Abstract
Menstrual migraine (MM) is a highly prevalent condition associated with considerable disability. Migraine attacks occur exclusively around the menstrual period in approximately 10% of women with migraine, that is, pure menstrual migraine, while at least 50% of them also experience migraine at other times of the month, that is, menstrually related migraine (MRM). The therapeutic approach to patients with MRM is based on treatment of the attack, or prophylactic strategies. Triptans are recommended as first-line treatments for moderate to severe migraine attacks, including MM. Frovatriptan is one of the newest triptans. Its high affinity for 5-HT1B/1D receptors and long half-life contribute to its distinctive clinical effect, characterized by a more sustained and prolonged effect than other triptans. Indeed, frovatriptan proved to be effective in treating the acute attack, but was particularly effective in the short-term preventive therapy of MM. In addition, frovatriptan is one of the safest triptans, with the lowest risk of treatment-emergent adverse events. Following extensive evidence from randomized pharmacological trials, frovatriptan has now gained a grade A recommendation from the guidelines for short-term prophylaxis of MM. Recent post-hoc analyses of direct comparative trials also suggest that frovatriptan might have an important role in the acute treatment of MRM. In these studies, frovatriptan showed pain relief and pain-free rates similar to those of zolmitriptan, rizatriptan, and almotriptan, but with significantly lower recurrence rates. More well-designed, randomized, prospective studies, specifically enrolling women with MM, will be needed in the near future to confirm the efficacy of frovatriptan in this migraine subtype.
Introduction
The International Headache Society defines menstrually related migraine (MRM) as attacks of migraine without aura in a menstruating woman, occurring on day 1 ± 2 of menstruation (namely between 2 days before menstruation and the first 3 days of bleeding) in at least two out of three menstrual cycles and additionally at other times of the cycle [Headache Classification Subcommittee of the International Headache Society, 2004]. Menstruation is usually defined by endometrial bleeding resulting from either the normal menstrual cycle, or from the withdrawal of exogenous progestogens, as in the case of combined oral contraceptives and cyclical hormone replacement therapy. Migraine attacks occur exclusively around the menstrual period in approximately 10% of women, that is, pure menstrual migraine (PMM), while at least half of women with migraine experience MRM [MacGregor, 2008; Martin and Lipton, 2008; Taylor, 2009].
Migraine attacks occurring during menses, generally classified as menstrual migraine (MM), represent a challenge for both the patient and the headache specialist as they are particularly difficult to manage and more disabling than migraines outside the menstrual period [Allais et al. 2007].
The purpose of this review is to provide an overview on the efficacy of frovatriptan in the acute and preventive treatment of MM in the light of recent publications and indications from guidelines [Silberstein et al. 2012; Steiner et al. 2007].
Current therapeutic approach to MM
The therapeutic approach to patients with MRM is based on treatment of the symptoms of attack, or with prophylactic strategies [Silberstein et al. 2012; Steiner et al. 2007].
The goal of acute treatment is to attenuate or remove symptoms, and this approach is ideal for infrequent attacks. No drugs are specifically approved for the acute treatment of MRM. However, treatments licensed for migraine are usually indicated also for the treatment of MM [Headache Classification Subcommittee of the International Headache Society, 2004]. The current therapeutic armamentarium against acute MM is the same as for nonmenstrual attacks, and includes triptans, nonsteroidal anti-inflammatory drugs, ergot derivatives, or a combination of analgesics with or without prokinetic antiemetics [MacGregor, 2010].
Prophylaxis may be an option for treating MM, but its choice depends on the frequency, duration, and severity of attacks, as well as on the response to acute treatment [Tepper, 2006]. The goal of preventive treatment is to reduce attack frequency, severity, and duration, improve responsiveness to treatment of acute attacks, ameliorate function, and reduce disability [Pringsheim et al. 2008]. Currently suggested effective prophylaxis is based on nonsteroidal anti-inflammatory drugs, perimenstrual administration of estrogen or triptans, but no treatment is at the moment licensed for MM prevention. The current opinion is that an empirical approach is usually necessary, trying different interventions before considering the effective prophylactic program. Prophylaxis seems to be particularly suited for those women experiencing inadequate relief from the usual forms of acute therapy, or who are troubled by headache recurrence and require multiple doses of acute migraine medications [Tepper, 2006; Pringsheim et al. 2008], or for those women with regular menses and predictable crisis [Allais et al. 2007]. The choice between short-term (perimenstrual) and continuous prophylaxis depends on the individual woman’s type of migraine, regularity of menstruation, other menstrual problems, and need for contraception [MacGregor, 2010]. Usually, short-term prevention is preferred to long-term prophylaxis, because it potentially reduces the risk of adverse events.
Triptans and migraine
Triptans are serotonin 5-HT1B/1D receptor agonists acting on several areas, both around vasculatures and centrally [Loder, 2010]. They provide relief from migraine pain by cranial vasoconstriction, most likely through action at postsynaptic 5-HT1B receptors on the smooth-muscle cells of meningeal blood vessels. They also act at presynaptic 5-HT1D receptors on trigeminal nerve terminals projecting peripherally to the dural vasculature and centrally to the brainstem trigeminal nuclei [Loder, 2010; Kalra and Elliott, 2007]. 5-HT receptors are also localized on glutamate-positive trigeminal neurons: triptans could reduce glutamate release by acting through these receptors and this may contribute to their therapeutic effect [Ramadan et al. 2003].
Seven triptans are available worldwide, in different strengths and formulations, including oral tablets, orally disintegrating tablets or wafers, nasal sprays, subcutaneous injections, and suppositories (Table 1). Specific pharmacological and clinical differences among the triptans exist [Loder, 2010; Ferrari et al. 2002; Bigal et al. 2003; Johnston and Rapoport, 2010; Pascual et al. 2007]. The first triptan to be developed, sumatriptan, offers the most rapid and complete pain relief when administered subcutaneously, but is also associated with a higher incidence of adverse events. Pharmacology of second-generation triptans was modeled on sumatriptan, aiming at developing oral formulations with improved bioavailability, and increased rate of drug absorption potentially to hasten and improve migraine headache relief [Rapoport et al. 2003]. All triptans are superior to placebo in clinical trials, and some (zolmitriptan, rizatriptan, almotriptan, and eletriptan) are similar to each other and the prototype, sumatriptan (Table 1). These triptans are known as fast-acting triptans and are characterized by a faster onset of action, but also a higher risk of adverse events and a higher recurrence rate (Table 1). Naratriptan and frovatriptan belong to the group with lower recurrence and a better tolerability (Table 1).
Pharmacologic and clinical characteristics of triptans and main efficacy results from clinical trials.

Tmax, time to reach peak plasma concentration.
Use of triptans in MM
Triptans are recommended as first-line treatment for moderate to severe migraine, hence including MM [Taylor, 2009; Mannix and Files, 2005].
Data from randomized, placebo-controlled or active-treatment comparative studies for the acute treatment of MM are available for all seven triptans currently approved and licensed. Evidence of triptan efficacy for short-term prophylaxis is consistent only for naratriptan [Mannix et al. 2007; Moschiano et al. 2005; Newman et al. 2001; Brandes et al. 2007], and frovatriptan [Silberstein et al. 2004, 2009; Brandes et al. 2009; Guidotti et al. 2007], while it is limited for sumatriptan [Newman et al. 1998], and zolmitriptan [Tuchman et al. 2006, 2008], and lacking for other triptans.
Main efficacy and safety endpoints of acute treatment studies reporting on conventional efficacy endpoints (i.e. pain relief, pain free, sustained pain free, and recurrence) are summarized in Table 2. Data reported in this table were indentified through a Medline database search from inception to May 2012. Retrieved publications were evaluated for relevance to this review. No formal meta-analytic approach was deliberately applied, and the review was kept intentionally descriptive.
Summary of studies evaluating efficacy according to traditional endpoints and safety of acute treatment of various triptans orally administered in menstrual migraineurs.

Drug-related AE.
AE, adverse event; CO, cross over; DB, randomized double blind; OL, nonrandomized open label; PC, placebo-controlled; PF, pain free; PG, parallel group; PR, pain relief; R, recurrence; SPF, sustained pain free.
All triptans seem to be effective in the acute treatment of MRM, with no significant difference in their efficacy in the treatment of menstrual versus nonmenstrual attacks. Also the trials using triptans for perimenstrual prophylaxis have suggested efficacy, particularly for naratriptan and frovatriptan. However, most of the studies were post-hoc analyses of efficacy trials, which were not originally specifically undertaken in women with MM, and in which treatment within or outside a specified temporal menstrual window is reported. Some trials evaluated MRM attacks, others PMM attacks. Some studies treated only moderate or severe attacks, while other studies also treated mild attacks. Heterogeneity in drug doses and treatment duration also limited the robustness of the interpretation of results.
Evidence from recent systematic reviews and meta-analyses of randomized trials supports a grade B recommendation for the use of sumatriptan 50 mg and 100 mg, or sumatriptan 85 mg plus naproxen 500 mg, or rizatriptan 10 mg for the acute treatment of MM [Pringsheim et al. 2008; Mannix and Files, 2005]. With respect to prophylaxis, there is grade A evidence of efficacy for short-term treatment with frovatriptan 2.5 mg twice daily, and grade B for naratriptan 1 mg twice daily [Silberstein et al. 2012]. In the case of preventive treatment, attention should be paid to avoid treatment overuse, which may defer attacks or even result in rebound migraine following withdrawal.
Pharmacologic features of frovatriptan
Frovatriptan is one of the newest agents of the triptan class. The drug was developed in order to provide a triptan with the clinical potential for a long duration of action, and a low likelihood of side effects and drug interactions [Balbisi, 2006]. The drug has distinctive pharmacologic features compared with other triptans, which make it particularly well suited to patients with prolonged migraines and those suffering migraine recurrence, such as patients with MM (Table 1) [Balbisi, 2006].
Frovatriptan has a potent affinity for 5-HT1B and 5-HT1D receptors, the highest among agents of the same class, and a higher selectivity for the cerebral vasculature, with minimal effects on coronary circulation [Comer, 2002]. The mean blood terminal deposition half-life of frovatriptan is approximately 26 h, the longest among agents within its class, and is not affected by gender, dose, or route of administration [Buchan et al. 2002]. In addition, due to the lack of inhibitory or inducing effects on cytochrome P450 isoenzymes, frovatriptan retains a low risk of drug interactions, and dosage adjustment is unlikely to be warranted. This is particularly relevant, since combination treatment of frovatriptan with analgesic drugs is sometimes required.
All these elements seem to contribute to the distinctive pharmacologic properties of frovatriptan, reflecting in particular clinical efficacy and safety in MM, summarized below.
Efficacy of frovatriptan in the prevention of MM: evidence from randomized controlled studies
Scheduled short-term preventive frovatriptan therapy effectively reduced the occurrence of MM in women with attacks occurring in association with menstruation in a number of randomized, controlled trials (Table 3).
Summary of randomized controlled and open-label studies evaluating preventive or acute efficacy of frovatriptan for the treatment of menstrual migraine.

CO, cross over; DB, randomized double blind; F, frovatriptan; MRM, menstrually related migraine; OCMM, oral contraceptive-induced menstrual migraine OL, nonrandomized open label; PG, parallel group; PMM, pure menstrual migraine.
In a randomized, double-blind, placebo-controlled, three-way cross-over study, 546 women treated three perimenstrual periods with placebo, frovatriptan 2.5 mg once daily, and frovatriptan 2.5 mg twice daily [Silberstein et al. 2004]. Treatment started 2 days before the anticipated start of MM headache and lasted for 6 days. Both frovatriptan regimens were superior to placebo, with a dose-dependent reduction in the incidence of migraine. Overall, the incidence of headache was 67% in subjects receiving placebo, 52% in those receiving frovatriptan 2.5 mg once daily, and 41% in those receiving frovatriptan 2.5 mg twice daily. Both active treatment regimens significantly (p < 0.001) reduced the incidence of all-severity headache, but also the incidence of moderate or severe headache (28–37% frovatriptan versus 51% placebo, p < 0.0001), duration (16.6–20.3 h frovatriptan versus 31.1 h placebo, p < 0.0001), and the use of rescue medication (34–44% frovatriptan versus 53% placebo, p < 0.01) compared with placebo. The incidence and type of adverse events were similar between active treatment (42.9% frovatriptan 2.5 mg once daily and 44.3% frovatriptan twice daily) and placebo (40.2%), and consistent with those reported for short-term migraine management.
More recently the same authors run a post-hoc analysis of the same study in a subgroup of 179 women with PMM [Silberstein et al. 2009]. Also in this sample of migraineurs, a preventive regimen with frovatriptan significantly reduced the occurrence of headache to 37.7% with frovatriptan 2.5 mg twice daily (p < 0.001 versus placebo), to 51.3% with frovatriptan 2.5 mg once daily (p = 0.002), and to 67.1% with placebo. A significant dose response was noted also in this case.
Treatment with frovatriptan 2.5 mg once or twice daily was associated with a significantly increased number of headache-free perimenstrual periods (0.92 and 0.69, p < 0.001 and p < 0.02, respectively) compared with placebo (0.42) in a double-blind, placebo-controlled, parallel-group trial, enrolling 410 women with difficult-to-treat MM [Brandes et al. 2009]. Frovatriptan also reduced migraine severity.
From all this evidence, a grade A recommendation for short-term treatment with frovatriptan 2.5 mg twice daily has recently been made [Silberstein et al. 2012].
Frovatriptan in the acute treatment of MM: evidence from randomized controlled studies
The efficacy of frovatriptan for the treatment of acute migraine attack has been recently assessed with that of other triptans in three different randomized, double-blind, cross-over studies. All studies had an identical design. Post-hoc analyses of such studies were recently published [Allais et al. 2012]; they were performed in women with MRM, defined according to International Headache Society criteria [Headache Classification Subcommittee of the International Headache Society, 2004]. Each patient received frovatriptan 2.5 mg or the comparator (rizatriptan 10 mg, zolmitriptan 2.5 mg, or almotriptan 12.5 mg) in a randomized sequence: after treating three episodes of migraine in no more than 3 months with the first treatment, the patient had to switch to the other treatment. Subjects were encouraged to treat one to three attacks for a maximum period of 6 months. Pain relief was defined as a decrease in migraine intensity from severe or moderate to mild or none at 2 h and 24 h, pain free as absence of migraine episodes at 2 h and 24 h after intake of one dose of the study drug, and recurrence as a pain-free episode at 2 h and headache of any severity returning within 24 h and 48 h. As shown in Figure 1, the rate of pain relief and pain free at 2 h and 24 h were not significantly different between frovatriptan and each individual comparator. In order to obtain pain free at 2 h the number of additional patients to be treated with frovatriptan was 33 versus rizatriptan, 25 versus zolmitriptan, and 10 versus almotriptan. Recurrence at 24 h (Figure 1), and even more so at 48 h (Figure 2) was always significantly lower with frovatriptan than with the other triptans. The numbers needed to treat to reduce the risk of recurrence at 24 h were favorable for frovatriptan (5 fewer than rizatriptan, 14 fewer than zolmitriptan, and 8 fewer than almotriptan). Similarities and differences between treatments were more striking when data were pooled (bottom panel of Figures 1 and 2). The numbers needed to treat to obtain pain free at 2 h were 14 versus comparators, while recurrence at 24 h was prevented treating 9 fewer patients with frovatriptan.

Frequency (%) of PR at 2 h and 4 h, PF at 2 h and 4 h, and R at 24 h in patients treated with frovatriptan (open bars) or rizatriptan, zolmitriptan, almotriptan or the three triptans pooled together (full bars). Data refer to three different double-blind, randomized, cross-over studies. p-values indicate the statistical significance of the between-treatment difference [Savi et al. 2011; Allais et al. 2011b; Bartolini et al. 2012]. PF, pain free; PR, pain relief; R, recurrence.
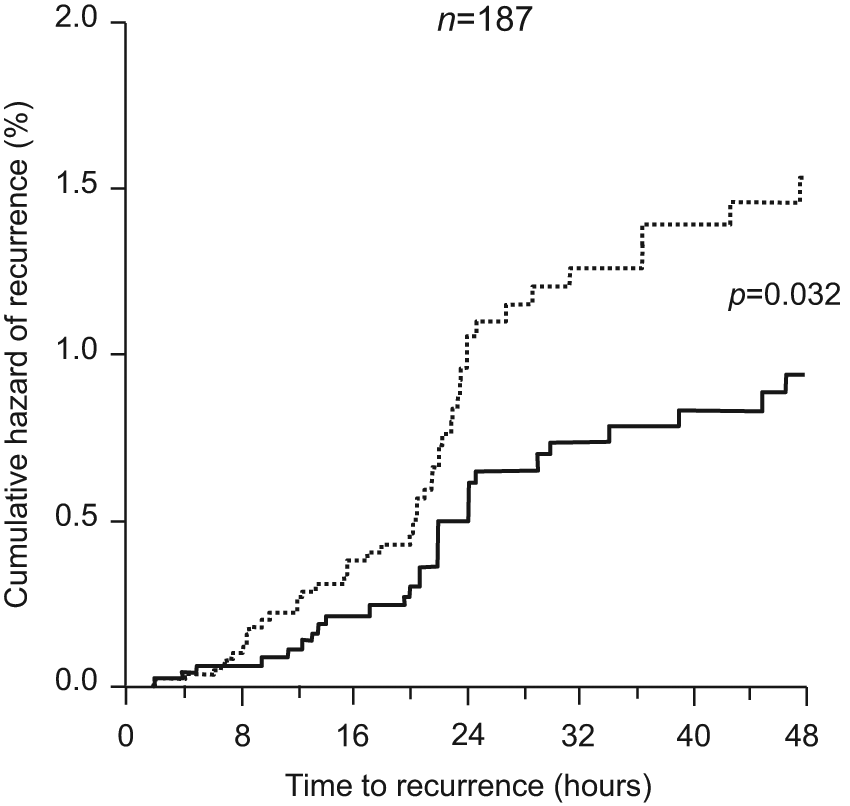
Cumulative hazard of recurrence over the 48 h of treatment with frovatriptan (continuous line) or comparators (dashed line) in 187 patients of three double-blind, randomized, cross-over studies. p-value refers to the statistical significance of the between-treatment difference [Allais et al. 2012].
Interestingly, the response to treatment with frovatriptan or the comparators was less potent in menstruating women than in the whole study population, confirming that MM attacks are particularly difficult to treat (Table 4).
Demographic and clinical characteristics and main efficacy parameters for the whole population of three randomized controlled studies comparing frovatriptan with rizatriptan, zolmitriptan, or almotriptan, and for the subgroup of women with menstrually related migraine (Allais et al. [2012]; Cortelli et al. [2011]).

NS, not significant; SD, standard deviation.
Efficacy of frovatriptan in MM: evidence from open-label studies
There are also some open-label studies confirming the efficacy of frovatriptan as preventive or acute treatment for MM attacks.
A small pilot, non-randomized, parallel group study compared the preventive efficacy of frovatriptan versus that of transdermal estrogens or naproxen in 38 women with MM [Guidotti et al. 2007]. Daily incidence of migraine was significantly (p = 0.045) lower under frovatriptan than under control treatments, this being particularly evident for the subgroup of patients with PMM (60% frovatriptan versus 79% estrogens and 78% naproxen sodium). During treatment the median score of headache severity was significantly (p = 0.049) lower under frovatriptan (2.5) than under estrogen (3.0) or naproxen treatment (3.9).
In an open-label study 151 menstrually capable patients were enabled to take up to three tablets of frovatriptan 2.5 mg over a 24-h period to treat an acute attack. Migraine relief within 24 h was achieved in 82% of menstrual attacks and 87% of nonmenstrual attacks. The time to overall relief was longer in menstrual attacks (5.5 h versus 3.6 h nonmenstrual attacks) [MacGregor et al. 2010].
Another open-label, uncontrolled pilot study, evaluated the efficacy of frovatriptan 2.5 mg in 20 women with oral contraceptive-induced MM [Allais et al. 2008]. Frovatriptan was administered during the 7 days off oral contraceptive at the onset of a moderate or severe migraine attack. A second dose could be taken if symptoms were alleviated but recurred within 24 h. After administration of frovatriptan the headache intensity progressively and significantly (p = 0.0001) decreased from 2.4 at onset to 0.8 after 24 h. A total of 10% of subjects were pain free at 2 h, 35% at 4 h, and 60% at 24 h, while pain relief was reported after 2 h by 55% of patients and by 75% after 4 h and 24 h. Headache recurred within 24 h in 36% of patients.
Finally we should mention an open-label postmarketing surveillance study conducted in 1931 women with MM and 2080 women with nonmenstrual migraine [Newman et al. 2009]. The study was a retrospective analysis of an acute treatment study. Previous medications were rated at baseline as good or very good for effectiveness by only 20% of menstrual migraineurs and 19% of nonmenstrual migraineurs, while the corresponding figure after frovatriptan treatment was 93% and 91%, respectively.
All the randomized controlled and open-label studies based on frovatriptan and described in the previous paragraphs were financially supported by the manufacturers of frovatriptan (Berlin-Chemie AG, Berlin, Germany; Endo Pharmaceuticals Inc., Chadds Ford, PA, USA; Vernalis Development Ltd.,Winnersh, UK; Istituto Lusofarmaco d’Italia, Milan, Italy).
Safety of frovatriptan in MM
Safety and tolerability of frovatriptan as short-term preventive therapy or acute treatment for MM have been extensively studied.
A pooled analysis of three studies including 1487 women with MM has evaluated the long-term safety of frovatriptan prophylactic treatment in up to 12 perimenstrual periods over a 12- to 15-month period [MacGregor et al. 2009; MacGregor et al. 2010]. Adverse events were reported in 58% of women treated with frovatriptan 2.5 mg twice daily, in 63% of those treated with frovatriptan once daily, and in 63% of those receiving placebo, with no between-group differences. There were no consistent differences in adverse events reported by patients with potential cardiovascular risk. In no cases was a dose relationship for adverse events observed. No cardiovascular safety issues resulted from repeated dosing, nor was there evidence of rebound migraine after withdrawal of frovatriptan at the end of the treatment period.
In the pooled analysis of the three direct comparative studies of frovatriptan versus other triptans, a total of 18 drug-related adverse events were recorded in 401 menstrually related attacks treated [Allais et al. 2012]. No statistically significant differences were observed in the rate of attacks associated with drug-related adverse events between frovatriptan (5%) and the comparators (4%).
Conclusion
Frovatriptan has been found in a number of trials to be effective in the acute treatment of MRM. In particular, direct comparative studies evaluating approximately 200 women with MM have shown a similar immediate antimigraine efficacy profile of frovatriptan, zolmitriptan, rizatriptan, and almotriptan. However, frovatriptan treatment was associated with a more sustained relief effect on migraine with lower headache recurrence rates over 24 h, and even more so over 48 h [Savi et al. 2011; Allais et al. 2011b; Bartolini et al. 2012]. Such differences might be explained, at least in part, by differences in the pharmacokinetics of frovatriptan in respect to the other triptans. Frovatriptan has a longer elimination half-life than rizatriptan, zolmitriptan, and almotriptan, possibly explaining why frovatriptan, unlike the other tested triptans, greatly reduced the risk of recurrence [Loder, 2010].
Frovatriptan given prophylactically for 6 days was effective in reducing the incidence of MM in approximately half of the 1100 women treated with the drug [Silberstein et al. 2004; Silberstein et al. 2009; Brandes et al. 2009; Guidotti et al. 2007]. These findings are consistent with the prolonged duration of action of frovatriptan that has been observed in studies of acute migraine treatment.
Unfortunately, most of the studies performed so far with frovatriptan in MRM share a post-hoc nature. However, their positive results encourage the design and implementation of large, double-blind, randomized, prospective clinical trials specifically evaluating frovatriptan efficacy in MRM.
Frovatriptan was as well tolerated as the other triptans, with a low incidence of drug-related effects. This reinforces evidence from previous placebo-controlled or head-to-head trials in non menstrually related migraine, namely that frovatriptan, used for immediate or repeated sustained use, is one of the best tolerated triptans [MacGregor et al. 2010; Cady et al. 2009; Géraud et al. 2002; Kelman et al. 2010; Poolsup et al. 2005; Wallasch, 2010].
In conclusion, MRM poses a unique set of challenges for the clinician, including the choice of the therapy. Among the variety of effective modalities and strategies available, frovatriptan represents an interesting therapeutic option. Its sustained effect and good tolerability profiles show considerable potential in the acute and prophylactic management of MRM.
Footnotes
Acknowledgements
Special thanks to Stefano Omboni, MD for his collaboration.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
GA has occasionally served as scientific consultant for the manufacturers of frovatriptan.
