Abstract
Background:
In multiple sclerosis (MS), the frequency of hypogammaglobulinemia is unknown. We aimed to evaluate the frequency of reduced immunoglobulin (Ig) concentrations and its association with immunotherapy and disease course in two independent MS cohorts.
Methods:
In our retrospective cross-sectional study, MS patients and control patients with head or neck pain from Bern University Hospital (Bern, Switzerland) and Eginition University Hospital (Athens, Greece) were included. The lower limits of normal (LLN) for serum Ig concentration were IgG < 700 mg/dl, IgM < 40 mg/dl, and IgA < 70 mg/dl. Mann–Whitney U test, analysis of variance test, and multiple linear regression analysis were employed.
Results:
In total, 327 MS patients were retrospectively identified (Bern/Athens: n = 226/101). Serum IgG concentrations were frequently under LLN in both MS cohorts (Bern/Athens: 15.5%/14.9%), even when considering only untreated patients (Bern/Athens: 7.9%/8.6%). MS patients (n = 327) were significantly more likely to have IgG concentrations below LLN and below 600 mg/dl in comparison with controls (n = 58) (p = 0.015 and 0.047, respectively). Between both patient groups, no significant differences were found in frequencies of IgA and IgM concentrations under LLN [n (MS patients/controls): IgA 203/30, IgM 224/24]. Independently of age, secondary progressive MS patients had lower IgG concentrations than relapsing–remitting and primary progressive patients (both: p ⩽ 0.01). After adjusting for sex, age, and disease course, IgG concentrations were lower in patients treated with rituximab (p = 0.001; n = 42/327), intravenous corticosteroids (p < 0.001; n = 16/327), natalizumab (p < 0.001; n = 48/327), and fingolimod (p = 0.003; n = 6/327).
Conclusion:
Our study demonstrated high prevalence rates of reduced serum IgG concentrations in MS patients with and without disease-modifying treatments. The significance of lower IgG concentrations at the levels noted is unclear considering that infections or interference with antibody production generally occur when IgG levels are much lower, at or below 400 mg/dl. However, the information is useful to monitor IgG levels especially with anti-B-cell therapies and consider IgG substitution when levels drop below 400 mg/dl.
Introduction
In the general population, primary immunodeficiency is a rare disease. The most prevalent forms are immunoglobulin (Ig) M deficiency and IgA deficiency (0.37% and 0.2%, respectively).1,2 Regarding patient populations with autoimmune diseases, Ig deficiency appears to be more common because 1.7% of patients with celiac disease and 5.2% of patients with systemic lupus erythematosus had IgA deficiency.3,4 In patients with multiple sclerosis (MS), however, the prevalence of hypoimmunoglobulinemia is unknown.
In autoimmune disorders such as celiac disease, Grave’s disease, and rheumatoid arthritis, Igs are regarded as pathogenic contributors.5–7 Similarly, central nervous system lesion pathology provides evidence for the role of antibodies in MS because lesions pattern II is dominated by complement activation presumably being caused by autoantibodies. 8 Furthermore, the importance of Igs in the diagnostic workup of MS is reflected by the recent introduction of intrathecal cerebrospinal fluid specific oligoclonal bands into the 2017 McDonald criteria to support dissemination in time.9–11
In the treatment of MS, therapies targeting CD20 such as ocrelizumab and rituximab are effective immunotherapeutic options.12–15 Decreased serum IgG concentrations caused by these therapies may pose an increased risk of infection, which was found in patients with hematologic malignancies or autoimmune diseases, who developed hypogammaglobulinemia while being treated with rituximab. 16 Furthermore, reduced serum IgG concentrations may interfere with standard serological testing as the John Cunningham virus (JCV) antibody index decreased alongside IgG concentrations in patients treated with rituximab. 17
Owing to the potential clinical implications of reduced serum Ig concentrations in MS, our study aimed to evaluate its frequency in two independent European MS cohorts from the University Hospitals in Bern (Switzerland) and Athens (Greece) and to study the association between reduced IgG concentrations, immunotherapy, and MS disease course.
Methods
Study design, participants, and procedure
In our retrospective cross-sectional study, two independent European MS cohorts from the following institutions were analyzed: Bern University Hospital (Bern, Switzerland) and Eginition University Hospital (Athens, Greece). MS patients with available serum Ig concentrations measured between January 2015 and April 2018 were included in this study. At Bern University Hospital, the hospital-wide computer program identified electronically all MS patients with available Ig concentrations regardless of immunotherapy. At Eginition University Hospital, all MS patients with natalizumab treatment or without disease-modifying therapy were identified using a manual registry and the patient records were manually checked for available Ig concentrations. Furthermore, we used a control cohort consisting of patients with head or neck pain and available Ig concentrations sampled between January 2016 and April 2019. At both institutions, medical records were evaluated retrospectively. Serum Ig concentrations were analyzed in the respective main laboratories using the c502 module of Cobas 8000 (Roche; Basel, Switzerland) in Bern and Behring Nephelometer II (Behring; Marburg, Germany) in Athens. In the case of multiple Ig measurements, the most recent serum Ig sample was used for the analysis. With reference to treatment status, patients were indexed as having received corticosteroids if administered intravenously ⩽4 weeks before blood sampling. All data were acquired during routine clinical practice. The respective ethics committees approved the analyses conducted in Bern (registration no. KEK-BE 2017-01369) and Athens (registration no. 511; 12.07.2018). The following cut-offs were used to define the lower limit of normal (LLN) of Ig concentrations: IgG < 700 mg/dl, IgM < 40 mg/dl, and IgA < 70 mg/dl. 18
Statistical analysis
Data of both MS cohorts were pooled for the analysis of the effects of immunotherapy and MS disease course on serum IgG concentrations and for the comparison between MS and control patients. In all other analyses, both MS cohorts were investigated separately. Continuous variables are expressed as median and 25th to 75th percentile. If continuous variables are classified into different groups (e.g. IgG concentrations under the LLN), they are expressed as frequencies. The Mann–Whitney U test (MWU) was employed to identify differences between two independent groups. If more than two groups were compared, the analysis of variance (ANOVA) with Dunn’s multiple comparison test was used. To investigate the effect of immunotherapy on serum IgG concentration, linear regression analyses were performed with IgG concentration as the dependent variable and immunotherapy as the independent variable. These analyses were adjusted for sex, age at serum Ig sampling, and MS disease course. As each regression analysis was performed for seven different treatment conditions, p values were adjusted for multiple comparisons following Bonferroni’s procedure. Therefore, a p value of <0.007 was defined to be significant. In all other analyses, a p value of <0.05 was defined to demonstrate a significant finding. Spearman’s Rho correlation was employed to investigate the correlation between treatment duration and IgG concentration.
Results
In total, 327 MS patients were included in this study (Bern: n = 226, Athens: n = 101). Baseline characteristics, serum IgG concentrations, and frequency of IgG concentrations under the LLN are presented in Table 1. From 327 MS patients, one had a common variable immunodeficiency (CVID) as comorbidity. This was a female relapsing–remitting MS patient who was 61 years of age at first Ig sampling in Bern with a serum concentration of IgG 384 mg/dl, IgM 26 mg/dl, and IgA 92 mg/dl.
Patient characteristics of the MS cohorts from Bern and Athens.

Injectables include interferons and glatiramer acetate.
Patients were indexed as having received corticosteroids if administered intravenously ⩽4 weeks before blood sampling.
25th–75th, 25th–75th percentile; EDSS, Expanded Disability Status Scale; Ig, immunoglobulin; Mdn, median; MS, multiple sclerosis; PPMS, primary progressive multiple sclerosis; RRMS, relapsing–remitting multiple sclerosis; SPMS, secondary progressive multiple sclerosis.
A trend toward lower IgG concentrations was observed in the MS group compared with the control group, which consisted of patients with head or neck pain [median (25th–75th, n): MS 931 mg/dl (782–1100, n = 327)] versus control group 997 mg/dl (823–1175, n = 58), MWU: p value = 0.055). Furthermore, in the control cohort, 2/58 patients (3.5%) had serum IgG concentrations < 700 mg/dl and none (0%) < 600 mg/dl compared with 50/327 (15.3%) and 21/327 (6.4%), respectively, in the MS cohort. Serum IgG concentrations below the LLN (< 700 mg/dl) and below 600 mg/dl were significantly more frequent in MS patients compared with controls [chi-squared (χ2) p value = 0.015 and 0.047, respectively]. IgA and IgM concentrations below the respective LLN were slightly more frequent in the MS cohort (IgA: 6/203 = 3%; IgM: 28/224 = 12.5%) than in the control cohort (IgA: 0/30 = 0%; IgM: 1/24 = 4.2%) in absolute and relative terms. However, these differences were not significant.
In MS patients without disease-modifying treatment, IgG concentrations were frequently under the LLN [Bern: 7.9% (11/140), Athens: 8.6% (5/58)] (Table 1) and they were 2.3 times more likely to have IgG concentrations < 700 mg/dl compared with the control patients. Nonetheless, the χ2 statistic comparing IgG concentrations under the LLN in untreated MS patients and control patients was not significant.
We demonstrated that secondary progressive (SP) MS patients [median (25th–75th): 750 mg/dl (690–850), n = 28] had significantly lower IgG levels than RRMS [median (25th–75th): 950 mg/dl (800–1110), n = 276) (p < 0.001] and primary progressive (PP) MS patients [median (25th–75th): 940 mg/dl (830–1120), n = 23) (p ⩽ 0.01) (Figure 1(a)).
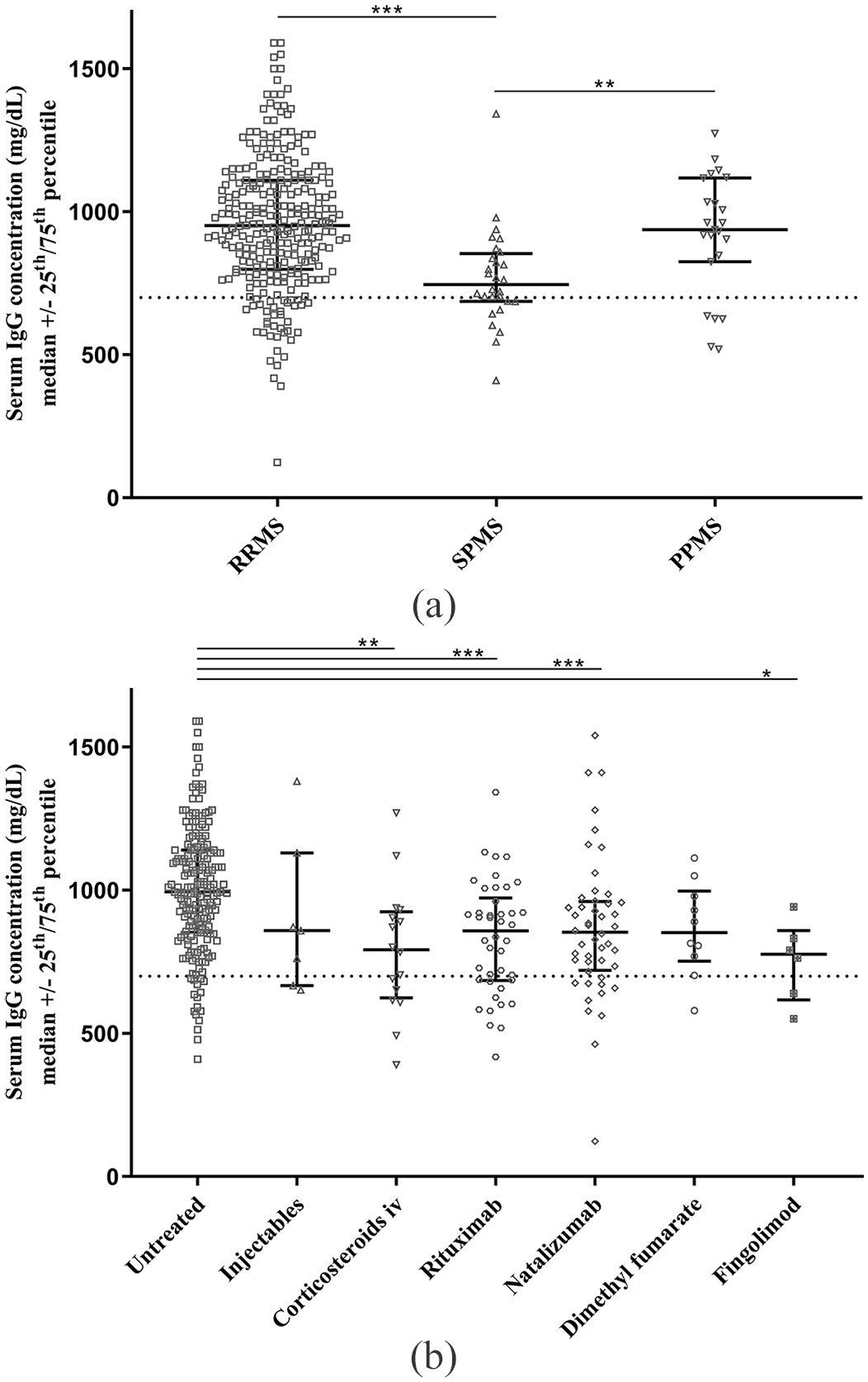
Serum immunoglobulin G concentrations in MS patients stratified by disease course (a) and treatment (b).
Employing the univariate ANOVA, we found that patients treated with rituximab, intravenous corticosteroids, natalizumab, and fingolimod had significantly lower serum IgG concentrations than untreated patients (Figure 1(b)). The multiple linear regression analyses confirmed the significant effects of rituximab [coefficient: −1.38, 95% confidence interval (CI) −2.18 to −0.57, p = 0.001, n = 42/327], intravenous corticosteroids (coefficient: −2.09, 95% CI: −3.22 to −0.96, p < 0.001, n = 16/327), natalizumab (coefficient: −1.57, 95% CI: −2.28 to −0.86, p < 0.001, n = 48/327), and fingolimod (coefficient: −2.68, 95% CI: −4.41 to −0.95, p = 0.003, n = 6/327) on serum IgG concentrations. However, none of the immunotherapies showed significant correlations between treatment duration and IgG concentrations (supplementary table).
Discussion
Our retrospective analysis of two independent European cohorts demonstrated that a substantial proportion of MS patients had serum IgG concentrations below the LLN, which was influenced by MS disease course and certain immunotherapies (rituximab, intravenous corticosteroids, natalizumab, and fingolimod). The expected normal distribution of serum IgG concentrations would result in 2.5% of the general population being below the LLN (700 mg/dl). This frequency was substantially exceeded by our treated as well as untreated MS patients [treated: 26.4% (34/129), untreated: 8.1% (16/198)]. Overall, the frequency of IgG concentrations under the LLN was significantly higher in the MS cohort than in the small control group of patients with head or neck pain. Nonetheless, when comparing only untreated MS patients with the control patients, the χ statistic showed no significant differences in the frequency of reduced serum IgG concentrations, even though IgG concentrations under the LLN were more frequent in the untreated MS group than in the control group. Furthermore, no significant differences between our MS cohort and the control cohort were found in IgA and IgM concentrations under the LLN. Missed significances may be explained by lower sample sizes.
Our findings are supported by a recent study, which demonstrated lower serum IgG concentrations in drug-naïve patients recently diagnosed with the clinically isolated syndrome (n = 20) compared with healthy controls (n = 10). 19 In that study, however, no significant differences in IgG concentrations were found between MS patients and healthy controls, which could be explained by the relatively small number of MS patients included (n = 8). 19
In our study, SPMS patients had lower IgG concentrations than RRMS and PPMS patients, which could be related to the older age of SPMS compared with RRMS patients (p < 0.001). However, since this age difference was not significant when comparing SPMS with PPMS patients and since we found only a weak correlation coefficient between age and serum IgG concentration in MS patients (Pearson correlation coefficient: r = –0.11, p = 0.046, n = 327), the influence of age does not appear to sufficiently explain the difference in IgG concentrations between disease courses. In addition to age, the difference in immunotherapies may also be an influencing factor. Nevertheless, when considering only RRMS and SPMS patients without disease-modifying treatment (n = 179 and n = 8, respectively), the significant difference in IgG concentrations remained present despite the small sample size in the SPMS group (MWU: p ⩽ 0.001).
Furthermore, we demonstrated that rituximab treatment was significantly associated with lower serum IgG concentrations in comparison with untreated MS patients, which is in line with a phase II, double-blind trial involving RRMS patients, in which rituximab treatment was associated with a higher rate of reduced Ig concentrations (7.9%) than placebo (3%). 15 In addition to predisposing to infections, another clinical implication of low serum IgG concentrations is false-negative antibody testing during rituximab therapy, which was reported recently. 17 During rituximab therapy, not only IgG concentrations but also the JCV antibody index decreased. 17 Since reduced serum IgG concentrations may lead to false-negative JCV antibody index test results in patients treated with anti-CD20 therapies, this assay must be interpreted cautiously when assessing PML risk, for example, when planning to switch from anti-CD20 therapies to natalizumab.
Furthermore, we demonstrated reduced serum IgG concentrations in MS patients treated with natalizumab in comparison with untreated MS patients, which is consistent with a previous study. 20 A pathophysiological explanation may be that natalizumab impairs B-cell differentiation into plasma cells, which consequently decreases Ig synthesis. 21
Finally, our finding that intravenous corticosteroids reduce serum IgG concentration is in line with previous studies.22,23 In healthy volunteers exposed to 96 mg of methylprednisolone for 3–5 days, a 22% reduction of serum IgG concentration was observed within 2–4 weeks after drug administration. 23
In our MS cohort, 1/327 (0.31%) patients had CVID as comorbidity. The frequency of CVID in our MS patient cohort exceeds the CVID prevalence in the general population by 31- to 310-fold, which is estimated between 1:10,000 (0.01%) and 1:100,000 (0.001%). 24 However, since the patient number is low and, therefore, chance could be an important confounding factor, this finding needs re-evaluation in larger MS cohorts.
The strength of our study is the use of two independent MS cohorts with corroborating results. Furthermore, a high number of patients without disease-modifying treatments were investigated permitting the hypothesis that reduced IgG concentrations could be genuinely associated with MS. A limitation of our study, however, is the heterogeneous distribution of immunotherapies between both centers, which can cause a center related inclusion bias. Missing data on antibody testing and the incidence of infections is another weakness of our study, which limits the clinical significance of our findings and should be addressed in future studies. Furthermore, only 16 patients treated with oral disease-modifying drugs were included. This small patient number prohibits drawing final conclusions in this patient population.
Altogether, our study demonstrated high prevalence rates of reduced serum IgG concentrations in MS patients with and without disease-modifying treatments. Interpretation of antibody levels in MS patients has not been studied extensively and is, therefore, mostly a case based decision. Nonetheless, some recommendations from other conditions and diseases may be useful. Agarwal and Cunningham-Rundles argue that adequate amounts of antibodies for the successful immune response to pathogens are still available if antibody responses of patients with mild to moderate reductions in IgG concentrations (300–600 mg/dl) are normal. 25 In MS patients, the significance of lower IgG concentrations at the levels demonstrated in our study is unclear considering that infections or interference with antibody production generally occur when IgG levels are much lower, at or below 400 mg/dl. However, the information is useful to monitor IgG levels especially with anti-B-cell therapies and consider IgG substitution when the levels drop below 400 mg/dl.
Supplemental Material
Correlation_coefficient – Supplemental material for Reduced serum immunoglobulin G concentrations in multiple sclerosis: prevalence and association with disease-modifying therapy and disease course
Supplemental material, Correlation_coefficient for Reduced serum immunoglobulin G concentrations in multiple sclerosis: prevalence and association with disease-modifying therapy and disease course by Greta Zoehner, Andrei Miclea, Anke Salmen, Nicole Kamber, Lara Diem, Christoph Friedli, Maud Bagnoud, Farhad Ahmadi, Myriam Briner, Nazanin Sédille-Mostafaie, Constantinos Kilidireas, Leonidas Stefanis, Andrew Chan, Robert Hoepner and Maria Eleftheria Evangelopoulos in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Greta Zoehner and Andrei Miclea contributed equally to this work. Robert Hoepner and Maria Eleftheria Evangelopoulos contributed equally to this work.
Author contributions
G Zoehner collected the data and contributed to the design of the study, the analysis and the interpretation of the data, and the writing and revision of the manuscript.
A Miclea contributed to the design of the study, the analysis, and interpretation of the data, and the writing and revision of the manuscript.
A Chan, L Stefanis, C Friedli, N Kamber, C Kilidireas, M Briner, M Bagnoud, F Ahmadi, L Diem, N Sédille-Mostafaie, and A Salmen contributed to the interpretation of the data, and the writing and revision of the manuscript.
R Hoepner contributed to the design of the study, the analysis, and interpretation of the data, and the writing and revision of the manuscript.
ME Evangelopoulos collected the data and contributed to the analysis, the interpretation of the data, and the writing and revision of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
G Zoehner, A Miclea, N Kamber, C Friedli, F Ahmadi, M Bagnoud, and N Sédille-Mostafaie report no disclosures.
A Salmen received speaker honoraria and/or travel compensation for activities with Almirall Hermal GmbH, Biogen, Merck, Novartis, Roche, and Sanofi Genzyme, none related to this work.
L Diem has received travel grants from Bayer, Biogen, Roche, and Merck, as well as speaker honoraria from Merck.
M Briner received travel grants from Merck and Biogen.
C Kilidireas has received travel grants and consulting fees from Biogen, Sanofi, Novartis, Roche, Teva, and Serono.
L Stefanis has received the following grants: MULTISYN European Program (EU, FP7-HEALTH.2013.1.2-1, number 602646), PPMI (supported by the Michael J. Fox Foundation), IMPRIND-IMI2 Number 116060 (EU, H2020), SANTE 2017 and 2019 Research Grants in Biomedical Sciences, NO-MND (EU-FP7-PEOPLE-2013-IRSES), NPF 2015 Investigator Award (Collaborator), ‘PBMC and urine collection in LRRK2 and idiopathic PD’ Grant by the Michael J. Fox Foundation (Collaborator). He has served on an Advisory Board for Abbvie. He is conducting clinical trials supported by Abbvie and Sanofi.
A Chan has received personal compensation for activities with Bayer, Biogen, Genzyme, Merck, Novartis, Roche, and Teva. He received research support from the Swiss National Fonds No. 310030_172952), Genzyme and UCB. He serves on the editorial board for Clinical and Translational Neuroscience and the Journal of International Medical Research.
R Hoepner received research and travel grants from Novartis and Biogen Idec. He also received speaker honoraria from Biogen, Novartis, Merck, and Almirall. He is supported by the Swiss MS Society.
ME Evangelopoulos has received travel grants and consulting fees from Biogen, Novartis, Teva, Genzyme, Roche, and Merck.
Data sharing statement
The authors are willing to share the data of this study for collaborations. Please contact the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
