Abstract
Introduction
Historical vignette
Intracranial aneurysms (IAs) account for the most challenging benign diseases of the human brain. Although they have been known since the early 1900s, aneurysms of the cerebral vessels were thought to be untreatable except for ligation of the carotid artery (Cushing). Yet, Cushing described in detail the ‘subarachnoid haemorrhage’ (SAH) and, furthermore, designed the ‘silver clip’ that was later used for the treatment of aneurysms [Cushing, 1911]. It was Dandy who first successfully clipped a cerebral aneurysm [Dandy, 1938]. He and Norman Dott pioneered the neurovascular era [Dott, 1969]. The implementation of the operating microscope into the operating theatre enabled neurosurgeons to diligently dissect the cerebral vessels and their pathologies. From the 1970s until the 1990s, Donaghy and Yasargil established one of the world’s largest series of microsurgically treated cerebral aneurysms of the anterior and posterior circuit [Donaghy and Yasargil, 1968]. One decade later Guglielmi and colleagues invented electrolytically detachable platin coils for endovascular thrombotic occlusion of aneurysms [Guglielmi et al. 1991a, 1991b]. Today, neurosurgeons and neurointerventionalists have further extended their therapeutic armamentarium with numerous clips, coils, stents, balloons, intraoperative fluorescence angiography and (intraoperative) biplanar digital angiography.
Recently, both neurosurgeons and neurointerventionalists are facing a growing number of innocent aneurysms of the cerebral vessels, incidentally found on magnetic resonance imaging (MRI) mainly performed for indications other than screening for aneurysms [Vernooij et al. 2007]. The statistical chance of revealing an aneurysm of the cerebral vessels in a routinely performed cerebral MRI is as low as 0.34% [Morris et al. 2009].
Epidemiology of intracranial aneurysms
The incidence of IAs is estimated to be between 1.8% and 2% [International Study of Unruptured Intracranial Aneurysms Investigators, 1998; Rinkel et al. 1998], a number that is supported by recent MRI investigations [Vernooij et al. 2007]. About 3–6% of the population harbour an IA according to angiographic and autopsy studies [Iwamoto et al. 1999; Menghini et al. 1998]. The wide range of prevalence of IAs might be due to the bias of population-based examinations, as there were great differences reported depending on the intent of the study [Rinkel et al. 1998]. They are more common in females, increase in frequency with age and are independently associated with nicotine abuse and alcohol, probably hypertension, obesity and the use of oral contraceptives [Juvela et al. 2000; Wardlaw and White, 2000]. Multiple aneurysms are found in 17–33.2% of patients sustaining a SAH [Sonobe et al. 2010; Inagawa, 2009; Lai et al. 2009; Kaminogo et al. 2003]. Interestingly, 19% of the first-degree relatives of patients with SAH are at risk of harbouring an aneurysm [Brown et al. 2008]. Familial inheritance of IAs is reported with 8.1% (without confounding disorders such as polycystic kidney disease), and it seems that these IAs tend to bleed more frequently than spontaneous aneurysms [Broderick et al. 2009; Ronkainen et al. 1997].
Incidence of SAH
From the International Study of Unruptured Intracranial Aneurysms Investigators (ISUIA) the yearly rupture rate for IAs was calculated to be as low as 0.05% [International Study of Unruptured Intracranial Aneurysms Investigators, 1998]. Recent publications have raised the risk figure to 1.3–1.4%, respectively [Ishibashi et al. 2009; Juvela et al. 2000]. Risk factors of SAH include the size of the aneurysm, patient’s age, hypertension and smoking [Juvela et al. 2000]. The risk of rupture of the aneurysms is calculated by the size and the aspect ratio (dome/neck ratio) [Steinmetz, 2008; Weir et al. 2003; Bederson et al. 2000]. Smaller aneurysms (<5 mm) account for 13% of SAH [Sonobe et al. 2010]. Prime target after SAH for the neurovascular specialists is to prevent rebleeding, which will occur in up to 9.7% within 21 days following haemorrhage and is associated with a mortality rate of 40% [Beck et al. 2006; Ross et al. 2002]. Owing to the increasing occurrence of vasospasm and ischaemia after day three following initial haemorrhage it is advised to occlude IAs within 72 hours following initial SAH [Seifert et al. 1988; Kassell and Torner, 1983]. In the case of multiple aneurysms it can be difficult to identify the aneurysm responsible for the bleeding. In CT scans haemorrhages might be difficult to be linked to a distinct aneurysm, if a close location of the aneurysms is given by their parent vessels’ proximity, e.g. tip of the basilar artery and posterior communicating artery.
Treatment modalities for intracranial aneurysms
The introduction of electrolytically detachable coils by Gugliemli and colleagues in 1991 has changed medical practice of the previously ‘state of the art’ surgical aneurysmal occlusion [Guglielmi et al. 1991a, 1991b]. The ISAT study (International Subarachnoid Aneurysm Trial) favoured endovascular intervention for IAs bearing a lower morbidity and mortality for the patients suffering from aneurysmal SAH when compared with the surgically treated patient group [Molyneux et al. 2002]. However, long-term follow up showed that endovascular occlusion of aneurysms is associated with a higher rate of regrowth and consecutive necessity of re-intervention [Molyneux et al. 2009]. Nevertheless, numerous studies have been published with the aim of comparing surgical and endovascular aneurysm treatment in order to investigate the superiority of one over the other [Klompenhouwer et al. 2011; de Oliveira et al. 2007; Fraser et al. 2006; Lanzino et al. 2006; Taha et al. 2006; Sandalcioglu et al. 2005; van der Schaaf et al. 2005; Hoh et al. 2004; Wanke et al. 2002].
Study philosophy
It is the goal of the present article to suggest a modern treatment philosophy of a specialized neurovascular centre that treats all neurovascular abnormalities and their complications (haemorrhage, hydrocephalus, etc.) interdisciplinarily 24 hours a day all year. The question of whether coiling or clipping is potentially better has been overcome, as it is our understanding that both treatment modalities are still necessary in a synergistic effort to achieve the best possible occlusion of an aneurysm. The modern treatment philosophy therefore must consist of an optimized interdisciplinary treatment that is tailored to each individual patient. In our personal experience, numerous highly difficult clinical conditions (multiple and regrowing aneurysms, etc.) may occur, which is why such a philosophy is necessary.
From our own data we show typical treatment algorithms for single and multiple aneurysms as well as for recurrent aneurysms. We try to provide thoughts and advices from our lessons learned how to approach these pathologies adequately. Our personal experience with various difficult conditions (namely: (1) late recurrent haemorrhages after aneurysmal occlusion; (2) de novo appearance of aneurysms; (3) incomplete occlusion of complex, broad-based aneurysms; and (4) the still growing experience with possible treatment algorithms for multiple innocent and/or symptomatic aneurysms) establish the basis for the present study, which is supported by a retrospective single-centre cross-sectional analysis of a consecutive series of 321 patients.
Methods
From September 1st, 2008, until December 31st, 2010, medical charts of all consecutive patients undergoing treatment for their IAs in our centre were reviewed by a cross-sectional analysis. Treatment modality (clip or coil) was discussed in all cases interdisciplinarily. In patients with multiple aneurysms, the treatment strategy and timing of aneurysmal occlusion was planned very carefully. All patients were clinically examined by a neurosurgeon prior to treatment. Patients were informed about the interdisciplinary tailored procedure by both a neurosurgeon and a neurointerventionalist. Informed consent was granted for elective procedures. In the case of acute haemorrhage the patient’s next of kin were informed about the treatment.
For all patients digital subtraction angiography (DSA) was the gold standard to decide treatment modality. Computed tomography angiography (CTA) was performed in patients with either thrombosed or partial calcified aneurysms, or if DSA could not be performed immediately in patients with SAH. In the latter, DSA was only suspended if intracranial mass lesions of an associated intracerebral or extra-axial haematoma required urgent surgical intervention (16 patients). Patients with occlusive hydrocephalus secondary to haemorrhage received an external ventricular drain prior to DSA.
Aneurysms of the posterior circulation were mainly submitted for endovascular treatment, especially those of the basilar artery. Aneurysms of the anterior circulation were analysed for their size, aspect ratio and probability of coiling the aneurysm without stenting.
For study purposes medical charts were analysed for allocation of aneurysms into anterior and posterior circulation, initial clinical conditions of the patient Hunt & Hess (HH), associated SAH/intracerebral haemorrhage (ICH) and multiplicity of IAs.
The occlusion of aneurysms was ascertained by DSA after 1 week for surgical interventions and after 6 months for endovascular-treated aneurysms. Of the clip ligated patients, 128 underwent re-angiography during their stay (94%). A total of 141 patients treated endovascularly could be followed at 6 months (75%). This lower rate is explained by the cross-sectional character of the present study, thus a higher rate at 6 months follow up is expected for the longitudinal analysis of the interventionally treated patients.
Results
Between September 2008 and December 2010 a total of 321 patients with cerebral aneurysms were treated consecutively. During this period 132 patients (41.1%) were clipped (with 151 aneurysms), and 189 individuals (58.2%) were designated for coiling (with 213 aneurysms) (Figure 1).
This histogram gives an overview on total number of patients, treatment modalities and subgroup distribution with regard to innocent intracranial aneurysms (IAs) and aneurysmal subarachnoid haemorrhage (SAH).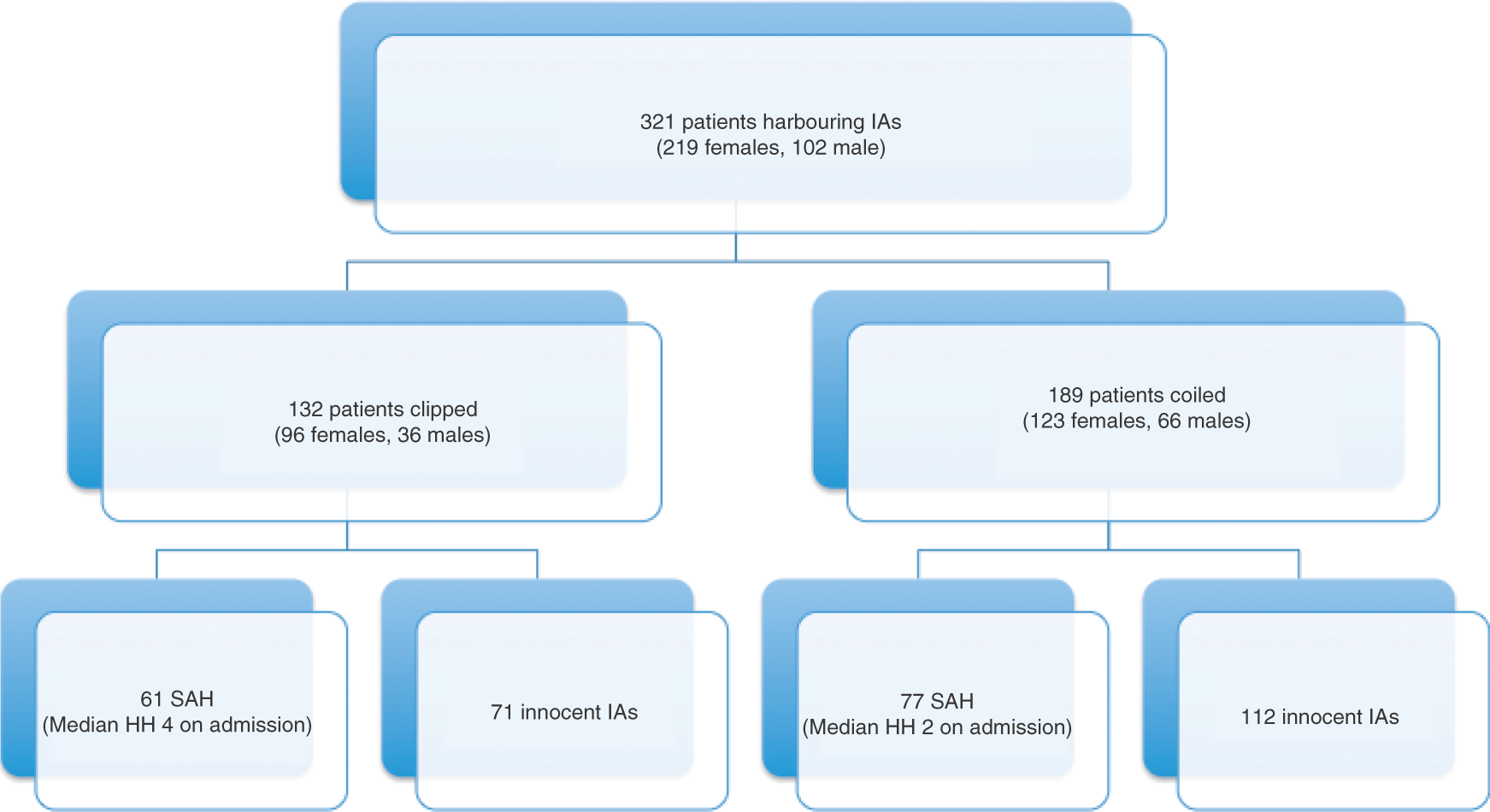
Multiple aneurysms
Individuals with multiple aneurysms were primarily treated for their symptomatic aneurysm. A total of 98 patients harbouring 266 multiple aneurysms (range 2–5 aneurysms/per patient) were treated for at least one of their aneurysms (30.2%). Seventy-one aneurysms of the 266 had been already treated prior to the study period. Of all multiple aneurysms, a proportion of 21.4% (n= 57/266 aneurysms) were submitted to continuous observation. Such a ‘conservative’ treatment was chosen if: (1) patients were suffering from a severe comorbidity; (2) if the aneurysms were localized extradurally; (3) if the aneurysms were very small (<5 mm, see comment below); or (4) if the patients declined further treatment. We recommended treatment to all patients in a good clinical state with additional innocent aneurysms larger than 3 mm.
Treatment data
In the ‘clip group’, aneurysms were allocated in the anterior circulation in 130/132 cases (98.5%), whereas 147/213 aneurysms of the ‘coil group’ were located in the anterior circulation (69.0%). The middle cerebral artery (MCA) dominated with 88/132 cases, followed by aneurysms of the anterior communicating artery (AcomA, n = 31) and the internal carotid artery (ICA, n = 26).
Patients submitted for coiling harboured mostly ICA aneurysms (n = 64), followed by aneurysms of the AcomA (n = 50). Aneurysms of the basilar artery (trunk and tip) were treated in 41 cases and MCA aneurysms in 38 cases. In 42 procedures, the occlusion of the aneurysm was secured by the additional use of endovascular stents.
Aneurysmal subarachnoid haemorrhage
In the subgroup of patients with SAH (n = 138), 61 (44.2%) were treated surgically, whereas 77 (55.8%) underwent endovascular intervention. Patients undergoing surgery were in a poorer clinical condition (HH median 4) when compared with patients allocated to a coiling procedure (HH median 2) which was statistically not significant (Student’s t-test, H0, p = 0.001). Furthermore, these patients presented more often with multiple aneurysms (31.7% in the clip group versus 28.6% in the coil group).
In patients treated surgically, SAH was mainly caused by ruptured MCA aneurysms (n = 34; 56.7%), whereas patients receiving interventional occlusion of their aneurysm suffered predominately from Acom aneurysms (n = 32; 41.6%).
The outcome on cross-sectional analysis did not differ significantly between the two groups Glasgow Outcome Scale (GOS median 3 vs. 3.5, respectively, in the group of coiled patients). The mortality rate was equal in both groups (GOS 1; eight patients vs. nine patients in the coil group)
Recurrent aneurysms
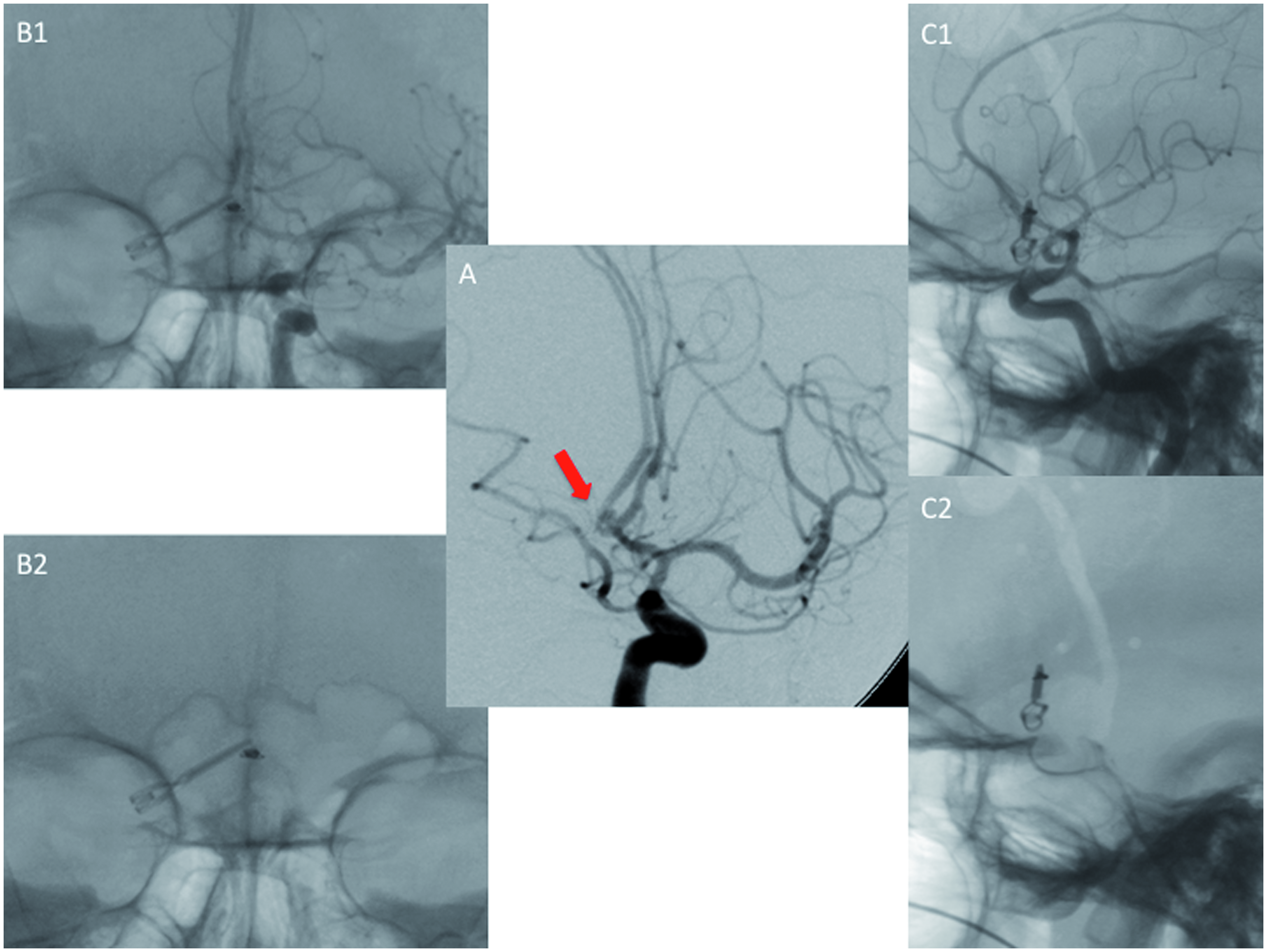
A total of 43 recurrent aneurysms were treated during the study period (34 coiled, 9 clipped). Of these, only 12 aneurysms were initially occluded between September 2008 and December 2010. Eight endovascularly occluded aneurysms (4.2%) recurred at the 6-month follow-up DSA, and were considered for re-intervention. Four surgically occluded aneurysms (2.9%) were identified with a significant rest at the aneurysm base on the postoperative DSA requiring re-intervention. All of these latter were initially treated without DSA due to an accompanying ICH and impending uncal herniation. Special attention was givento ‘cross-over’ aneurysms, aneurysms previously treated by clip or coil, changing procedure modality for their recurrence. These aneurysms were identified by the posttreatment routine DSA. Eight aneurysms, previously coiled, were clipped after regrowth that was diagnosed on follow-up imaging (Figure 2). Five patients, previously clipped, underwent endovascular intervention for their recurrent aneurysm (Figure 3).
Cross-over aneurysm of the anterior communicating artery (AcomA): after clipping, an incomplete occlusion of the aneurysmal base was diagnosed (A, bold arrow depicting the reminiscent aneurysm); the aneurysm could be occluded interventionally without stents (B1/B2, frontal view after coil embolization; C1/C2, lateral view after successful occlusion with the coils projecting underneath the clip).
Discussion
Increasing incidence of aneurysms?
The incidence of innocent cerebral aneurysms seems to be increasing over the past few decades due to the wide availability of MRI. Yet, the trend of increasing interventions for unruptured aneurysms cannot be verified by prospective studies so far. Vernooij and colleagues found an incidence of 1.8% in 2000 scanned people, which is equal to the prevalence estimated from the ISUIA study [Vernooij et al. 2007; International Study of Unruptured Intracranial Aneurysms Investigators, 1998]. MRA sensitivity is estimated to be as high as >95% for aneurysms >6 mm, with rapidly decreasing sensitivity for the reduction of aneurysmal size (60% sensitivity for aneurysms <5 mm) [Atlas et al. 1997; Korogi et al. 1996]. Local resolution of tomographic imaging techniques does not grant assured detection of aneurysms smaller than 3 mm in size [Forsting, 2005]. Today, widespread employment of MRI for preclinical diagnostic use will lead to the detection of more unruptured aneurysms. As for diagnostic use, CTA is a very useful tool for preoperative evaluation of atherosclerotic plaques at the base of the aneurysms, which might be missed by DSA. In particular, in large aneurysms with a broad-neck CTA is helpful in planning surgical or endovascular occlusion. Furthermore, the latest generation of CT scanners (Dual Source CT) create less artefacts caused by clips applied to aneurysms. Thus, if a radiological follow up is required (e.g. remnant base after clipping) these scanners might replace DSA for follow up, favouring its reduced invasiveness and fewer procedural complications.
Furthermore, routine high-resolution posttreatment DSA evaluation as performed in the present study additionally increases the number of detected incompletely occluded aneurysms.
The increasing numbers are reflected in Figure 4, which depicts registered cases of aneurysmal haemorrhage (blue line) and of unruptured aneurysms (red line) in Germany from 2000 to 2009. While the cases of SAH due to ruptured IA rather decreases over the last decades, the total number of unruptured IA treated increases from about 2500 to nearly 6000. It can only be speculated whether this trend is set by optimized coding of DRGs in the medical sector or whether it truly reflects a reduction of SAH due to early detection of IA prior to eventual rupture.
The total number of registered cases of both aneurysmal haemorrhage (ICD 10 of I60.x and I67.1; blue line) and unruptured aneurysms (red line) in Germany from 2000 to 2009. Note that even though the absolute number of SAH is decreasing, the frequency of newly diagnosed, unruptured aneurysms raises over the last decade (data source: Federal Statistical Office, branch Bonn).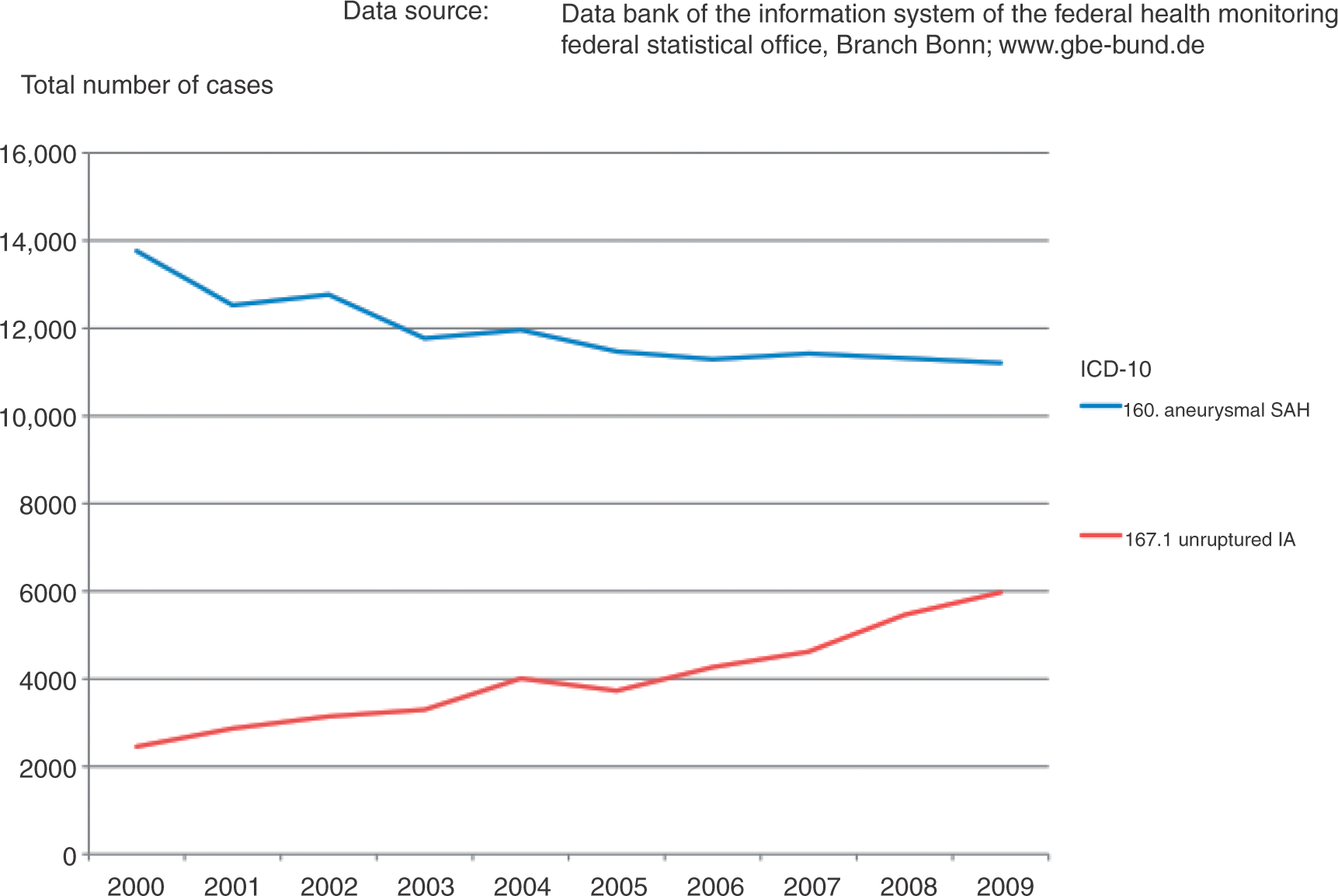
The incidental diagnosis of IA in a MRI of the neurocranium for reasons other than vascular pathologies is 0.34% from data in the literature [Morris et al. 2009; Vernooij et al. 2007]. Furthermore, familial aneurysm syndromes have been described, leading to a more frequent screening of relatives of patients suffering from cerebral aneurysms [Brown et al. 2008; Wills et al. 2003; Ronkainen et al. 1997; Lozano and Leblanc, 1987].
Treatment recommendations
Recommendations for the treatment of unruptured aneurysms have been given with regard to the size (≥7 mm) and with respect to the patient’s age [Steinmetz, 2008; Raabe et al. 2003; Bederson et al. 2000]. Calculating an annual bleeding risk for approximately six to 15 patients out of 100,000 people per year we can estimate about 4800 to 12000 SAHs in Germany each year [Rinkel et al. 1998; van der Schaaf et al. 2005]. The commonly used code (International Classification of Disease [ICD10]) I60.x resembles a SAH otherwise not specified. It was charted nearly 15,000 times in 2010 (Information System of the Federal Health Observation; Federal Statistical Office, branch Bonn, Figure 4).
Treatment strategies
ISAT shifted treatment of aneurysms towards coiling due to the superior short-term outcome of coiled patients with a favourable morbidity and mortality [Molyneux et al. 2002]. In further trials the excellent data from ISAT were reproduced, especially for aneurysms of the posterior circulation [van der Schaaf et al. 2005]. In the mid- to long-term follow-up of coiled patients from ISAT, it was found that the rate of recurrence of previously sufficiently coiled aneurysms was increasing over the years [Molyneux et al. 2005]. Thus, this lead to the recommendation that IAs of the anterior circulation in young patients (<40 years) should be suggested for clipping if applicable [Molyneux et al. 2009; Mitchell et al. 2008]. Aneurysms of the posterior circulation, in particular aneurysms of the basilar artery, are generally accepted to be treated preferably by coiling. Yet, recent studies still outline the importance of surgical occlusion of these aneurysms, if an endovascular approach is limited by the aneurysmal configuration [Sandalcioglu et al. 2005; Ogilvy et al. 2002]. From our data we show that it is important to offer both treatment modalities, clipping and coiling, to the patients. Of 321 patients presenting to our centre more than 40% required surgical intervention, as their aneurysms were not suitable for coiling for various reasons, regardless of whether an unfavourable base/dome ratio or an accompanying mass lesion accounted for surgery. In elective cases, the decision for treatment has to be made deliberately to prevent any harm to the patient that may occur by withholding one of the treatment options.
In an emergency situation (e.g. with SAH) it seems advisable that a neurosurgeon and a neurointerventionalist are neurointerventionalist, both, are available for a prompt treatment decision. In our series, nearly 56% of aneurysms causing SAH could be treated endovasculary, leaving 44% aneurysms for surgery. In particular, MCA aneurysms are prone to surgery as they tend to present more frequently with accompanying ICH and with a larger base/neck ratio so are less suitable for coiling. In any case, all patients suffering from SAH should be treated for occlusion of their aneurysm within the first 72 hours, because such undelayed treatment lowers the risk of early rebleeding, yet avoiding the time onset of vasospasms and ischaemia [Seifert et al. 1988]. A flowchart for treatment decisions is suggested in Figure 5, with all of the limitations that a simplified histogram brings.
A simplified flowchart is suggested dividing decision making on IAs into two categories. (i) Innocent intracranial aneurysms (IAs) subgrouped into single and multiple IAs; an interdisciplinary discussion should occur as to whether coiling or surgical ligation would be most suitable for aneurysmal occlusion is mandatory. Potential complications and presumed morbidities (surgical approach, thromboembolic complication by remodelling or stenting) are not embedded in the flowchart, even though they have to be discussed in detail prior to intervention. (ii) The subgroup of aneurysms presenting with SAH; similar to the situation for innocent aneurysms, an interdisciplinary evaluation of the best treatment option should occur. Any treatment option should be carried out within 72 hours to minimize the risk of an early re-bleeding and avoiding the time of onset of vasospasms and ischaemia. In the case of an accompanying haemorrhagic mass lesion intra- or extra-axially with impending uncal herniation, urgent surgical intervention remains the only treatment option and is, therefore, not embedded in the histogram.
Single versus multiple aneurysms
Multiplicity of aneurysms occurs in 17–32% of all cases [Juvela et al. 2000; Rinkel et al. 1998; Rinne and Hernesniemi, 1993]. In the case of multiple aneurysms and SAH, identification of the ruptured aneurysm is difficult, even more so if the aneurysms are located close to each other, e.g. ICA and the anterior cerebral artery (ACA) or proximal M1 aneurysm of the same side. The CT scan might be helpful in this regard, if the distribution of blood clots is suggestive for one particular aneurysm (Figure 6). Challenging situations are characteristically given if (1) the side of the bleeding cannot be located or (2) multiple aneurysms are diagnosed that cannot be treated in one single procedure (either clip or coil). In the latter situation one or more aneurysms may require complex endovascular occlusion (stenting or a balloon remodelling technique) requiring an intensive and prolonged anticoagulation therapy.
Initial CT scan showing SAH in the quadrigeminal cisterns (B), DSA elucidating an aneurysm of the pericallosal artery (C, bold circle) and a second aneurysm of the right MCA (D); the blood clot in the interhemispheric fissure (A, bold circle) was highly suspicious of a bleeding from the pericallosal aneurysm.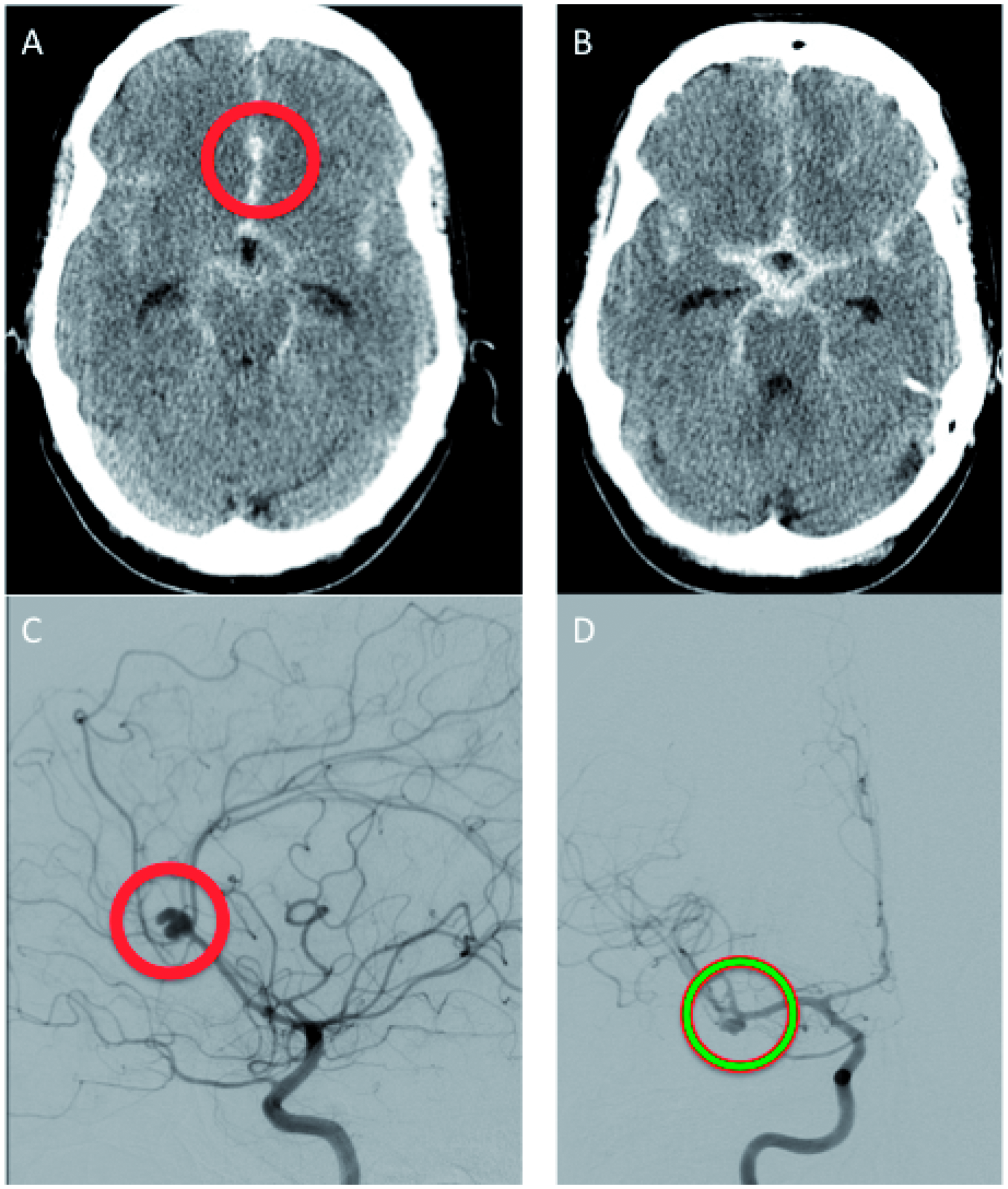
In these patients in particular, a very close interdisciplinary cooperation is imperative. Timing and timely scheduled order of surgical and interventional aneurysm occlusion is mandatory. As shown in Figure 7, a deliberate decision may need to be made that an initially incomplete occlusion of the aneurysm without stenting (the basilar artery aneurysm was accused of having caused the SAH) should be performed. Such a preplanned strategy allows the surgical treatment of the interventionally untreatable MCA aneurysms. Treatment algorithms based on such diligent and prospective thoughts are mandatory for all patients with multiple aneurysms in order to offer the occlusion of all aneurysms being at risk for SAH.
Massive subarachnoid haemorrhage (SAH) in the quadrigeminal cisterns (A); digital subtraction angiography (DSA) revealing at least five aneurysms: basilar artery (D), left posterior communicating artery (PcomA) (C) and right middle cerebral artery (MCA) (B, two aneurysms: M1 and M2 segment); the distribution of blood was indicative of either basilar artery aneurysm or PcomA aneurysm as bleeding source; after discussion the basilar and PcomA aneurysms were interventionally occluded without remodelling or stenting, leaving the MCA aneurysms for second stage surgery (lower row: left, frontal view of loosely packed aneurysm of the superior cerebellar artery and dense packing of the basilar tip aneurysm; right, lateral view, bold arrows pointing to the coil-occluded aneurysms of the internal carotid artery [ICA] and basilar artery).
Recurrent aneurysms
Aneurysms tend to recur, especially if they are incompletely occluded or if they are localized at branching vessels with specific mechanical strength/shear stress, e.g. carotic bifurcation, ACA/AcomA complex and MCA bifurcation [Schaafsma et al. 2009; Wermer et al. 2005; Tsutsumi et al. 2001]. We identified a total of 34 recurrent aneurysms that were previously coiled. Nine aneurysms that were clip ligated recurred. Of these, four were identified by early postoperative DSA bearing a remnant base requiring re-intervention. Of the recurrent aneurysms, 12 aneurysms were suggested for ‘cross over’. This means that the recurrent aneurysm was switched to the other treatment modality after interdisciplinary discussion. Decisive for a ‘cross over’ is either: (1) the feasibility of placing a clip over the regrown base of an aneurysm (Figure 2) or (2) the coiling (and stenting) in a previously clipped aneurysm (Figure 3).
Treatment decisions on the recurrent aneurysm should be performed in an interdisciplinary fashion just like in de novo aneurysms and require an estimation of the individual treatment risk (morbidity, mortality) as well as the risk of a ‘conservative’ treatment [Krisht et al. 2006; Wanke et al. 2002]. Such counterbalancing of risks should be performed in consultation with the patient. If the compacted coils allow for clip placement, it might be prudent to reconstruct the wall of the vessel by clip (Figure 2), since clip occlusion is less often associated with aneurysm regrowth when compared with coil occlusion. On the other hand, revision surgery of an (incompletely clipped or regrown) aneurysm is usually more difficult (and more dangerous) than primary surgery due to the usual perivascular scar formation. In particular, if an applied clip in a patient with aneurysm reperfusion prohibits sufficient visualization of the aneurysm base, it might be safer to recommend an interventional therapy.
Conclusion
Nowadays, both surgical and interventional occlusion of cerebral aneurysms are performed successfully. The advanced imaging techniques lead to a more precise and thus more frequent diagnosis of cerebral aneurysm. Such an increase in the precision of diagnostics means that patients suffering from recurrent and/or multiple aneurysms present more frequently. For these patients, an individually tailored interdisciplinary treatment plan should be delineated.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
