Abstract
Multiple sclerosis (MS) shares an immune-mediated origin with psoriasis. Long-term safety and efficacy data generated in Europe from usage of fumaric acid formulations in the latter disease constituted grounds to investigate their effects in MS patients. Dimethyl fumarate (DMF) was found to be the active principle in those formulations and in vitro studies have demonstrated that DMF has immune-modulatory properties exerted through abilities to divert cytokine production toward a Th2 profile, both on lymphocytes and microglial cells. More importantly, DMF was discovered to impact the anti-oxidative stress cell machinery promoting the transcription of genes downstream to the activation of the nuclear factor (erythroid derived 2)-like2 (NRF2). DMF exposure increases the cytosol concentrations of NRF2, which besides immune regulatory effects, has the potential for cytoprotection on glial cells, oligodendrocytes and neurons. Extensive and rigorous clinical trials have assessed the efficacy and safety of DMF at the dose of 240 mg twice and three times a day in relapsing-remitting MS patients during one phase IIb and two phase III trials. Robust, positive results were obtained across a number of clinical and paraclinical parameters. In one study (DEFINE), the relative reductions of the adjusted annualized relapse rate of the low and high dose regimens in comparison with placebo were 53% and 48%, respectively (p < 0.001 for both comparisons). In the other trial (CONFIRM), DMF decreased the annualized relapse rate in comparison with placebo by 44% in the lower and by 51% in higher dosage group (also p < 0.001). The number and size of lesions as detected by magnetic resonance imaging were also significantly decreased in comparison with the patients receiving DMF at every dosage. Multiple post hoc and subgroup analyses corroborated the clinical data, rendering DMF an appealing medication whose potential for impacting the degenerative aspects of MS remains to be explored.
Keywords
Introduction
The pace at which advances in medicine are translating into new therapies is particularly fast for the field of neurology and even more so for multiple sclerosis (MS). This is a disease that had no treatments that would modify its course until the early 1990s, when the first breakthrough occurred with the introduction of interferon β. Injection and infusion drugs remained the mainstay of MS treatments for almost two decades when finally oral therapies were developed [Haghikia et al. 2013]. Dimethyl fumarate (DMF), marketed as Tecfidera® [Biogen Idec, 2013], has now been granted indication for MS by the US Food and Drug Administration (FDA) [US Food and Drug Administration, 2013]. In the European Union, the medication received approval by the European Medicines Agency (EMA) in early 2013 [European Medicines Agency, 2013].
Like many others medicines, DMF was not developed out of a process of molecule screening for drug discovery. Besides a brief overview of its history as a medication, this review focuses on the data and the clinical studies leading to its commercial use for MS. This is a therapy that combines safety and efficacy, and has potential that goes beyond the anti-inflammatory effects [Fox et al. 2014].
Early medical use
DMF is a simple molecule derived from fumaric acid. Its utility as treatment for psoriasis was conceptualized on the erroneous assumption dating back to the 1950s that skin disease was caused by an alteration of the citric acid cycle and that exogenous administration of fumaric acid, an intermediate component of that metabolic pathway (Figure 1), could restore its balance [Moharregh-Khiabani et al. 2009]. While fumaric acid is poorly absorbed by the gastrointestinal tract, its ester derivatives, monomethyl fumarate (MMF) and DMF proved to be beneficial in treating psoriasis administered both as topical and orally [Altmeyer et al. 1994]. In Germany, the combination of three ethylhydrogen fumarates and DMF is used in the preparation Fumaderm®, DMF being an approximate 60% of the compounds. This medication obtained indication for the treatment of moderate and severe forms of psoriasis in 1994 and DMF was ultimately found to be the effective principle in the preparation.
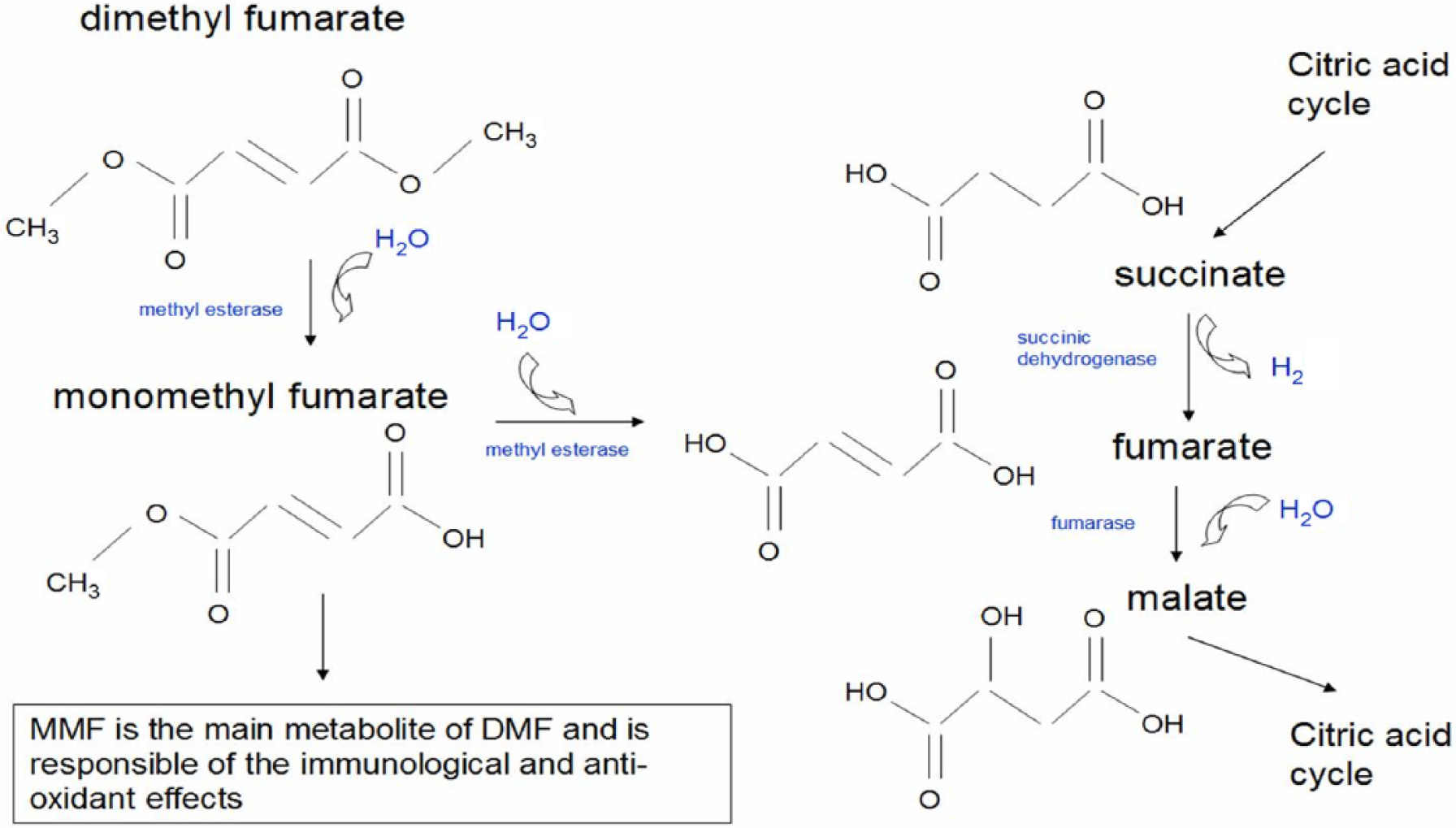
Metabolism of dimethyl fumarate (DMF). Orally administered DMF is not detected in the blood stream as it is rapidly converted in the gastrointestinal tract by hydrolases, such as methyl esterases, into monomethyl fumarate (MMF), which is the active principle exerting the therapeutic effects.
As the immunological disturbances underlying the pathogenesis of psoriasis were unveiled, it became clear that DMF had immunomodulatory properties. Also prompted by the overall good safety profile observed over the extended period that fumarate esters have been prescribed for psoriasis [Reich et al. 2009], DMF has been proposed in other immune-mediated diseases [Meissner et al. 2012]. However, while general practitioners and dermatologists have relied on case reports and case series as evidence for efficacy in a variety of conditions [Meissner et al. 2012], the rigorous testing conducted in patients with MS has provided class I evidence to support its use in MS [Fox et al. 2014].
Metabolism and mechanisms of action
DMF is recognized as the component responsible for the clinical effects of the fumaric acid ester preparation. In vitro studies have demonstrated that DMF is rapidly metabolized at the level of the gastrointestinal tract to its primary, active metabolite MMF by the abundant esterases present in the tissues [Werdenberg et al. 2003]. While DMF is not traceable in the systemic circulation [Meissner et al. 2012], the concentration of MMF in the blood stream peaks at approximately 2–2.5 hours (Tmax) after ingestion and the area-under-the-curve (AUC) proved to be proportional to the dosages [Biogen Idec, 2013]. Tmax is delayed up to a few hours with co-administration of a fat-rich, high calorie meal that, however, does not modify the AUC while it decreases the side effects related to the metabolism of the medication, and less than 0.1% of the DMF dosage can be detected unchanged in the urine [Burness and Deeks, 2014].
To allow it to bypass the stomach and be released in the intestine, DMF has been packed in an oral delayed release formulation known as BG12 [Electronic Medicines Compendium, 2014]. In light of the rapid metabolism of the molecule (Figure 1) and the initial uncertainty on the ability to reach a clinical effect, dosages ranging between 120 and 720 mg per day were pursued in the clinical trials (see below).
Scientific investigations are still in progress to clarify the ultimate mechanism of action responsible of the treatment effects of DMF. What has become clear thus far is that, similarly to other medications such as interferon, DMF does not have a single mechanism of action but rather has a multitude of biological effects. In vitro studies have revealed that DMF has anti-inflammatory properties linked to its ability to promote a Th2 immune response. Added to cultures of stimulated peripheral mononuclear blood cells, MMF enhanced the production of interleukin-4 (IL-4) and IL-5, cytokines characteristic of the Th2 phenotype, in a dose-dependent fashion [de Jong et al. 1996]. Furthermore, besides affecting T lymphocytes, a shift to a Th2 profile was confirmed and replicated in dendritic cells [Litjens et al. 2004, 2006], and it is fair to say that directing the immune response away from Th1 is a likely mechanism by which DMF exerts some of its immuno-modulatory effects.
Additional in vivo and in vitro experiments have further clarified the impact of DMF on type II dendritic cells, providing more details on the cascades of events that follow exposure to DMF [Ghoreschi et al. 2011]. In the end, the impact on T lymphocytes seems to be just a portion of the modifications induced by DMF, which influences several other cells, including macrophages, microglia, astrocytes and neurons [Moharregh-Khiabani et al. 2009; Linker et al. 2011]. In fact, an interesting property that was also largely elucidated on preclinical grounds is the ability of DMF to positively impact the natural anti-oxidative stress machinery of cells. In resting states, nuclear factor (erythroid derived 2)-like2 (NRF2), the major transcription factor for genes involved in anti-oxidative responses, is sequestered in the cytoplasm by the Kelch-like erythroid cell-derived (ECH) associated protein-1 (KEAP-1). MMF has been shown to bind to KEAP-1 and enable the nuclear translocation of NRF2, resulting in transcription of anti-oxidative genes such as hemoxygenase-1 (HMOX1), nicotinamide adenine dinucleotide phosphate (NADPH), quinoline oxidoreductase-1 (NQO1) and others [Chen et al. 2014].
While the details of the interaction between DMF, its membrane receptor and the downstream events continue to be unveiled [Chen et al. 2014], a key message has already emerged: quite remarkably, the biological effects of DMF on the NRF2 pathway are what mediates its immune regulatory properties and lend to the implication that DMF has the potential for being a cytoprotecting agent, a role that at least in animals DMF has been proven to exert [Linker et al. 2011; Chen et al. 2014].
DMF in MS clinical trials
Early experience
The prospect of utilizing DMF as an immune therapy for disorders other than psoriasis was envisioned based on the prolonged experience with good evidence of efficacy in the treatment of psoriasis [Reich et al. 2009], the long-term safety with its use [Hoefnagel et al. 2003) and the appealing mechanisms of actions [Litjens et al. 2006; Moharregh-Khiabani et al. 2009]. All of these functioned as incentives that combined with the positive results obtained in the animal model of MS, the experimental autoimmune encephalomyelitis [Schilling et al. 2006], paved the way to the clinical studies in MS patients.
MS is thought to be a disease of the immune system that is characterized by infiltration into the central nervous system of autoreactive immune cells; these, with varying degrees of severity, cause demyelination, gliosis, neuronal loss and eventually cerebral atrophy [Frohman et al. 2006]. A relapsing-remitting course, which is experienced by the majority of the patients affected by the disease, coincides with recurrent bouts of more or less self-limited inflammatory activities. When eloquent parts of the central nervous system are involved, attacks of neurological deficits, or relapses, manifest clinically [Frohman et al. 2006].
Assessing the ability of a drug to modify the rate of clinical relapses has been an outcome measure for pretty much every medication thus far tested for efficacy and ultimately approved for use in MS. Similarly, all clinical trials carried out with DMF included patients classified as relapsing-remitting MS (RRMS), and the annualized relapse rate was either the primary or a secondary study outcome. And, recognized as a powerful tool for diagnosing and monitoring MS disease activity, magnetic resonance imaging (MRI) techniques have now assumed a key role in the evaluation of the efficacy of treatments under investigation.
Thus, the first exploratory experience in a controlled setting, where a few MS patients were treated with the fumaric acid esters contained in Fumaderm®, was reported in 2006, and both clinical and radiological parameters were tested [Schimrigk et al. 2006). A total of 10 RRMS patients with Expanded Disability Status Scale (EDSS) scores between 2.0 and 6.0, and at least one gadolinium positive (Gd+) lesion on brain MRI were initially enrolled in this open-label pilot study. The investigation was designed with four stages: (1) a 6-week run-in period; (2) an 18-week treatment period during which the dose was escalated up to 720 mg/day; (3) 4 weeks off therapy; and (4) 48 weeks with target dose of 360 mg/day. Three patients withdrew during the initial baseline period. After stage 2, a significant reduction of number (p < 0.05) and volume (p < 0.01) of the Gd+ lesions was detected in comparison with baseline and this effect persisted through the stage 3 or wash out period, and into the treatment phase at 360 mg/day. Other clinical parameters, such as EDSS, ambulation index and nine-hole peg test remained stable. Safety was evaluated by monitoring blood and urine tests, obtaining electrocardiograms (EKG) and recording side effects. No safety concerns were raised on the laboratory measures and no adverse events manifested besides gastrointestinal complaints and flushing, common occurrences in prior experience with the medication [Schimrigk et al. 2006].
Large clinical trials
Multicenter, international, placebo controlled, double blind, clinical trials involving all together almost 2900 MS patients have been completed. A summary of the characteristics and the results of the phase II [Kappos et al. 2008] and the two phase III trials [Gold et al. 2012; Fox et al. 2012] are given in Table 1.
Prospective data from the phase IIb and the two phase III clinical trials of dimethyl fumarate (DMF) in multiple sclerosis (MS) patients.

ITT: intention to treat analysis is intended for patients who after randomization received at least one dose.
BID, twice a day; CONFIRM, Comparator and an Oral Fumarate in Relapsing–Remitting Multiple Sclerosis; DEFINE, Determination of the Efficacy and Safety of Oral Fumarate in Relapsing-Remitting Multiple Sclerosis; EDSS, Expanded Disability Status Scale; GA, glatiramer acetate; MRI, magnetic resonance imaging; N/A, not applicable; QD, daily; RRMS: relapsing-remitting MS; TID, three times a day.
In 2008, the results of a phase IIb clinical trial that tested the safety and efficacy of three different dosages of BG12, the DMF delayed release formulation, were published [Kappos et al. 2008]. A total of 257 patients with RRMS were randomized to receive 120 mg daily, 120 mg three times daily (TID), 240 mg TID or placebo for 24 weeks. Monitoring of all patients was continued for 24 more weeks, while the patients who had been on placebo were switched to the 240 mg TID dosage. The primary outcome was new postcontrast enhancing lesions on MRI brain assessed at 12, 16, 20 and 24 weeks. Besides additional radiological parameters, the annualized relapse rate was compared between treatment and placebo groups. At week 24 relative to week 12, the treatment with DMF 240 mg TID resulted in a reduction of the mean total number of Gd+ lesions by 69% (1.4 versus 4.5; p < 0·0001). Both the number of new or enlarging T2 lesions and new T1 hypointensities were also significantly reduced compared with placebo, and from the clinical point of view, the effect of DMF consisted of an improvement in the annualized relapse rate of 32% (0.44 versus 0.65 for placebo; p = 0.272). Subsequent analyses of the MRI data confirmed the efficacy of the medication on various subgroups of patients [Kappos et al 2012].
The side effects recorded during the study were in line with the expected gastrointestinal symptoms and flushing and, in the absence of safety concerns, the stage was set for the two larger phase III studies [Papadopoulou et al. 2010].
In its 2 year duration, the Determination of the Efficacy and Safety of Oral Fumarate in Relapsing-Remitting Multiple Sclerosis (DEFINE) study [Gold et al. 2012] was set to assess as the primary aim the proportion of patients who developed clinical relapses after random assignment to either 240 mg of DMF twice a day (BID) or 240 mg TID or placebo (Table 1). A statistically significant difference with a p value of 0.001 was observed in the comparison between the two treatment and the placebo groups, the proportion of patients with relapses being 27%, 26% and 46% for the BID, TID and placebo groups, respectively. The relative reduction of the annualized relapse rate of the low and high dose DMF regimens in comparison with placebo was 53% and 48%, respectively (p < 0.001 for both comparisons).
Along with meeting the primary endpoint, the results were positive for radiological parameters and other secondary outcome measures (see Table 1 for details). Of particular interest is the effect of treatment on disability progression; at the 2 year mark, the percentage of patients who had manifested progression in their severity of EDSS as estimated by Kaplan–Meier curve was 16% for the BID and 18% for the TID dosages, and 227% for the placebo group. The treatment reduced the risk of disability progression in comparison with placebo by 38% for the BID and 34% for the TID dosages. Taken as a whole, the data led to the conclusion that DMF had a robust treatment effect at both dosages.
The results of the other trial, the Comparator and an Oral Fumarate in Relapsing–Remitting Multiple Sclerosis (CONFIRM) study [Fox et al. 2012] were published simultaneously to the DEFINE study. The intention to treat analysis for CONFIRM was performed on 1417 patients who had been assigned to receive DMF 240 mg BID, 240mg TID, placebo or open label, subcutaneous 20 mg daily glatiramer acetate (GA) (Table 1). The study was designed to compare DMF with placebo and not to test superiority or noninferiority to GA, which functioned as active comparator. The investigators remained blinded to the treatments by having distinct treating and assessing raters, and the annualized relapse rate was computed throughout the 2 years of the study. The treatment with DMF resulted in a reduction of the annualized relapse rate over placebo by 44% for the lower dose and 51% for the higher dose group (p < 0.001). Suggestive of robustness was the ability of GA to lower the annualized relapse rate by 29% relative to placebo, data consistent with the original pivotal trial [Johnson et al. 1995]. The disability progression, one of the secondary outcome measures, was not significantly different at study end; compared with placebo, each treatment arm showed a relative reduction of 21% (p = 0.25), 24% (p = 0.20) and 7% (p = 0.70) for DMF BID, DMF TID and GA, respectively. A subset of 681 patients had MRI scans and, at 2 years, DMF BID, DMF TID and GA proved to be superior to placebo, reducing the mean number of new or enlarging lesions on T2 weighted images by 71%, 73% and 54% (p < 0.001 for all comparisons), respectively.
To further define the impact of the treatment with DMF on patients with MS, several investigations and post hoc analyses have been pursued. The benefits of DMF were confirmed on reduction of clinical relapses, slowing of disability progression and various radiological outcome measures [Havrdova et al 2013; Hutchinson et al. 2013; Arnold et al. 2014a, 2014b; Kawalec et al. 2014]. Besides positive results on several radiological parameters, noteworthy are the data on brain atrophy obtained in the cohort of patients who had MRI scans in the DEFINE study [Arnold et al, 2014a]. Although the placebo group had a low rate of brain atrophy and despite the difficulties with accurate and reliable measurements of brain atrophy, treatment with DMF at the BID dosage led to a reduction by 21% with p = 0.0449 when baseline was compared with those at 2 years, and 30% (p = 0.0214) when data at 6 months were compared with those at 2 years, also in the BID patient group.
Moreover, MS appeared to afflict the quality of life in a direct relationship to the disease stage and to the occurrence of relapses. In addition, the health-related quality of life was shown to be favorably impacted by the treatment with DMF in both the DEFINE [Kappos et al 2014] and CONFIRM [Kita et al. 2014] trials.
The similarities and the homogeneity of the patient populations enrolled in the DEFINE and the CONFIRM trials permitted a combined analysis to be performed on the results for patients with newly diagnosed disease [Gold et al. 2014a). Focusing on the treatment-naïve patient population who had been diagnosed with MS within 1 year prior to entering the studies, the investigators identified 221, 234 and 223 patients assigned to the 240 mg BID, the 240 mg TID and placebo, respectively. By the end of the 2 years, DMF reduced the annualized relapse rate in comparison with placebo by 56% for the BID and 60% for the TID groups (both p < 0.0001). The risk of 12 week confirmed disability progression was also lower in the treatment groups by 71% (p < 0.0001) and 47% (p = 0.0085) for the BID and TID groups, respectively. A subset of about 100 patients in each group had data for radiological analysis and, in comparison with placebo, DMF decreased the adjusted mean number of new or enlarging T2 lesions or the mean number of Gd+ lesions in the range of 80 to 92% at either the low or high dosage [Gold et al. 2014a].
Demonstrating that DMF has an effect in early stages of the disease that is perhaps even stronger than in the global cohort has an implication on the application of an established paradigm in the treatment of MS. Initiation of therapy early on has a greater impact on the disease activity and, for better long-term results, it would make sense to apply an effective therapy before damage has occurred [Bomprezzi et al. 2012]. This concept allows DMF to be considered a valid first-line agent for the treatment of MS [Havrdova et al. 2013; Nicholas et al. 2014].
Safety profile and side effects of DMF
The safety profile of a medicament or any medical intervention constitutes an essential element for it to be considered a suitable treatment option, and in the neurology clinical practice, the variability of the courses of MS demands decision making that involves computing risks and benefits [Bourdette and Whitham, 2010; Carrithers, 2014; Nicholas et al. 2014]. Faced with a disease with devastating potential, chemotherapies with overt toxic side effects [Bourdette and Whitham, 2010] have been part of the armamentarium available to practitioners. However, the expectations are that, in parallel with better understanding of disease pathogenesis, come better therapies defined by improved efficacy and diminished potential harm.
With decades of experience with obvious benefits and no significant ill effects [Reich et al. 2009; Hoefnagel et al. 2003], DMF has aspired to the role of an immune therapy with broader applications [Meissner et al. 2012]. It has now undergone the most rigorous clinical testing for patients with MS and its safety has been established [Fox et al. 2014]. The most common symptoms experienced by patients taking DMF in the context of the clinical trials were flushing and gastrointestinal complaints (inclusive of diarrhea, nausea, upper abdominal pain, vomiting and abdominal discomfort) that combined reached an incidence up to 38% for the treated groups and 2–6% for the placebo or the active comparator GA groups (Table 2) [Gold et al. 2012; Fox et al. 2012; Salmen and Gold, 2014; Burness and Deeks, 2014]. It is worth noting that the severity was rated at mild-to-moderate and the percentage of patients complaining of the side effects dropped to about 5% after the first month on the medication; the percentage of patients who withdrew from the studies due to any gastrointestinal side effects ranged between 1 and 6% in the two phase III clinical trials [Gold et al 2012; Fox et al. 2012].
Prospect of the most common adverse events (AEs) in the DEFINE and CONFIRM trials.
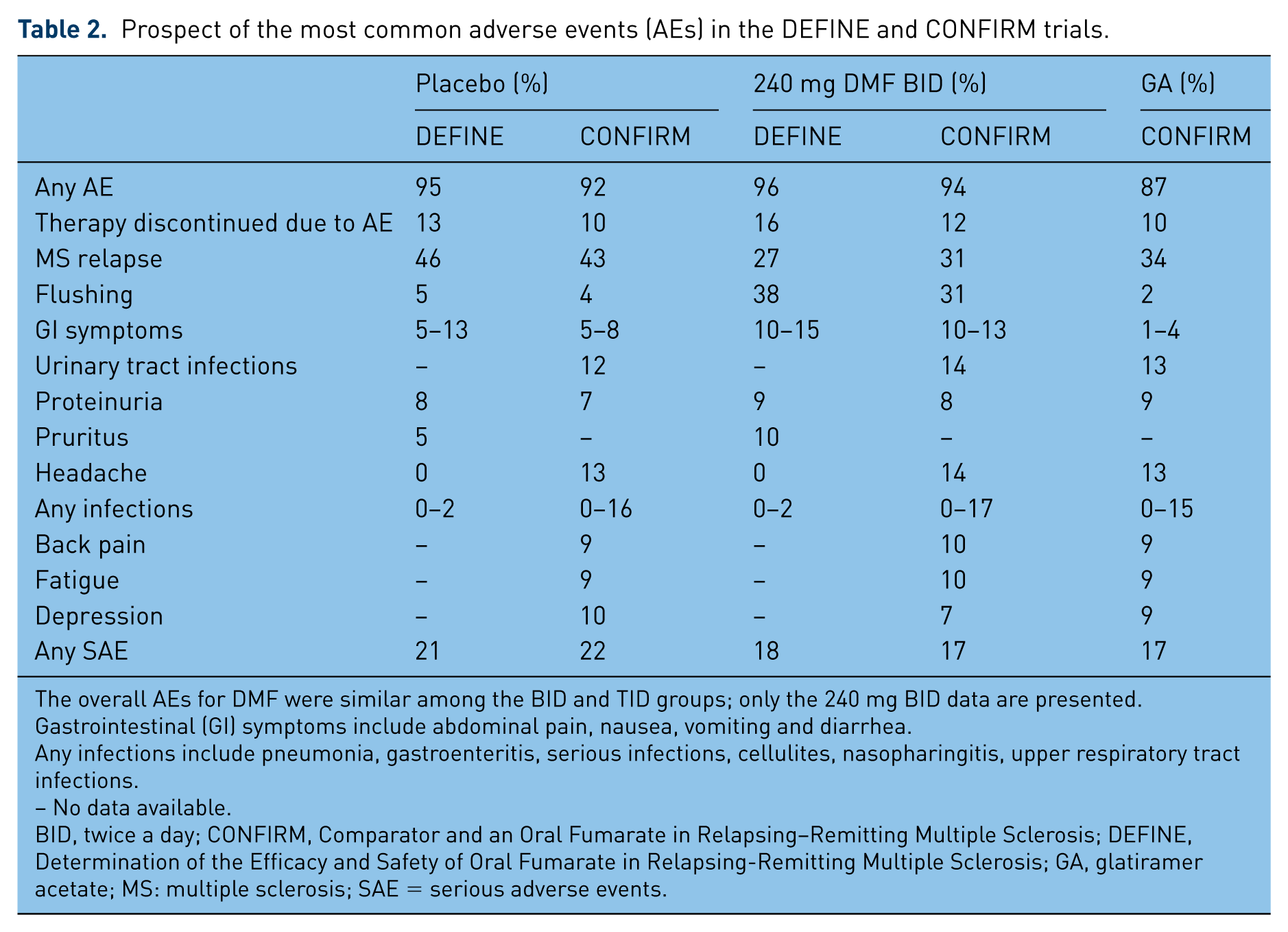
The overall AEs for DMF were similar among the BID and TID groups; only the 240 mg BID data are presented.
Gastrointestinal (GI) symptoms include abdominal pain, nausea, vomiting and diarrhea.
Any infections include pneumonia, gastroenteritis, serious infections, cellulites, nasopharingitis, upper respiratory tract infections.
– No data available.
BID, twice a day; CONFIRM, Comparator and an Oral Fumarate in Relapsing–Remitting Multiple Sclerosis; DEFINE, Determination of the Efficacy and Safety of Oral Fumarate in Relapsing-Remitting Multiple Sclerosis; GA, glatiramer acetate; MS: multiple sclerosis; SAE = serious adverse events.
Patients who have suffered from the gastrointestinal symptoms have sometimes improved their tolerability of DMF by taking the medication with a full stomach and by the co-administration of fat-rich food. However, as reported in a recent update from the extension study of DEFINE and CONFIRM trials, the discontinuation rate due to side effects remains in the range of 4–6% at 4 years [Phillips et al. 2014], and a trial is ongoing to test the possibility of mitigating the side effects by pretreatment with aspirin [ClinicalTrials.gov identifier: NCT02090413]. An investigation in animals has provided the evidence for the rationale for a nonsteroidal anti-inflammatory drug to lessen the flushing associated to DMF administration [Hanson et al. 2010].
The occurrence of infections (including nasopharyngitis, other upper airway or urinary tract infections) back pain, headaches, fatigue, malignancies and other symptoms was similar between treatment and placebo groups (Table 2). MS relapses were more frequent among the patients on placebo, causing up to 8% of patients to drop out of the studies [Gold et al. 2012].
As discussed above, the metabolism of DMF does not involve the CYP450 system and chances are low that DMF and MMF have drug–drug interactions, including with oral contraceptives [Burness and Deeks, 2014]. As a matter of fact, liver enzyme elevation did not appear to be associated with the administration of DMF, while the medication was found to cause a decrease in the total mean white blood cell and the mean lymphocyte counts. Lower numbers of those circulating cells up to 32% of the baseline range were detected starting at week 8 and persisted for the duration of the studies [Gold et al. 2012; Fox et al. 2012]. This led to the recommendation to obtain a complete cell count within 6 months for patients interested in treatment with DMF and the subsequent monitoring while on therapy [US FDA, 2013]. However, a fatality case secondary to progressive multifocal leukoencephalopathy (PML) in a MS patient who had been treated with BG12 for 4.5 years was published on 23 October 2014 [UK Multiple Sclerosis Society, 2014]. The patient had been followed in the context of an open-label extension study [ClinicalTrials.gov identifier: NCT00835770] and persistent lymphopenia related to the administration of DMF was documented in that patient. In the posmarketing setting there had been precedent instances of patients with psoriasis manifesting PML while being treated with Fumaderm® and compounded fumarates [Ermis et al. 2013; van Oosten et al. 2013; Sweetser et al. 2013; Nicholas et al. 2014]. The common denominator to those cases was the high degrees of lymphopenia; this correlates with an incompetent immune system, the premise for opportunistic infections to occur [Mrowietz and Reich, 2013]. The suggestion to monitor lymphocyte counts for patients on DMF predates this incident in an MS patient [Salmen and Gold, 2014]. The German Multiple Sclerosis Society has now issued new recommendations that advise repeating blood cell counts every 6–8 weeks with discontinuation of treatment in case of persistent lymphopenia [German Multiple Sclerosis Society, 2014].
With regards to pregnancy, there are limited data to draw a conclusion on the teratogenicity in humans of DMF. The medication is labeled as category C [Lu et al. 2014] and, being a small molecule, DMF has the ability to cross the placenta. However, from the analysis of a registry, the inadvertent exposure to DMF during the first trimester of pregnancy did not result in increased risk of fetal abnormalities nor increased incidence of spontaneous abortions [Gold et al. 2014b] and participation in pregnancy registries is likely to yield more definite answers on this topic [National Multiple Sclerosis Society, 2014].
In the end, the principle that there are no intrinsically toxic substances but there are invariably toxic doses applies to DMF, too; when it was used as an antifungal on leather products and furniture, DMF caused an outbreak of allergic dermatitis [Basketter et al. 2013] and it was the serendipitous utilization as an oral agent for psoriasis that changed the fate of DMF as a medication. Now, the overall safety of DMF continues to inspire a confidence that already within the first year from release into the market has made it a widely prescribed medication for RRMS patients.
Conclusive remarks
Through consistency across all of the studies, DMF has proven to have robust treatment effects with acceptable safety, and the data have permitted approval of delayed-release DMF for RRMS patients at the dose of 240 mg BID by the FDA [US Food and Drug Administration, 2013] and the EMA [European Medicines Agency, 2013].
Although the list of MS therapies continues to grow, the greatest challenge in the treatment of MS lies ahead. Decent results have been obtained in controlling the inflammatory component of the disease, whereas the attempts to effectively abate the degenerative processes and promote repair of the central nervous system damage, the ultimate cause of the disabilities related to the illness, have been unsuccessful. Positive effects have been observed on disability progression with DMF [Gold et al. 2012], but those data that include the MRI findings on brain atrophy could be interpreted as benefits obtained through the reduction of the destructive inflammatory activities exerted by the medication, while no actual impact has been proven on the cell degenerative process once it is set in motion. Addressing this latter issue, arguably the holy grail of MS therapy, has been the focus of intense research and it looks like uncontrolled oxidative stress has a role in mitochondrial damage, cell degeneration and disease progression [Gilgun-Sherki et al. 2004; Arnold et al. 2014c]. As discussed above, DMF has been shown to be an activator of the cellular, intrinsic response mechanisms to oxidative stress [Arnold et al. 2014c], and it has the potential to prevent cell damage and hence promote cell survival [Linker et al. 2011]. Recently, the results of a pilot study on the impact of compounded fumarates and Fumaderm® in a limited number of patients with progressive MS was reported [Strassburger-Krogias et al. 2014]. During the mean follow up of 13.2 ± 7.5 months (range 6–30 months), the majority of the patients showed stability of their neurological status in the face of an overall good tolerability, and additional investigations to assess the potential benefits of DMF in patients with progressive stages of MS were proposed on the basis of those observations. Indeed, it seems that what it is already known about DMF is far from sufficient to provide a complete picture of the potentials of this medication. In parallel with more randomized trials, there is a need for prolonged monitoring for long-term safety and tolerability, and observational studies have already been implemented [Phillips et al. 2014; ClinicalTrials.gov identifiers: NCT02047097, NCT00835770].
Footnotes
Conflict of interest statement
The author declares no conflicts of interest in preparing this article.
Funding
This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
