Abstract
Background:
Endoscopic variceal treatment (EVT) in cirrhotic patients carries a risk of periprocedural bleeding, making anticoagulation management challenging in those with acute portal vein thrombosis (PVT).
Objectives:
The first randomized controlled trial to evaluate the safety and efficacy of continuous versus intermittent anticoagulation during EVT in cirrhotic patients with acute PVT.
Methods:
In this trial, cirrhotic patients with acute PVT and esophageal varices requiring EVT were assigned to either a continuous or intermittent anticoagulation group. In the continuous anticoagulation group, anticoagulation began immediately after PVT diagnosis and continued during EVT. In the intermittent group, anticoagulation was resumed 1 week post-procedurally. Incidences of post-procedural bleeding and hepatic decompensation events were observed during a 1-year follow-up period. Potential confounders were adjusted for using multivariable binary logistic regression.
Results:
Among 120 enrolled patients, 112 were analyzed (continuous anticoagulation group, n = 55; intermittent anticoagulation group, n = 57). The rate of early rebleeding within 6 weeks did not differ between groups (p > 0.99). However, continuous anticoagulation significantly reduced the incidence of late rebleeding (p = 0.037) and all-cause bleeding (p = 0.020). The cumulative incidences of late rebleeding (hazard ratio (HR) = 0.386, 95% confidence interval (CI): 0.150–0.994; log-rank p = 0.041) and all-cause bleeding (HR = 0.396, 95% CI: 0.173–0.904; log-rank p = 0.023) were significantly lower in the continuous anticoagulation group. The continuous anticoagulation group had significantly lower incidences of hepatic decompensation events (p = 0.006), and ascites (p = 0.015). After adjustment for liver function, indications for EVT, and history of splenectomy, continuous anticoagulation remained significantly associated with superior outcomes compared with intermittent anticoagulation.
Conclusion:
Continuous anticoagulation during EVT in cirrhotic patients with acute PVT is safe and does not increase early rebleeding risk. It significantly reduces late and overall bleeding events and may improve long-term outcomes.
Trial registration:
Chinese Clinical Trial Registry, ChiCTR1800017430.
Plain language summary
Liver cirrhosis patients can simultaneously suffer from both esophageal varices and portal vein thrombosis (PVT) as complications. Previous guidelines recommended that anticoagulation therapy should only be initiated after complete resolution of esophageal varices in cirrhotic patients with varices and PVT. However, increasing evidence supports the safety of starting anticoagulation therapy before the varices have been fully resolved. The discontinuation protocol for anticoagulation during endoscopic variceal treatment remains unclear. This study investigated the impact of continuous versus intermittent anticoagulation (i.e., resuming anticoagulation one week post-procedurally) during endoscopic treatment of esophageal varices on bleeding and liver decompensation events. The results showed that continuous anticoagulation, compared to intermittent anticoagulation, did not increase the early rebleeding rate and had a potential benefit in reducing the late rebleeding and all-cause bleeding events at 1 year post-procedurally. Additionally, continuous anticoagulation reduced the occurrence of liver decompensation events other than bleeding, particularly improving ascites. This is the first study to compare continuous and intermittent anticoagulation during endoscopic variceal treatment in patients with acute PVT in cirrhosis, and the findings suggest that continuous anticoagulation is safe and effective.
Introduction
Portal vein thrombosis (PVT) and esophageal variceal bleeding (EVB) are common and serious complications of cirrhosis. The incidence of esophagogastric variceal bleeding (EGVB) accompanied by PVT in cirrhotic patients has been reported to be 24.1%–32.5%. The presence of PVT aggravates portal hypertension, thereby increasing the risk of variceal bleeding.1,2 Anticoagulation therapy is generally considered safe and effective for the management of PVT in cirrhosis.3–5
According to the guidelines of the British Society of Gastroenterology and the European Society of Gastrointestinal Endoscopy, low-molecular-weight heparin (LMWH) bridging is recommended for patients at high risk of thrombosis who undergo high-risk endoscopic procedures, such as endoscopic variceal treatment (EVT), whereas anticoagulation may be temporarily withheld in those with lower thrombosis risk. 6 Early initiation of anticoagulation after the onset of PVT facilitates portal vein recanalization;7,8 however, in cirrhotic patients requiring EVT, the optimal management of anticoagulation remains uncertain and clinically challenging.
According to the American Association for the Study of Liver Diseases 9 guidelines, anticoagulation can be initiated before complete eradication of varices in cirrhotic patients with PVT. Previous studies mainly compared outcomes between patients receiving anticoagulation and those who did not, demonstrating that anticoagulation does not increase the risk of post-procedural rebleeding and may improve liver function.10,11 However, no study has directly compared continuous versus interrupted anticoagulation during the entire course of EVT. In addition, evidence remains limited by relatively short follow-up durations, with the longest reported follow-up being only 6 months. 10 To address this issue, we conducted a randomized controlled trial comparing continuous anticoagulation throughout EVT versus intermittent anticoagulation resumed 1 week after EVT in cirrhotic patients with acute PVT. All patients were prospectively followed for 1 year to evaluate the impact of these two anticoagulation strategies on post-procedural bleeding and hepatic decompensation.
Patients and methods
Patients
This study was reported in accordance with the CONSORT 2025 statement (Supplemental Material). 12 This single-center, randomized, open-label trial was conducted between January 2019 and May 2025 at the Department of Gastroenterology, First Affiliated Hospital of Anhui Medical University. The study protocol was approved by the Institutional Ethics Committee (Approval No. PJ2018-07-18) and conducted in accordance with the Declaration of Helsinki (2013). Written informed consent was obtained from all participants. This study represents a secondary analysis of a registered clinical trial (ChiCTR1800017430), which was originally designed to evaluate the effects of different endoscopic treatments for esophageal varices on post-procedural bleeding and hepatic decompensation. During the course of the trial, a substantial proportion of enrolled patients were identified as having acute PVT. Given the limited evidence regarding the optimal timing of anticoagulation initiation in this population, a prespecified secondary analysis focusing on this subgroup was performed. All patient-identifiable information in the text, tables, and figures was completely removed to ensure confidentiality.
Eligible patients met the following criteria: (1) age 18–80 years; (2) diagnosis of cirrhosis based on clinical, laboratory, and imaging findings; (3) acute PVT, requiring early anticoagulation 13 ; and (4) acute EVB or secondary prophylaxis for EVB, requiring EVT.
Exclusion criteria included: (1) poor overall health, including uncontrolled active bleeding, multiple organ failure, grade III–IV hepatic encephalopathy (HE) and malignant tumors; (2) cirrhosis caused by venous occlusion or thrombosis-related diseases, such as Budd-Chiari Syndrome; (3) coagulation disorders unrelated to cirrhosis, such as myeloproliferative diseases, and hereditary thrombophilia, hemophilia; (4) platelet count below 30 * 10^9/L; (5) serum creatinine level above 170 μmol/L; (6) poor compliance.
Randomization
Patients were continuously and randomly allocated (1:1) to either the continuous or intermittent anticoagulation group using computer-generated random numbers (Research Randomizer). Following enrolled screening, a research coordinator entered the baseline data of the patients into the allocation system. The system generated a unique study identification number along with a corresponding treatment number for either continuous anticoagulation or intermittent anticoagulation. This study was an open-label trial with blinded outcome assessment, in which the researchers responsible for performing the endoscopic and anticoagulant treatments were aware of the patient’s group allocation, while the researchers responsible for outcome assessment and data analysis remained blinded to the allocation.
As no prior data were available to estimate the effect size for long-term outcomes for continuous anticoagulation, a pilot study was first conducted, showing 1-year all-cause bleeding rates of 15.0% in the continuous group and 38.5% in the intermittent group. Based on these results, the required sample size was calculated using PASS 15.0 software (NCSS, LLC, Kaysville, UT, USA), with α = 0.05 and 80% power. A minimum of 52 patients was required in each group. Allowing for 10% attrition, at least 58 patients were needed per group. Based on this, we planned to recruit 60 patients per group.
Procedures
Endoscopic treatment
EVT was performed by two experienced endoscopists, each with 10 years of experience, including endoscopic variceal ligation (EVL) and endoscopic injection sclerotherapy. EVs are classified as F1, F2, and F3. 14 The choice of appropriate endoscopic treatment method was based on the appearance of varices, with EVL applied to EVs F1 and F2, and a modified endoscopic injection sclerotherapy technique with balloon-occluded esophageal variceal obliteration used for F3. 15 Because of concerns about potential progression of PVT, 16 nonselective β-blockers were not administered to any patients during the study follow-up period.
Anticoagulation protocol
Anticoagulation was administered for 6 months or until PVT recanalization.
Continuous anticoagulation group
Anticoagulation was maintained throughout the peri-endoscopic period.
Intermittent anticoagulation group
Anticoagulation was postponed or paused around the procedure and resumed 1 week post-procedurally.
The anticoagulant used 1 week before and after the endoscopic treatment was LMWH, with no anticoagulation administered within 12 h before and after the procedure. LMWH (enoxaparin; Nanjing Jianyou Biochemical Pharmaceutical Co., Ltd, Nanjing, China) was injected subcutaneously at 4000 U every 12 h during the first month, followed by oral warfarin or once-daily LMWH. Warfarin was initiated 5 days before discontinuing LMWH, with dose adjustments to maintain the international normalized ratio (INR) of 2–3. Anticoagulation was temporarily withheld in cases of bleeding until hemostasis was achieved for 24 h.
Follow-up
All patients were followed for 1 year after EVT or until death. Follow-up visits were scheduled monthly. The first visit included repeat endoscopy; if residual varices were identified, additional sessions were performed. For patients on warfarin, INR was checked weekly during the first month, then monthly. Liver function tests and Doppler ultrasound of the portal system were performed every 3 months. Bleeding events within 1 year after endoscopy were recorded, including time, manifestations, cause, and D-dimer, INR, prothrombin time, as well as the occurrence of other hepatic decompensation events.
Endpoints and definitions
The primary endpoint was rebleeding from esophagogastric varices (EGVs), defined as recurrent melena or hematemesis due to EGVs requiring hospitalization, transfusion, ⩾3 g/L hemoglobin drop, or resulting in death within 6 weeks post-procedure. 17 Early rebleeding occurred within 6 weeks; late rebleeding occurred thereafter. 18
Secondary endpoints included all-cause bleeding and hepatic decompensation events. Hepatic decompensation events included ascites, infections, HE, hepatic hydrothorax, hepatocellular carcinoma, and other cirrhosis complications, as well as death. Infections comprised respiratory tract infections, urinary tract infections, and spontaneous bacterial peritonitis.
Acute PVT was diagnosed based on imaging evaluation performed prior to EVT. The diagnostic criteria for acute PVT were as follows: (1) PVT confirmed by Doppler ultrasound, computed tomography, or magnetic resonance imaging; (2) presence of acute symptoms, including abdominal pain, fever, nausea, diarrhea, or rapid worsening of ascites, with onset within 60 days; and (3) imaging features consistent with acute thrombus formation and excluding chronic PVT. In this study, patients with imaging findings suggestive of chronic PVT—including cavernous transformation of the portal vein, thrombus calcification, well-established collateral circulation, or long-standing portal vein narrowing and fibrotic changes—were excluded first. On ultrasound, acute PVT typically appears as heterogeneous intraluminal material associated with portal vein dilatation, and the thrombus may be isoechoic or hypoechoic, sometimes resembling the blood-flow signals. On computed tomography, acute PVT is characterized by slightly increased attenuation on non-contrast images and absence of enhancement after contrast administration, occasionally accompanied by arterial-phase hyperperfusion and portal-phase hypoperfusion in affected hepatic segments. On magnetic resonance imaging, acute PVT appears iso- to mildly hyperintense on T1-weighted images and hyperintense on T2-weighted images, without enhancement after contrast administration; peripheral enhancement may be observed in some cases. 19 In this study, clinical symptoms, symptom duration, and imaging findings were integrated to accurately identify patients with acute PVT as accurately as possible. Classification of PVT is defined according to the Yerdel classification system. 3
Statistical analysis
Data were analyzed using IBM SPSS Statistics 27.0 and R version 4.4.1. Continuous variables were expressed as mean ± SD or median (interquartile range) and compared using Student’s t test or Mann–Whitney U test, respectively. Categorical variables were expressed as counts (%) and compared using Chi-square or Fisher’s exact test. Ordinal data were analyzed with the Wilcoxon signed-rank test. Kaplan–Meier analysis was used to estimate cumulative bleeding rates, and comparisons were made with the log-rank test, with hazard ratio (HR) and 95% confidence interval estimated by Cox proportional hazard model. A p value <0.05 was considered statistically significant. Potential confounders, including liver function, indications for EVT, and history of splenectomy, were adjusted for using multivariable binary logistic regression.
Results
Baseline characteristics
A total of 162 patients met the inclusion criteria, of whom 42 were excluded. Thus, 120 patients were enrolled and randomized. After accounting for patients who discontinued anticoagulation or were lost to follow-up, 112 patients were included in the final analysis, with 55 in the continuous anticoagulation group and 57 in the intermittent anticoagulation group (Figure 1).

Flowchart of patient inclusion.
Baseline characteristics were comparable between groups, with no statistically significant differences (Table 1).
Baseline characteristics of the study groups.

Categorical data are presented as frequencies (%), normally distributed continuous data are expressed as mean ± standard deviation (x¯ ± SD), and non-normally distributed continuous data are presented as median (interquartile range).
CRE, serum creatinine; DD, D-dimer; EIS, endoscopic injection sclerotherapy; EVL, endoscopic variceal ligation; EVT, endoscopic variceal treatment; Hb, hemoglobin; INR, international normalized ratio; MELD, model for end-stage liver disease; PHG, portal hypertensive gastropathy; PT, prothrombin time.
Endoscopic treatment
In this study, acute PVT was diagnosed based on imaging evaluation performed prior to endoscopic treatment for esophageal varices. The interval between acute PVT diagnosis and EVT did not differ significantly between the continuous anticoagulation group and the intermittent anticoagulation group (2 (2–3) vs 2 (2–3) days, p = 0.565).
Anticoagulation regimens
In the continuous and intermittent anticoagulation groups, 87.3% and 86.0% of patients, respectively, received LMWH monotherapy, while the remainder underwent LMWH-to-warfarin sequential therapy. The distribution of anticoagulation regimens did not differ significantly between groups (p = 0.839). During the anticoagulation treatment period, in the intermittent anticoagulation group, 9 patients required 1 endoscopic procedure, 22 required 2, 25 required 3, and 1 required 4, with a mean of 2.31 ± 0.76 procedures per patient.
Post-procedural bleeding
Bleeding severity
The mean follow-up duration was 390.0 (380.0–400.0) days for the continuous anticoagulation group and 391.0 (384.5–400.0) days for the intermittent anticoagulation group, with no significant difference (p = 0.679). During the 1 year post-procedurally, 27 patients (24.1%) experienced bleeding events. One patient (0.9%) in the intermittent group died from circulatory failure following massive hematemesis on post-procedural day 91. This patient had a baseline Child–Pugh score of 9 with heart failure and pulmonary infection. Five patients (4.5%) developed hemorrhagic shock on post-procedural days 41, 56, 70, 87, and 240; three were due to EVB, one to portal hypertensive gastropathy (PHG), and one to rectal ectopic varices. Eleven patients (9.8%) experienced bleeding events that required transfusion.
The severity of bleeding was assessed according to clinical manifestations, including melena, hematemesis or hematochezia, shock, and death, with severity increasing in this order. No significant differences were observed between the two groups in bleeding severity (p = 0.622), transfusion requirements (3 (37.5%) vs 8 (42.1%), p > 0.99), or hemoglobin levels (86.75 ± 28.33 vs 71.68 ± 22.45 g/L, p = 0.153) at the time of bleeding.
Bleeding causes
All bleeding events in the continuous anticoagulation group originated from EGVs, whereas in the intermittent group, three patients had bleeding from other sources (PHG, rectal varices, and hemorrhoids). One patient in the intermittent anticoagulation group experienced PHG bleeding on post-procedural day 56 and EVB on post-procedural day 330.
Comparison of bleeding rates
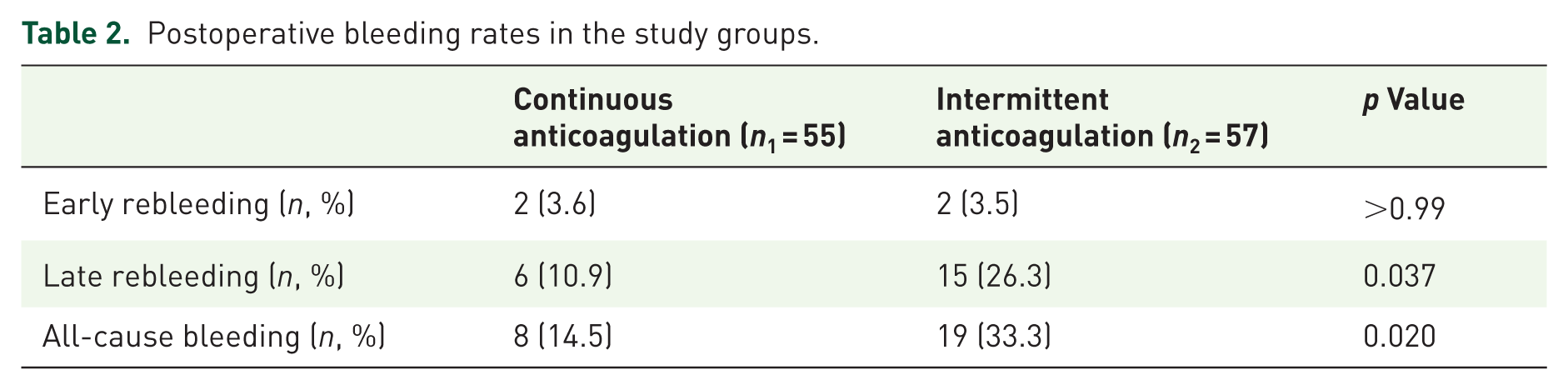
Post-procedural bleeding rates within 1 year are summarized in Table 2. Early rebleeding rates were similar between the groups (3.6% vs 3.5%, p > 0.99). However, the late rebleeding rate was significantly lower in the continuous group (10.9% vs 26.3%, p = 0.037), as was the overall all-cause bleeding rate (14.5% vs 33.3%, p = 0.020). Kaplan–Meier analysis further demonstrated a significantly lower 1-year cumulative incidence of late bleeding (HR = 0.386, 95% confidence interval (CI): 0.150–0.994; log-rank p = 0.041) and all-cause bleeding (HR = 0.396, 95% CI: 0.173–0.904; log-rank p = 0.023) in the continuous anticoagulation group (Figures 2 and 3).
Postoperative bleeding rates in the study groups.

Kaplan–Meier curve analysis of the post-procedural 1-year cumulative incidence of late rebleeding in the study groups.
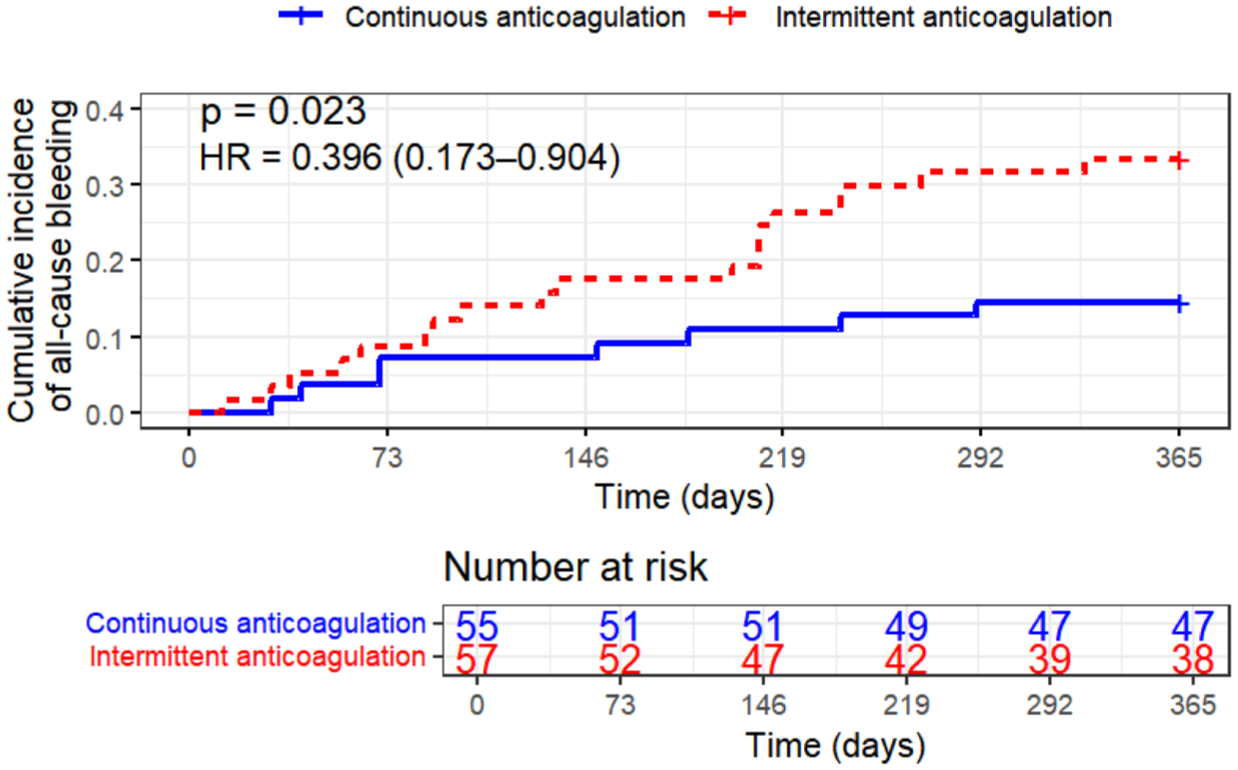
Kaplan–Meier curve analysis of the post-procedural 1-year cumulative incidence of all-cause bleeding in the study groups.
Post-procedural hepatic decompensation events
During the follow-up, hepatic decompensation events excluding bleeding occurred in 15 patients (27.3%) in the continuous group and 30 patients (52.6%) in the intermittent group (p = 0.006). Ascites occurred less frequently in the continuous group (13 (23.6%) vs 26 (45.6%), p = 0.015; Table 3). One year after surgery, there was one death, one case of hepatocellular carcinoma, and one case of HE, all of which occurred in the intermittent anticoagulation group.
Postoperative hepatic decompensation events in the two groups of patients.

Categorical data are presented as absolute numbers (%).
Multivariable logistic regression analysis
Because liver function, indications for EVT, and history of splenectomy may substantially affect bleeding risk and the natural course of cirrhosis, multivariable binary logistic regression was performed to adjust for these potential confounders (Table 4). The model included anticoagulation timing group, baseline Child–Pugh grade, Child–Pugh score, MELD score, indications for EVT, and history of splenectomy.
Effect of anticoagulation timing on clinical outcomes after controlling liver function, endoscopic treatment indications, and splenectomy history using binary logistic regression.

Adjusted Child–Pugh grade, Child–Pugh score, MELD score, indications for endoscopic variceal treatment, and history of splenectomy.
CI, confidence interval; MELD, model for end-stage liver disease; OR, odds ratio.
The analysis showed that anticoagulation timing was an independent factor associated with late rebleeding (odds ratio (OR) = 0.315, 95% CI: 0.106–0.936, p = 0.038), all-cause bleeding (OR = 0.302, 95% CI: 0.112–0.818, p = 0.019), hepatic decompensation events (OR = 0.386, 95% CI: 0.170–0.875, p = 0.023), and ascites (OR = 0.413, 95% CI: 0.177–0.964, p = 0.041) within 1 year after EVT.
Discussion
Managing anticoagulation during EVT in cirrhotic patients with PVT remains clinically challenging, particularly in the setting of acute PVT. This study represents the first randomized controlled trial directly comparing continuous versus intermittent anticoagulation during EVT in this patient population. Compared with intermittent anticoagulation, continuous anticoagulation was associated with reduced long-term bleeding risk and fewer hepatic decompensation events. Although early post-procedural rebleeding rates were comparable between groups, late and overall rebleeding rates were significantly lower in the continuous anticoagulation group (p < 0.05), suggesting a benefit in preventing long-term rebleeding. The 1-year incidence of hepatic decompensation, particularly ascites, was also reduced (p < 0.05), indicating a potential prognostic benefit. Multivariable analysis further confirmed anticoagulation timing as an independent factor associated with these outcomes. These findings suggest that continuous anticoagulation may represent a safe and beneficial management strategy in this population.
In this study, no procedure-related hemorrhage occurred in either group. No bleeding was observed from ulceration following EVL. The 6-week early rebleeding rates were 3.6% and 3.5% in the two groups, respectively, which were comparable to those reported in previous studies.10,11 Early rebleeding rates were low and comparable, suggesting that continuous LMWH administration during EVT does not increase perioperative bleeding risk. This aligns with previous findings indicating that LMWH is a relatively safe anticoagulant during the perioperative period.6,20,21
In the intermittent anticoagulation group, one patient died from hemorrhagic shock following EGVB. The patient had undergone mitral valve replacement, with a history of heart failure and lung infection. The risk of endoscopic treatment was high, and conservative treatment failed, leading to death. Overall, five patients (4.46%) experienced hemorrhagic shock from major bleeding: three EGVB, one PHG bleeding, and one rectal ectopic variceal bleeding. EGVB occurred within 3 months post-procedurally during anticoagulation in patients with F3 esophageal varices and was controlled endoscopically. Non-EGVs bleeding with hemorrhagic shock cases were successfully managed conservatively. No significant difference in post-procedural bleeding severity was observed between the two groups (p = 0.643). This aligns with a multicenter study showing anticoagulation does not worsen bleeding severity, transfusion requirement, or mortality among cirrhotic patients. 22 The patients having a hepatic venous pressure gradient ⩾20 mmHg show increased uncontrollable bleeding. 23
In cirrhotic patients with PVT undergoing anticoagulation therapy, bleeding events are primarily related to portal hypertension rather than anticoagulation itself. A meta-analysis reported a very low rate of anticoagulation-related bleeding (3.3%). 24 All bleeding events observed in this study were related to portal hypertension, with no spontaneous mucosal or systemic bleeding. This may be related to the choice and dosage of anticoagulants. LMWH was the primary anticoagulant used in this study and is associated with a relatively lower bleeding risk.20,25 In addition, the administered dose was lower than the guideline-recommended standard dose, which may have further enhanced its safety profile. Importantly, in the intermittent group, several bleeding events originated from PHG, rectal varices, or hemorrhoid lesions associated with worsening portal hypertension. This underscores that continuous anticoagulation, by preventing PVT progression, may alleviate portal pressure and thus reduce the risk of portal hypertension-related bleeding.5,8
Continuous anticoagulation significantly reduced the risk of late and all-cause bleeding compared with intermittent anticoagulation. Current studies26,27 have revealed the tendency of anticoagulation in reducing variceal bleeding. Other studies have explicitly shown that anticoagulation can decrease portal hypertension-related bleeding and variceal bleeding.28–30 Given that all bleeding events observed in our study were portal hypertension-related bleeding, the possible mechanisms include: on one hand, initiating and maintaining anticoagulation promotes portal vein recanalization, reduces portal pressure, and prevents hypertension-related bleeding. 7 Early anticoagulation in acute PVT has been associated with higher recanalization rates, while interruption increases the risk of thrombus progression or recurrence. Turnes et al. 13 reported that the recanalization rate for acute PVT decreased from 62.5% when anticoagulation therapy was started within the first week to 18.2% when started after 1 week (p < 0.05). A study found that 70% of patients who discontinued anticoagulation experienced PVT recurrence or progression. 31 On the other hand, continuous anticoagulation improves liver function and prognosis, thereby reducing the risk of portal hypertension-related bleeding. 32 In this study, the lower incidence of hepatic decompensation events observed in the continuous anticoagulation group further supports this view, which has also been demonstrated in previous studies that anticoagulation can delay the onset of liver decompensation and improve survival rates.33–35 However, some studies did not observe a benefit of anticoagulation in reducing portal hypertension-related bleeding.10,35 The positive findings of the present study may be attributable to the inclusion of patients with acute PVT, who may be more responsive to anticoagulation therapy, as well as the relatively longer follow-up period, which may have facilitated detection of long-term clinical benefits.
In our cohort, patients in the continuous anticoagulation group had significantly fewer hepatic decompensation events, especially ascites. Moreover, there was one death, one case of hepatocellular carcinoma, and one case of HE among all patients, all of which occurred in the intermittent anticoagulation group. These findings support growing evidence that anticoagulation improves prognosis. Previous studies have shown that anticoagulation reduces the incidence of ascites36,37 and benefits liver function and prognosis in cirrhosis.5,33 These benefits may stem from anticoagulant therapy, which can directly improve hepatic microvascular thrombosis and reduce the activation of coagulation proteases on intrahepatic cells, particularly stellate cells. 38 Serper et al. 34 found that anticoagulation was associated with reduced hepatic decompensation and all-cause mortality in patients with liver cirrhosis and atrial fibrillation. Improved portal venous flow following recanalization may also contribute to reduce hepatic decompensation events indirectly. 39
However, this study has limitations. First, this study represents a secondary analysis of a registered clinical trial (ChiCTR1800017430). Although anticoagulation timing was randomized, this specific analysis was not prospectively registered and should therefore be considered exploratory. Accordingly, the findings should be interpreted with caution and regarded primarily as hypothesis-generating. Second, it was a single-center, open-label study with a modest sample size, which may limit the generalizability of the findings. Future large-scale, multicenter prospective randomized studies with prespecified stratification according to key factors (e.g., cirrhosis severity and indications for EVT) are needed to validate and extend these results. In addition, the absence of systematic imaging follow-up and standardized assessment of recanalization precluded complete evaluation of PVT recanalization rates. Prospective studies incorporating standardized imaging follow-up are required to address this limitation. Moreover, the advantages of continuous anticoagulation may be less pronounced in patients with chronic PVT, in whom the potential for thrombus resolution is limited.3,8
The anticoagulation regimen also has limitations. The enoxaparin dose used in this study was lower than the guideline-recommended dose in China, 40 primarily because all patients had decompensated cirrhosis and required anticoagulation during the peri-EVT period, during which standard dosing may increase bleeding risk. Previous studies have also used LMWH at 70 U/kg twice daily during the periprocedural period. 11 Therefore, a reduced dose was adopted in the present study. In addition, enoxaparin is supplied in our hospital as 4000 U/syringe, which is more convenient in clinical use and may improve patient adherence. Future studies with standard-dose anticoagulation are warranted. In the present study, anticoagulation in the intermittent group was resumed 1 week after EVT, which was more conservative than routine clinical practice, where interruption may often last only 1–2 days, and this may have contributed to poorer outcomes. Future studies directly comparing continuous anticoagulation, short interruption, and delayed resumption strategies are needed to determine the optimal periprocedural anticoagulation management. Although our findings strongly support the use of continuous LMWH during EVT, the optimal protocol for bridging therapy, particularly in patients on long-term warfarin, requires further investigation. 7 The safety and efficacy of direct oral anticoagulants in patients undergoing EVT also require further prospective evaluation.
Conclusion
In patients with cirrhosis and acute PVT undergoing endoscopic EVs treatment, continuous LMWH anticoagulation is safe and advantageous. It does not increase the risk or severity of early post-procedural bleeding but significantly reduces late and all-cause bleeding, as well as hepatic decompensation events such as ascites.
These findings provide new evidence-based support for maintaining anticoagulation during EVT, especially in patients requiring urgent treatment for acute PVT or those at risk of mesenteric ischemia. Continuous anticoagulation should be considered a safe and effective management strategy to improve long-term outcomes in this challenging patient population.
This is the first randomized trial to provide evidence for the safety of continuous anticoagulation during EVT in cirrhotic patients with acute PVT, challenging the conventional practice of anticoagulation interruption.
Our findings may help refine future clinical guidelines on periprocedural anticoagulation management in cirrhosis.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261439217 – Supplemental material for Continuous versus intermittent anticoagulation during endoscopic treatment of esophageal varices in cirrhosis: a randomized controlled trial
Supplemental material, sj-docx-1-tag-10.1177_17562848261439217 for Continuous versus intermittent anticoagulation during endoscopic treatment of esophageal varices in cirrhosis: a randomized controlled trial by Yingying He, Zhihong Wang, Jiayi Zhang, Qianqian Zhang, Yaxian Kuai, Yuchuan Bai, Xuecan Mei and Derun Kong in Therapeutic Advances in Gastroenterology
