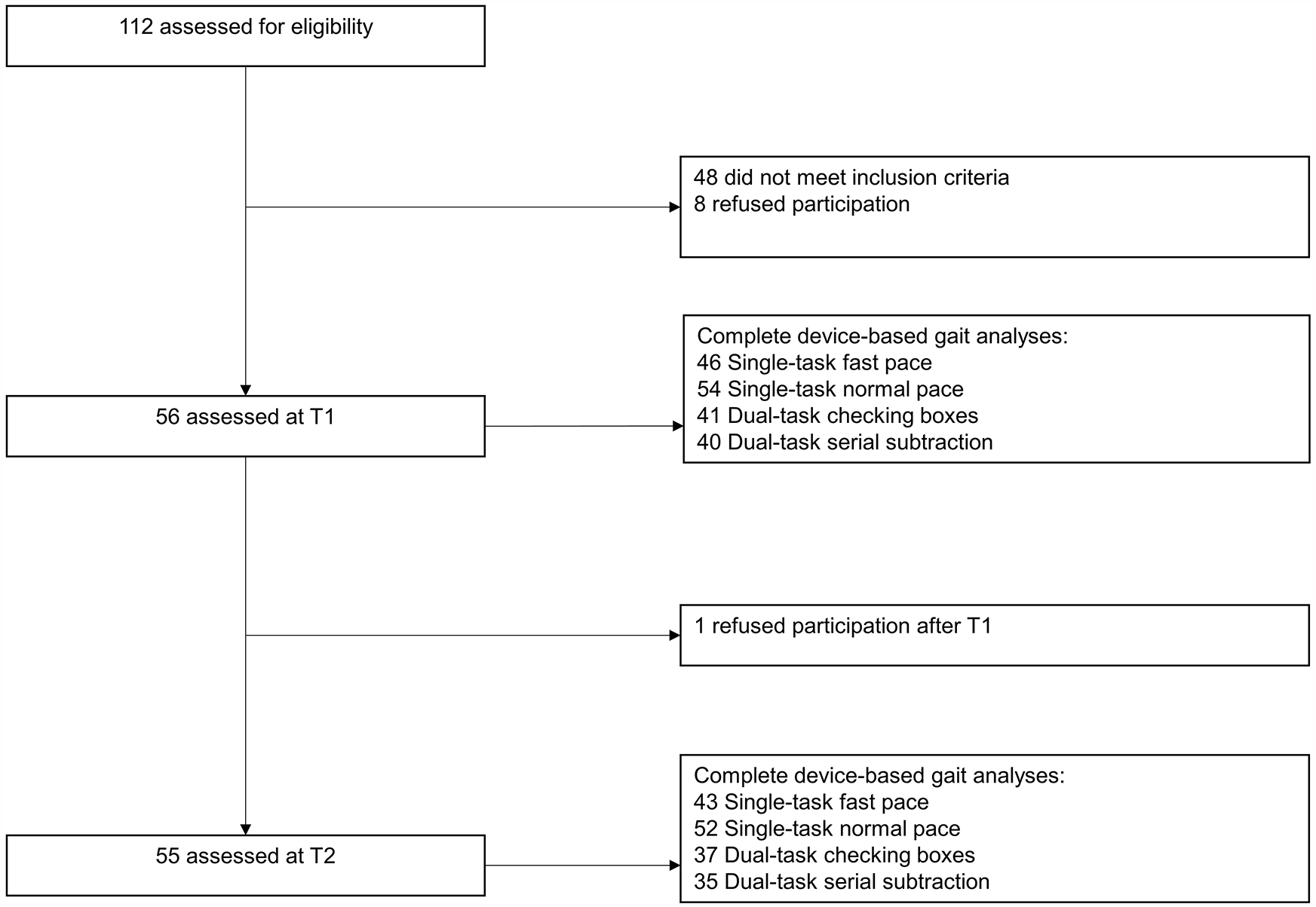
Park-Move study flowchart of participants.
The participants received treatment from various disciplines. The average amounts of treatment hours are shown in Table 2.
Amount of treatment hours per various disciplines.

PD-MCT, Parkinson’s Disease Multimodal Complex Treatment.
The age of participants was between 48 and 82 years (mean ± SD age 66.32 ± 9.46). 28.6% of the participants were female, 71.4% male.
More than 96.4% of the participants had a bilateral involvement (stage 2 or higher) according to modified H&Y with a median stage of 2.5. The average duration of the disease was 8.57 (SD ± 5.6) years. A total of 32.1% had a tremor dominant, 12.5% an indeterminate, and 55.4% a postural instability/gait difficulty (PIGD) phenotype. 60 A total of 72% of participants had a mild cognitive impairment (MCI) according to the MoCA T1 results. 52 Study population characteristics are summarized in Table 3.
Study population characteristics.

N = 56.
TD, tremor dominant; PIGD, postural instability/gait difficulty; H&Y, modified Hoehn & Yahr scale; PD-MCT, Parkinson’s Disease Multimodal Complex Treatment; IQR, interquartile range.
In a binomial logistic regression model, young age, high perceived impairment of ADL (MDS-UPDRS II), low step time variability (DBP), and low impairment of fine motor skills (PPT) independently predicted a better outcome concerning experience of ADL after PD-MCT (Table 4; Figure 3).
Binomial logistic regression model: predictors of benefit in activities of daily living after PD-MCT.

Significant changes are highlighted in bold.
aOR, adjusted odds ratio; B, regression coefficient; df, degrees of freedom; LL/UL, lower/upper limit; MDS-UPDRS II, Movement Disorder Society Unified Parkinson’s Disease Rating Scale Part II; PDQ-39, Parkinson’s Disease Questionnaire; SE, standard error; 95% CI, confidence interval.

Predictors of ADL benefit after 14-day PD-MCT.
Four of the five variables entered into the regression model significantly contributed to the prediction of perceived ADL outcome after PD-MCT. The binomial logistic regression model was overall statistically significant (X2 = 31.66, p < 0.001) with a large amount of explained variance shown by Nagelkerkes R2 = 0.651 and an overall percentage of accuracy in classification of 89.8%. The model was checked using a ROC curve with an AUC of 0.921.
Concerning clinical scores (Table 5), we found significant improvements in motor experience of daily living (MDS-UPDRS II) and health-related quality of life (PDQ-39 and EQ-5D-5L). In addition, motor symptoms (MDS-UPDRS III), motor complications (MDS-UPDRS IV), balance (BBS), functional capacity of the lower extremities (SPPB), and fine motor skills (PPT) improved significantly. Moreover, experience of nonmotor symptoms as measured by MDS-UPDRS I improved in a significant manner. The mean levodopa equivalent daily dose increased throughout PD-MCT.
Clinical data: T1 and pre–post differences.
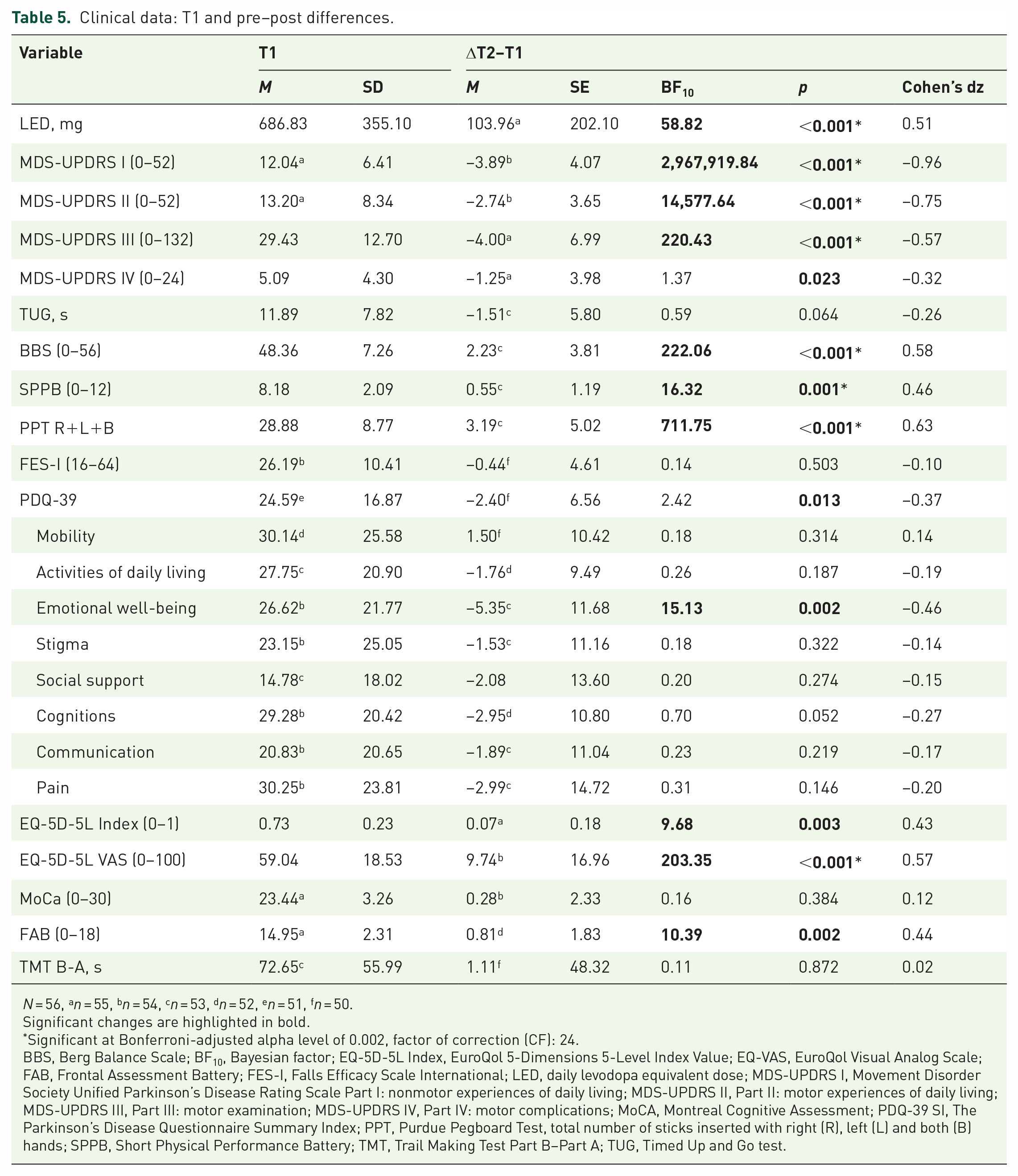
N = 56, an = 55, bn = 54, cn = 53, dn = 52, en = 51, fn = 50.
Significant changes are highlighted in bold.
Significant at Bonferroni-adjusted alpha level of 0.002, factor of correction (CF): 24.
BBS, Berg Balance Scale; BF10, Bayesian factor; EQ-5D-5L Index, EuroQol 5-Dimensions 5-Level Index Value; EQ-VAS, EuroQol Visual Analog Scale; FAB, Frontal Assessment Battery; FES-I, Falls Efficacy Scale International; LED, daily levodopa equivalent dose; MDS-UPDRS I, Movement Disorder Society Unified Parkinson’s Disease Rating Scale Part I: nonmotor experiences of daily living; MDS-UPDRS II, Part II: motor experiences of daily living; MDS-UPDRS III, Part III: motor examination; MDS-UPDRS IV, Part IV: motor complications; MoCA, Montreal Cognitive Assessment; PDQ-39 SI, The Parkinson’s Disease Questionnaire Summary Index; PPT, Purdue Pegboard Test, total number of sticks inserted with right (R), left (L) and both (B) hands; SPPB, Short Physical Performance Battery; TMT, Trail Making Test Part B–Part A; TUG, Timed Up and Go test.
Regarding cognitive tests, only executive functions on FAB showed a significant improvement while mean MoCA and TMT scores remained unchanged. No significant changes were found in functional mobility (TUG), fear of falling (FES-I), and several domains of health-related quality of life (PDQ-39 subscales).
Largest effects (absolute Cohen’s d ⩾ 0.8) were found for nonmotor and motor experience of activities of daily living (MDS-UPDRS I and II), medium effects (0.5 ⩽ Cohen’s d < 0.8) for fine motor skills (PPT), balance (BBS), motor symptoms (MDS-UPDRS III), and quality of life as assessed by EQ-VAS, and small effects (0.2 ⩽ Cohen’s d < 0.5) for emotion-related quality of life (PDQ-39 subscale), the functional capacity of the lower extremities (SPPB), executive function (FAB), motor complications (MDS-UPDRS IV), and quality of life as measured by PDQ-39 and EQ-5D-5L.
During PD-MCT, gait improved toward a faster and more dynamic gait (Table 6).
Device-based gait parameters: T1 and pre–post differences.

Significant changes are highlighted in bold.
Significant at Bonferroni-adjusted alpha level of 0.004, factor of correction (CF): 14.
BF10, Bayesian factor.
Parameters connected to pace improved, as shown by a significant increase in gait speed in single- and dual-task gait analyses and a reduction of the number of steps, whereas step length significantly decreased in STNP and DTS7 analyses.
Concerning the rhythm of gait, step time and stride time decreased significantly in three walking test paradigms (STNP, STFP, and DTS7) with the cadence increasing correspondingly.
Gait parameters associated with the phases of the gait cycle improved significantly in three of the four walking test paradigms, as shown by decreasing stance and double-limb support (DLS) time.
Variability and asymmetry did not change significantly in any paradigm.
During motor-motor dual-tasking (DTCB), no significant changes in gait parameters were found.
While significant differences in gait parameters showed small effects during SWFP, a large proportion of parameters during SWNP and DTS7 changed with medium-effect sizes.
Discussion
The inpatient multidisciplinary Parkinson’s Disease Multimodal Complex Treatment (PD-MCT) is a widely and increasingly implemented therapeutic approach in Germany that is a key element of multidisciplinary and patient-centered PD care, as internationally recommended.12,13 This exploratory cohort study aimed at identifying predictive factors of perceived ADL benefit after PD-MCT and evaluating PD-MCT effectiveness using objective device-based parameters (DBP). We identified young age, high baseline impairment of perceived ADL, high fine motor skills, and low step time variability as characteristics of promising PD-MCT candidates.
Study population characteristics such as age, disease stage, motor disability, and LED were similar to those of the study validating the applied DBP extraction algorithm. 43 The distribution of PD phenotypes is comparable to previous studies on PD-MCT15,16 and the original publication on MDS-UPDRS-based phenotypes. 60 Importantly, more than half of the participants had a PIGD phenotype that is associated with higher impairment of gait and balance. 63
Perceived ADL benefit from PD-MCT: Analysis of the most promising factors
Regarding the primary objective, our analysis revealed characteristics of PwP who had a clinically meaningful perceived ADL benefit from PD-MCT. With a mean difference of −2.74 points, our study population showed a statistically significant improvement in MDS-UPDRS II but, on average, did not reach the margin of a clinically meaningful difference of −3.05 points. 62 Nevertheless, a total of 36.4% of participants reached this MCID. Experience of daily function as measured by MDS-UPDRS II was used as the primary outcome in only one previous study identifying predictors of PD-MCT benefit. 18 However, MDS-UPDRS II seems very suitable for the evaluation of PD-MCT as a form of short-term rehabilitation as both the patient-rated outcome assessment and the aims of the intervention are related to everyday performance (“does do”) rather than capacity (“can do”) of ADL. 38 Yet, as a patient-reported outcome assessment, MDS-UPDRS II is limited by recall bias, subjectivity, and low ecological validity.
Other analyses focused on predictors of improvement in motor outcomes as measured by MDS-UPDRS III as a capacity measure. They identified higher motor symptom load (MDS-UPDRS III score), lower age, and lower depression scores as predictors of motor improvement.15 –17 For the first time, and including the more relevant MDS-UPDRS II score, this analysis takes objective DBP of gait into account when identifying predictors of perceived ADL benefit from PD-MCT.
Younger age and higher initial perceived ADL impairment as predictors identified in this study are similar to those found by a large, monocenter cohort study by Ziegler et al. 18 Young age seems to be an important indicator for clinically meaningful outcomes concerning both ADL and motor benefit.17,18 Regarding the perceived baseline ADL limitations, we found a 1.4-fold chance for people with high initial limitations to achieve a clinically meaningful treatment response. The about sixfold higher chance found by Ziegler et al. 18 could be a result of differences in the baseline MDS-UPDRS II data as with their 22.3 points, the perceived activity limitations at the beginning of PD-MCT were considerably higher than in our study population (mean baseline MDS-UPDRS II score 13.2). As a further predictor, dexterity is considerably affected by PD due to its main symptoms. 1 This is illustrated by worse fine motor skills on a pegboard (PPT) as performed by the study population compared with those of healthy elderly people described in the original PPT publication (mean PPT scores of 28.9 vs 35.6 in healthy males aged 60–69 years). 49
During PD-MCT, dexterity training is a part of occupational and physiotherapy.64,65 At the end of PD-MCT, the PwP’s PPT score significantly improved which confirmed previous analyses of PD-MCT effects.16,20 In our model, people with high fine motor skills at baseline had an about 1.2-fold higher chance of achieving a clinically relevant perceived ADL benefit.
Variability of gait is already increased in early PD stages.5,44 It correlates with disease severity, 66 is associated with increased fall risk29,67 and affects ADL and quality of life of PwP through gait impairment.6,7 Device-based assessment of gait offers an easily feasible option to objectively measure variability parameters in daily clinical routine. In addition, variability is described as a nondopa-responsive parameter and seems promising for evaluating nonpharmacological treatment. 68 When integrating gait variability into our model, we selected the values from the single-task walking paradigm for the least confounding possible, as gait variability increases under dual-task conditions independently of age or disease.69,70 According to our logistic regression model, people with low step time variability have a significantly better chance for clinically meaningful perceived ADL benefit from PD-MCT. Nevertheless, results have to be interpreted carefully because gait variability is also affected by factors like age and cognitive impairment.71,72
We added the PDQ-39 mobility subscore to our model because there have been promising results using this subscale as a relevant measure for health-related quality of life in PD. 73 It turned out that the subscale does not contribute to our model in a significant way.
Interestingly, most of the predictors of our perceived ADL benefit model have a motor component which can be explained by the strong influence of motor symptoms on quality of life and ADL.7,74
The motor focus of the predictors underlines the relevance of motor skills for PwP and emphasizes the importance of therapeutical units with a focus on individual motor and gait components during PD-MCT. These findings can contribute to individual and patient-centered therapy concepts in the form of, for example, adjusted gait training.
Effects of PD-MCT—clinical and device-based aspects
Concerning the secondary objective of evaluating device-based gait parameters and clinical scores at the beginning and the end of PD-MCT, we could confirm most of our previous results 20 and found additional evidence of improved gait parameters.
As already shown in several PD-MCT studies15,16,20 we found a significant improvement in the clinical motor-scores MDS-UPDRS III and IV, BBS, SPPB, and PPT. MDS-UPDRS III improved on average in a clinically meaningful way. 75 In addition, we also found a clinically relevant improvement 76 of motor complications after PD-MCT (MDS-UPDRS IV). These improvements might reflect the effective and constant motor training and pharmacological optimization during PD-MCT.
Regarding experience of nonmotor aspects of daily living (MDS-UPDRS I), the mean difference can be considered clinically meaningful according to Horváth et al. 62 Also the PDQ-39 improved significantly but on average still below a clinically meaningful response. 77
Similar to previous results, 20 we found no significant changes in the MoCA and TMT scores while FAB improved in a statistically significant way. Because of the relatively short treatment duration and a lack of explicit cognitive activating training, we did not expect improvements in cognition. Nevertheless, physical activity is a promising nonpharmacological approach in diseases with cognitive impairment. 78 General fitness with long-term application seems to have more effects on cognition than short-term activation with local effects on brain structures in MRI studies. 78 The duration over several months and the frequency of training are essential elements and cannot be covered by a 14-day PD-MCT short-term treatment.
Regarding the effect of PD-MCT measured by device-based parameters, we found a significant improvement of gait speed, step and stride time, stance time, and DLS time, whereas step length, cadence, variability, and asymmetry showed no improvement. Dual-task analysis presented inconsistent outcome results.
A careful selection of gait parameters for analysis is mandatory because not every parameter is suitable for all neurodegenerative diseases. 41 Usually, PwP present a slower and insecure gait.79 –81 Gait speed is easy to register and has already found its approved diagnostic place in the clinical routine 79 but interacts with factors like age, disease progression (H&Y), and motor symptoms.80,82 The gait speed of our study population is slower at T1 in comparison to healthy adults. 83 In our further analyses, gait speed constantly showed a significant improvement in single-task and in DTS7 measurement.
However, the need for individual therapeutic concepts requires a more detailed analysis of the patient’s gait. Using wearable digital devices enables a broader consideration of temporal (e.g., gait speed) and spatial gait parameters (e.g., step length) and provides a strong discriminative power when used in combination. 79
As well as gait speed, step length is a promising pace parameter5,29,41,84 that is considered to reflect PD progression, especially at higher stages of disease (H&Y > 3). 84 Surprisingly, we found no improvement in step length. In contrast, we even found an aggravation with significantly reduced step length under STNP and DTS7 conditions, whereas an aggravation of step length during dual-tasking has been described previously. 70 In our previous analysis, step length increased in half of the participants, while decreasing in the other half. 20 Interestingly, Vila et al. 85 also found inconsistent results of step length differentiating between left and right foot in context of H&Y stages. The sample size and a median H&Y of 2.5 might limit the generalizability of this finding and ask for further investigations if step length is a suitable parameter for monitoring short-term effects of PD-MCT.
Cadence of PwP is increased and the highest progression of cadence is described between H&Y stages 2 and 3. 85 We found no improvement of cadence in all walking tasks and registered a statistically significant increase of cadence in STNP and DTS7. This result may correspond to the finding of Vila et al. 85 concerning our H&Y median stage of 2.5.
Considered reliable and promising for rhythm monitoring are also step time and stride time,29,41 which changed in three of four gait analyses (STNP, STFP, DTS7) toward a stable and continuous gait.
Previous analyses identified swing and stance time as promising29,41 which changed both significantly during normal pace walking. The reduction of stance time is about four times higher than of swing time. A reduction of swing time is characteristic for PD and associated with disease severity, whereas a reduction of stance time conversely stands for an improved gait. 66 In addition, a significant reduction of stance time can also be found in DTS7.
As a result of reduced step length, shorter swing time, and reduced gait speed, DLS time is affected by PD and prolonged. 81 Under STNP, STFP, and DTS7, it reduced significantly. Interestingly, DLS and single-limb support do not seem to be influenced by disease severity (H&Y), age, or disease duration. 81
It is remarkable that in STNP analysis, we had the most significant results and high BF10. This gait task also registers the most participants which can be explained by the composition of this task: it is low-threshold and performable even for patients with severe disease progression, MCI or with low physical condition, and can be implemented easily into clinical routine.
Daily life often requires simultaneous tasks, 86 whereas PwP report about difficulties absolving more than one task at the same time. 87 Dual task can reveal gait deficits that are not seen during single task and can raise fall risk. 88 Even analyses with healthy adults showed a reduced gait speed.69,88 Regarding PD, further changes in gait under dual-task conditions like shorter strides, increased DLS and increased variability are recognizable. 89 We can acknowledge these findings: in comparison to STFP results patient’s gait presented a slower gait speed, increased number of steps, step time, DLS, and stance during dual-tasking. Simultaneously, the interpretation of dual task is challenging because of inconsistent results.
Physiotherapy and specialized dual-task training improve dual-tasking performance86,90 and an analysis of dual-task gait outcome seemed promising. We found no significant changes in DBP during motor-motor dual-task, which is contrary to our previous findings, 20 while there is a significant improvement of several parameters during cognitive dual-task. This might be explainable with the neural networks that are involved 88 and the discussion if PwP prioritize gait over the cognitive task. 91 A potential cofounder is the cognitive performance of our sample, 52 and executive functions which are important for performing dual tasks. 88
According to the inconsistent results of our analysis and potential cofounders mentioned above, further research and detailed analysis of cognitive performance are needed before implementing it as a reliable monitoring parameter of PD-MCT. Additional information on dual-task performance as performance time, numbers of attempts or efficiency might be useful in future studies.
Of note, PD-MCT seems to have a positive impact on gait speed, number of steps, step time, stance time, and DLS in which changes of step time, stance time, and DLS are independent from gait speed and design (single- and dual task) of the analyses.
Clinical use
Device-based gait analysis allows a monitoring of effects gained through the PD-MCT. Concerning our findings, we suggest the use of device-based gait analysis to examine individual baseline characteristics at the beginning of PD-MCT to specify therapy according to individual needs. Also, monitoring of PD-MCT effects on gait seems promising but still needs additional investigation. In our analysis, we considered the results of STNP gait analysis as the most promising outcome parameter of PD-MCT which can be performed independently from disease stage or progression. However, the use of wearable digital devices also requires patient motivation. Short-term results of measurement and for patients understandable parameters may help to strengthen patient compliance and empowerment as an active player.21,31 A frequently discussed aspect is the potential of device-based home assessment which was already investigated in different studies.33,92 To monitor the long-term effects of PD-MCT outside the clinical environment, a home assessment should be implemented in future research and clinical practice.
Assessing the true performance of functional tasks including gait or dual-tasking using home-based rather than inpatient-based assessments will yield higher ecological validity and outcomes more relevant to the lives of PwP.
Regarding the growing number of PD8,93 and the need for constructive individualized therapies10,11 technologies like wearable digital devices can bring specific, continuous, quantitative, and objective data collection into clinical routine. 24 A complex disease like PD needs a complex treatment—to capture PD in its complexity wearable digital devices offer a promising approach for clinical daily routine.
Generalizability and limitations
Although our analysis refers to the German PD-MCT, results can be transferred into the context of comparable and internationally recommended multidisciplinary complex treatments.12,13
The results refer predominantly to male people with a moderate disease progression of PD (median H&Y stage 2.5), a mean age of 66.32 years, and a mean disease duration of 8.57 years.
There is a varied number of participants of the different gait tasks due to technical problems and infections with COVID-19 during PD-MCT. Governmental regulations for healthcare systems during the pandemic led to PD-MCT postponements and prioritization of patients with severe PD progression. 94
Participants were selected carefully according to inclusion criteria and are often highly motivated to participate in extra assessments. 95 Several other studies proofed differences in mobility between supervised and unsupervised conditions96,97 according to the Hawthorne effect.
Statistical aspects like the risk of overfitting of the binomial logistic regression model have also to be considered. We tried to avoid overfitting by a careful selection of the number of parameters contributing to the model.
Due to the explorative study design, there is a lack of a control group for evaluating the PD-MCT effects and claiming casual relations. Additionally, no power analysis or calculation of sample size were done for this subanalysis. Results should be interpreted carefully keeping the explorative character in mind.
Nevertheless, the impact of levodopa on motor functions must be kept in mind. The dopaminergic medication improves assured gait speed and stride length.68,72,98 Gait analysis was performed in medication ON state, potential overlapping effects of nonpharmacological treatments and medication adjustment as a regular part of PD-MCT have to be considered.
Furthermore, our results represent short-term effects immediately after the treatment. Studies with long-term follow-up examinations proved that motor improvements were still consistent in long-term follow-ups.15,16 Long-term follow-up of effects on DBP is still needed. It is important to note that PD-MCT focuses rather on capacity than performance. 38
In addition, the results refer to perceived ADL benefit rather than real-life ADL performance. Patient-reported outcomes have the potential for bias due to recall bias and subjectivity. 96 Future studies should evaluate the logistic model and gait parameters for PD-MCT with bigger sample sizes, control groups, a broader range of variability parameters and should use less artificial-functional task assessments taking potential differences between clinical and home assessment into account.
The borders between the inpatient, outpatient, and home-based sectors of health care may contribute to fading effects after discharge from PD-MCT. To ensure maintained effects on ADL, self-management—being active as PwP and changing health-related behaviors—and specialized outpatient nonpharmacological treatments delivered via networks of integrated PD care could play a crucial role. Further research into the role of proactive or reactive inpatient rehabilitation stays in the context of integrated PD care is pending.
Conclusion
Our results identified predictors of perceived ADL benefit from the widely implemented multidisciplinary inpatient PD-MCT considering clinical and device-based parameters. Young age, high initial perceived ADL impairment, high dexterity skills, and a steady gait are important factors of perceived ADL benefit after PD-MCT.
Using wearable technology in the clinical context of PD-MCT is promising, can support the information gained through clinical tests, and may reflect a more true-life condition.24,26
The findings of improved clinical scores, gait speed, step time, stance time, and DLS toward a faster, more fluid, and steadier gait might be a result of intensive motor training during inpatient PD-MCT and contribute to individual therapeutical concepts. At the moment, gait parameters still have to undergo more detailed research and have to be evaluated carefully 84 before being integrated as outcome parameters of PD-MCT.
With our research, we contribute to the evaluation of different gait parameters as potential monitoring parameters.
