Abstract
Background:
Western lifestyle has been associated with an increase in relapsing–remitting multiple sclerosis (RRMS). In mice, dietary wheat amylase–trypsin inhibitors (ATIs) activate intestinal myeloid cells and augment T cell-mediated systemic inflammation.
Objective:
The aim of this study was to assess whether a wheat- and thus ATI-reduced diet might exert beneficial effects in RRMS patients with modest disease activity.
Methods:
In this 6-month, crossover, open-label, bicentric proof-of-concept trial, 16 RRMS patients with stable disease course were randomized to either 3 months of a standard wheat-containing diet with consecutive switch to a > 90% wheat-reduced diet, or vice versa.
Results:
The primary endpoint was negative, as the frequency of circulating pro-inflammatory T cells did not decrease during the ATI-reduced diet. We did, however, observe decreased frequencies of CD14+ CD16++ monocytes and a concomitant increase in CD14++ CD16− monocytes during the wheat-reduced diet interval. This was accompanied by an improvement in pain-related quality of life in health-related quality of life assessed (SF-36).
Conclusion:
Our results suggest that the wheat- and thus ATI-reduced diet was associated with changes in monocyte subsets and improved pain-related quality of life in RRMS patients. Thus, a wheat (ATI)-reduced diet might be a complementary approach accompanying immunotherapy for some patients.
Registration:
German Clinical Trial Register (No. DRKS00027967).
Keywords
Introduction
The contribution of dietary factors to the pathogenesis of multiple sclerosis (MS) and the disease-modifying potential of dietary interventions are topics of increasing interest. Recent studies demonstrated immunomodulatory effects of diets enriched in propionic acid 1 or conjugated linoleic acid, 2 and that a ketogenic diet improved signs of fatigue and depression in patients with relapsing–remitting multiple sclerosis (RRMS). 3 Furthermore, 5.5% of American MS patients follow a gluten-free diet (GFD) and believe it to be effective. 4
Wheat has become the most widely consumed food staple worldwide, which has been accompanied by an increase in hypersensitivities to wheat. 5 With gluten being the clearly identified trigger of celiac disease, it has long been suggested that gluten proteins also mediate the other hypersensitivities. However, recent findings have highlighted the important role of non-gluten wheat proteins, the amylase–trypsin inhibitors (ATIs), in the underlying pathogenesis of intestinal and extra-intestinal inflammatory diseases.6–12
ATIs constitute a small proportion of the protein fraction of various gluten-containing cereals (~2–4%). Thus, a GFD can also be considered ATI-free.8,13 ATIs act as triggers of the innate immune system. In myeloid cells, the binding of ATIs to the toll-like receptor 4 (TLR4) results in the activation of both the classical and the non-classical nuclear factor kappa B (NF-ĸB) pathway. 6 Moreover, ATIs promote a pro-inflammatory intestinal dysbiosis by a direct interaction with the microbiota. 14 We hypothesize that the mild pro-inflammatory intestinal signal induced by nutritional ATI is propagated toward extra-intestinal organs by migration of activated myeloid cells out of the gut where they can co-stimulate T cell-mediated preexisting inflammation. 8 This can induce stronger antigen-specific adaptive immune responses with a consecutive exacerbation of disease activity. 7 Indeed, in murine models of intestinal or allergic airway inflammation, and non-alcoholic fatty liver disease, ATI feeding in quantities comparable to the average consumption of humans, promoted airway, liver, and adipose tissue inflammation.8-11,14 In a recent first clinical pilot trial in patients with familial Mediterranean fever, we could show a marked pro-inflammatory effect of a wheat- and thus ATI-based diet compared to a wheat-free diet, both in clinical symptoms and immune-relevant parameters. 15 As the described effects of ATIs are dose-dependent, a reduction of daily ATI ingestion by 90–95% or even less may be sufficient to abolish their co-stimulatory effect on chronic T cell-mediated inflammation. 7
In experimental autoimmune encephalitis (EAE), the murine model of MS, a markedly more severe disease course paralleled by a shift toward a pro-inflammatory immune cell phenotype, was observed in mice that were fed purified ATI concentrations comparable to a standard western diet. 16 The aim of our current study was thus to evaluate if these promising preclinical findings could be translated into clinical strategies to modulate the immune response in MS by reducing dietary ATIs.
Materials and methods
Subjects
Subjects diagnosed with RRMS 17 were enrolled into this 6-month, crossover, open-label, bicentric pilot study. Among the 20 subjects initially included in the study, the 3- and 6-month visits were completed by 16 patients. Four patients decided to quit the study due to personal reasons, such as moving and extended traveling.
To be included, patients had to be aged between 18 and 60 years and to maintain an unchanged mild or moderate disease-modifying therapy (DMT). Furthermore, patients had to be relapse-free for at least 3 months prior to study enrollment. Exclusion criteria were the presence of other autoimmune diseases, previous limiting dietary habits, organ transplantation, or the intake of any other immunosuppressive drugs apart from the MS medication.
The primary study endpoint was defined as a decrease in pro-inflammatory T cell populations in the peripheral blood on a wheat/gluten- and therefore ATI-reduced diet (W/G/A−). Secondary study endpoints included the decrease in other pro-inflammatory immune cell populations in peripheral blood, an improvement in health-related quality of life assessed by the 36-item short form survey (SF-36), clinical disease stability assessed by the Expanded Disability Status Scale (EDSS) score, and a reduction of the annualized relapse rate (ARR). In addition, serum neurofilament light chain (sNfL) levels as a biomarker of neuroaxonal damage were assessed.
This study was registered in the German Clinical Trial Register (No. DRKS00027967).
Study procedures
At the baseline visit, patients were randomly assigned to one of the two study arms in a crossover study design (Figure 1). They were instructed to either continue on their normal wheat/gluten- and thus ATI-containing diet (W/G/A+) for 3 months and then switch to the largely wheat- and ATI-free diet (reduction of at least 90%) for the following 3 months (W/G/A−), or started with 3 months of the W/G/A− diet with a subsequent switch to their normal W/G/A+ diet. Wheat consumption was assessed and monitored by structured food questionnaires (see supplement) before study entry, at diet switch and at study end.

Study design.
Clinical and patient-reported outcomes
At baseline visit, demographic and MS-related data, including disease history, history of relapses, and past and current DMTs, were collected. The baseline visit and every consecutive study visit in 6-week intervals included general physical and neurological examination and the assessment of self-reported quality of life. Weight and height were measured, and the body mass index (BMI) was calculated. EDSS scores were assessed by an experienced neurologist blinded to the diet assignments. 18 The occurrence of new neurological symptoms indicative for a relapse was documented at each visit. Quality of life was determined using SF-36, which is a widely used patient-reported outcome covering eight domains of physical and mental quality of life (physical functioning, role limitations due to physical health, role limitations due to emotional problems, energy and fatigue, emotional well-being, social functioning, pain, and general health). 19 Item reports were summed without weighting separately for each domain, and ranges were transformed to have a common range of 0 (worst health) to 100 (best health) as described previously. 20
Measurement of gluten immunogenic peptides
Diet adherence was monitored by the measurement of gluten immunogenic peptides (GIPs) in 4-h urine samples at every study visit (Glutenostics, Indianapolis, USA). GIPs are degradation products of gluten and can be detected in urine for 3–34 h after the consumption of at least 25–50 mg of gluten. 21
sNfL single-molecule array
sNfL levels of study participants were measured by sNfL single-molecule array (SiMoA) technology as previously described 22 (see supplemental material).
Immunophenotyping
High-quality peripheral blood mononuclear cells (PBMCs) were isolated from all patients at baseline, and at the end of the respective diet intervals at the 3- and 6-month time points. Multiparameter flow cytometry of immune cells in peripheral blood was performed as described previously. 23 Supplementary Figure 1 illustrates the immune cell parameters as determined by conventional gating.
Statistical analysis
The data from all subjects completing the 3- and 6-month time points of the crossover period were included into statistical analyses (per protocol analysis). Normality of distribution was assessed by Kolmogorov–Smirnov test. Changes in clinical and laboratory outcomes between the two dietary intervals were assessed using two-tailed, paired t-tests in case of normally distributed variables, and Wilcoxon test for not normally distributed variables. A two-sided p-value of less than 0.05 was defined as statistically significant. Of note, due to the explorative character of this pilot study, no correction for multiple testing was performed. Statistical analyses were conducted using SPSS 23.0 software (IBM Corp., USA); figures were generated using GraphPad Prism 8.0 for Windows (GraphPad Software, USA).
Results
Patient characteristics
At the baseline visit, patients were randomly assigned to one of the two study arms in a crossover study design (Figure 1). In total, 16 patients completed the 3- and 6-month visits [15 females, median 42.0 (interquartile range, IQR 35.5–51.5) years, details in Table 1].
Baseline characteristics of study cohort.

ARR, annualized relapse rate; DMT, disease-modifying treatment; EDSS, Expanded Disability Status Scale; IQR, interquartile range.
Effects of wheat-reduced diet on cell populations of the adaptive immune system
The primary study endpoint defined as a decrease in pro-inflammatory T cell populations in the peripheral blood on W/G/A− diet was negative. We observed no significant changes in the frequency of pro-inflammatory Th1 [Figure 2(a)] and Th17 cells [Figure 2(b)] on the wheat-reduced diet. However, the CD8+ TEMRA (terminally differentiated effector memory T cells re-expressing CD45RA) subset (gated by CD3+ CD8+ CD45RO− CD27−) displayed significant alterations during the wheat-reduced diet, as the frequency of granulocyte-macrophage colony-stimulating factor (GM-CSF)-producing [Figure 2(c)] and interleukin (IL)-17A-producing [Figure 2(d)] TEMRA cells was lower on the W/G/A− diet. However, the overall proportion of these cell populations was low and the data of six patients had to be excluded from the analysis since cell counts were too low for cytokine measurement (Supplementary Figure 2).

Effects of dietary ATI reduction on peripheral blood cell subsets of the adaptive immune system in patients with RRMS. There was no significant change in (a) Th1 (CCR4−CCR6−CXCR3+) cells or (b) Th17 cells during the ATI-reduced diet interval. The proportion of (c) GM-CSF-producing and (d) IL-17-producing TEMRA cells was lower during the ATI interval. (e) Proportions of transitional B cells were decreased during the ATI-reduced diet interval, whereas (f) the proportion of class switch memory B cells (CD20+ CD19+ CD27+ IgD− IgM−) was increased.
Within the B cell compartment, there was a decrease in transitional B cells (gated by CD19+ CD27+ IgD+ IgM+ CD38+ CD21+ CD24+) [Figure 2(e)] and an increase in class switch memory B cells (CD20+ CD19+ CD27+ IgD− IgM−) [Figure 2(f)] during the wheat-reduced diet.
Effects of wheat-reduced diet on cell populations of the innate immune system
While there was no difference in the overall CD14+ monocyte frequency [percentage of lineage-negative (linX−) HLA-DR+ cells] between the two diet intervals, we observed relevant alterations of their composition. There was a reduction of circulating CD14+ CD16++ non-classical monocytes during the wheat-reduced compared to the wheat-containing diet [Figure 3(a)]. Correspondingly, the frequency of classical CD14++ CD16− monocytes increased [Figure 3(b)], whereas intermediate CD14++ CD16+ monocytes remained unchanged [Figure 3(c)].
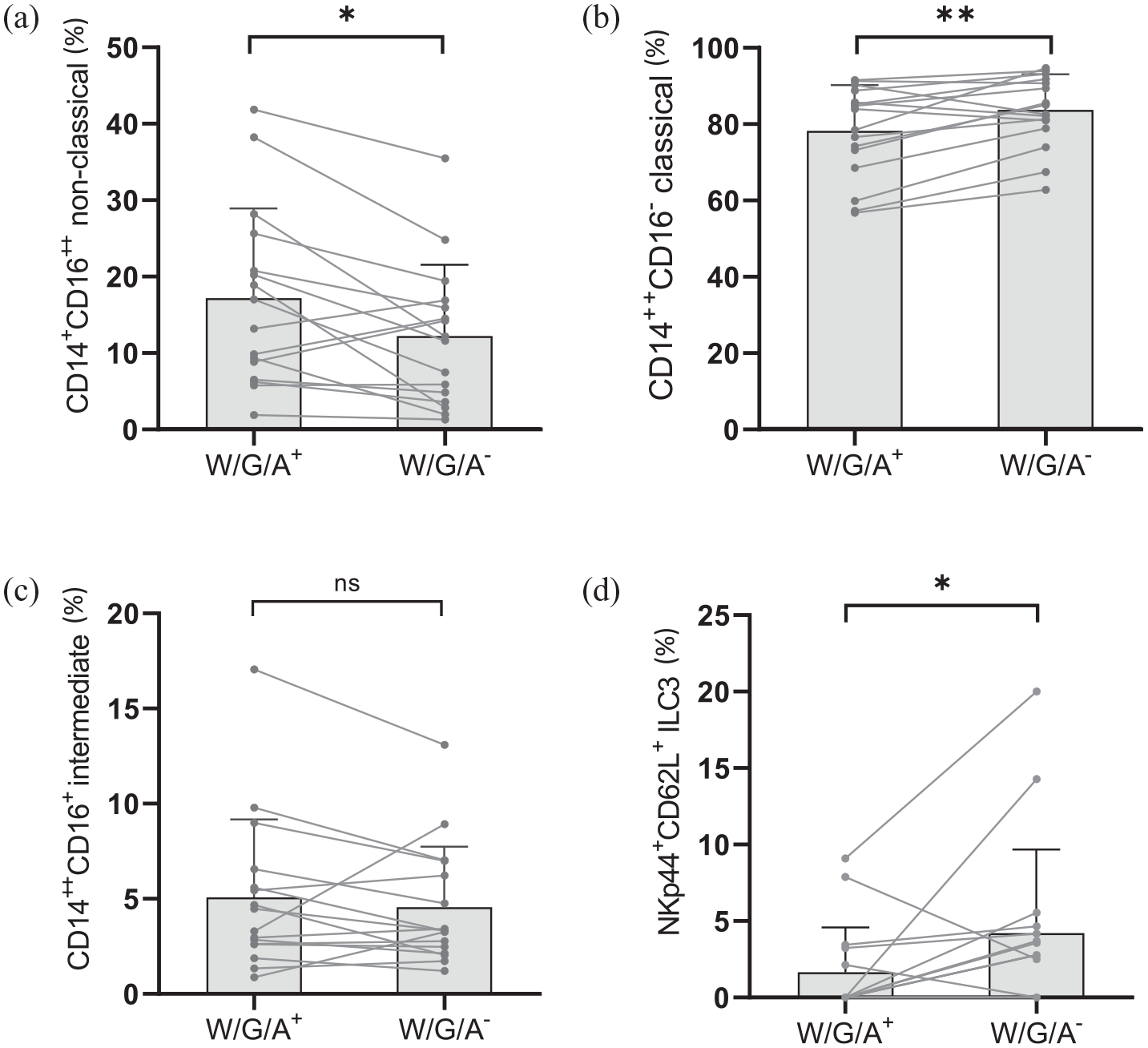
Dietary ATI reduction impacts peripheral blood cell subsets of the innate immune system in patients with RRMS. A reduction of dietary wheat/gluten and ATIs (W/G/A−) led to (a) a decreased proportion of CD14+ CD16++ non-classical monocytes (% of CD14+ monocytes), whereas (b) the proportion of CD14++ CD16− monocytes increased. (c) No change in the proportion of intermediate CD14++ CD16+ monocytes was observed. (d) The proportion of circulating type 3 innate lymphoid cells (ILCs) expressing NKp44 and CD62L (percentage of ILC3) was increased by ATI reduction.
Furthermore, there was an increase in type 3 innate lymphoid cells (ILCs) bearing the natural cytotoxicity receptor (NCR+ ILC3, gated by CD45+ linX− NCR+ CD117+) expressing both NKp44 and CD62L [Figure 3(d)]. There were no significant changes within the dendritic cell compartment.
Patient adherence
In the dietary questionnaires, all patients declared a reduced consumption of wheat and other gluten-containing foods (as proxy for ATI consumption since both are highly correlated) by about 88% (from an estimated 6.6 ± 6.3 to an estimated 0.8 ± 2.4 g of gluten daily). However, 3 patients went on a strict GFD and 10 consumed gluten-containing foods less than once a week. Another 3 patients reported to be consuming gluten-containing foods more than once a week. Urinary GIP levels of 10 patients were reduced during the W/G/A− interval compared with the W/G/A+ interval, while there was no difference in GIP levels between the W/G/A− diet and W/G/A+ diet in 4 patients. Two patients had GIP levels below the detection limit at both time points. Overall, the median GIP levels were lower during the W/G/A− diet compared with the W/G/A+ diet (Supplementary Figure 3).
Wheat-reduced diet is tolerated well and leads to an improvement in pain-related quality of life in RRMS patients
The EDSS scores during the wheat-reduced diet interval did not differ from those during the wheat-containing diet interval. One patient suffered from an MS relapse with new gait ataxia during the W/G/A+ diet interval, while no MS relapses were reported during the W/G/A− diet interval. Levels of sNfL remained stable throughout the study independent of diet assignments. In total, the occurrence of 11 adverse events was reported (Table 2).
Comparison of outcome measures between wheat-containing (W/G/A+) and wheat-reduced (W/G/A−) diet.

BMI, body mass index; EDSS, Expanded Disability Status Scale; SD, standard deviation; SF-36, 36-item short form survey; sNfL, serum neurofilament light chain; W/G/A, wheat/gluten/ATI.
Comparison of mean clinical outcome measures during the W/G/A+ and the W/G/A− diet interval. During the W/G/A− diet interval, patients scored higher in the category ‘Pain’ of the SF-36 questionnaire, indicating a higher quality of life with regard to pain. If not stated otherwise, results within this table represent mean ± SD. Statistically significant differences are marked in bold.
In the SF-36, we observed a significant improvement in the category ‘pain’ during the wheat-reduced diet (72.3/100 ± 30.4 versus 79.5/100 ± 25.6, p = 0.008). The self-reported perception of the the other categories did not differ significantly between the wheat-reduced and the wheat-containing diets.
A comparison of clinical outcome measures between wheat-reduced and wheat-containing diet is reported in Table 2.
Discussion
In this proof-of-concept study, we were able to translate some of our preclinical findings on the immunomodulatory effects of reducing dietary wheat consumption, and thus nutritional ATIs, to a potential clinical application in RRMS patients. While the study’s primary endpoint (decrease in circulating pro-inflammatory T cell populations) was negative, we observed a reduction in non-classical monocytes, an ILC subset (ILC3), and two small TEMRA subpopulations in the peripheral blood of RRMS patients during the > 90% wheat- and thus ATI-reduced diet interval. Furthermore, the wheat-reduced diet was tolerated well and led to an improvement in pain-related quality of life.
Rodent studies have shown that oral ingestion of wheat or purified ATIs as active ingredient leads to the activation of pro-inflammatory monocytes, macrophages, and dendritic cells (DCs) in the intestine via activated TLR4-signaling, resulting in elevated numbers of (activated) myeloid cells in the intestinal lamina propria and mesenteric lymph nodes as compared to animals on a wheat- and ATI-free control diet6,8–12,14 Moreover, this activation exacerbated inflammatory diseases of the intestine, the liver, and the lungs in mice, including mice with a humanized immune system10–12,14 This supported the hypothesis that after local activation of intestinal myeloid cells by nutritional ATIs, these myeloid cells may migrate to the mesenteric lymph nodes, where they may encounter already primed autoreactive T cells to aggravate tissue-specific inflammation at distant sites. 8 In our preliminary data on the role of ATI in EAE, this translated into a more severe disease course. 16
Depending on their expression of CD14 and CD16, myeloid cells can be subdivided into classical CD14++ CD16−, non-classical CD14+ CD16++, and intermediate CD14++ CD16+ monocytes. 24 Functionally, the roles of these monocyte subsets are still a matter of controversial debate. Some have described the classical monocytes as mainly phagocytic with few inflammatory attributes, 25 while others have suggested that they are critical for initial inflammatory response.26,27 Non-classical monocytes display antigen-presenting properties and are usually described to express an inflammatory phenotype, 25 although they can also be viewed as anti-inflammatory as they were found to maintain vascular homeostasis.28,29 Intermediate monocytes show both moderate phagocytic and inflammatory features. 25 In the current study, the reduction of dietary ATIs was associated with decreasing frequencies of circulating non-classical monocytes and a concomitant increase in classical monocytes. This is of interest as in comparison to healthy controls, MS patients show an elevation of non-classical and a reduction of classical monocytes in peripheral blood. 24 Furthermore, a recent study identified higher numbers of classical monocytes as a predictor of therapeutic efficacy in dimethyl fumarate-treated MS patients, 30 which might argue for a favorable effect of avoiding wheat/nutritional ATIs in MS. Notably, classical monocytes display a lower expression of toll-like receptors than intermediate and non-classical monocytes. 25 Since ATIs stimulate myeloid cells via TLR-4 signaling, this provides a mechanistic link to our observation of a shift from non-classical to classical monocytes during the diet low in TLR4-activating ATI.
Apart from monocytes and DCs, the innate immune system also comprises ILCs, which function as potent immune effector cells during inflammation. ILC3 can promote an immunologically tolerogenic state in the intestine in response to nutrients, commensal bacteria, or bacterial metabolites that limits the magnitude of potentially damaging T cell responses. 31 In the current study, the reduction of dietary ATIs led to an increase in activated (NKp44+) ILC3, which are prominent in the intestine and which are able to invade lymphoid tissues via CD62L+. While the exact consequence of this finding is unclear, it might indicate that the reduction of the modest intestinal immune cell activation during the wheat (ATI)-reduced diet, which has been observed in murine models, might be partly mediated by a recruitment of ILC3 to the mesenteric lymphoid tissue. Studies on the contribution of ILCs in the pathogenesis of MS remain inconclusive so far.23,32,33
Based on the aforementioned rodent studies on the effects of wheat or ATI consumption,6,8–12,14 we hypothesized that reducing dietary ATIs might diminish pro-inflammatory T cell responses in the periphery. Although we did not observe a general reduction of peripheral inflammatory T cell populations, there was a decrease in two small T cell subpopulations, namely GM-CSF- and IL-17A-producing CD8+ TEMRA cells. These cells, which represent the most differentiated type of memory cells, express high levels of cytotoxic effector molecules, such as perforin and Fas ligand. 34 An increased occurrence of clonally expanded CD8+ TEMRA cells in the blood of MS patients has been reported, 35 and the decreased proportion of subsets producing the pro-inflammatory cytokines GM-CSF and IL-17A on the ATI-reduced diet may reflect a downregulation of detrimental adaptive immune responses. However, the absolute number of TEMRA cells was low and therefore our findings should be interpreted with caution.
Clinically, we did not detect any evidence of increased MS disease activity, with stable EDSS scores and unchanged sNfL levels as a marker of neuroaxonal damage. There were no reported relapses during the wheat-reduced diet interval, whereas one patient suffered from a relapse during the wheat-containing diet (difference not statistically significant). However, in the patient-reported quality of life assessment, the wheat-reduced diet was associated with a reduction of pain. A plausible pathomechanistic explanation might be a reduction of ATI-mediated activation of intestinal myeloid cell TLR4 and downstream adaptive effector functions. This is in line with growing evidence suggesting that TLR4-signaling plays an important role in the induction, conversion, and maintenance of chronic systemic and neuropathic pain in MS. 36 Therefore, our current finding argues for a complementary role of reducing dietary ATIs in the symptomatic treatment of chronic pain and possibly also long-term sequelae in MS patients.
In an effort to verify dietary compliance, we used a validated test to quantify urinary gluten peptides (GIP). These peptides were indeed decreased in the majority of patients on the wheat-reduced versus the wheat-containing diet, but still detectable in the majority of patients on the wheat-reduced diet. Recent studies showed that urinary GIP have a high sensitivity – but a low specificity – to detect minor gluten ingestion in patients with celiac disease who need to comply with a strict gluten-free diet.37,38 In contrast, participants of our study were allowed to consume some gluten as long as the total amount was reduced by at least 90%. This explains why we were able to detect GIP in the majority of patients even during the wheat-reduced diet and underlines that urinary GIP cannot be considered a reliable biomarker to monitor dietary compliance in studies where minor amounts of wheat and gluten are allowed.
It should be noted that due to the explorative character of this pilot study, we did not employ corrections for multiple testing. Furthermore, the observed changes in immune cell populations are partly small or driven by outliers. Thus, statistically significant differences in these immune cell populations do not necessarily imply physiological relevance. Therefore, a larger-scale clinical trial is necessary to validate the currently observed shifts within the PBMC compartment and to evaluate the impact of a reduction of wheat consumption, and thus dietary ATIs on the clinical disease course of MS. This trial should also enroll patients with more severe MS and follow-up MRT to further classify CNS disease activity.
To conclude, this pilot study suggests that reducing dietary wheat/ATIs may be helpful for some RRMS patients as a complementary treatment. Along with good tolerability, following a wheat/ATI-reduced diet improved pain-related quality of life and exerted a potentially immunomodulating effect, which was most pronounced in the myeloid cell compartment.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231170928 – Supplemental material for Attenuation of immune activation in patients with multiple sclerosis on a wheat-reduced diet: a pilot crossover trial
Supplemental material, sj-docx-1-tan-10.1177_17562864231170928 for Attenuation of immune activation in patients with multiple sclerosis on a wheat-reduced diet: a pilot crossover trial by Sinah Engel, Luisa Klotz, Timo Wirth, Ann-Katrin Fleck, Geethanjali Pickert, Melanie Eschborn, Samia Kreuzburg, Valentina Curella, Stefan Bittner, Frauke Zipp, Detlef Schuppan and Felix Luessi in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pptx-2-tan-10.1177_17562864231170928 – Supplemental material for Attenuation of immune activation in patients with multiple sclerosis on a wheat-reduced diet: a pilot crossover trial
Supplemental material, sj-pptx-2-tan-10.1177_17562864231170928 for Attenuation of immune activation in patients with multiple sclerosis on a wheat-reduced diet: a pilot crossover trial by Sinah Engel, Luisa Klotz, Timo Wirth, Ann-Katrin Fleck, Geethanjali Pickert, Melanie Eschborn, Samia Kreuzburg, Valentina Curella, Stefan Bittner, Frauke Zipp, Detlef Schuppan and Felix Luessi in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
