Abstract
Background:
The consideration of patient preference for a certain drug route of administration (RoA) plays an important role in promoting patient adherence in chronic diseases. Natalizumab is an established treatment for relapsing-remitting multiple sclerosis (RRMS) and can be administered as intravenous (IV) infusion or subcutaneous (SC) injection developed to enable a shorter and easier administration versus IV RoA.
Study objectives:
Primary objective is to compare patients’ preference for RoA and satisfaction with SC versus IV natalizumab at baseline and subsequent visits up to 12 months. Secondary objectives include drug utilization, clinical outcomes, safety, and treatment satisfaction in a usual care setting.
Design and methods:
SISTER (Subcutaneous: Non-Interventional Study for Tysabri Patient Preference – Experience from Real World) is an ongoing, prospective, observational study where natalizumab is utilized according to local label. RRMS patients are included in three natalizumab cohorts: Patients switching from current IV to SC administration (switcher) and patients newly starting natalizumab on either SC or IV route (starter SC/IV). This interim analysis includes 262 patients (184 switchers, 39 SC starters, and 39 IV starters), median observation period was 9 months.
Results:
80.8% IV starters and 93.9% SC starters reported at baseline that they prefer the assigned RoA. Although initial satisfaction with chosen RoA was maintained over time from baseline through Month 12 in all three cohorts, the wish for change of the current RoA after 6 and 12 months was more frequently expressed among IV starters than in either SC cohort. Consistently, six patients (23.1%) starting with IV changed their RoA from IV to SC route.
Mean global treatment satisfaction according to TSQM-II score at baseline remained high in the switcher group and increased through Month 12 in both IV and SC starter cohorts.
Conclusion:
Based on current data, there is a trend toward patients’ preference for the natalizumab SC route over the IV route, which provides valuable insights into patients’ preference for natalizumab RoA in routine care and complements available data from clinical studies with real-world data on SC natalizumab.
Trial registration:
This observational (non-interventional) study was registered in the local German PEI register for non-interventional studies (NIS-No. 611) and in the international CTgov register (NCT05304520).
Keywords
Introduction
Multiple sclerosis (MS) is a degenerative, autoimmune-inflammatory disease of the central nervous system (CNS) characterized by various areas of demyelinating lesions in the CNS and, thus, by a variety of clinical manifestations and courses. The etiology of MS is unknown, but most likely results from complex interactions between genetic and environmental factors. 1
MS is the most common form of inflammatory, degenerative, demyelinating CNS disease and is the most common cause of non-traumatic neurological disability in young adults. It affects more than 280,000 patients in Germany 2 and more than 2.8 million patients worldwide. 3 MS is often disabling, leading to a wide range of burdening symptoms, such as loss of vision, ataxia, tremors, bowel incontinence and/or urinary incontinence, generalized pain, fatigue, memory and learning problems, depression, and anxiety. 4 Due to the wide range of manifestations, debilitating nature, and onset during patients’ most active and productive years, MS has an enormous impact on patients’ physical, psychological, social, and economic well-being. MS causes a progressive reduction in patients’ physical and cognitive functions until patients need continuous assistance, thereby causing a high economic burden of MS on society and healthcare systems. 4 These disease characteristics underline the medical need for disease awareness and effective treatment options in all stages of the disease.
Although there are still no curative treatments available, research in the past years has led to major advances in therapies and treatment strategies for MS, especially for the most common type, relapsing-remitting multiple sclerosis (RRMS). Treatment options include disease-modifying therapies (DMTs) to prevent relapses and slow the accumulation of disability, relapse management therapies to shorten the duration and reduce the severity of acute disease exacerbations (mostly by means of corticosteroids), and symptomatic treatments to counteract specific symptoms of MS, such as pain or fatigue. 5
DMTs, in general, seek to suppress or modulate the immune system and reduce inflammation, slowing disease progression and helping prevent relapses. These drugs are administered via injection (e.g. beta interferons and glatiramer acetate), infusion (e.g. ocrelizumab and alemtuzumab), or as oral formulation (e.g. fingolimod, siponimod, and dimethyl fumarate).
As there is no consensus regarding the choice of one DMT versus another within defined efficacy categories, patient education and shared decision-making are important when establishing the best treatment plan for each individual patient. In addition, patient preferences, and patient-reported outcomes (PROs) should be considered when choosing a DMT, which also may improve treatment acceptance and adherence. PROs and patient preferences are captured directly from patients and offer a complementary perspective to that of clinician assessments and may provide greater insights into health status, function, symptom burden, adherence, health behaviors, and quality of life. 6
Natalizumab (Tysabri®, Biogen, GmbH, Munich, Gemany) is a recombinant monoclonal humanized anti-α4 integrin antibody. As α4β1-integrin was demonstrated to mediate the attachment of immune-competent cells to inflamed brain endothelium, the therapeutic effect of natalizumab is attributed to the inhibition of immune cell extravasation and inflammation in the CNS. 7 Intravenous (IV) natalizumab has been extensively investigated in clinical studies with a well-known efficacy and safety profile in patients with RRMS both in pivotal randomized clinical trials versus placebo8–10 and real-world scenarios. 11 Multiple cohort studies have proven positive effects of natalizumab on health-related quality of life (hrQoL).12,13 In real-world studies, patients treated with natalizumab experienced significant improvements in overall health and hrQoL.14–16 Natalizumab-treated patients more commonly reported improvements in disease activity and physical, emotional, and cognitive benefits than those treated with other DMTs.14,17–19 Natalizumab-treated patients reported significant improvements across a wide range of MS symptoms and functions, including working ability, fatigue, depression, and bladder/bowel disability.15,16,18,20–23
Since the European Medicines Agency (EMA) approval of IV and subcutaneous (SC) natalizumab in June 2006 and March 2021, respectively, through 31 July 2023, natalizumab (IV or SC) has been administered to 264,276 patients with a total of 1,117,808 patient-years of exposure, and SC natalizumab has been administered to 21,406 patients (corresponding to 24,659 patient-years of exposure). Natalizumab has a firm place as a DMT in the therapeutic armamentarium among patients with highly active RRMS according to current national 24 as well as international guidelines issued in the EU 25 and in the United States. 26
The World Health Organization (WHO) recognizes patient adherence in chronic diseases as one of the most important factors contributing to effective therapy. Poor adherence to long-term therapy severely compromises the effectiveness of treatment, making this a critical issue in population health both from the perspective of hrQoL and health economics. Interventions aimed at improving adherence would provide a significant positive return on investment through primary prevention (of risk factors) and secondary prevention of adverse health outcomes. 27 In this context, five different dimensions have been defined by the WHO that are known to affect adherence both favorably and unfavorably. One of these dimensions is a cluster of ‘therapy-related factors’, for example, those related to the complexity of the medical regimen, duration of treatment, previous treatment failures, frequent changes in treatment, early onset of efficacy, side effects, and the availability of medical support to deal with them. 27 This cluster also covers the route of administration (RoA) of treatment. A SC injection of natalizumab, administered in shorter time compared with established IV treatment, could simplify administration and improve patient convenience, thereby potentially promoting adherence of patients scheduled for long-term treatment with natalizumab.
First, clinical results of natalizumab SC compared with IV administration were generated in two clinical studies demonstrating comparable pharmacokinetic/pharmacodynamic properties and similar efficacy and safety outcomes.28,29 Subsequently, the SC route of natalizumab administration was approved by the EMA in March 2021.
Study objectives
According to the Summary of Product Characteristics (SmPC) of IV natalizumab, the diluted solution containing 300 mg natalizumab is to be infused intravenously over 1 h at a rate of approximately 2 mL/min. An SC formulation of natalizumab can be administered in shorter time and under simplified conditions as compared with the IV route and thus could improve patient adherence and convenience.
The choice of treatment and RoA for natalizumab should be based on shared decision-making between patient and healthcare professional (HCP) and can be based on a variety of different aspects. Real-world data on the interactions between HCPs and patients and the underlying decision processes are currently lacking in the literature.
To better understand the reasons for patients’ preference for RoA and general utilization patterns of SC natalizumab in routine care, additional and systematically collected clinical experience is needed. Therefore, the current prospective, observational non-interventional study (NIS) with three natalizumab treatment cohorts (i.e. natalizumab-naïve patients starting with IV and SC natalizumab, respectively or pre-treated patients switching from natalizumab IV to SC RoA) aims to collect and evaluate data on patients’ preference and therapy satisfaction with natalizumab, as well as clinical data on safety, immunogenicity, and effectiveness of SC and IV natalizumab administered in a routine healthcare setting. These study data are expected to provide additional information for a well-balanced decision-making and individually tailored use of the best suitable administration route to improve patients’ convenience and to contribute to the current knowledge on the clinical safety and effectiveness of SC natalizumab administered under real-world conditions.
Design and methods
Study design, setting, and regulatory requirements
SISTER (Subcutaneous: Non-Interventional Study for Tysabri Patient Preference – Experience from Real World) is a multicenter, prospective, non-controlled, non-interventional, observational cohort study conducted in Germany and Austria to evaluate the patients’ preference, utilization, safety, effectiveness, and PROs of SC and IV natalizumab over a treatment period of up to 1 year in RRMS patients treated in a usual care setting.
The study was registered (NCT05304520; PEI-No. 611) and approved by an independent Ethics Committee and by the national competent authority (Paul-Ehrlich Institute – PEI) and is conducted based on the Declaration of Helsinki according to all applicable laws and regulations.
Patients and treatment
A total of 500 adult patients with highly active RRMS according to the revised McDonald criteria 30 – and in other respects eligible for treatment with SC or IV natalizumab according to the local label as well – are planned to be enrolled and documented at about 60 study sites in Germany and Austria. No study-specific instructions relating to drug administration and patient management were made, and patients who have consented in writing to participate in the study were treated according to the SmPC for Tysabri. Patients with progressive forms of MS or with contraindications listed in the current natalizumab SmPC were excluded from enrollment.
To be eligible for study documentation, RRMS patients have to be treatment-naïve for natalizumab prior to starting SC or IV treatment or have to be on IV treatment and considering a switch from IV to SC RoA. Consequently, the three natalizumab treatment cohorts in the study are either patients switching from IV natalizumab to SC natalizumab (SC switchers; 300 patients planned) or patients starting natalizumab intravenously (IV starters; 100 patients planned) or patients starting natalizumab subcutaneously (SC starters; 100 patients planned; see Figure 1).
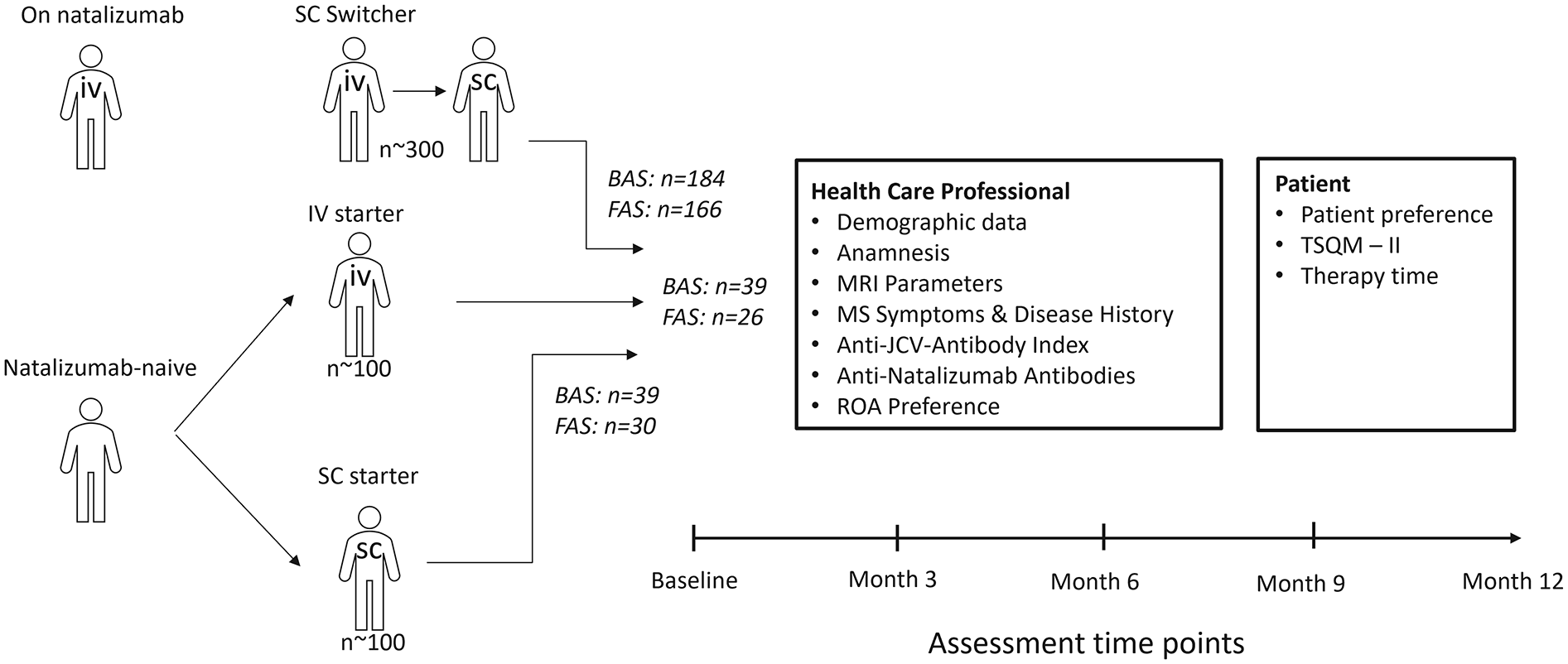
Study design.
A maximum of three SC natalizumab administrations are allowed prior to study inclusion, meaning that patients may have already been treated with 1–4 administrations at the time of the baseline assessments. During the observation period, a change of the initially chosen RoA (i.e. from SC to IV or vice versa) is possible and will be documented as an endpoint.
The individual study duration per patient is approximately 12 months. Patients who permanently discontinue natalizumab treatment before completing 12 months of observation are withdrawn from the study.
Criteria for evaluation
The main study objective is the evaluation of the patient preference between SC and IV cohorts. For this reason, four questionnaires were designed to evaluate both investigators’ and patients’ perceptions and treatment preferences (for wording of the four questionnaires, see Table 1; the two patient preference questionnaires are abbreviated as PPQ1 and PPQ2, respectively). Other objectives include the immunogenicity of SC and IV natalizumab in natalizumab-naïve patients [i.e. frequency of anti-drug antibody (ADA)-positive patients and persistently positive patients], course of disease activity [MS relapse rate, time to first relapse, disability improvement and progression according to Expanded Disability Status Scale (EDSS), magnetic resonance imaging, if available], patients’ treatment satisfaction [measured using Treatment Satisfaction Questionnaire for Medication (TSQM-II) 31 ], tolerability of treatment [occurrence of injection site reactions (ISRs) and other adverse events], and utilization of natalizumab (e.g. dosing intervals, duration of application, proportions of patients with treatment discontinuation, and switch of RoA during the study).
Study-specific questionnaires aiming at treatment preference.

Only in patients starting natalizumab (SC or IV) and assessed only at baseline.
Adapted from Lazaro Cebas et al. 32
At the time of site activation (Question 1 + 2) and intervals of 6 months afterwards (Question 2).
Assessed at baseline and at intervals of 6 months afterwards.
Adapted from Pivot et al. 33
HCP, healthcare professional; IV, intravenous; PPQ, patient preference questionnaire; SC, subcutaneous.
Observation time points in the study are aligned to clinical routine visits and cover a baseline visit and follow-up visits after approximately 3, 6, 9, and 12 months post-baseline (as far as routinely performed). Results of ADA testing are to be documented if tests have been routinely performed at the study sites. A sandwich/bridging enzyme-linked immunosorbent assay (ELISA) method is used at a central laboratory to determine the concentration of natalizumab antibodies in serum relative to a monoclonal anti-natalizumab antibody calibrator (I2C4).
Statistical analysis
No hierarchical hypothesis testing is sought in this study, and thus, all confidence intervals (CIs) and test results are of exploratory nature. However, for orienting considerations, it was assumed that preference rates of patients would be similar to those reported in the Her2+ breast cancer literature for the groups of trastuzumab SC starters versus IV starters, that is, 86% versus 60%.32,33 Therefore, this study will descriptively test for the potential superiority of the SC route versus the IV route in terms of patient preference at study start (primarily based on PPQ2) and over time at months 6 and 12 (based on PPQ1). Likewise, the sample size estimation for IV and SC starters in this study was roughly adapted to the aforementioned preference rates, with an estimate of 93 patients per starter arm needed to show a difference in preference rates of at least 10% with a power of 80%. Thus, 100 patients per starter arm (including drop-out reserve) were planned to be enrolled, while the planned number of 300 SC switchers was primarily based on feasibility considerations.
Generally, continuous variables are analyzed using suitable descriptive measures [e.g. mean, standard deviation (SD), median, first and third quartiles, minimum, and maximum], while categorical variables are presented in tabulated summaries showing missing values and absolute and relative frequencies. If not otherwise specified, percentages were calculated as adjusted percentages (i.e. without inclusion of missing values). For selected frequencies, two-sided exact 95% CIs according to Clopper-Pearson are displayed. Patient preference rates are compared using Fisher’s exact test (for dichotomous variables) or Chi-square test (for categorical variables with >2 categories). Time-to-event variables (e.g. time to first relapse) are presented by means of Kaplan-Meier estimates; a Poisson regression model adjusted for duration of disease and EDSS baseline score is used for the calculation of the annual relapse rate (ARR). No imputation of missing data is planned.
This report describes the results of a formal interim analysis performed with the data obtained by cut-off date 14 February 2023, where first patient’s first visit was on 29 September 2021 and last patient’s last visit on 10 February 2023. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) cohort reporting guidelines were followed for report preparation. 34 More comprehensive study results (including full analysis of adverse events) will be presented with the final study analyses.
Analysis populations are the ‘safety set’ (SAF), including all patients treated with natalizumab; the ‘baseline analysis set’ (BAS), which includes all patients who received at least one natalizumab dose after study enrollment and have at least one baseline documentation, and the ‘full analysis set’ (FAS), which includes all BAS patients with at least one post-baseline documentation. All data described in the current analyses are based on the BAS/SAF (N = 262), unless otherwise specified.
Results
Patient disposition and current duration of observation
From September 2021 through the data cut-off in February 2023, a total of 270 patients were enrolled in 33 centers (32 in Germany, 1 in Austria); 262 patients were eligible for inclusion in the SAF and BAS, respectively. Of these, 184 patients (70.2%) were allocated to the SC switcher cohort and 39 patients (14.9%) each to the IV and SC starter cohorts, respectively. The FAS included 222 patients (166 in the SC switcher cohort, 26 in the IV starter cohort, and 30 in the SC starter cohort; see Figure 1). A total of 216 patients (82.4%) have completed Month 3, 200 (76.3%) have completed Month 6, 155 patients (59.2%) have completed Month 9, and 96 patients (36.6%) have completed Month 12. So far, nine patients (3.4%) have discontinued the study prematurely due to treatment discontinuation (three in the switcher cohort, four in the IV starter cohort, and two in the SC starter cohort), resulting in a total treatment persistence of 96.6% by cut-off date. The current proportion of any patients who withdrew from the study is 5.3% (14 patients).
Currently, the mean duration of observation in the total study population is 7.8 ± 4.3 months (median: 9 months; range: 0.0–13.9 months), where the observation period is remarkably longer in the SC switcher cohort than in the IV and SC starter cohorts (median of 9.8 months versus 5.6 and 5.8 months, respectively).
Demographic and other baseline characteristics
The demographic data of the 262 patients at baseline (see Table 2) reflected a typical RRMS population characterized by predominance of women at a relatively young age. Here, 79% of study patients were women, and the mean age of the study population at baseline was 38.4 ± 10.0 years, ranging from 18 to 70 years (median: 38 years). As could be expected due to the longer disease history, patients in the switcher cohort were on average older than patients in the two starter cohorts (median age of 39.0 years versus 30.5 and 33.5 years, respectively).
Demographic data and other baseline characteristics (BAS/SAF).

Number of patients with missing values not included in this table.
Relative to time point of informed consent.
Time elapsed between occurrence of first symptoms and first diagnosis.
BAS, baseline analysis set; BMI, body mass index, BW, body weight; EDSS, Expanded Disability Status Scale; Gd, Gadolinium; NAT, natalizumab; Q, quartile; SAF, safety analysis set; SD, standard deviation.
Most common initial symptoms of RRMS (prevalence of ⩾10%) were sensory symptoms (53.3%), visual disturbances (34.2%), and pyramidal symptoms (17.7%). The mean time between the occurrence of first symptoms and study entry was 10.7 ± 7.5 years (median: 9.4 years), and the mean time since the first RRMS diagnosis was 9.5 ± 7.0 years (median: 8.5 years). Inherently, these periods were roughly longer by half among patients in the switcher cohort than among patients in the two starter cohorts (see Table 2). The mean difference between the occurrence of the first symptoms and the final diagnosis suggested a rather small therapeutic time loss of 1.1 ± 2.3 years (median: 0.1 years; third quartile: 1.0 years).
The mean EDSS at baseline was comparable among the cohorts with 2.2 ± 1.4 in switchers, 2.4 ± 1.3 in IV, and 2.2 ± 1.2 in SC starters. Most study patients (n = 167; 67.3%) were relapse-free in the year before enrollment.
The treatment with natalizumab prior to study entry in the switcher cohort showed a lower mean number of relapses in the past year (0.2 ± 0.5 versus 1.5 ± 1.4 in IV starters and 1.2 ± 0.9 in SC starters, respectively) and a smaller proportion of patients with gadolinium (Gd)-enhancing lesions on T1w scan at baseline in the switcher cohort versus the starter cohorts (21.3% versus 50.0% IV starters and 44.0% SC starters, respectively; see Table 2).
The proportion of patients negative for anti-JCV antibodies at baseline was 84.7% (216 patients); 30 patients had an anti-JCV-antibody-index ⩽0.9, 3 patients >0.9 to ⩽1.5, and 6 patients >1.5. 35
Prior MS treatments
Prior MS treatment at any time was reported for 216 BAS patients (82.4%); the mean number of treatments given before study entry was 1.7 ± 1.3 treatments (median: 1; range: 0–7); 46 patients (17.6%) had no prior treatment reported (see Figure 2). The last previous treatment of MS prior to study entry included the DMTs (if administered to ⩾5.0% of patients; N = 262; based on trade name): Copaxone (glatiramer acetate) (41 patients, 15.6%), Gilenya (fingolimod) (39 patients, 14.9%), Tecfidera (dimethyl fumarate) (39 patients, 14.9%), Aubagio (teriflunomide) (19 patients, 7.3%), and Rebif (interferon beta-1a) (19 patients, 7.3%).
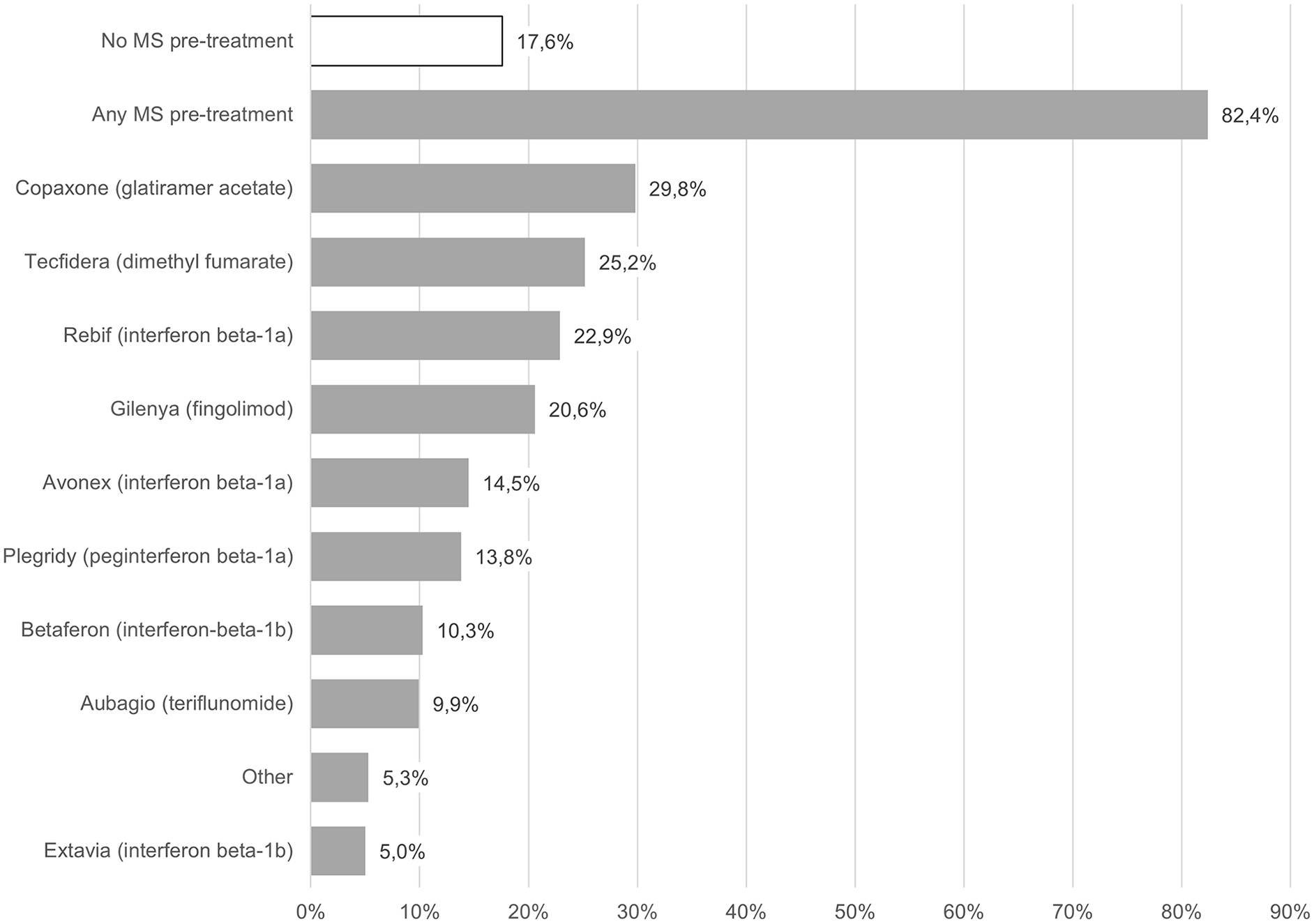
Any MS pre-treatments administered before start of natalizumab (BAS/SAF).
Utilization of natalizumab
During the current study observation period of 7.8 ± 4.3 months, 9.9 ± 4.0 natalizumab injections/infusions on average were administered to the 262 study patients in the BAS (median: 10; range: 1–14 injections/infusions).
The mean time on natalizumab treatment before study start in the switcher cohort was 4.8 ± 4.1 years (median: 3.6 years), and the mean number of infusions administered in the past 12 months was 8.5 ± 2.6 (median: 9).
Almost all IV/SC starters were planned to receive natalizumab at intervals of 4 weeks, while the proportions of patients with extended intervals were higher in the SC switcher cohort (see Table 2). At Month 3, 49.1% (80 patients) were on every 4 weeks, 13.5% (22 patients) on every 5 weeks, 36.8% (60 patients) on every 6 weeks, and 0.6% (1 patient) on every 8 weeks dosing intervals in the SC switcher cohort. In the IV and SC starter cohorts, the majority of patients were on 4-weekly dosing intervals (91.7% and 92.9%, respectively).
Natalizumab administration modalities showed that the procedure time (i.e. preparation, administration, and follow-up duration at the site) at baseline was distinctly shorter in the two SC cohorts (median of 1 h each) compared with the IV starter cohort (median of 3 h; see also mean values in Figure 3). Currently, available post-baseline visits suggested similar results.

Mean natalizumab administration time (h) at baseline (BAS/SAF).
Preliminary analyses of RoA preference
Initial patient preference at baseline
The analysis of patients’ initial preference at baseline (by PPQ2; for wording, see Table 1) showed that the vast majority of patients in both starter cohorts had preferred their actual RoA (80.0% in the IV starter cohort and 93.9% in the SC starter cohort; no statistically significant difference between cohorts). No particular preference was expressed by five patients (14.3%) in the IV starter cohort and by two patients (6.1%) in the SC starter cohort. Thus, two patients in the IV starter cohort (5.7%) were apparently treated against their preferences. The most commonly specified reason for the preference in the two starter cohorts was a more comfortable administration among IV starters (46.2%) and time-saving among SC starters (53.8%). A ‘very strong’ preference for their chosen treatment was reported by five IV starters (17.2%) and nine SC starters (30.0%; see Table 3).
Analysis of patients’ preference at baseline – PPQ2 (BAS/SAF).
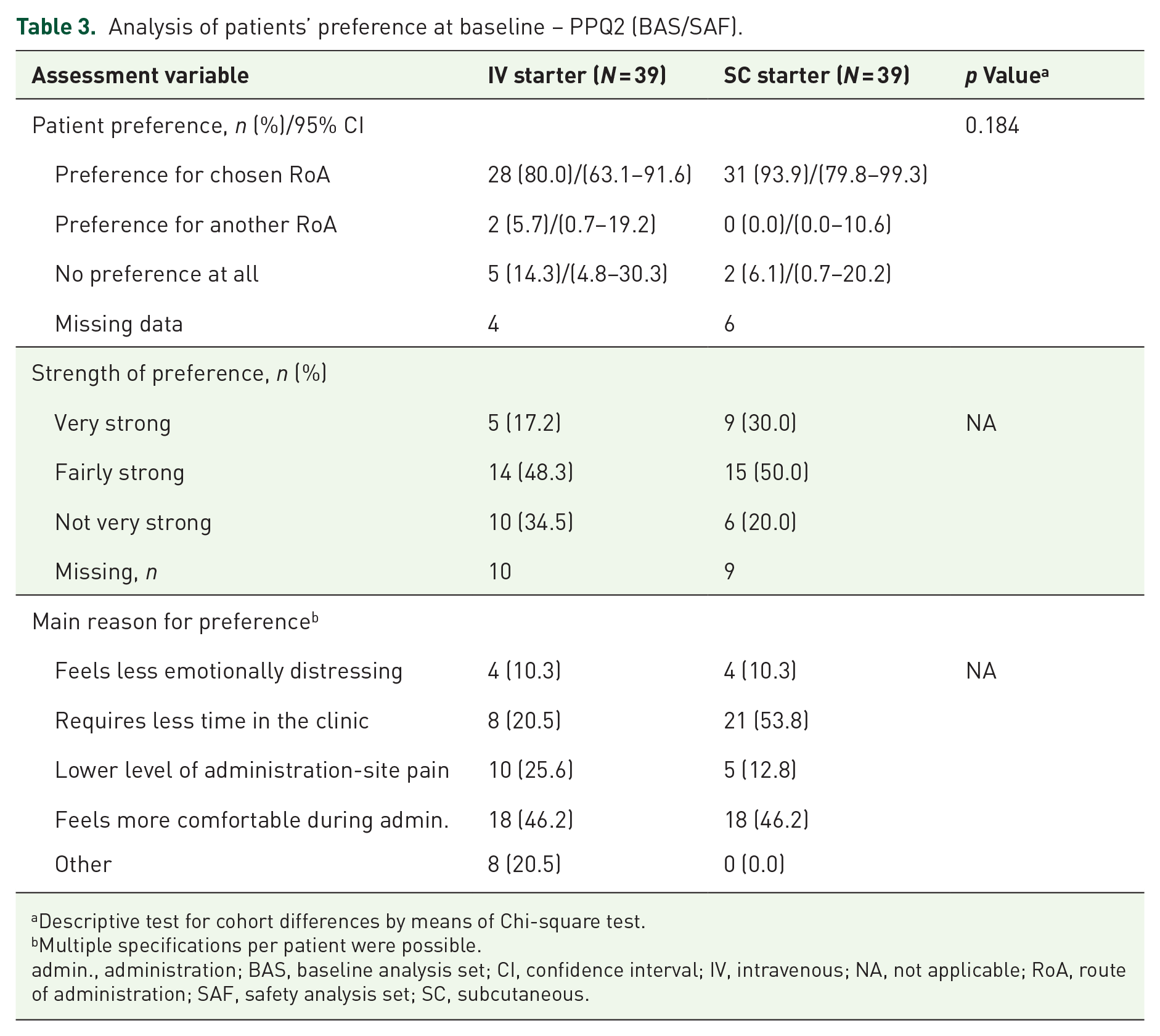
Descriptive test for cohort differences by means of Chi-square test.
Multiple specifications per patient were possible.
admin., administration; BAS, baseline analysis set; CI, confidence interval; IV, intravenous; NA, not applicable; RoA, route of administration; SAF, safety analysis set; SC, subcutaneous.
Changes in patient preference over time
PPQs documented every 6 months allow the assessment of changes over time reflecting the satisfaction with treatment and the wish to change the RoA at a given time point (see Table 1). While the satisfaction with the current RoA remained high at Months 6 and 12 in all three cohorts [see Figure 4(a)], the proportions of patients who confirmed their initially chosen RoA as satisfactory were numerically lower (and the proportions of patients considering a change of the initially chosen RoA thus higher) in the IV starter cohort than in the two SC cohorts [see Figure 4(b)]. These data indicate a trend toward the preferred use of SC natalizumab also among patients who were initially treated with IV natalizumab.

Patient preference at baseline and Months 6 and 12 (BAS).
Proportions of patients with change of initial RoA
Consistent with the aforementioned trend, 6/26 patients with post-baseline data (FAS; 23.1%) in the IV starter cohort have changed their RoA from IV to SC, while none of the SC starter patients were switched to the IV RoA to date. These changes were all due to the patient’s wish. In addition, 7/184 patients (3.8%) in the SC switcher cohort had a change of RoA during the current study period (mainly due to patients’ wish).
HCP Preference assessments at natalizumab initiation
Questions addressed to the HCPs are listed in Table 1. For the 78 natalizumab starters (IV or SC), the majority of HCPs considered both RoAs as equivalent treatment options (n = 73; 93.6%). Most common reasons for HCPs’ preference when prescribing the SC RoA (11 patients overall) were ‘administration less complicated/less time consuming than IV’ (5 patients, 45.5%) and ‘efficacy and safety considered comparable’ (3 patients, 27.3%).
One of the most common reasons for HCPs’ preference when prescribing the IV RoA (31 patients overall) was ‘more experience with administration route’ (13 patients, 41.9%).
Experience of nursing staff at baseline (N = 17) showed that all sites had experience with SC natalizumab, 16 sites (94.1%) had experience with IV natalizumab, and 13 sites (76.5%) each had experience with other IV and SC drugs. At baseline, most of the 17 study sites stated that they would prefer the SC route over the IV route in terms of both preparation (15/17 sites, 88.2%) and administration (14/17 sites, 82.4%). By Month 6 (data from 10 centers available), 9 centers (90%) still prefer both the preparation and the administration of SC natalizumab.
Global treatment satisfaction measured with TSQM-II
Mean TSQM-II global satisfaction score values at baseline were 83.0 ± 24.3 in the SC switcher cohort (N = 163), 66.4 ± 25.3 in the IV starter cohort (N = 29), and 67.8 ± 20.5 in the SC starter cohort (N = 29). Thus, baseline treatment satisfaction was higher in the switcher cohort than in the two starter cohorts. While the global treatment satisfaction remained stable in the SC switcher cohort, there were similar increases in the two starter cohorts visible already at Month 6, which remained stable through Month 12 (IV starter: 80.0 ± 15.7; SC starter: 82.4 ± 15.6; see Figure 5).
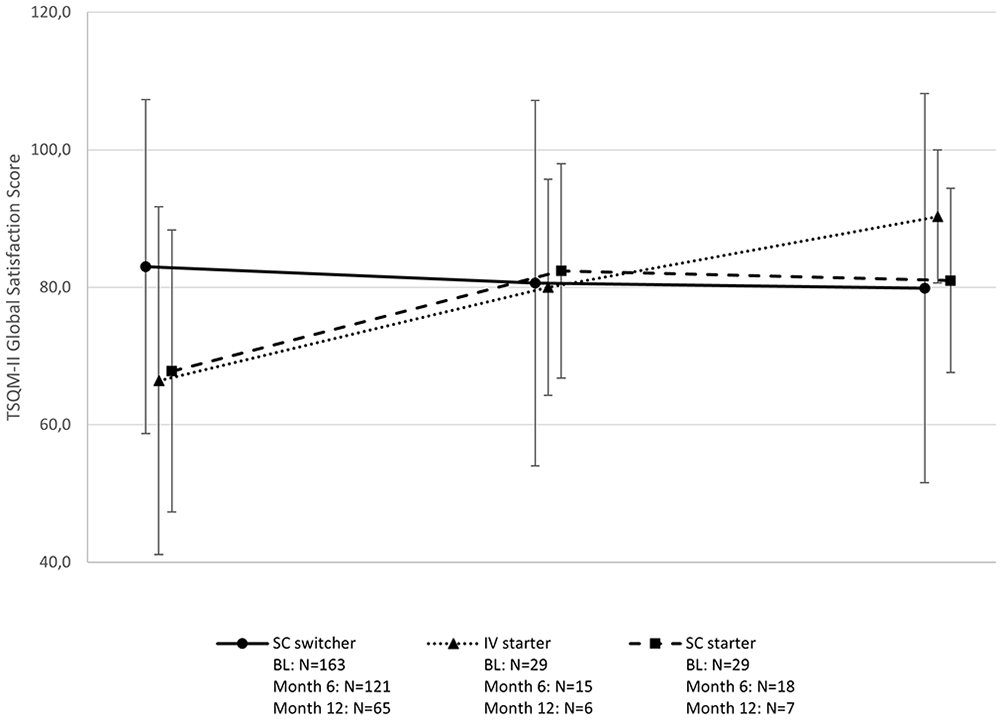
TSQM-II global satisfaction score by visits (BAS/SAF).
Preliminary effectiveness outcomes
MS relapses occurred in 5/166 patients in the SC switcher cohort (ARR: 0.03389), 1/26 patients in the IV starter cohort (ARR: 0.04840), and 3/30 patients in the SC starter cohort (ARR: 0.1749; data are for the 222 FAS patients). Due to the small number of patients with relapse, no median time-to-event can be calculated.
The mean post-baseline EDSS values remained almost identical through Month 12 in all cohorts. Consistently, most patients with available data showed ‘stable disease’ up to Month 12 based on the individual changes in the EDSS over time. By trend, individual changes in EDSS (both improvement and progression) were observed in the two starter cohorts, while in the SC switcher cohort (where patients had already been on IV treatment with natalizumab prior to study entry), the EDSS remained stable. In terms of occurrence of new symptoms during the entire study, two patients (7.7%) in the IV starter cohort, four patients (13.3%) in the SC starter cohort, and three patients (1.8%) in the SC switcher cohort presented new symptoms during the observation period. Due to low patient numbers and short observation time, these results should be considered exploratory.
Preliminary safety outcomes: Immunogenicity and injection-related adverse events
None of the patients were ADA-positive at baseline (after 1–4 natalizumab administrations), and 157/160 patients with available data (123 switcher, 15 starter IV, and 22 starter SC) remained ADA-negative at all time points.
Injection-related AEs as reported in patients’ questionnaires focused on the patients treated with SC natalizumab (184 patients in the SC switcher cohort and 39 patients in the SC starter cohort; 223 patients in total). These events were reported for 33 patients (14.8% based on all 223 patients), and in 17 patients (7.6%) the events occurred at the injection site. Most common local ISRs were injection site pain (16 events in 13 patients), followed by redness (11 events in 10 patients), pruritus (3 events in 3 patients), and inflammation (2 events in 2 patients). The majority of the 32 local ISRs were mild (16 events) or moderate (9 events); the remaining 7 events had a ‘high’ intensity (see Table 4) as assessed by patient and confirmed by HCP.
Analysis of injection site reactions – PPQ1, Question 3 (BAS/SAF).

SC switcher cohort and SC starter cohort combined. No clinically meaningful differences were observed between the two cohorts. Percentages are adjusted, that is, the denominator is based on patients with available observation (i.e. with distinct statement of yes or no).
Given is the severity by number of events using the categories 1 = mild, 2 = moderate, 3 = high, 4 = very high, and 5 = intolerable (see Table 1).
Worthy of note, no cases of progressive multifocal leukoencephalopathy were observed during the course of the study so far.
Discussion
The non-interventional, observational SISTER study evaluates treatment preferences and clinical course of patients with RRMS treated with intravenously or subcutaneously administered natalizumab up to 12 months in a real-world setting. For the current analysis, data from 262 patients enrolled at 33 study sites with a mean observation period of 7.8 ± 4.3 months (median: 9 months, range: 0–13.9 months) were analyzed in a descriptive manner.
The observation period was remarkably longer in the SC switcher cohort than in the IV and SC starter cohorts. This imbalance can be explained by a higher recruitment rate in the switcher cohort compared with the two starter cohorts, where the available pool of suitable de novo patients is inherently smaller.
The HCPs’ experience and preference from 17 currently active sites with documentation at baseline (N = 17) indicated that all sites had experience with SC natalizumab, 16 sites (94.1%) had experience with IV natalizumab, and 13 sites (76.5%) each had experience with other IV/SC drugs. Interestingly, most of the 17 study sites documented that they prefer the SC over the IV route in terms of both preparation (88.2%) and administration (82.4%). Thus, there appeared to be a high general acceptance of SC treatment among the participating sites. By Month 6 (data from 10 centers are currently available), 9 centers (90%) still stated that they would prefer both the preparation and the administration of SC natalizumab, suggesting that the general preferences of study sites in favor of the SC administration had not changed over time.
Shared decision-making with patients seems to be the key to optimum care for MS patients, given the preference-sensitive nature of decisions to be made between HCPs and MS patients and the increasing numbers of available DMTs. 36 Within this context, the focus of the SISTER study is the patients’ preference between IV versus SC RoA. Current study data indicated that the patients starting natalizumab initially have a high preference for their assigned RoA, where this preference was more pronounced in the SC cohort compared with the IV cohort (93.5% patient preference for SC and 80.0% for IV). Among SC starters, both time-saving aspects (53.8%) and administration convenience (46.2%) were the most frequently specified, patient-reported reasons for their preference for the SC route. Administration convenience (46.2%) and lower pain levels during administration (25.6%) were the most common reasons among the IV starters.
The course of patient preferences over time indicated that the initial satisfaction with current RoA was maintained over time from baseline through Month 12 among the three treatment cohorts (always >90% of patients were satisfied), whereas the wish for a change of the current RoA at Months 6 and 12 was more frequently expressed by patients in the IV starter group than in the two SC cohorts. Thus, there appeared to be a trend towards a change from IV to SC administration post-baseline among the patients who were initially treated with the IV route, but this finding remains to be strengthened with more patients and mature data.
This trend was supported by the observation that six FAS patients (23.1%) in the IV starter cohort had changed their RoA from IV to SC route; these changes were solely triggered by patient’s wish. No SC starters had changed the RoA by the end of data cut-off date, while seven patients (3.8%) in the SC switcher cohort had changed back to IV during the current study period (due to patients’ wish in six patients and due to physician’s decision in two patients; no cases of ISR or needle fatigue were reported as reason).
Importantly, the treating HCPs considered the two available routes of natalizumab as equal treatment options for almost all starting patients (93.6%), suggesting a broad leeway for a free choice of RoA in the majority of patients since potential absolute restrictions (e.g. injection anxiety or poor venous access) are obviously infrequent. An important factor for the HCPs’ choice of the IV route was the familiarity with that administration route (41.9% of patients), while the choice of the SC route was triggered (among others) by the easy and time-sparing administration (45.5%) and clinical similarity of the IV and SC routes (27.3%).
These results provide insight into shared decision-making, weighting of HCPs’ and patients’ preferences and demonstrate improved patients’ involvement in therapy decisions.
Inherent limitations of open-label, non-randomized, single-arm studies such as the SISTER study contain an inherent risk of selection bias and attrition bias. Moreover, this single-arm study with three natalizumab cohorts does not employ a randomized parallel control group, and potential correlations between treatment and outcome variables can only be interpreted as descriptive associations. Another limitation of the study is that the required sample sizes for treatment starters could only be roughly estimated based on a different patient population (breast cancer patients treated with trastuzumab), which was not necessarily a reliable reference population for the MS population enrolled in this study. In addition, the PPQs used in this study were adapted from other studies but are not formally validated instruments (in contrast to TSQM-II). On the other hand, NISs allow the assessment of clinical outcomes and potentially rare AEs in larger and more heterogeneous populations in real-world settings than those in clinical trials and thus can be useful as complementary data to corroborate the results of pivotal clinical trials.
In addition, it should be considered in the SISTER study that patients in the SC switcher cohort had already been on IV treatment with natalizumab and thus may represent a rather positive selection of relatively stable patients who have benefitted from prior IV natalizumab treatment, while the treatment response of the de novo starters with either IV or SC natalizumab is uncertain and thus more dynamic after treatment initiation. This imbalance between switchers and starters (e.g. mean age, disease duration, and proportions of patients with relapses in the past year and Gd-enhancing lesions at baseline in the switcher cohort) is likely to explain the descriptive post-baseline differences between these cohorts in terms of current effectiveness outcomes (i.e. relapse rate, EDSS score). Moreover, it has to be taken into account that, due to recruitment issues, the sample size and duration of study observation were distinctly higher in the switcher cohort as compared with the starter cohorts, and that the results seen among the switchers are more robust than in the smaller-sized starter cohorts, particularly at later visit time points.
Preliminary relapse rates were small in all treatment cohorts and lowest in the SC switcher cohort, suggesting a smooth switch from IV to SC treatment [213/222 patients overall (95.9%) remained relapse-free]. Generally, ARRs and proportions of patients with non-stable disease (i.e. progression on EDSS) were slightly higher in the de novo starter cohorts than in the switcher cohort. However, different patient characteristics and small patient numbers, particularly in the starter cohorts, need to be considered for the interpretation of the results.
No differences in clinical effectiveness were seen between the IV and SC starter cohorts based on preliminary study data, consistent with the pivotal studies DELIVER 28 and REFINE. 29 The incidence of local ISRs among the 223 patients receiving the SC route was 7.6%, with the majority of the 32 reported ISRs being mild or moderate.
In the immunogenicity analysis, 157/160 patients (123 switchers, 15 starter IV, and 22 starter SC) were anti-natalizumab antibody-negative at all available time points. This suggests low immunogenic potential of natalizumab and comparable immunogenicity between both RoAs.
From a prospective, observational, single-arm study performed in the United States, Foley et al. reported physical and psychological hrQoL improvements (SF-12, MSIS-29, TSQM-9) over 3 years of IV natalizumab treatment in 120 RRMS patients, supporting the long-term efficacy of natalizumab in real-world settings. 12 Treatment satisfaction (measured using the TSQM-9) increased after natalizumab initiation (mean for global satisfaction: 67.38 points) and remained high over 3 years of treatment (Year 1: 84.59 points). These data are similar to those currently observed in the two starter cohorts in the SISTER study, which underline high treatment satisfaction with natalizumab given on either route.
The treatment discontinuation rate by the end of the cut-off date (overall 3.4% treatment discontinuations, 5.3% study withdrawals) is considered low in all cohorts, thereby suggesting a high treatment persistence (96.4% during a median observation period of 9 months).
Extended dosing intervals of >4 weeks were more frequently planned and observed in the SC switcher cohort than in the two starter cohorts. This difference might be explained by the fact that patients in this cohort were already on established therapy, had a longer exposure and stable disease as compared with the de novo starters, and thus were considered more suitable for extension of dosing intervals.
Overall conclusion
This is the first report of real-world data collected with SC natalizumab in Germany and Austria. Patient preference for the initially chosen RoA, which is mandatory for adherence and thus long-term treatment success, is high among both IV and SC natalizumab starters at baseline. However, preliminary results suggest that natalizumab patients have stronger preference for the SC RoA after treatment initiation. ISRs were consistent with the known safety profile of SC natalizumab.
Overall, SC natalizumab is a widely accepted and preferred alternative to IV natalizumab. The time savings of about 2 h during the administration procedure compared with IV administration are regarded as an additional benefit for both patients and medical staff. It is anticipated that the use of SC natalizumab fulfils the users’ expectations and might further improve patients’ adherence to therapy, which is an important prerequisite for treatment success in the long term. These early interpretations will be corroborated with the more mature, final SISTER study results (expected in Q4 2024) as well as with data from other, currently ongoing studies (e.g. the NOVA Phase 3b extension study with a cross-over design; NCT03689972).
Footnotes
Acknowledgements
Statistical analysis (Elisabeth Grünewald) and medical writing support (Dr. Bernd Graulich) were provided by the Contract Research Organization Winicker Norimed GmbH, Nuremberg, Germany. Manuscript submission was supported by Winicker Norimed GmbH, Nuremberg, Germany. This assistance was funded by Biogen GmbH, Munich, Germany. All listed authors have authorized the submission of their manuscript via a third party and approved any statements or declarations, for example, conflicting interests, funding, etc.
