Abstract
Objectives:
New metrics for clinical spasticity are needed to assess motor performance, since scales such as the Ashworth and Tardieu are unreliable. Here, we assessed outcomes of baclofen treatment in patients with multiple sclerosis (MS) using biomechanical analysis of voluntary movements.
Methods:
Patients with MS and symptomatic limb spasticity were recruited for a pre–post baclofen titration study, along with age-matched healthy controls. Oral baclofen was titrated to optimize spasticity symptoms in all MS cases over 4 weeks. Clinical assessments included the Modified Ashworth Scale (MAS), Tardieu Scale (TS); elbow kinematics were measured via the Transient Acceleration Measurement Interface (TAMI); performance was measured as the score at 4 weeks minus the baseline score in all measures. Movement proficiency within TAMI was quantified through a scale-free smoothness measure, according to the regional excursion deviation (RED) from a constant-velocity approximant.
Results:
Twelve patients with MS [age: 47.8 ± 9.8 years; women: 4; disease duration: 20 ± 10 years; disease-modifying therapy use: 7; Expanded Disability Status Scale (EDSS): 6.8 ± 1.4] and eight age-matched healthy controls were evaluated concurrently (mean age: 49.5 ± 13.1 years; women = 3). In MS cases, no significant improvement in arm spasticity was observed with main effects: MAS: −41.6 ± 72.6 (p = 0.09); EDSS: −1.6 ± 10.4% (p = 0.49); and TS: −8.3 ± 2.1% (p = 0.32), −24.9 ± 63.6% (p = 0.42), and −30.7 ± 79.9% (p = 0.06), at slow, moderate, and fast speeds, respectively. However, voluntary motion smoothness, as measured by TAMI: RED, decreased significantly: 0.62 ± 0.08 versus 0.54 ± 0.09, p < 0.001, indicating significant increase in movement smoothness post treatment.
Conclusion:
A simple biomechanical analysis of voluntary movements revealed a significant reduction of spasticity after 30 days of baclofen therapy in patients with MS that was not detected by clinical assessments.
Introduction
Patients with multiple sclerosis (MS) frequently suffer progressive loss of limb control, characterized by spasticity, which usually leads to muscle rigidity and pain, and in about one third of patients, limits or curtails their activities [Mitchell et al. 2005]. A study of 68 patients with MS found that 53 (78%) were receiving antispasticity medication, whereas 32 (47%) remained impaired [Solari et al. 1999]. Reducing spasticity with medication, even if only temporarily, can help the patient to perform physical exercise, improving their strength, mobility, and coordination [Crayton et al. 2004; de Sa et al. 2011; Stuifbergen, 1997]. Antispasticity medications, however, must be administered carefully, as their dose–response characteristics are idiosyncratic; there is a fine line between muscle release and the ‘rag doll’ effect. Compounding management is the unpredictable nature of relapses and pseudo exacerbations in MS. Thus, a quick, reliable assessment is needed to identify the small changes in motor skill that occur early in an intervention. The commonly used clinical scales, Modified Ashworth Scale (MAS) and Tardieu Scale (TS), are not always appropriate or reliable for the task: they show inconsistent association with electromyogram parameters and have weak inter-rater agreement [Ansari et al. 2008; Blackburn et al. 2002; Fleuren et al. 2010; Pandyan et al. 2003].
Here we report the initial results of a pre- and post-baclofen titration study of MS cases and age-matched healthy controls. Treatment responses were measured by both standard clinical measures, and analysis of kinematic data collected through the Transient Acceleration Measurement Interface (TAMI), during isolated single-joint flexions about the elbow. We tested the hypotheses that conventional clinical measures will not show significant improvement in spasticity, and at least one objective measure will show significant improvement in spasticity.
Methods
Participants
All study participants were recruited from the Washington, DC Veterans Affairs Medical Center (DC VAMC).Twelve patients met the McDonald diagnostic criteria [Polman et al. 2011] and had mild to moderate limb spasticity that was either currently or previously treated with standard anti-spasticity medications. Patients were excluded on the basis of history of other neurological disorders (e.g. stroke), history of severe psychiatric disturbance (e.g. schizophrenia), an MS exacerbation within the last month, and current participation in rehabilitation therapies or interventions for the hand. The research protocol was approved by the Institutional Review Board at the DC VAMC.
Study design
Upon entering the study, participants were weaned from their baclofen over a period of 1 month. After this washout, a baseline clinical evaluation was performed, consisting of the following assessments: the MAS of muscle spasticity [Ansari et al. 2012], TS for the measurement of spasticity [Rivard et al. 2009], and the Kurtzke Expanded Disability Status Scale (EDSS) [Kurtzke, 1983]. These three clinical morbidity scales were administered by a trained neurologist. For the MAS and TS, a single representative score was obtained in the major muscle groups of the upper extremity by averaging the two independent scores. TAMI robotic assessments were also obtained at baseline by a trained research assistant. After titration of baclofen to optimize control of spasticity in each patient (30–90 mg/day), the MAS, TS and TAMI assessments were repeated 1 month from the baseline assessment. We retained only the triceps and biceps + triceps content from the MAS, and elbow and elbow + wrist content from the TS. Both instruments were administered in their entirety, per protocol, but we selected only those aspects which we anticipated would have greatest relevance to the kinematic measure (i.e. gross motor control about the elbow), and discarded data which might obscure this effect. The EDSS is a broader measure without such specificity, and so all data were retained from the EDSS.
TAMI instrumentation
TAMI kinematic performance was measured with TAMI via single-joint extension about the elbow. As shown in Figure 1, participants were seated with their arms supported against gravity and comfortably fastened with Velcro straps by the Mechanical Arm Supporter and Tracker (MAST) [Wininger et al. 2009], with the elbow positioned on axis with the goniometer. Resistive torque of 9 N-m was constantly applied [Pousson et al. 2001]. Elbow angular position was routed from the MAST to a real-time biofeedback display (Figure 1).

Exhibit of the Mechanical Arm Support and Tracker in support of the left arm.
Experimental protocol
Participants were seated comfortably in the MAST and strapped in securely. Measurements were collected from both left and right arms at three speeds: slow, moderate and fast, corresponding to approximate angular velocities of 50º/s, 100º/s and 150º/s, similar to conditions established elsewhere to test the effects of concentric training on muscle hypertrophy [Farthing and Chilibeck, 2003]. During each trial, subjects were instructed to flex and extend their arm in synchrony with a moving target on the computer display. Each trial lasted approximately 60–90 s and comprised at least six cycles of the computer cursor movement in order to ensure that a sufficient amount of data were collected. Participants were instructed to perform discrete motions, stopping briefly at the end of each flexion and extension. Three trials were performed per side (×2) per speed (×3) per day (×2); thus for each participant, 36 trials were collected. Participants were invited to practice the exercise to their own satisfaction before recording; warm-up exercises were offered before the day’s observations, and ample rest was allowed between each session.
Signal processing
Signals were sampled at 225 Hz and processed using the LabVIEW signal processing tools. Post processing of all signals was accomplished via first-order low-pass Butterworth’s filter with 100 Hz cutoff, via the Matlab (The Mathworks, Natick, MA, USA) signal processing toolbox. Individual flexion cycles were segmented manually by picking onset and cessation points. Since fixed criteria could not reliably detect the angular extrema in impaired subjects, who often stalled within a traverse, movement cessation was defined as an epoch of zero velocity followed by a sustained reversal, identified manually.
Analysis
We compared the effects of baclofen treatment while patients extended their elbow within a range of speeds. Here, we chose a family of measures based on the residual excursion deviation (RED). RED measures transient departures of a single-joint trajectory from an ideal constant velocity [Wininger et al. 2009]. According to the general formulation:
where Δθ is the total range of motion and N is the total number of data subdivisions: data were divided into two subdivisions: 0 < t ⩽ 0.5·T and 0.5·T < t ⩽ T, where T is total movement time, and then into progressively smaller subdivisions (indexed by j), until the minimally informative subdivision: data triads. This integration evaluates the area under the curve of
are vectors describing error value Ek at locus
To minimize bias in the RED measure due to factors unrelated to movement smoothness and sensitivity to size factors, such as total range of motion, movement time, or peak or average velocity, we implemented three variations of RED. These variations were exact reproduction of the original RED measure [Wininger et al. 2009]; elimination of the interpolation step, which creates a condition of equal subdivisions N among all movement cycles and thus a uniform number of contributions to S; and reintroduction of a counter measure to control for movement time, but via division of total movement time T, as opposed to interpolation. These variations tested whether temporal normalization was either suboptimally performed in the original formulation of RED or was completely unnecessary. In addition, in order to account for possible bias due to scale in amplitude, we introduced a normalization coefficient 1/Δθ. When the original formulation of RED did not normalize for range of motion, we calculated each measure with and without this coefficient. Thus there were six total implementations of RED.
Statistical analysis
Clinical scores were evaluated at baseline and after 30 days of baclofen treatment for each patient. Cumulative scores for the MAS were calculated by summing scores across applicable categories into a single score. These scores were tested via a paired t test within each of the nine datasets (two MAS, six TS and one EDSS); we did not correct for multiple hypothesis testing for this observational study [Bender and Lange, 2001]. Effect size was calculated as proportional difference: post treatment minus pretreatment, normalized to pretreatment score. Kinematical measures were tested via a repeated measures analysis of covariance, accounting target speed and side of test (right versus left) as covariates.
Results
Clinical characteristics
The demographic and clinical characteristics of the study cohort are outlined in Table 1.
Baseline characteristics of the study population.
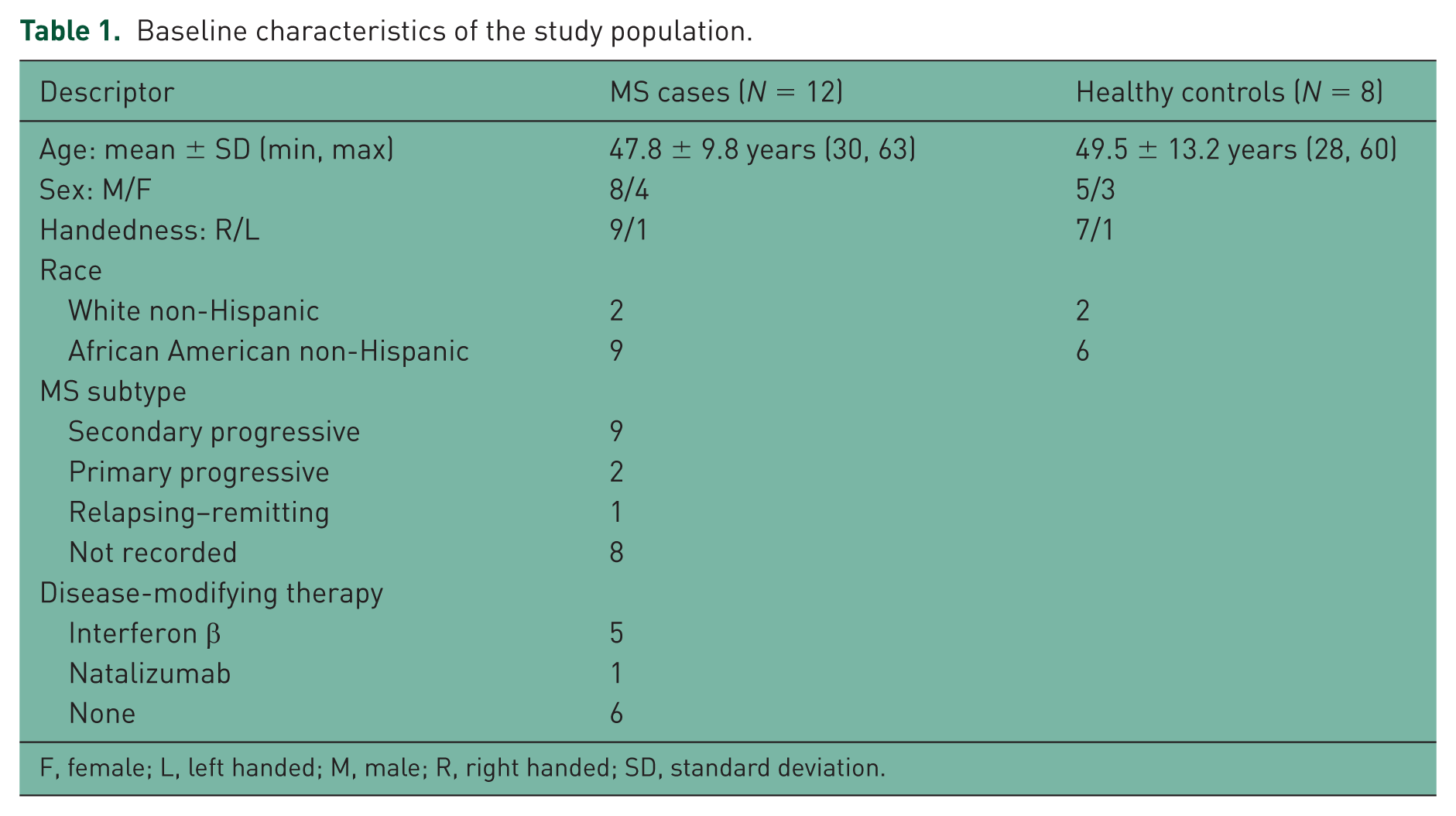
F, female; L, left handed; M, male; R, right handed; SD, standard deviation.
These groups did not significantly differ in terms of age (Wilcoxon rank sum: p = 0.58), sex (χ2: p = 0.77) or handedness (χ2: p = 0.56). The diversity of these cohorts reflects the patient population from which participants were recruited; handedness is approximately commensurate with the general population [Galobardes, 2001; Messinger et al. 1994; Peters, 1992]. From this we conclude that cohorts are adequately representative, and there is no need to include additional covariates in the subsequent hypothesis tests.
Clinical measures
As shown in Table 2, the clinical performance measures did not show statistically significant change (Table 2).
Functional status outcomes following Baclofen administration on patients with multiple sclerosis (N = 12).

EDSS, Kurtzke Expanded Disability Status Scale; MAS, Modified Ashworth Scale; TS, Tardieu Scale; V1–3, Velocity 1–3.
We note that the TS was marginally insignificant at high speeds, with spasticity improvement of over 30%, but p not less than 0.05; the effect as measured by MAS was also large but highly variable, and the marginal significance disappears when wrist data are removed. Similarly, we see a nearly significant effect on TS velocity 1 in the elbow data, but the significance disappears when the wrist data are added. Thus, the null hypothesis that 30-day treatment with baclofen did not improve functional status following treatment cannot be rejected.
Metric selection
The power of six variations of a single performance measure were tested to correctly identify motoric restoration of arm extension following administration of an antispasticity drug. The single measure with the greatest power to discriminate kinematic data between day 1 and day 30 was the original RED measure with a correction for range of motion: metric 4 out of 6:

Whereas none of the other measures yielded significant differences, we conclude that there is a single valid measure for further use in this study. Relative effect was calculated as the proportional change of day 30 to day 1 (i.e. the difference between days, divided by the value at day 1). Exemplars are shown in Figure 2.

Kinematic portraits (left), regional excursion deviation (RED) portraits (center), and heat map visualizations of the RED portraits (right) for a single subject: left arm, medium speed, pre (top) and post treatment (bottom). Time is percent of total movement time, angle is in degree units, and error is a dimensionless error value (departure from linear fit line), normalized by time and angle (units: per degree second).
We see within Figure 2 that the pretreatment data show a reduced range of motion, with multiple narrow bands of high error (substantial departure from a straight-line trajectory, top panels), versus the post-treatment sample, which shows a larger range of motion, with fewer bands of high error; the peak error is approximately half that of the pretreatment sample (bottom panels).
Kinematic measures
Baseline measure of kinematic performance showed substantial impairment in the patients with MS relative to the control group: RED = 0.62 ± 0.08 versus 0.54 ± 0.09, that is, 13% relative difference; cohort effect: p = 2.3 × 10−5. Following treatment with baclofen, the patients showed substantial improvement: 0.55 ± 0.08, that is, 12.7 ± 19.8% relative effect; p = 2.1 × 10−4, thus confirming our hypothesis that the treatment effect would be measurable through kinematic assessment (Table 3).
Effect of 1 month of Baclofen administration on kinematic measures.

For patients with MS, baseline is equivalent to ‘pretreatment’, and 4 weeks is equivalent to ‘post treatment’. Comparison controls were not treated.
MS, multiple sclerosis.
We note that for both groups, there appears to be an improvement in RED at 4 weeks, relative to baseline; this is an interesting result, especially considering that the comparison controls were not exposed to therapeutic intervention. While not an explicit hypothesis of this study, we tested for significance of this difference via a mixed model including the same covariates, and found that there was still a significant difference (p = 0.01) in main effect (not shown in Table 3).
Discussion
Implications
Biomechanical analysis revealed improvements in arm spasticity in patients with MS following baclofen treatment of those with MS who were moderately impaired, while standard clinical measures MAS and TS did not. These results show that the objective measurement by TAMI of voluntary movements produced are more sensitive to the effects of clinical intervention, consistent with results from previous studies of movement smoothness as in patients after stroke [Natarajan et al. 2012; Wininger et al. 2009, 2012; Wininger, 2012]. The question remains: why; what is the difference?
One possible explanation is scope: we consider smoothness (or other basic parameters of movement that can be captured by a rehabilitation device, e.g. movement speed) as the basic rudiments of skilled movement, and therefore a valuable prognostic of health [Hogan, 1984; Mussa-Ivaldi and Bizzi, 1997; Todorov and Jordan, 1998]. In this way, it may be that paradigms like TAMI are reductionistic enough to detect signals of early recovery, and that the clinical measures too comprehensive to detect change except in cases where treatment has become broadly and substantially therapeutic. In contrast with clinical measures, TAMI may have higher resolution for detecting early recovery, since it captures motions at higher frequencies than is possible by human sensation. Moreover, composite data collected over several repetitions is likely more reliable than single observations. Lastly, we observe that while the kinematic measure yields statistical significance when the clinical measures do not, the effect size in the kinematic measures are substantially less than that of the clinical measures. As can be seen in Tables 2 and 3, the effects reported via the kinematic measures are much more consistent versus those reported via the clinical measures, which are much more variable. Whether the variability observed in the clinical measures reflects inter-subject variability in the sensitivity to treatment or vagaries in the assessment process is not known. Nevertheless, we believe that the favorability of the kinematic measure not only supports its use in the neurological evaluation, but also provides evidence of a patient treatment response early in the treatment period.
Study relevance
The efficacy of antispasticity treatments for MS is influenced not only by dose, but also by administration schedule, comorbidity burden, and the types and intensities of patients’ physical activities. Thus, tracking spasticity dynamically with TAMI would be useful in MS management. In addition, exercise training could be initiated at a point when spasticity is optimally managed. Though training at low to moderate intensity is historically favored in MS [Petajan et al. 1996; Schulz et al. 2004], high-intensity exercise for patients with MS is an emergent area of research [Hayes et al. 2011]. Vigorous exercises, particularly those incorporating resistance training, have yielded improvements in strength and power for patients with MS [Dalgas et al. 2009; DeBolt and McCubbin, 2004], but functional gains may be limited by compound mobility issues, weakness and fatigue [Petajan et al. 1996].
Technologies like TAMI provide a convenient, efficient vehicle for measuring spasticity independent of a human observer. As patient load continues to grow beyond the capacity of most clinics, there is incumbent need to find rapid, accurate measures of treatment efficacy so as to place minimal burden on the patient, the provider, and the healthcare system [Birkmeyer et al. 2002]. Where we have shown standard clinical measures to be insensitive to the changes of baclofen in a 30-day treatment window and a kinematic measure capable of reporting measurable change, we believe we extend the basis of evidence to the suggestion that automated performance measurements can impact efficiency and customizability of care delivery [Bosecker et al. 2010].
Study limitations
The findings of this study must be taken in their proper context: a 30-day treatment in a somewhat narrow patient population (patients with MS who are moderately impaired), following de novo titration on a single study medication (baclofen). We acknowledge that the small sample size of this study limits its scope to that of a preliminary pilot investigation. Furthermore, though it is not possible to comprehensively test all potentially relevant subgroupings of elements among the clinical measures, it is possible that some of the variables captured by the RED metric have a particularly strong association with a subset of these items. As such, we propose this as an opportunity for future work.
From a study design standpoint, it may be argued that our six device-based measures were twice as likely to yield a significant effect compared with the three clinical measures, especially in a study without corrections for multiple hypothesis testing. Such corrections, however, would not change the outcome of our primary analysis: even the most aggressive correction, such as the Bonferroni correction, when p is divided by the number of tests (six tests), would yield a significant result. Moreover, the hypothesis of the clinical measures was tested not three but nine times (including the three speeds for the Tardieu and two formulations of the TS and MAS).
By not randomizing the order of assessments, we ran greater risk of systematic bias. However, it was decided to implement the clinical measures in strict order (MAS, then EDSS, then TS V1, TS V2 and lastly TS V3) for the sake of consistency. Moreover, since baclofen is but one of a wide class of possible interventions for the treatment of spasticity, and we neither controlled for, nor analyzed, concomitant medications, no one medication was taken by enough subjects to support a statistical analysis. Thus, the results of this study must be understood in their proper context, that is, each patient presents their own unique clinical history, symptoms, sensitivity/tolerability, compliance to therapies, and disease progression [Bishop et al. 2008].
In addition, we observe that the control subjects showed small but significant improvement at the follow-up evaluation. Whereas these subjects did not receive formal training in the TAMI system, we cannot immediately conclude ‘motor learning’ has transpired. At the same time, whereas these subjects did not receive the study intervention, we can rule out a therapeutic effect. It may not be possible to identify the cause of this decrease in RED. However, clearly the presence of a significant improvement in the smoothness among the control group suggests that the effect observed in the patient cohort may be due to more factors than the baclofen administration.
Lastly, the particular formulation of RED used here is novel; it has not been used in any previous clinical studies. However, this RED measure is similar to the original formulation [Wininger et al. 2009], which has been regularly replicated and studied [Hogan and Sternad, 2009; Hutin et al. 2011; Natarajan et al. 2012; Wininger et al. 2012]. Any measure of kinematic smoothness must undergo rigorous testing before it can be reliably used to test hypotheses related to treatment efficacy; these validations include demonstration of lack of association between the smoothness measure and size factors (duration, amplitude and velocity), and ability to reliably discriminate a known impaired cohort (or, as here, before and after treatment with a known therapeutic agent). The scope of this study does not permit such testing, so while we can assert the viability of the measure for our use in this instance, we cannot yet fully assert its goodness for use beyond this study
Future work
This study reports new information on the effects of baclofen on motion smoothness of the arm but can readily be extended to other joints. Our study design presents a potentially fruitful approach to testing motor control hypotheses and a novel paradigm for the development of performance measures, including identification of a potentially useful modification to a smoothness measure currently in use, and for association between clinical measures and kinematical measures in a targeted training task. Further studies would extend our analysis to limb extension as well as flexion in the upper and lower extremities, for use in spasticity management and as an outcome for physical rehabilitation [Hortobágyi et al. 1996].
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this study was provided by a National Multiple Sclerosis Society Pilot Study Grant PP1658 and the VA Multiple Sclerosis Center of Excellence-East.
Declaration of Conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.


