Abstract
Background:
The effect of gene variants and their interactions on response to aspirin and clinical adverse outcomes after an acute ischemic stroke (IS) is not fully understood. The aim of this study was to investigate the association of aspirin-relevant gene variants and their interactions with clinical adverse outcomes in IS patients taking aspirin.
Methods:
A total of 14 variants from six genes encoding COX enzymes (COX-1, COX-2), platelet membrane receptors (TXAS1, P2Y1, P2Y12) and glycoprotein receptor (GPIIIa) were examined in 850 acute IS patients. Gene–gene interactions were analyzed using generalized multifactor dimensionality reduction (GMDR) analysis. All patients were followed up for 1 year after admission. Primary outcome was a composite of recurrent ischemic stroke (RIS), myocardial infarction (MI) and death.
Results:
The primary outcome occurred in 112 (13.5%) patients (81 RIS, 16 MI and 15 deaths). There were no significant differences in the frequencies of the genotypes of the 14 variants between the patients with and without primary outcome using single-locus analytical approach. However, there was significant gene–gene interaction among rs20417, rs1371097 and rs2317676. The high-risk interactive genotypes of rs20417, rs1371097 and rs2317676 were independently associated with primary adverse outcome of RIS, MI, and death after acute IS.
Conclusion:
The three-loci interactions are associated with sensitivity of IS patients to aspirin and aspirin-induced adverse clinical events. The combinatorial analysis used in this study may be helpful to elucidate complex genetic risk of aspirin resistance (AR).
Clinical trial registration:
The study described here is registered at http://www.chictr.org/ (unique identifier: ChiCTR-OCH-14004724).
Introduction
Stroke is a significant worldwide health problem and is one of the leading causes of death among the elderly [Wang et al. 2007]. Patients with ischemic stroke (IS) are at a high risk of developing a recurrent ischemic stroke (RIS) [Zhao et al. 2008]. Aspirin is highly recommended for these patients, in order to reduce their risk of clinically important atherothrombotic events [Kernan et al. 2014], and has shown beneficial effects on adverse clinical events [International Stroke Trial Collaborative Group, 1997; Chen et al. 2000]. However, a proportion of IS patients, despite taking aspirin, did experience RIS or other vascular events in the subsequent years after their first stroke [Eikelboom and Hankey, 2003; Yi et al. 2013a], and this was referred to as aspirin resistance (AR), that is, poor or no response to aspirin treatment [Floyd and Ferro, 2015]. Thus, further research on this individual variability could help us develop more effective drugs to control or prevent IS in clinic.
The mechanisms leading to a poor response to aspirin have not yet been fully elucidated and are most likely multifactorial [Schwartz et al. 2005; Cotter et al. 2004; Frelinger et al. 2006], such as lack of compliance, reduced absorption, possibly with increased metabolism of aspirin, and the biosynthesis of thromboxane A2 (TXA2) via pathways not inhibited by aspirin as well as alternative pathways involved in platelet activation not blocked by aspirin [e.g. those involving adenosine 5’-diphosphate (ADP), collagen, epinephrine, and thrombin]. The mechanism of action of aspirin is the inhibition of cyclooxygenase enzymes (COX), leading to the prevention of TXA2 generation from arachidonic acid (AA). TXA2 binds to its glycoprotein-coupled receptor (GPIIb/IIIa) leading to activation of platelet aggregation [Nakahata, 2008]. The platelet membranes receptors (P2Y12, P2Y1) have also played a key role in platelet aggregation and thrombosis [Storey, 2006; Dorsam and Kunapuli, 2004]. There is growing evidence showing that aspirin sensitivity may be influenced by pharmacokinetic or pharmacogenetic variables, such as activation and aggregation of platelets, all of which are further affected by gene single nucleotide polymorphisms (SNPs). For example, some studies showed that COX-1, COX-2, GPIIIa, P2Y1, P2Y12 polymorphisms may lead to AR [Goodman et al. 2007, 2008; Sharma et al. 2013; Halushka et al. 2003; Li et al. 2007; Grinshtein et al. 2013]. However, our previous study and some other studies did not find the association between AR and these candidate gene variants in patients with symptomatic vascular disease [Yi et al. 2013b; Kunicki et al. 2009; Lev et al. 2007]. These conflicting results indicated that the genetic factors for AR might be very complex. It may be that genetic variants at individual loci only contribute to AR by interaction with other gene variants. Thus, the investigation of multiple gene–gene interactions is necessary to enable us to understand the genetic etiology of AR in IS patients using the generalized multifactor dimensionality reduction (GMDR) approach [Lou et al. 2007; Yi et al. 2015b]. However, the potential effects of gene–gene interactions of relevant genes on clinical outcomes after acute IS in patients receiving aspirin are unknown.
To address this issue, we hypothesized that interactions among aspirin-relevant genetic variants may affect aspirin responsiveness and clinical adverse outcomes in IS patients. This hypothesis was tested by using the GMDR approach with 14 variants from six genes to explore these gene–gene interactions, and whether they related to clinical adverse outcomes during a 1-year follow-up period after acute IS.
Materials and methods
Study population
A protocol for this prospective study was reviewed and approved by the Ethics Committees of The People’s Hospital of Deyang City and The Third Affiliated Hospital of Wenzhou Medical College. Each of the participants provided informed consent before participating in this study. The study was registered at http://www.chictr.org/ with the unique identifier of ChiCTR-OCH-14004724.
We consecutively enrolled 850 patients who underwent a first-ever stroke and were admitted to the above two hospitals within 72 hours of their index stroke between August 2010 and August 2014. The inclusion criteria were: (1) age ⩾ 40 years old; (2) diagnosis of IS, based on both clinical findings and results of brain magnetic resonance imaging; (3) stroke in all cases was due to atherothrombotic or small artery disease, according to the Trial of ORG 10172 in the Acute Stroke Treatment (TOAST) classification system [Han et al. 2007]. Exclusion criteria were: (1) allergic to aspirin; (2) treatment with aspirin plus clopidogrel; (3) cerebral embolism and other determined or undetermined etiologies of IS; (4) administration of warfarin, dipyridamole, clopidogrel within 2 weeks prior to admission; (5) usage of heparin or low-molecular-weight heparin within 24 hours before enrollment; (6) fever, hypoxia, alterations in consciousness on admission; (7) any major surgical procedure within 2 weeks; (8) if the patient had a history of carotid endoartectomy or carotid stent therapy, or underwent carotid endoartectomy or carotid stent therapy during the follow-up period. (9) platelet count < 100 × 109/l or >450 × 109/l; (10) asthma or severe cardiovascular, liver, or renal disease.
All enrolled patients received standard therapies based on guidelines [Kernan et al. 2014], including aspirin (Bayer Healthcare Company Ltd., Beijing, China) 200 mg/day for 14 days and 100 mg/day thereafter for at least 1 year. Vascular risk factors, including body mass index, body weight, tobacco smoking, diabetes mellitus and hypertension were recorded; and fasting blood sugar, total plasma cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) were measured.
Assessment of clinical outcomes
The primary outcome was a composite of RIS, myocardial infarction (MI) and death during the first 1 year after admission. RIS was defined as a new focal neurologic deficit of vascular origin lasting for at least 24 hours, which was proved to be nonhemorrhagic by either computed tomography or magnetic resonance imaging scanning. MI was defined as the presence of at least two of these criteria: prolonged angina > 30 min; total creatinine kinase isoenzyme elevation more than twice the upper limit of normal; electrocardiographic evidence of infarction. Death was defined as vascular mortality due to MI, IS, and other vascular causes.
During the 1-year follow-up period, interviews were conducted every month by investigators who were blinded to the results of other data such as genotyping data. After discharge, scheduled follow-up telephone calls were made biweekly to encourage compliance, to answer any queries, and to record any side effects. Clinical data were collected from patients’ medical records or via phone interview by the referring physicians.
Genotyping
In the present study, a total of 14 variants from six genes, including COX-1 (rs1236913, rs3842787), COX-2 (rs689466, rs20417), TXAS1 (rs194149, rs2267679, rs41708), P2Y1 (rs701265, rs1439010, and rs1371097), P2Y12 (rs16863323, rs9859538), and GPIIIa (rs2317676, rs11871251) were selected from the NCBI database (http://www.ncbi.nlm.nih.gov/SNP), based on the following criteria: (i) SNPs that have been assessed in previous studies [Goodman et al. 2007, 2008; Sharma et al. 2013; Halushka et al. 2003; Li et al. 2007; Grinshtein et al. 2013; Yi et al. 2013b; Kunicki et al. 2009; Lev et al. 2007], (ii) SNPs with minor allele frequency > 0.05, (iii) SNPs leading to amino acid changes.
Whole blood (3 ml) was drawn from an arm vein into a sterile tube containing ethylenediaminetetraacetic acid and stored at −80°C for genotype analysis. Genotypes of the 14 variants were examined using a matrix-assisted laser desorption/ionization time-of-flight mass spectrometry method according to our previous study [Yi et al. 2015b]. In brief, each SNP was amplified using two specific polymerase chain-reaction primers and one extension primer. Genotype call was performed in real time with Mass ARRAY RT software version 3.0.0.4 and analyzed using Mass ARRAY Typer software, version 3.4 (Sequenom Inc., San Diego, CA, USA). Each allele of these SNPs was classified by its known effect on function. For each gene, subjects were dichotomized a priori into two groups, based on whether or not they possessed at least one mutant allele.
Platelet aggregation tests
Venous blood (3 ml) was drawn from an antecubital vein prior to the initial dose of aspirin and at day 7–10 of therapy. Platelet aggregation was measured by light transmittance aggregometry (LTA). The procedures and consistency tests were performed as described in our previous studies [Yi et al. 2013a, 2013b]. Platelet aggregation was recorded as changes in light transmission. The results of optical platelet aggregometry are presented as the amplitude of light transmittance at 5 minutes after addition of the agonist 0.5 mm AA and 10 μm ADP with a BioData PAPS-4 platelet aggregometer (Helena Laboratories, Beaumont, TX, USA). According to our previous studies [Yi et al. 2013a, 2013b], the measurement of platelet aggregation using LTA was established to be highly valid and reproducible.
Statistical analysis
Based on a suggested sample size requirement for detecting gene–gene interactions [Wang and Zhao, 2003], we speculated that a sample size of 820 patients would sufficiently provide 80% power to detect a relative risk increment of 10% in the percentage of primary outcome in patients carrying high-risk interactive genotypes, with a two-sided type I error of 0.05, assuming an event rate of 12% in patients without carrying high-risk interactive genotypes.
All statistical analyses were performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA). The χ2 test was used to analyze the deviation of Hardy–Weinberg equilibrium for genotype frequencies and compare genotype frequencies. Discrete variables were compared using χ2 test or Fisher’s exact test. Continuous variables were compared using Student’s t test.
Gene–gene interaction was assessed using the GMDR program (beta version 0.7, www.healthsystem.virginia.edu/internet/addiction-genomics/Software) according to our previous reported [Lou et al. 2007; Yi et al. 2015b]. The GMDR computed the maximum likelihood estimates and the scores of all individuals under the null hypothesis. This model with the minimum prediction error, the maximum cross-validation consistency score, and a p value of 0.05 or less (derived automatically from the sign test in the GMDR software) was considered as the best model.
Cox proportional hazard regression analysis was performed after adjusting some risk factors to assess the independent contribution of gene–gene interactions for a risk of primary adverse outcome and reported as values of the hazard ratio (HR) with 95% CI. All tests were two sided, and the threshold level of p < 0.05 denoted statistical significance.
Results
Characteristics of the patients
Among 850 acute IS patients, 829 (97.5%) patients completed the 1-year follow-up, 9 (1.1%) patients were lost to follow up, and 12 (1.4%) patients discontinued the study medication because of patients’ early quit. The primary outcome occurred in 112 (13.5%) patients (81 RIS, 16 MI and 15 died). Compared with patients without primary outcome, the patients with primary outcome were older, had a higher prevalence of diabetes mellitus and higher fasting plasma glucose levels or LDL-C levels at baseline (Table 1).
Baseline characteristics of the study patients.
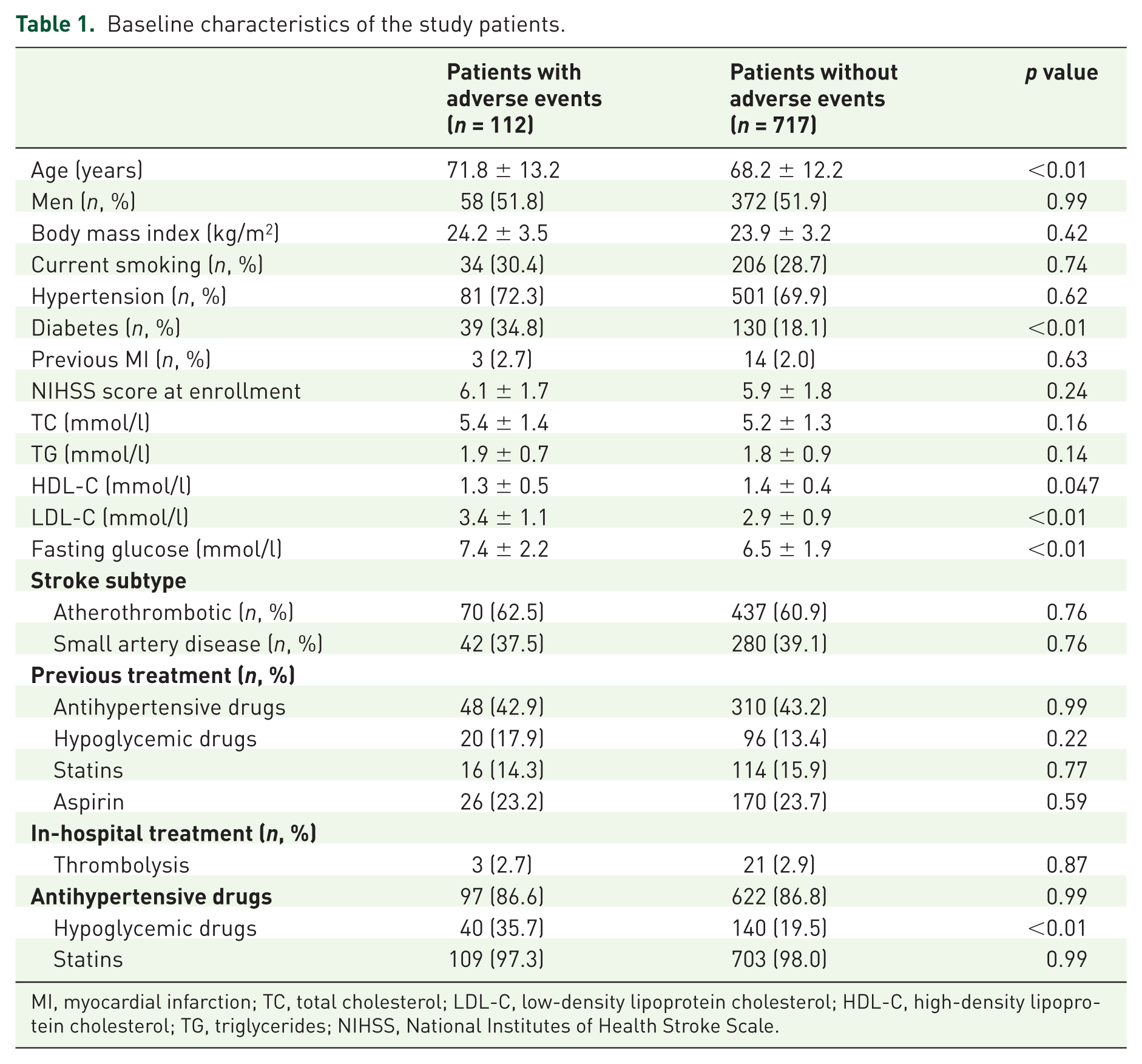
MI, myocardial infarction; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides; NIHSS, National Institutes of Health Stroke Scale.
Association of allelic frequencies with primary outcome
The genotype distributions of the 14 variants examined in this study were in Hardy–Weinberg equilibrium (p > 0.05). The allelic frequencies of 14 variants did not differ significantly between the patients with and those without primary outcome using single-locus analytical approach (all p > 0.05, Table 2).
Allelic frequencies of single nucleotide polymorphisms among the study patients.
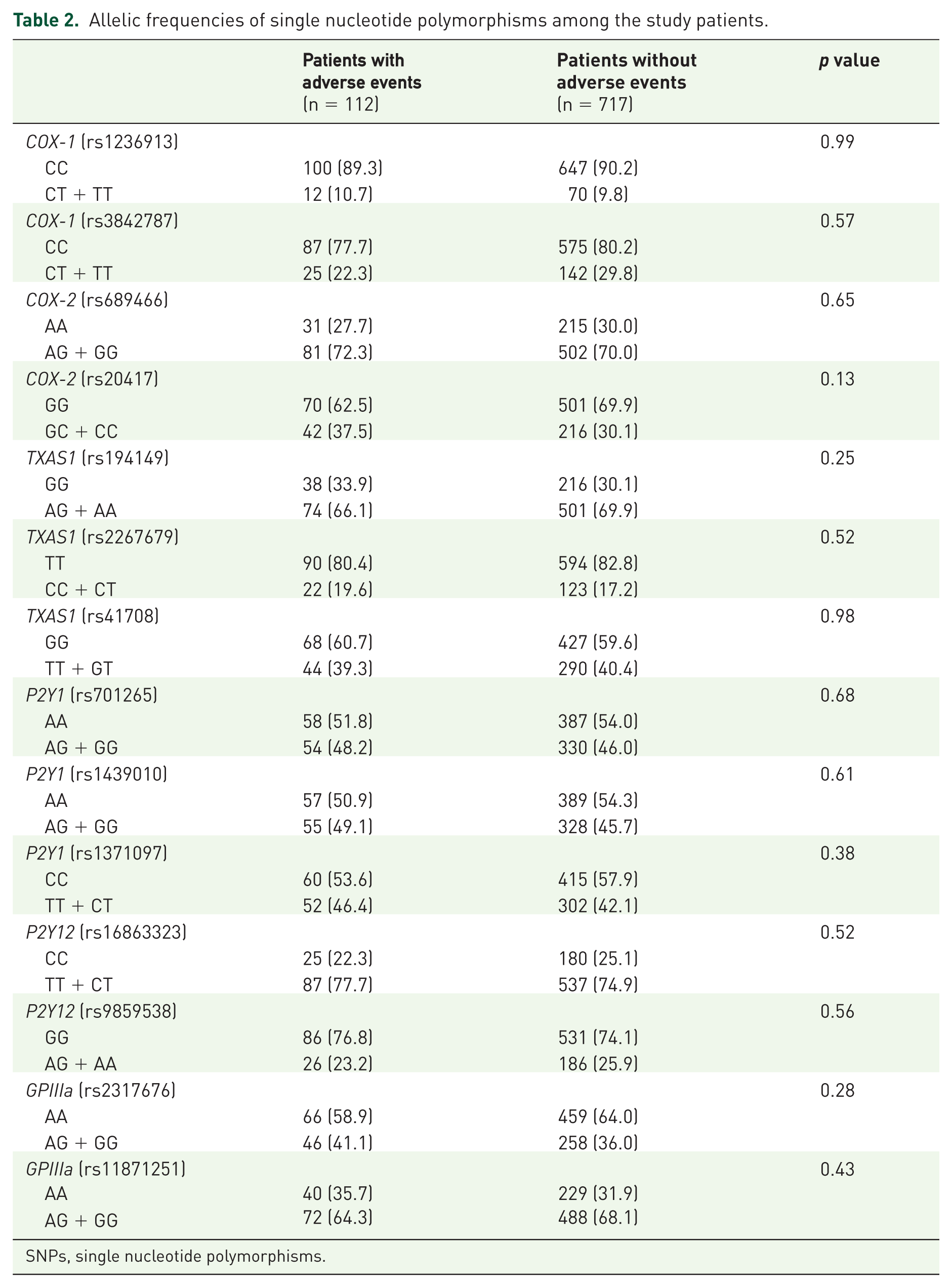
SNPs, single nucleotide polymorphisms.
Gene–gene interaction and its association with primary outcome
We then investigated the association of the high-order interactions of SNPs with primary outcome using the GMDR method. There were significant gene–gene interactions. The best models for primary outcome were rs20417, rs1371097, and rs2317676 after adjusting the covariates, which scored 10 out of 10 for cross-validation consistency and 9 out of 10 for the sign test (p = 0.018, Table 3). The one-locus model was also computed for each variant. The prediction accuracies of these one-locus models by GMDR were 0.5462, 0.6234 and 0.5532 (for rs20417, rs1371097, and rs2317676, respectively), yielding a minimum p value of 0.9426. The significance of this interaction was further confirmed by a permutation test (p = 0.023), suggesting that the three genetic variants together significantly contributed to primary outcome.
Comparison of the best models, prediction accuracies, cross-validation consistencies, and p values for primary outcome identified by generalized multifactor dimensionality reduction analysis.

rs20417, rs1371097, rs2317676, rs1236913, rs3842787, rs689466, rs194149, rs2267679, rs41708, rs701265, rs1439010, rs16863323, rs9859538, rs11871251 are symbolized as 1–14, respectively.
Compared with patients harboring wild-type genotypes rs20417GG, rs1371097CC, and rs2317676AA, the three SNP interactions making the larger contributions to this model were those occurring among rs20417CC, rs1371097TT, and rs2317676GG; rs20417CC, rs1371097CT, and rs2317676AG; rs20417CC, rs1371097TT, and rs2317676GG/AG (Table 4). These results indicate that the high-risk interactions among the three genetic variants together significantly conferred a higher risk for adverse events than did any single variant alone.
Associations between genotype combinations and primary outcome.
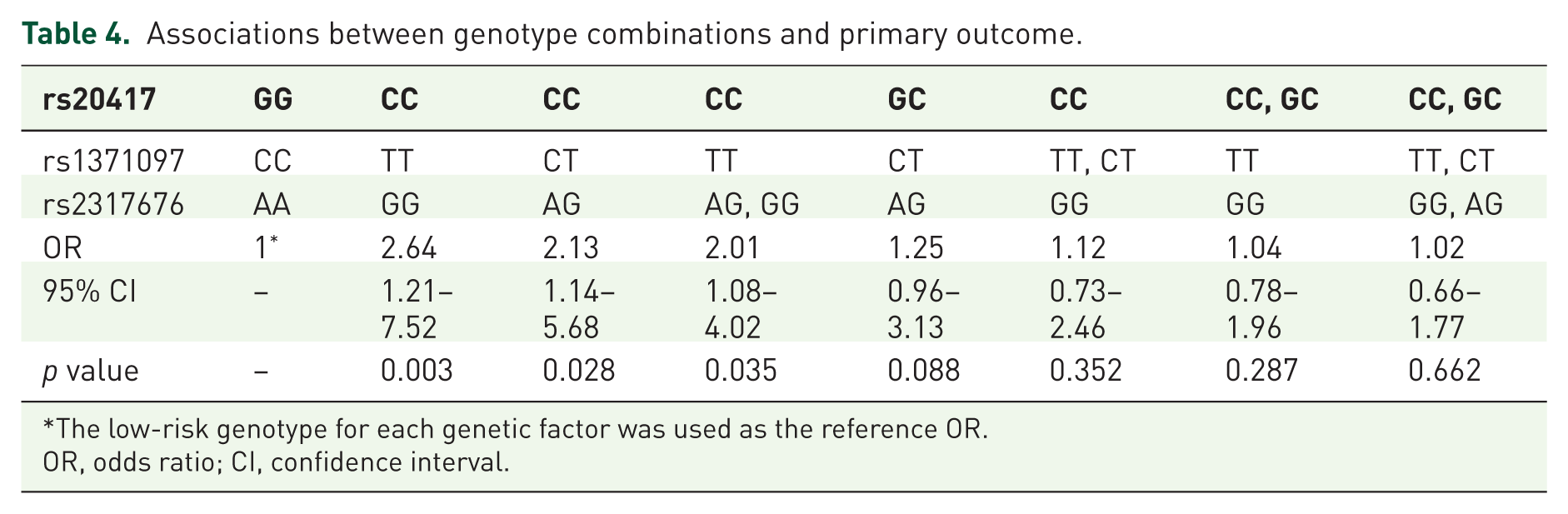
The low-risk genotype for each genetic factor was used as the reference OR.
OR, odds ratio; CI, confidence interval.
Analysis of risk factors for primary outcome
Furthermore, the relative risk conferred by the combinations of variants in the three genes was considered as an interactive variable. The combinations of rs20417CC, rs1371097TT, and rs2317676GG or rs20417CC, rs1371097CT, and rs2317676AG or rs20417CC, rs1371097TT, and rs2317676GG/AG were considered as high-risk interactive variables, were assigned as one; and other combinations of rs20417, rs1371097, and rs2317676 as low-risk interactive variables, were assigned as zero. Cox regression analysis revealed that the high-risk interactive genotypes were independent risk factors for the primary outcome (adjusted for age, diabetes mellitus, LDL-C, and fasting blood glucose) (Table 5).
Cox regression analysis of independent predictors for primary outcome.

LDL-C, low-density lipoprotein cholesterol; HR, hazard ratio; CI, confidence interval.
Association of SNPs with platelet aggregation activity
There were no significant differences in the activity of platelet aggregation among the 14 variants before treatment. After 7–10 days of treatment, the percentage of reduced platelet aggregation, whether AA induced or ADP induced, presented no significant differences among the 14 variants. However, the percentage of reduced platelet aggregation, whether AA induced or ADP induced, was significantly lower in patients with high-risk interactive genotypes than in patients without high-risk interactive genotypes (59.6 ± 11.2 versus 81.1 ± 13.2, and 31.1 ± 9.8 versus 42.4 ± 12.6, respectively, p < 0.01).
Discussion
In this study, we aimed to determine whether previously identified SNPs known to alter the pharmacokinetics of aspirin or the ex vivo ability of platelets to aggregate were associated with clinical outcomes during the first year after acute IS. Our data showed that there were no significant associations between genotype distributions of the 14 variants and primary adverse outcome using the single-locus analytical approach. However, the most noteworthy finding in the present study was that there were significant gene–gene interactions among rs20417, rs1371097 and rs2317676 using GMDR analysis, and the high-risk interactive genotypes of rs20417, rs1371097 and rs2317676 had an independent impact on clinical adverse events. Our results indicated that the sensitivity of IS patients to aspirin and adverse clinical events may be multifactorial, and is not determined by single-gene polymorphisms.
Thrombosis and arteriosclerosis are the major causes of IS, and platelets play a crucial role in triggering arterial thrombosis [Ruggeri, 2002]. Our previous studies and some other studies have shown that platelet activation plays a key role in the pathogenesis of IS and RIS [Marquardt et al. 2002; Wang et al. 2015; Yi et al. 2015a]. Aspirin acts by inhibiting COX enzymes, thereby preventing TXA2 generation and thrombosis. However, the antiplatelet effects of aspirin may not be equal in all individuals. A proportion of patients prescribed aspirin suffer recurrent thromboembolic vascular events. With advances in next-generation sequencing and genetic association analysis, accumulating evidence has suggested that genetic factors may be associated with response to aspirin and its adverse clinical outcome. COX and platelet receptors or glycoprotein receptor are involved in platelet activation and aggregation, and are inhibited by aspirin. Polymorphisms in COX-2 rs 20417 and COX-1 C50T, have been reported to be associated with impaired responses to aspirin [Sharma et al. 2013; Halushka et al. 2003]. However, in our previous study [Yi et al. 2013b] and this study, we did not find these polymorphisms to be associated with response to aspirin in acute IS patients. The GPllb/llla receptor is a key regulator of platelet aggregation. Upon platelet activation, GPllb/llla binds fibrinogen and cross-links adjacent platelets. Some studies have suggested that GPllb/llla variants are associated with AR [Goodman et al. 2008; Sirotkina et al. 2007], whereas other studies have found no such effect of GPllb/llla polymorphisms on platelet activity [Frey et al. 2003]. P2Y12 and P2Y1 are platelet membrane receptors and play a major role in platelet aggregation, arterial thrombosis and the pharmacology of antiplatelet therapy [Storey, 2006]. Grinshtein and colleagues [Grinshtein et al. 2013] reported AR to be associated with P2Y12 and P2Y1 polymorphisms, but Goodman and colleagues [Goodman et al. 2008] did not find these polymorphisms to be associated with AR. Feher and colleagues [Feher et al. 2009] found the role of SNPs in COX-1, COX-2, GPIb, GPIIIa, P2Y1, P2Y12 in the context of antiplatelet drug resistance; they concluded that the results are difficult to replicate between different research groups. This lack of consistency in the literature makes it difficult to define the effects of these gene variants on aspirin responsiveness. There could be a number of potential reasons for the conflicting results, including divergent ethnicity-specific genetic profiles, population stratification, inadequate sample sizes, and the lack of adjustment for confounders [Niu and Qi, 2012]. These controversial results may also reflect the complexity of mechanisms by which genetic factors contribute to aspirin responsiveness. Genetic variants at individual loci might only contribute to AR in the presence of other gene variants. Therefore, it may be crucial to analyze the potential effects of gene–gene interactions on aspirin responsiveness.
In this study, the most noteworthy finding was that there were significant gene–gene interactions among rs20417, rs1371097 and rs2317676, and the high-risk interactive genotypes of rs20417, rs1371097 and rs2317676 were independently associated with clinical outcomes. The nature of these interactions is unclear. Our current study demonstrated that the percentage of reduced platelet aggregation, whether AA induced or ADP induced, was significantly lower in patients with high-risk interactive genotypes than in patients without high-risk interactive genotypes after 7–10 days of treatment. Thus, one possible explanation for the three-factor interaction is that the three genes encode key COX enzymes, platelet membranes receptors and glycoprotein-coupled receptors that participate in the modulation of aspirin pharmacokinetics and pharmacodynamics, a principal component of aspirin responsiveness. However, further studies will be required to investigate the mechanisms by which these gene–gene interactions modify aspirin activity.
However, the present study has several potential limitations. First, due to the limited sample size and two-center design of our study, our results may not represent the full spectrum of the Chinese population study. The findings must be validated in larger, multi-center studies. Second, although we genotyped multiple functional variants in known aspirin-relevant genes, some rare functional variants may have been overlooked; thus, we are not able to exclude the role of rare functional variants in the aspirin responsiveness. Third, we only investigated 14 variants from six aspirin-relevant genes. Several other gene variants, such as GPIa, GPVI and GPIba, may be associated with aspirin responsiveness [Goodman et al. 2008]; thus, future studies involving a larger set of genetic variants must be conducted to elucidate the full extent of gene–gene interaction effects on aspirin responsiveness and its clinical outcomes. Fourth, platelet aggregation activity was only measured using the LTA in the current study. Some studies showed that vasodilator-stimulated phosphoprotein and VerifyNow P2Y12 assay could be a better choice for assessment of platelet aggregation activity [Cuisset et al. 2010]. Future studies with vasodilator-stimulated phosphoprotein and VerifyNow P2Y12 assay are necessary to confirm our current data.
In conclusion, we investigated the contribution of 14 variants from six genes with aspirin responsiveness and its clinical outcomes in acute IS patients using the GMDR approach. The most noteworthy finding was that there were significant gene–gene interactions among rs20417, rs1371097 and rs2317676. The high-risk interactive genotypes of rs20417, rs1371097 and rs2317676 were independently associated with primary outcome of RIS, MI, and death after acute IS.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the Deyang City Science and Technology Research Foundation (#2014SZ035) and the Scientific Research Foundation of Chengdu University of Traditional Chinese Medicine (#YYZX1510).
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
