Abstract
Background:
The calcium channel has been considered to have great potential as a drug target for neuroprotective therapy in Parkinson’s disease (PD), but previous studies yielded inconsistent results.
Objectives:
This study aimed to conduct a systematic review and meta-analysis to assess the relationship between using calcium channel blockers (CCBs) and the risk and progression of PD.
Data sources and methods:
The terms such as ‘Parkinson’s disease’, ‘PD’, ‘calcium channel blockers’, and ‘CCB’ were used to search the literature published before 1 May 2023 in English databases, including PubMed, Embase, and Cochrane Library, for studies on CCB and PD. Data analysis was performed using Review Manager 5.3 software.
Results:
A total of 190 works of literature were preliminarily retrieved, and 177 works of literature were excluded by eliminating duplicates, reading abstracts, and reading full texts. A total of nine studies were finally included in the meta-analysis of the CCB and the risk of PD, and five studies were included in the systematic review of the CCB and the progression of PD. A total of 2,961,695 participants were included in the meta-analysis. The random-effects model was used for analysis due to significant heterogeneity. The main results of the meta-analysis showed that the use of CCB could reduce the risk of PD (relative risk 0.78, 95% confidence interval 0.62–0.99).
Conclusion:
CCB use was associated with a significantly reduced risk of PD. Whether CCB use has a disease-modifying effect on PD needs further study.
Registration:
PROSPERO: CRD42024508242.
Introduction
Parkinson’s disease (PD) is one of the most common neurodegenerative disorders characterized by progressive degeneration of dopamine-releasing neurons in the substantia nigra (SN), leading to motor symptoms such as bradykinesia, rigidity, tremor, and nonmotor symptoms such as autonomic disturbance, sleep disorders, and cognitive decline. 1 Up to now, the therapy strategies of PD are all symptomatic, and disease-modifying therapy is lacking. 2
Calcium ion flows, which can modulate neurotransmitter release, muscle contraction, hormone secretion, and gene expression, have been found to play an essential role in the pathogenesis of PD. 3 The calcium channel has been considered to have great potential as a drug target for neuroprotective therapy in PD, 4 but previous studies yielded inconsistent results. Some studies reported significantly reducing PD risk in individuals using calcium channel blockers (CCBs).5–9 However, other studies failed to establish such a relationship.10–13 One study found that the use of CCB could reduce the risk of death in patients with PD. 7 Another study found that using CCB was associated with better long-term cognitive performance in patients with PD. 14 However, a randomized trial failed to identify a significant treatment effect for the clinical progression of isradipine in patients with PD. 15
Therefore, we aimed to conduct a systematic review and meta-analysis to evaluate the association between CCB use and the risk and progression of PD.
Materials and methods
Search strategy
This study followed the Preferred Reporting Items for Systematic Review and Meta-analysis guidelines (Supplemental Table 1). The study was registered on PROSPERO (CRD42024508242; https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=508242). We searched PubMed, Embase, and Cochrane Library for English language manuscripts published up to 1 May 2023. The search strategy included terms for CCB (calcium antagonists, CCB, calcium channel blocker, and Ca-antagonist) and PD (Parkinson, Parkinson’s disease, PD, and Parkinson disease). We also searched the references of included articles for potential additional reports.
Eligibility criteria
We included those studies that met all of the following criteria: (1) Either observational (cohort or case–control) or experimental study; (2) investigate the use of CCB and the risk or progression of PD; and (3) report at least a risk [relative risk (RR), odds ratio (OR), or hazard ratio (HR)] and a precision estimate [95% confidence intervals (CIs)] relating the use of CCBs and the risk or progression of PD or enough data to calculate them.
Studies were excluded if they were any of the following: (1) case reports, review articles, editorials, and clinical guidelines; (2) studies that did not provide effect estimates in RR, OR, or HR or did not allow the computation of such effect estimates, as well as only provide an effect estimate with no means to calculate a CI; and (3) studies that were not published in the English language.
Data extraction and quality assessment
Eligible articles were screened and reviewed independently by two investigators (JL and DP), and any discrepancies were resolved by discussion or by a senior investigator (JL). The following items were extracted for each study: first author, publication year, research country, study design, sample size, risk estimates, and 95% CI. The authors of the investigators were also contacted for further information as necessary.
The quality of the included studies was assessed independently using the Newcastle–Ottawa Scale (NOS) by two investigators (JL and DP). The NOS contained eight items that were categorized into three major components, including selection (0–4 stars), comparability (0–2 stars), and exposure (case–control studies) or outcome (cohort studies) (0–3 stars). The NOS scores of 1–3, 4–6, and 7–9 indicated low, medium, and high quality of studies, respectively.
Statistical analysis
We pooled all included studies’ HR, RR, or OR to get an overall effect estimate with 95% CI. Potential sources of heterogeneity between studies were assessed using Cochran’s Q and I2 statistics. I2 > 50% or p < 0.1 represented significant heterogeneity, and a random-effects model was chosen under this circumstance. Otherwise, a fixed-effects model was utilized. We also conducted sensitivity analysis by excluding one study at a time, reanalyzing the remaining studies to examine whether the results were altered substantially by any individual study. Forrest plots were used to summarize results, and funnel plots were used to assess publication bias. Subgroup analysis was conducted to evaluate a specific type of CCB and the risk of PD. All analyses were performed using Review Manager (version 5.3, Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration), establishing the significance level at a two-tailed p value of <0.05.
Results
Research results, study characteristics, and quality assessment
The literature search yielded 190 citations, of which nine studies that met the eligibility criteria were finally included in the present meta-analysis of the use of CCB and the risk of PD (Table 1). Five studies were finally included in the current systematic review of the use of CCB and the progression of PD (Table 2, Figure 1). Among the nine studies included in the meta-analysis, four were cohort studies,5–7,10 and five were case–control studies,8,9,11–13 yielding a total sample size of 2,961,695 participants. All nine studies were of high quality as assessed by the NOS score (Supplemental Table 2).
List and features of the studies included in the meta-analysis of CCB use and PD risk.

CCB, calcium channel blockers; CI, confidence interval; HR, hazard ratio; NOS, Newcastle–Ottawa Scale; OR, odds ratio; PD, Parkinson’s disease; RR, relative risk.
List and features of the studies included in the systematic review for CCB use and PD progression.

CCB, calcium channel blockers; PD, Parkinson’s disease; RCT, Randomized Controlled Trial.
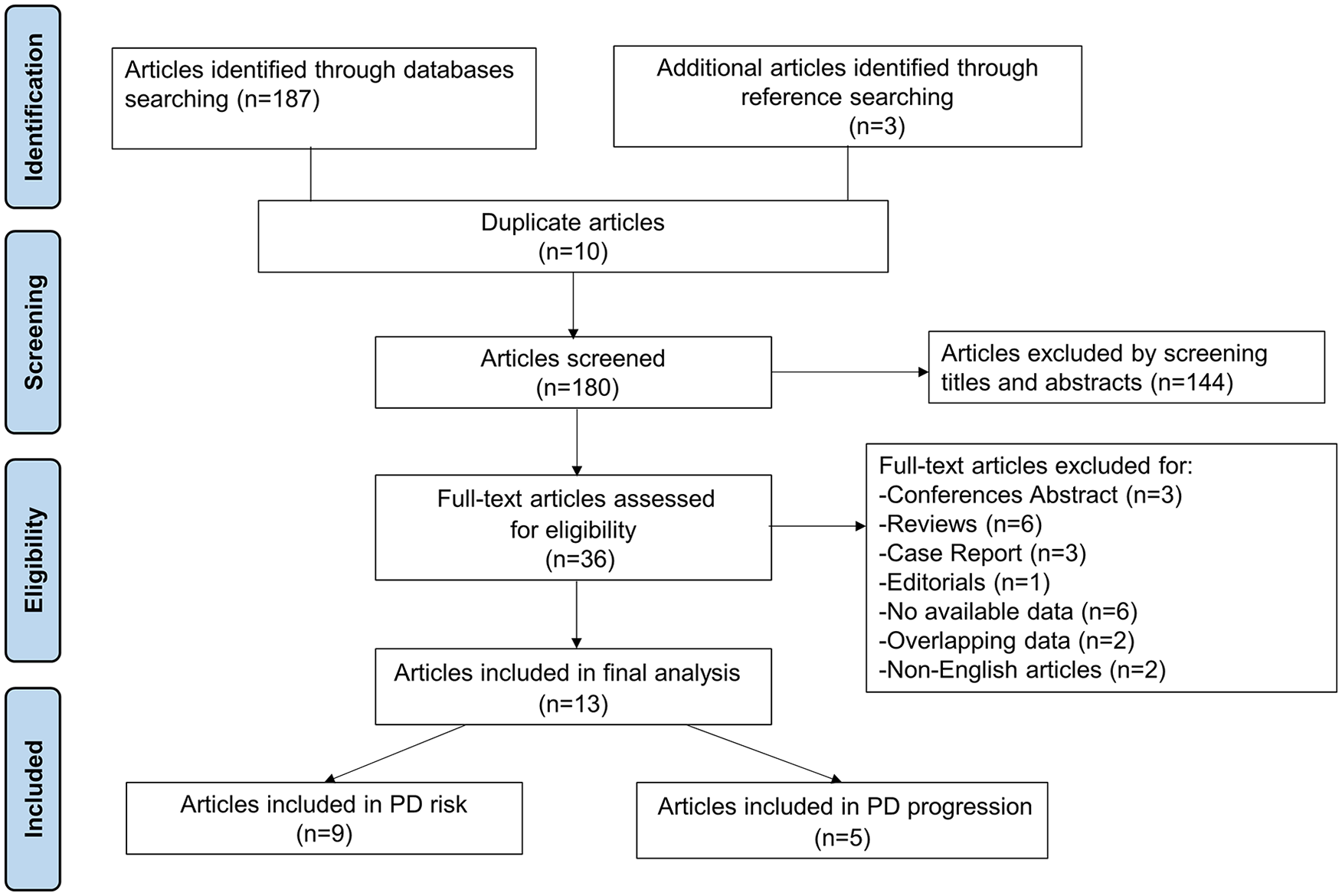
Flowchart representing the selection process.
Meta-analysis of CCB and the risk of PD
Since significant heterogeneity was observed in the included nine studies (I2 = 93%), a random-effects model was performed. The primary meta-analysis showed that the pooled effect estimate was RR 0.78, 95% CI 0.62–0.99 (Figure 2). The results confirmed a reduction effect of CCB use for PD risk. Sensitivity analyses showed that the main results remained robust. Inspection of the funnel plot did not suggest the presence of publication bias (Figure 3). Subgroup analysis showed that the pooled effect estimate for dihydropyridines (DHPs) use was RR 0.61, 95% CI 0.57–0.66 (Supplemental Figure 1), suggesting a reduction effect of DHP use for PD risk.

Forrest plots of the main results.
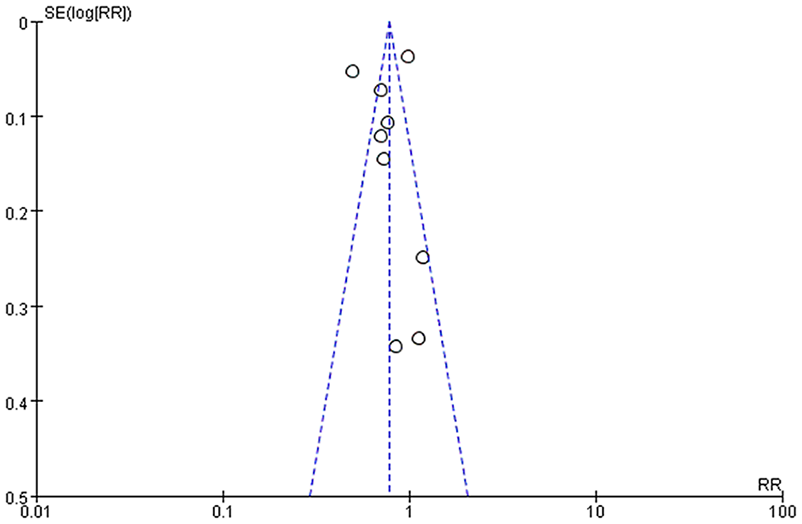
Funnel plots of the meta-analysis.
Systematic review of CCB and the disease progression of PD
Due to data limitations, we could not conduct a meta-analysis on the CCB use and the progression of PD. We have conducted a systematic review instead. As shown in Table 2, one study detected a protective role of CCB use on long-term cognitive outcomes of PD. 14 One study found an effect of CCB use on reducing the death risk of PD. 7 However, the other three studies failed to detect any impact of CCB use on the progression of PD.15–17
Discussion
By conducting a systematic review and meta-analysis, we found that CCB use could significantly reduce the risk of PD. However, whether CCB use could slow the progression of PD remains unclear.
The etiology of PD needs to be clarified. 18 Pathways such as mitochondrial dysfunction, oxidative stress, kinase pathways, calcium dysregulation, inflammation, protein handling, and prion-like processes have been considered to be implicated in PD so far. 19 The role of calcium in dopaminergic cell death associated with PD has broad experimental evidence. Neurons of the SN differ from other neurons in that they maintain autonomous electrical activity and, therefore, are exposed to increased calcium ion influx, which may increase Substantia nigrazona compacta (SNc) susceptibility to perturbations of calcium homeostasis. 20 CCBs may inhibit the Ca2+-dependent process of apoptosis. Based on such an antiapoptotic model, CCBs were subsequently hypothesized to be neuroprotective in PD and other conditions in which apoptosis contributes substantially to cell death. 21 The reduced risk of CCB use on the development of PD established in our meta-analysis was supported by most previously published studies.5–9
The L-type calcium channel with a pore-forming Cav1 subunit is the calcium channel that is widely targeted clinically, especially in cardiovascular disease. DHPs are negative allosteric modulators of these channels commonly used to treat hypertension because they can relax vascular smooth muscle. The Ca2+ channels underlying autonomous activity in dopaminergic neurons are closely related to the L-type channels found in the heart and smooth muscle. 20 The subgroup analysis of our study showed a reduction effect of DHP use for PD risk. Many experimental studies suggested a potential protective role of DHPs in PD. In neuronal cell culture, the DHP nimodipine significantly inhibited β-amyloid apoptotic neuronal injury. 22 In animal models of PD, nimodipine also prevented neurotoxicity induced by 1-methyl-4 phenyl-1,2,3,6-tetrahydropyridine (MPTP) in nonhuman primates 23 and mice. 24 One study found that nimodipine could attenuate mitochondrial dysfunction and rescue the loss of dopaminergic tyrosine hydroxylase positive neurons in the SN against MPTP-induced parkinsonism in mice. 25 Another study revealed that nimodipine is protective against dopaminergic neurons’ neurodegeneration via inhibiting the microglial-mediated oxidative stress and inflammatory response. 26 Another DHP felodipine had been found to have the ability to induce autophagy and clear diverse aggregate-prone, neurodegenerative disease-associated proteins and can clear mutant α-synuclein in mouse brains. 27 Another DHP, isradipine, can reduce dopaminergic cell damage from 6-Hydroxydopamine (6-OHDA) injection or MPTP administration in mice and suggested that using CCBs may confer protection against PD. 28
Multiple studies found that the use of central-acting DHPs, rather than peripheral-acting ones, was associated with a decreased risk of PD.6,8 A potential dose effect could also exist. One study found that DHPs reduced the risk of PD in a dose-dependent manner. 5 Another study found further decreased association for higher cumulative doses of DHPs (felodipine and amlodipine). 6 However, we could not conduct subgroup analysis on specific types or doses of DHP use due to the limited data. In addition, due to the limitations of the epidemiological database, most of the included studies were case–control studies or retrospective cohort studies. Further prospective studies with comprehensive information are needed to validate the hypothesis.
We identified two studies reporting a positive effect of CCB use on the progression of PD.7,14 Calcium interacts with α-synuclein and was a potential target for treating PD.29,30 A study indicated that CCB could potentially prevent spine enlargement by inhibiting the hypersensitivity of the synapse to excitatory inputs in PD and, therefore, could be a candidate for treating levodopa-induced complications in PD. 31 A phase III randomized trial failed to detect any effect on PD progression by using immediate-release isradipine. 15 However, another phase II randomized trial using controlled-release isradipine did show a significant disease-modifying impact with a smaller cohort of early-stage PD patients. 32 One possible explanation is that disease staging may affect the drug response. More studies are needed to identify whether CCB use has a treatment effect on PD in the future.
Our study has some strengths. First, we conducted a comprehensive systematic review and meta-analysis of the association between CCB and PD and provided evidence for the treatment potential of CCB on PD. Second, our subgroup analysis revealed that DHP could reduce the risk of PD. However, some limitations should also be acknowledged. First, there needs to be more data on the dose and duration relationship, which restrains us from conducting subgroup analysis on the dose and duration of CCB use. Second, we pooled the OR and RR values together due to the limited studies. Third, due to the data limitation, we could not conduct a quantified meta-analysis of CCB use and disease progression in PD. Finally, nonincluding articles published in other languages besides English might cause bias.
Conclusion
In conclusion, CCB use was associated with a significantly reduced risk of PD. Whether CCB use has a disease-modifying effect on PD needs further study.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241252713 – Supplemental material for Calcium channel blockers and Parkinson’s disease: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864241252713 for Calcium channel blockers and Parkinson’s disease: a systematic review and meta-analysis by Junyu Lin, Dejiang Pang, Chunyu Li, Ruwei Ou, Yujiao Yu, Yiyuan Cui, Jingxuan Huang and Huifang Shang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
