Abstract
Background:
The expanding therapeutic landscape of ulcerative colitis (UC) introduces uncertainty regarding the optimal positioning of new agents.
Objectives:
This study aimed to compare the real-world persistence of advanced therapies for UC.
Design:
This observational cohort study used a population-based national registry of biologic and small-molecule therapies for inflammatory bowel disease, CREdIT, to identify patients treated with advanced therapy for UC.
Methods:
The primary outcome was treatment persistence – defined as time from therapy initiation to discontinuation or last follow-up, whichever occurred first – and was analysed using mixed-effects Cox models and inverse probability weighting. Persistence was assessed overall, in first-line therapy, and in second-line therapy after adalimumab or infliximab exposure.
Results:
We included 3718 observations from 2525 patients with UC (48% female). Infliximab (40%), vedolizumab (27%) and adalimumab (17%) were most frequently used. At 5 years, 1852 of 3718 observations (49.8%) remained on the same therapy. The treatment line alone was not associated with treatment persistence after accounting for patient-level clustering using a mixed-effects model. Vedolizumab showed significantly greater persistence than infliximab or adalimumab, including in first-line use. In second-line settings post-anti-tumour necrosis factor (TNF) exposure, upadacitinib had the highest persistence compared to tofacitinib, vedolizumab and ustekinumab, whereas vedolizumab was more persistent than tofacitinib.
Conclusion:
Vedolizumab demonstrated longer persistence than infliximab or adalimumab, regardless of treatment line. Following anti-TNF failure, upadacitinib showed the highest persistence.
Plain language summary
Introduction
Treatment options for patients with inflammatory bowel disease (IBD) have expanded substantially over the last decade. A wide spectrum of drugs with different mechanisms of action is now available, especially for patients with ulcerative colitis (UC).1,2 Nevertheless, the expanding therapeutic landscape introduces uncertainty regarding the optimal positioning of new agents. To maximise therapeutic outcomes, gastroenterologists must rely on data from multiple sources to select the right drug at the right time. Head-to-head trials are the primary source for comparing different drugs. However, such studies are not available for the majority of drug pairs. Despite their inherent limitations – such as differences in therapeutic goals, patient populations, steroid-tapering regimens and trial designs – network meta-analyses are also employed to support treatment decision-making. 3
In addition, real-world data are increasingly being utilised.4–7 These cohorts do not exclude patients who would be ineligible for clinical trials, thus providing more representative insights into actual drug performance. The most commonly studied endpoint in such studies is treatment persistence, which reflects efficacy and safety to a large extent. However, these comparisons are susceptible to baseline differences between groups and therefore require appropriate methods to adjust for potential confounding. Inverse probability weighting (IPW) is one such method, using propensity scores to balance baseline patient characteristics by weighting each individual in the analysis according to the inverse probability of receiving the treatment they actually received. 8
Using a population-based national registry of patients receiving advanced therapy (CREdIT registry), we compared treatment persistence across various therapies for UC, overall, as first-line treatment, and as second-line treatment following exposure to adalimumab or infliximab. In addition, we investigated general predictors of persistence with advanced therapy, irrespective of the specific treatment.
Materials and methods
Study design and registry
This cohort study was designed as an analysis of prospectively collected data using a nationwide registry of patients with IBD treated with biologics or small molecules, referred to as CREdIT (https://credit.registry.cz). The registry included 46 centres providing advanced therapy across the Czech Republic. Established in 2016, the registry was approved by the Ethics Committee of the University Hospital Hradec Králové (reference number: 201806 S19R), and includes all eligible patients consecutively enrolled at the time of advanced therapy initiation, following written informed consent provided by the patient or their legal guardian. Data on patients who initiated advanced therapy prior to 2016 were collected retrospectively; from 2016 onward, patient enrolment, follow-up visits and drug administrations were recorded prospectively. Data were collected by a gastroenterologist or a designated nurse. The recorded data included demographic information, disease classification and characteristics at diagnosis; treatment details and disease activity at the time of therapy initiation and follow-up information on therapy and disease activity at each visit, as previously described. 9 Additional details are listed in Table 1. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 10
Baseline characteristics of all patients with ulcerative colitis from the CREdIT registry, regardless of the treatment line.
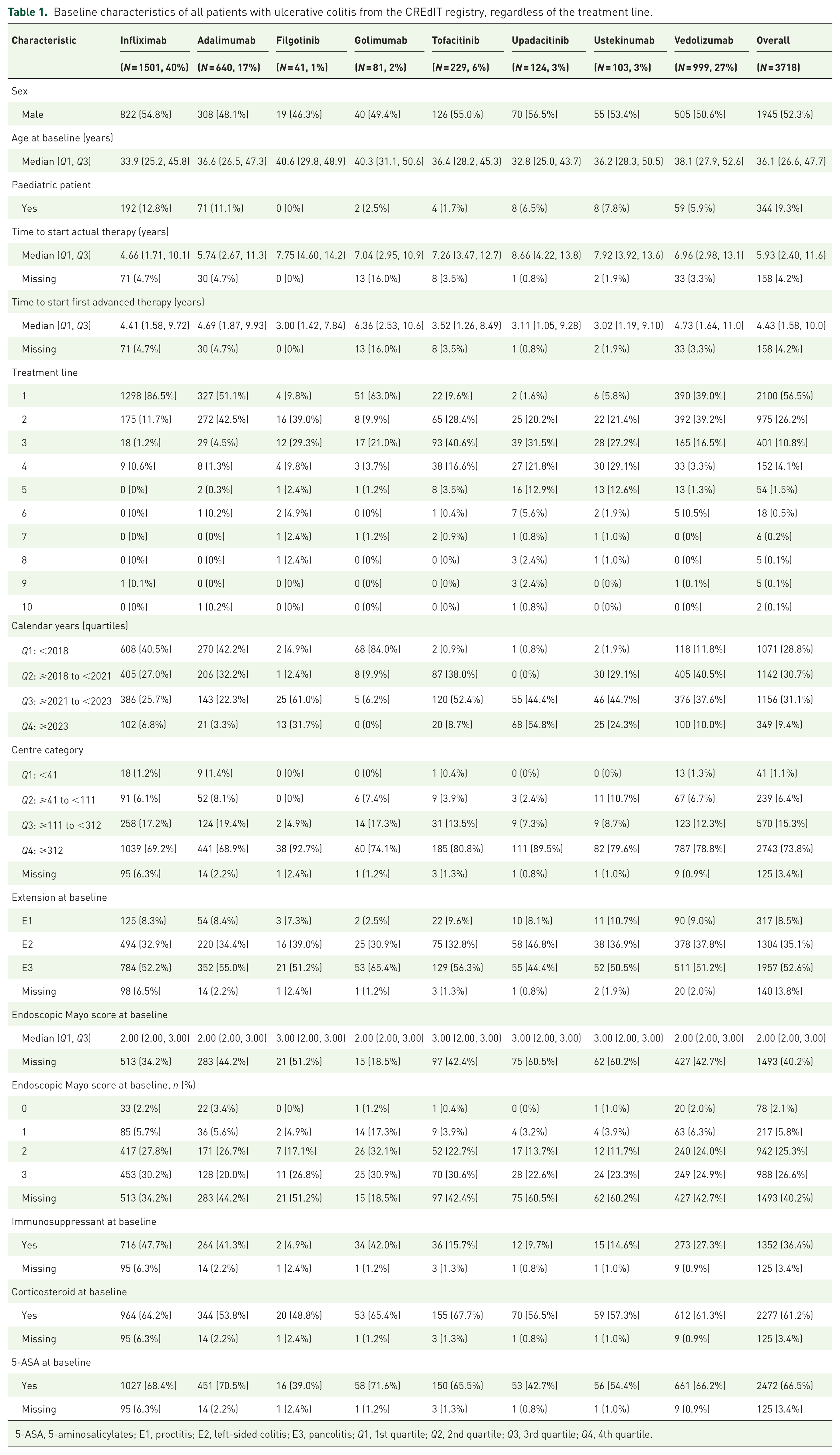
5-ASA, 5-aminosalicylates; E1, proctitis; E2, left-sided colitis; E3, pancolitis; Q1, 1st quartile; Q2, 2nd quartile; Q3, 3rd quartile; Q4, 4th quartile.
Study population
Each treatment course was considered a separate observation. Inclusion criteria encompassed treatment episodes in children (aged <19 years at the start of advanced treatment) and adult patients with UC who initiated advanced therapy at the discretion of the treating physician. Advanced therapy included biologic treatments (infliximab, adalimumab, golimumab, vedolizumab, ustekinumab) and small molecules (Janus kinase (JAK) inhibitors). Only treatment episodes in which patients received a single advanced treatment at any one time point were included. Exclusion criteria comprised treatment episodes with prior colectomy, rescue therapy (defined as treatment episodes being initiated and recorded as rescue therapy) and missing data related to treatment discontinuation events (cases in which it was not possible to determine whether treatment was continued or discontinued), time to event (defined as the time from initiation of advanced therapy to treatment discontinuation or censoring), treatment type or line of therapy. Treatment episodes initiated between 2002 and 2024 were included. Data were extracted from the registry on August 29, 2024. All treatment episodes that met the eligibility criteria were included in the analysis.
Outcome and definitions
The primary outcome was treatment persistence, defined as the time from therapy initiation to either the date of the last follow-up or drug discontinuation, whichever occurred first, without predefined evaluation time points. Any discontinuation was considered an event. Treatment persistence was assessed at the level of individual treatment episode and constituted the single primary outcome of the study. If an individual patient received more than one advanced treatment, all treatments that met the inclusion criteria were analysed, including repeated treatment with the same drug in separate episodes. Persistence was evaluated according to the type of advanced therapy initiated. Switching within the same biologic agent (between originators and biosimilars or from intravenous to subcutaneous formulations) was not considered treatment termination and was analysed as a single observation. For ongoing treatments, the date of the last recorded visit was used as the censoring point.
The primary analysis evaluated the overall treatment persistence of adalimumab, infliximab and vedolizumab across all treatment lines. Secondary analyses included: (1) assessment of overall treatment persistence and baseline predictors of persistence; (2) comparison of treatment persistence of adalimumab, infliximab and vedolizumab in first-line therapy; (3) comparison of treatment persistence of vedolizumab, ustekinumab, upadacitinib and tofacitinib following prior exposure to adalimumab or infliximab and (4) comparison of treatment persistence between paediatric and adult patients.
Immunosuppressive therapy at treatment initiation was defined as any dose of azathioprine, 6-mercaptopurine or methotrexate administered at the start of advanced therapy. Concomitant immunosuppressive treatment with anti-tumour necrosis factor (TNF) agents was defined as the use of azathioprine or methotrexate within the first 6 months after initiation of anti-TNF therapy, based on their documented presence at the 3- and/or 6-month follow-up visits when treatment status was assessed.
Statistical analysis
Median and interquartile range (IQR) were used for all continuous variables. Categorical variables were described as absolute frequencies and percentages. Owing to missing covariate values (Table 1), multiple imputation (10 sets, 10 iterations) was performed using the ‘mice’ R package. Analyses were performed on the following sets: (1) All treatment courses, (2) Infliximab, adalimumab and vedolizumab in all lines of treatment, (3) Infliximab, adalimumab and vedolizumab in first-line treatment only, (4) Vedolizumab, tofacitinib, ustekinumab and upadacitinib in ⩾2nd line only, if the patient had adalimumab or infliximab in first-line treatment. Comparisons of treatment persistence were made after balancing the groups using IPW, using the R ‘MatchThem’ package. Observations of patients treated with filgotinib and golimumab were not included in the separate set analyses because of the low number of observations and the difficulty of balancing the groups using IPW. The following baseline variables were considered potentially predictive of treatment outcome (‘predefined predictors’): sex, age, time from UC diagnosis to start of first advanced therapy, time from diagnosis to start of current advanced therapy, treatment line, calendar year of treatment initiation (quartiles, based on number of observations: Q1: <2018, Q2: ⩾2018 to <2021, Q3: ⩾2021 to <2023, Q4: ⩾2023), centre category (quartiles defined by the number of observations per centre, reflecting centre size and patient volume in the registry: Q1: <41, Q2: ⩾41 to <111, Q3: ⩾111 to <312, Q4: ⩾312), UC extension according to the Montreal classification, baseline endoscopic Mayo score (defined as the most recent endoscopic assessment performed within 2 months prior to initiation of advanced therapy), immunosuppressive therapy, corticosteroids (systemic or topical) and 5-aminosalicylates (5-ASA). Although faecal calprotectin and C-reactive protein were available in a subset of patients, they were not included among the variables used for covariate balancing; disease activity was assessed using the endoscopic Mayo score. Line of treatment was tested as a numeric variable and, in explicitly specified models, as a categorical variable (as follows: 1st, 2nd, 3rd, 4th and >4th advanced therapy for the whole dataset; 2nd, 3rd, 4th and >4th advanced therapy for comparison of treatment courses beyond the first line).
Kaplan–Meier curves were used to visualise time to treatment discontinuation. Cox proportional hazards regression models were used to test the association between treatment persistence and each predefined predictor. A multivariable Cox model was used to assess their independent associations. The same approach was used when comparing persistence within first-line treatment. For all other analyses, mixed-effects Cox regression models were implemented, including the patient factor as a random effect. Mixed-effects Cox proportional hazards models were fitted using the R package coxme. For the analysis of overall treatment persistence and its predictors, the fully adjusted mixed-effects Cox regression model including a patient-level random effect was considered the main model. Other model specifications, including models without a random effect and models with alternative parameterisations of the treatment line, were performed as supporting analyses. Comparisons of persistence were made between treatments with each other, and results were expressed using a heatplot for the following sets: Infliximab, adalimumab and vedolizumab in all lines of treatment; Infliximab, adalimumab and vedolizumab in first-line treatment only; Vedolizumab, tofacitinib, ustekinumab and upadacitinib in ⩾2nd line only if the patient had adalimumab or infliximab in first-line treatment. We also tested whether persistence differed between paediatric patients (<19 years at the time of initiation of advanced therapy) and adult patients.
Statistical significance was defined as a p-value <0.05. All estimates were reported with 95% confidence intervals (CIs). Figures were generated using the R package ‘ggplot2’. All data were analysed using the R statistical software (version 4.4.1; www.r-project.org). No formal a priori sample size calculation was performed, as this was a registry-based retrospective study. The sample size was determined by data availability and is comparable to or larger than similar published studies.
Results
A total of 4693 observations with a diagnosis of UC were defined in the CREdIT database (Supplemental Figure 1(a)). Of these, 975 (21%) were excluded for the following reasons: dual therapy (n = 30), history of colectomy or treatment indication as rescue therapy (n = 747) and missing data related to outcomes (n = 198).
Thus, 3718 observations (1773 female, 48%) of 2525 patients with UC were included in the analysis (Supplemental Figure 1(b), Table 1). Most observations were from the first (2100, 57%) and second (975, 26%) lines of advanced therapy and primarily involved infliximab (1501, 40%), followed by vedolizumab (999, 27%) and adalimumab (640, 17%). The remaining 589 observations (16%) involved tofacitinib, upadacitinib, ustekinumab, golimumab and filgotinib (Table 1). Due to the small number of observations with golimumab and filgotinib, we did not include these drugs in analyses comparing durability. Table 1 also shows that the proportion of drug use varied by period and centre. For example, 54% of observations with upadacitinib belonged to the last quartile, whereas adalimumab and infliximab were less frequently used during that time. Use of newer drugs – filgotinib and upadacitinib was more common in large centres (93% and 94% of observations, respectively). The median endoscopic Mayo score was 2 (IQR 2–3) for infliximab, adalimumab, golimumab and vedolizumab, and 3 (IQR 2–3) for the other drugs. Immunosuppressive therapy at the beginning of treatment was recorded in 1352 observations (36%), and was numerically more common with infliximab (48%), adalimumab (41%) and golimumab (42%) than with other drugs. Baseline corticosteroid therapy was numerically most common with tofacitinib (68%), golimumab (65%) and infliximab (64%), and lowest with filgotinib (49%) and adalimumab (54%; Table 1).
Overall persistence of UC treatment and predictors of persistence
The overall persistence of advanced therapies in patients with UC, regardless of treatment line, is presented in Supplemental Figure 2. At 5 years, 1852 of 3718 observations (49.8%) remained on the same therapy.
We first tested the predefined predictors – including sex, age, paediatric age, time from diagnosis to initiation of current advanced therapy, time from diagnosis to first advanced therapy, treatment line, calendar year of treatment initiation, centre category, UC extension, baseline endoscopic Mayo score, immunosuppressive therapy, corticosteroids and 5-ASA – individually, without adjusting for patient-related factors (supporting analyses; Supplemental Table 1(A)). Each additional treatment line was associated with a 9% decrease in the probability of treatment persistence (hazard ratio (HR) 0.91, 95% CI 0.86–0.95). Persistence was also positively associated with older age, shorter disease duration prior to current and first advanced treatment, later calendar periods, lower baseline endoscopic Mayo score, immunomodulator use and absence of corticosteroid therapy at treatment initiation.
However, when the patient factor was included as a random effect to account for multiple treatment cycles per patient (supporting analyses; Supplemental Table 1(B)), the association with treatment line disappeared (HR 0.99, 95% CI 0.94–1.04). The associations with older age, calendar period, lower endoscopic Mayo score, immunomodulator use and non-use of corticosteroids at baseline remained significant.
In the fully adjusted mixed-effects Cox regression model, which was considered the main model for this analysis, independent associations with persistence were observed for older age at diagnosis, later calendar periods, lower endoscopic Mayo score, immunomodulator use and absence of corticosteroids at treatment initiation (Table 2).
Independent predictors of persistence of overall observations in patients with ulcerative colitis from the CREdIT registry, regardless of treatment.

Calendar years: Q1: <2018, Q2: ⩾2018 to <2021, Q3: ⩾2021 to <2023, Q4: ⩾2023. Mixed-effects multiple Cox regression model with patient-specific factor added as a random effect.
At baseline, among patients receiving an immunosuppressant (methotrexate or azathioprine), 3.3% were treated with methotrexate.
CI, confidence interval; HR, hazard ratio; Q1, 1st quartile; Q2, 2nd quartile; Q3, 3rd quartile; Q4, 4th quartile; vs, versus.
Similarly, as a supporting analysis, in a model that included the patient factor and treatment line as a categorical variable, we did not find a statistically significant association between treatment line and persistence (Supplemental Table 2, Supplemental Figure 3).
Comparison of the overall persistence among adalimumab, infliximab and vedolizumab
This sub-analysis included 1501 observations with infliximab, 640 with adalimumab and 999 with vedolizumab. Clinical and demographic characteristics are summarised in Table 3. Owing to observed group differences, IPW was used to balance the cohorts (Supplemental Figure 4). Compared with infliximab and adalimumab, vedolizumab demonstrated greater treatment persistence (HR 3.42, 95% CI 2.90–4.04 vs infliximab; HR 3.77, 95% CI 3.13–4.54 vs adalimumab; Figure 1(a)). No significant difference was observed between infliximab and adalimumab (HR 0.91, 95% CI 0.80–1.03).
Baseline characteristics of patients with ulcerative colitis from the CREdIT registry treated with infliximab, adalimumab or vedolizumab, regardless of treatment line.
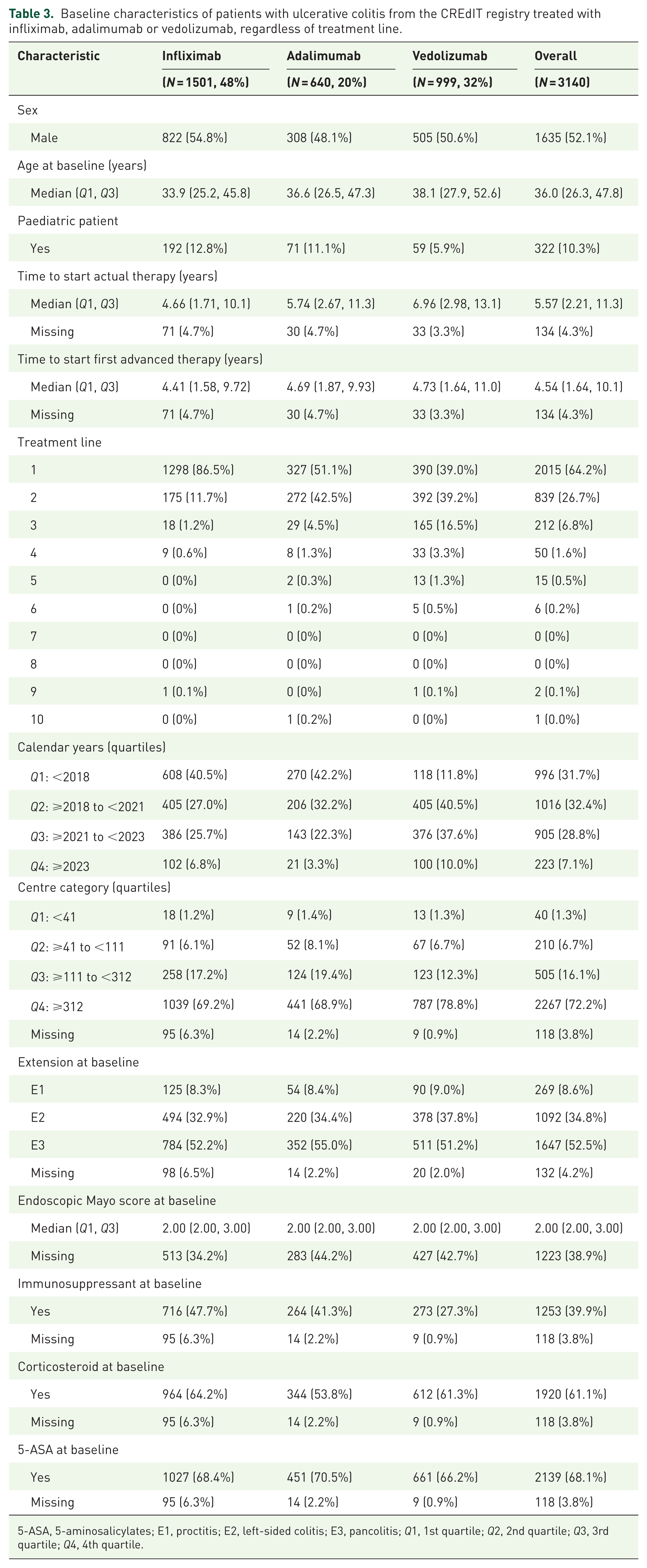
5-ASA, 5-aminosalicylates; E1, proctitis; E2, left-sided colitis; E3, pancolitis; Q1, 1st quartile; Q2, 2nd quartile; Q3, 3rd quartile; Q4, 4th quartile.

Kaplan–Meier curves showing treatment persistence of adalimumab, infliximab and vedolizumab in: (a) all patients with UC. In panel (a), the estimated treatment persistence at 1 and 5 years was 70.8% and 49.0% for adalimumab, 70.5% and 50.8% for infliximab and 75.7% and 51.9% for vedolizumab (Kaplan–Meier estimates). (b) Patients with UC treated with vedolizumab or anti-TNF agents (adalimumab and infliximab), with anti-TNF agents stratified by concomitant immunomodulator use. In panel (b), the estimated treatment persistence at 1 and 5 years was 70.8% and 48.0% for adalimumab monotherapy, 70.7% and 50.7% for adalimumab plus immunomodulator, 65.2% and 45.8% for infliximab monotherapy, 76.4% and 56.3% for infliximab plus immunomodulator and 75.7% and 51.9% for vedolizumab (Kaplan–Meier estimates). (c) Patients with UC receiving first-line advanced therapy. In panel (c), the estimated treatment persistence at 1 and 5 years was 71.1% and 51.1% for adalimumab, 72.2% and 52.5% for infliximab and 78.0% and 45.9% for vedolizumab (Kaplan–Meier estimates). (d) Patients with UC receiving first-line advanced therapy with vedolizumab or anti-TNF agents (adalimumab and infliximab), with anti-TNF agents stratified by concomitant immunomodulator use. In panel (d), the estimated treatment persistence at 1 and 5 years was 73.0% and 38.1% for adalimumab monotherapy, 71.3% and 53.5% for adalimumab plus immunomodulator, 65.4% and 38.5% for infliximab monotherapy, 77.7% and 51.4% for infliximab plus immunomodulator and 78.0% and 45.9% for vedolizumab (Kaplan–Meier estimates).
In the comparative analysis of treatments where anti-TNF therapy was stratified by concomitant immunomodulator use (Figures 1(b) and 2(a)), vedolizumab showed greater persistence compared with adalimumab and infliximab, regardless of whether the treatment was administered as monotherapy (adalimumab: HR 3.82, 95% CI 2.99–4.88; infliximab: HR 3.95, 95% CI 3.40–4.59) or combination therapy (adalimumab + immunomodulator: HR 3.38, 95% CI 1.99–5.71; infliximab + immunomodulator: HR 2.56, 95% CI 1.96–3.33). Infliximab combined with an immunomodulator was more persistent than infliximab monotherapy (HR 1.54, 95% CI 1.12–2.12) or adalimumab monotherapy (HR 1.50, 95% CI 1.02–2.19). However, we did not observe a statistically significant difference in persistence between infliximab + immunomodulator and adalimumab + immunomodulator (HR 1.32, 95% CI 0.86–2.04; Figure 2(a)). Similarly, no statistically significant difference in persistence was observed between infliximab monotherapy and adalimumab combined with an immunomodulator (HR 0.86, 95 % CI 0.48–1.52; Figure 2(a)).

Comparison of treatment persistence across advanced therapies in ulcerative colitis. Pairwise comparisons of treatment persistence between advanced therapies using HRs with 95% CIs. Therapies are compared pairwise, with the therapy in the row contrasted against the therapy in the column. Values above 1 indicate higher persistence of the therapy in the column compared to the therapy in the row. Anti-tumour necrosis factor agents are shown as monotherapy and in combination with immunomodulators. (a) All patients, regardless of treatment line. (b) Subgroup of patients receiving first-line advanced therapy.
Comparison of the persistence of adalimumab, infliximab and vedolizumab in first-line treatment
A total of 2015 observations from first-line treatment were included in the study (1298 infliximab, 327 adalimumab and 390 vedolizumab; Supplemental Table 3). After IPW (Supplemental Figure 5), adalimumab was not found to be more persistent than infliximab (HR 1.04, 95% CI 0.84–1.27). However, vedolizumab demonstrated greater persistence compared to infliximab (HR 1.67, 95% CI 1.33–2.11) and adalimumab (HR 1.45, 95% CI 1.10–1.91; Figure 1(c)).
When comparing individual treatments stratified by concomitant immunomodulator use in first-line treatment (Figures 1(d) and 2(b)), vedolizumab was more persistent than infliximab in monotherapy (HR 1.98, 95% CI 1.32 to −2.95) and combination therapy (HR 1.5, 95% CI 1.00–2.26), as well as more persistent than adalimumab monotherapy (HR 1.76, 95% CI 1.12–2.77). We did not observe a statistically significant difference in persistence between vedolizumab and adalimumab + immunomodulator (HR 1.57, 95% CI 0.94–2.64). Infliximab combined with an immunomodulator was more persistent than infliximab monotherapy (HR 1.31, 95% CI 1.08–1.60). In addition, persistence did not differ significantly between infliximab monotherapy and adalimumab combined with an immunomodulator in the first-line setting (HR 0.80, 95% CI 0.55–1.15).
Comparison of the persistence of vedolizumab, ustekinumab, upadacitinib and tofacitinib after prior use of adalimumab or infliximab
Following adjustment through IPW (Supplemental Figure 6), we compared the persistence of vedolizumab, ustekinumab, upadacitinib and tofacitinib in patients previously treated with adalimumab or infliximab (Table 4 and Figure 3). Compared to tofacitinib, vedolizumab (HR 1.55, 95% CI 1.25–1.92) and upadacitinib (HR 2.51, 95% CI 1.77–3.53) were associated with higher persistence. We did not observe a statistically significant difference in persistence between vedolizumab and ustekinumab (HR 1.30, 95% CI 0.96–1.75). Additionally, no difference in persistence was found between ustekinumab and tofacitinib (HR 1.20, 95% CI 0.89–1.61). Ustekinumab (HR 0.48, 95% CI 0.31–0.73) and vedolizumab (HR 0.62, 95% CI 0.45–0.85) were less persistent compared to upadacitinib (Figure 3, Supplemental Figure 7).
Baseline characteristics of patients with ulcerative colitis from the CREdIT registry treated with tofacitinib, upadacitinib, ustekinumab or vedolizumab (⩾2nd line), all with prior adalimumab or infliximab exposure.
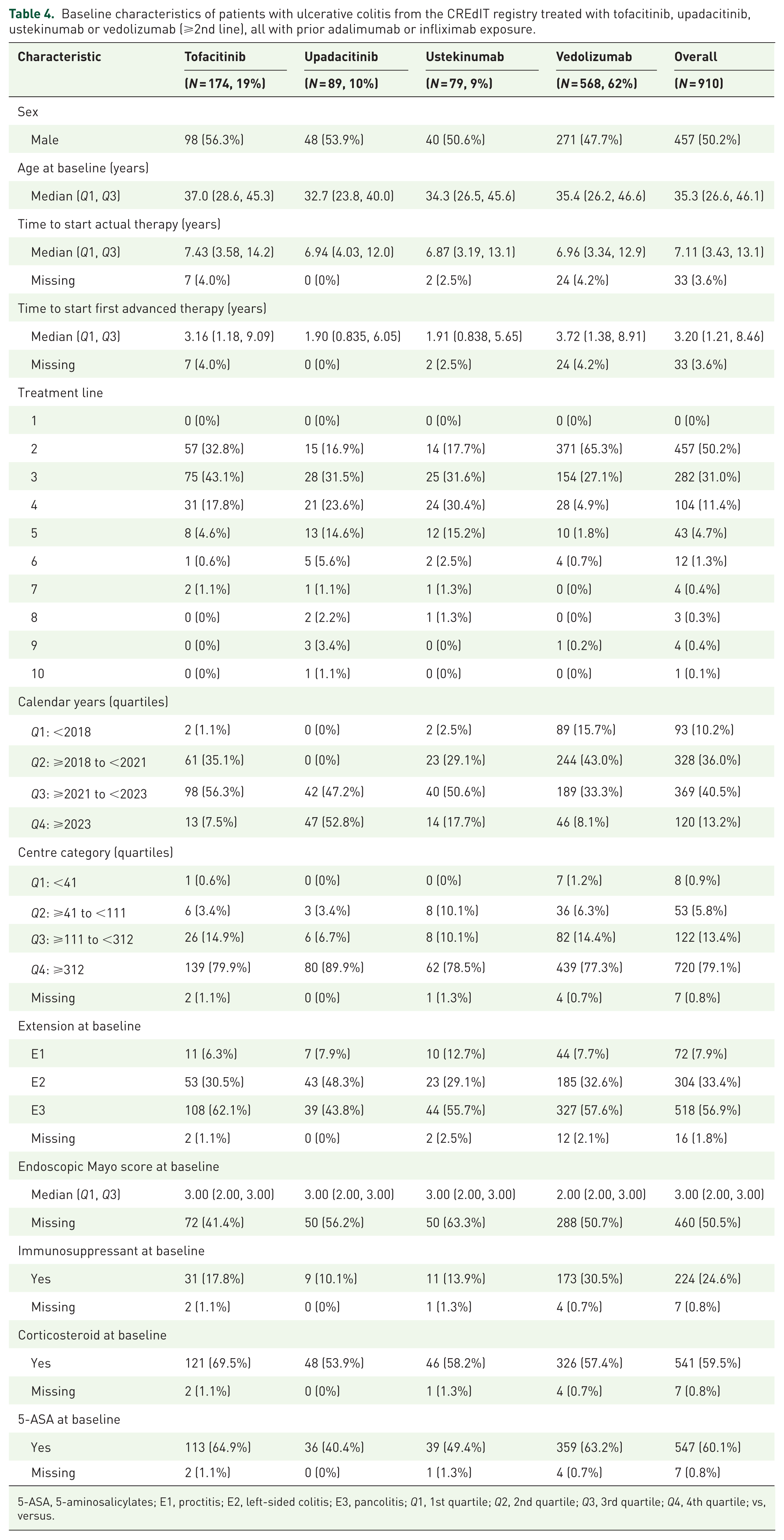
5-ASA, 5-aminosalicylates; E1, proctitis; E2, left-sided colitis; E3, pancolitis; Q1, 1st quartile; Q2, 2nd quartile; Q3, 3rd quartile; Q4, 4th quartile; vs, versus.

Comparison of treatment persistence in patients with ulcerative colitis from the CREdIT registry treated with tofacitinib, upadacitinib, ustekinumab and vedolizumab in second- and higher-line therapy following prior exposure to adalimumab or infliximab. Pairwise comparisons of treatment persistence between advanced therapies are presented as HRs with 95% CIs. Therapies are compared pairwise, with the therapy in the row contrasted against the therapy in the column. Values above 1 indicate higher persistence of the therapy in the column compared to the therapy in the row. Anti-TNF agents are shown as monotherapy and in combination with immunomodulators.
Comparison of paediatric and adult patients
A comparison of treatment courses involving infliximab, adalimumab and vedolizumab, stratified by age group (paediatric vs adult patients), did not reveal a statistically significant difference in overall persistence across treatment lines (HR 1.08, 95% CI 0.85–1.36; Supplemental Figure 8(a)) or in first-line treatment (HR 0.98, 95% CI 0.79–1.22; Supplemental Figure 8(b)).
Discussion
Utilising the National Registry of Advanced Therapies for patients with IBD (CREdIT), we demonstrated that in patients with UC, treatment with vedolizumab was more persistent compared to infliximab and adalimumab, in all treatment lines as well as in the first treatment line only. This effect was independent of concomitant immunomodulator therapy. In patients with prior exposure to anti-TNFs, upadacitinib (at least during early follow-up) was the most persistent therapy and superior to tofacitinib, vedolizumab and ustekinumab. Treatment persistence was not influenced by treatment line, but rather appeared to be a function of the patient’s inherent characteristics.
The current study revealed the better persistence of vedolizumab over infliximab and adalimumab in the treatment of UC. The only head-to-head study in UC, the VARSITY trial, 11 which compared vedolizumab and adalimumab, demonstrated that after 1 year of treatment, a greater proportion of patients treated with vedolizumab were in clinical remission. However, that study did not permit dose adjustment, which may have disadvantaged adalimumab. Furthermore, clinical remission without corticosteroids, a more important goal, was more frequently achieved by patients on adalimumab than on vedolizumab. Several network meta-analyses have also been published, with conflicting results depending on the outcome measures selected and the studies included.1,12–17 Therefore, even considering the known limitations of this approach, particularly important for anti-TNF trials, it is not possible to definitively determine whether vedolizumab is superior to anti-TNFs based on these analyses alone. 3 When considering treatment persistence as a treatment goal, a recent large-scale UK study reported, in line with our findings, the superiority of vedolizumab over anti-TNFs, with vedolizumab being three times more persistent. However, that study did not compare vedolizumab and anti-TNFs specifically in the context of immunomodulator use. 6 In contrast, recent data from the Israeli registry demonstrated that anti-TNFs exhibited greater persistence than vedolizumab when administered concurrently with immunomodulatory therapy. 4 In our study, vedolizumab was significantly superior to infliximab and adalimumab, regardless of concomitant immunomodulators, in the overall cohort and in the first treatment line subgroup. An exception was observed for adalimumab combination therapy, in which no significant difference in persistence was detected. The absence of a statistically significant difference between adalimumab plus azathioprine and vedolizumab in first-line therapy should be interpreted with caution, as a substantial decline in patient numbers in both groups over long-term follow-up, particularly beyond 5 years, may have reduced statistical power. The discrepancy between our findings and those of the Israeli study is difficult to explain. One notable difference was the later initiation of first-line advanced treatment in our patients (5 vs 2 years). However, another recent study found no difference in treatment efficacy whether vedolizumab was initiated before or after 4 years. 18 Furthermore, slight differences in group adjustment approaches between the two studies do not appear sufficient to account for the discrepancy. The evaluation of treatment persistence encompasses efficacy and safety and, from a long-term perspective, may be considered a more precise indicator of overall treatment effectiveness in clinical practice than efficacy alone. Indeed, a network meta-analysis specifically focused on the risk of serious infections in UC found vedolizumab to carry less risk than anti-TNF agents. 19 This may partially explain the higher persistence rates observed with vedolizumab. Despite growing evidence, it remains premature to draw definitive conclusions regarding the superiority of vedolizumab over anti-TNF therapy.
We also demonstrated that infliximab monotherapy was as persistent as adalimumab mono- or combination therapy in the first-line setting and regardless of treatment line. However, the combination of infliximab with an immunosuppressant resulted in longer persistence compared to infliximab monotherapy (in both analyses) and adalimumab monotherapy (in the analysis of all treatment lines only). Network meta-analyses have suggested that infliximab may be more efficacious than adalimumab.12–17,20 Nevertheless, these comparisons often did not account for concomitant therapy, despite the SUCCESS trial demonstrating a significant benefit of combining infliximab with immunomodulatory therapy. 21 Furthermore, the efficacy of infliximab and adalimumab has been assessed in several retrospective, administrative or real-world registry studies, with inconsistent findings.4,6,22–25 For instance, a recent large UK study with balanced groups found superior persistence with infliximab over adalimumab but did not control for immunomodulator use. Conversely, another population-balanced study that included immunomodulator use found that the persistence of adalimumab and infliximab did not differ when used as monotherapy. 6 However, combination therapy with infliximab was found to be superior to infliximab monotherapy. 4 There remains a need for head-to-head studies directly comparing infliximab and adalimumab to provide definitive conclusions. In our registry, concomitant immunomodulator therapy was used in 48% of patients treated with infliximab. Although current guidelines generally favour the use of infliximab in combination with an immunomodulator, 26 real-world practice appears more heterogeneous, as a recent nationwide cohort reported combination therapy in less than one-third of infliximab-treated patients. 4
An evaluation of treatment persistence in subsequent lines following anti-TNF failure showed higher observed persistence with upadacitinib compared with other advanced therapies during early follow-up. However, this finding should be interpreted with caution, as follow-up for upadacitinib was relatively limited. During early follow-up, upadacitinib showed higher persistence compared with tofacitinib, ustekinumab and vedolizumab, while long-term comparative estimates, especially in comparison with vedolizumab, remain uncertain. The advantage of upadacitinib over tofacitinib in terms of efficacy is supported not only by recent network meta-analyses,12,13,15 but also by data from real-world clinical practice, highlighting the distinct mechanisms of action of individual JAK inhibitors.27–29 Direct comparative data between upadacitinib and ustekinumab are limited 30 ; however, the available literature to date consistently favours upadacitinib.13,15,30 Similarly, results from network meta-analyses generally support the superiority of upadacitinib over vedolizumab,12,13,15 although real-world evidence on this comparison remains scarce. In light of these findings and the published real-world observations, as well as the previously mentioned network meta-analyses, it can be suggested that upadacitinib, at least in the short term, is among the most efficacious and persistent therapeutic options following anti-TNF exposure. The optimal positioning of other agents post-anti-TNF exposure remains to be determined in further studies.
In our extensive patient cohort, we observed that once individual patient-related factors were incorporated into the models, treatment line was no longer a significant predictor of persistence. Similarly, a recent nationwide Korean claims-based study did not identify treatment line as a predictor of treatment persistence in UC. 31 As clinical trials are designed to observe patients as new cases, the only transferable information is the number of previous therapies. Our data suggest that, rather than treatment line, the key predictor of failure is the patient’s intrinsic refractoriness, which may stem from a monogenic IBD defect, 32 other immune-mediated diseases mimicking IBD, 33 or simply a refractory disease phenotype. This approach has the advantage of enabling comparison of various therapeutic regimens within the same patient, offering unique insights. In addition, our findings suggest that drug persistence is comparable between adult and paediatric patients with UC, consistent with previous studies. 4
Our results also suggest a modest increase in persistence with increasing age at initiation of the current therapy. The impact of age on outcomes in UC remains inconsistent across studies and appears to depend on the age structure of the cohort, the specific therapy, the outcome of interest and the analytical approach.7,34–38 This heterogeneity likely reflects the interplay between a more aggressive disease course in patients diagnosed at a younger age 37 and increasing frailty in older patients, 38 with opposing effects on treatment durability.
The major strength of our study lies in the use of a large, unselected, national prospective registry with long-term follow-up. An additional strength is the availability of data on concomitant immunosuppressive therapy, which is particularly relevant for the interpretation of outcomes with anti-TNF agents. The utilisation of multiple patient observations with disparate therapeutic modalities enables a comparison of the efficacy of various agents in the same subjects. Furthermore, a comparison of newer therapeutic options (e.g. ustekinumab and upadacitinib) has not previously been made in such a cohort. It should be noted that the standard balancing procedure, in this case using IPW, was employed with a model that also included the period in which the data were recorded. This is because there is a clear shift towards ever more stringent therapeutic targets, which is also reflected in real-world practice. Some limitations must be addressed; first, our analysis assessed treatment persistence rather than direct efficacy. However, as discussed, treatment persistence, comprising efficacy and safety, might be viewed as an important and sometimes more accurate parameter of overall treatment effectiveness than just efficacy. Second, increasing availability of therapeutic options for UC over time, or physician belief in a particular product based on evolving evidence, might have influenced the decision/willingness of treating physicians to switch between treatments. Furthermore, non-medical factors such as costs or internal treatment policy of centres might have had an impact on the persistence of a particular drug. However, as noted above, the calendar period included in our statistical model could at least partially account for this. Other limitations are related to the type of study and the method of data collection. For instance, there is a lack of specific information regarding treatment intensification or more precise reasons for discontinuing therapy as well as a substantial proportion of missing data for the endoscopic score; moreover, the lack of consistently available biochemical data precluded the assessment of alternative outcomes, such as biochemical remission. Nevertheless, these conditions were consistent across all preparations. Finally, there was a shorter follow-up period for newer agents; however, we attempted to compensate for these differences between groups using IPWT.
Conclusion
In conclusion, the present study suggests greater treatment persistence of vedolizumab over infliximab and adalimumab in patients with UC, in the first-line setting and irrespective of treatment line. The persistence of infliximab and adalimumab as monotherapy appears to be similar. Overall, treatment persistence appears to be more a function of a patient’s inherent characteristics than of the treatment line itself.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261436156 – Supplemental material for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry
Supplemental material, sj-docx-1-tag-10.1177_17562848261436156 for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry by Ondrej Hradsky, Dana Duricova, Milan Lukas, Lenka Nedbalová, Barbora Pipek, Pavel Drastich, Premysl Falt, Tomas Vanasek, Jana Svikova, Milan Siroky, Ludek Bouchner, Pavel Svoboda, Jiri Bronsky and Martin Bortlik in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848261436156 – Supplemental material for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry
Supplemental material, sj-docx-2-tag-10.1177_17562848261436156 for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry by Ondrej Hradsky, Dana Duricova, Milan Lukas, Lenka Nedbalová, Barbora Pipek, Pavel Drastich, Premysl Falt, Tomas Vanasek, Jana Svikova, Milan Siroky, Ludek Bouchner, Pavel Svoboda, Jiri Bronsky and Martin Bortlik in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848261436156 – Supplemental material for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry
Supplemental material, sj-docx-3-tag-10.1177_17562848261436156 for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry by Ondrej Hradsky, Dana Duricova, Milan Lukas, Lenka Nedbalová, Barbora Pipek, Pavel Drastich, Premysl Falt, Tomas Vanasek, Jana Svikova, Milan Siroky, Ludek Bouchner, Pavel Svoboda, Jiri Bronsky and Martin Bortlik in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848261436156 – Supplemental material for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry
Supplemental material, sj-docx-4-tag-10.1177_17562848261436156 for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry by Ondrej Hradsky, Dana Duricova, Milan Lukas, Lenka Nedbalová, Barbora Pipek, Pavel Drastich, Premysl Falt, Tomas Vanasek, Jana Svikova, Milan Siroky, Ludek Bouchner, Pavel Svoboda, Jiri Bronsky and Martin Bortlik in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-5-tag-10.1177_17562848261436156 – Supplemental material for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry
Supplemental material, sj-docx-5-tag-10.1177_17562848261436156 for Persistence of advanced therapies in patients with ulcerative colitis: real-world data from the nationwide CREdIT registry by Ondrej Hradsky, Dana Duricova, Milan Lukas, Lenka Nedbalová, Barbora Pipek, Pavel Drastich, Premysl Falt, Tomas Vanasek, Jana Svikova, Milan Siroky, Ludek Bouchner, Pavel Svoboda, Jiri Bronsky and Martin Bortlik in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to thank Editage (![]() ) for English language editing. We also thank the Institute of Biostatistics and Analyses for operating the CREdIT registry. We thank all representatives of the centres participating in the CREdIT registry: Vladimír Zbořil, University Hospital Brno, Internal Department of Gastroenterology; Martin Pešta, University Hospital Bulovka, Department of Internal Medicine; Tomáš Douda, University Hospital Hradec Králové, 2nd Department of Gastroenterology; Premysl Falt, 2nd Department of Internal Medicine – Gastroenterology and Geriatrics, University Hospital and Faculty of Medicine, Palacký University, Olomouc, Czech Republic; Pavel Svoboda, Department of Internal Medicine and Cardiology, University Hospital Ostrava and Faculty of Medicine, University of Ostrava, Czech Republic; Jana Koželuhová, University Hospital Plzeň, 1st Department of Internal Medicine, Department of Gastroenterology; Luděk Bouchner, University Hospital Plzeň, Department of Internal Medicine; Jan Martínek, University Hospital St. Anne in Brno, 2nd Internal Medicine Clinic, Department of Gastroenterology; Pavel Kohout, Thomayer University Hospital, Department of Internal Medicine; Jan Hajer, University Hospital Královské Vinohrady, 2nd Department of Internal Medicine, Gastroenterology Outpatient Clinic; Václav Leksa, Gastromedic s.r.o., Gastroenterology Department, Pardubice; Tomáš Vaňásek, Hepato – Gastroenterology Hradec Králové; Pavel Drastich, IKEM, Clinic of Hepatogastroenterology; Milan Lukáš, ISCARE a.s., Clinical and Research Centre for Inflammatory Bowel Diseases; Lenka Nedbalová, Regional Hospital Liberec, Department of Gastroenterology; Vladimír Kojecký, Regional Hospital T. Baťa, Internal Medicine Clinic; Jan Valkovský, MAPO Gastro s.r.o., Prague; Jiří Stehlík, Masaryk Hospital Ústí and Labem, Department of Gastroenterology; Radka Košková, Municipal Hospital Ostrava, Gastroenterology Outpatient Clinic; Barbora Pipek, AGEL Hospital Ostrava-Vítkovice, Centre for Digestive Tract Care; Petr Konvička, AGEL Hospital Prostějov, Department of Gastroenterology; Jan Trna, Hospital Boskovice, Gastroenterology Outpatient Clinic; Martin Bortlík, Hospital České Budějovice, Department of Gastroenterology; Michal Voška, Hospital Hořovice, Department of Surgery; Marie Tomanová, Hospital Jihlava, Department of Internal Medicine; Štěpán Peterka, Hospital Jindřichův Hradec, Gastroenterology Outpatient Clinic; Blanka Zbořilová Pazdiorová, Hospital Karlovy Vary, Department of Internal Medicine – Gastroenterology; Magdalena Otrubová, Hospital of the Brothers of Mercy, Brno, Endoscopy Centre; Robert Mudr, Hospital of the Sisters of Mercy of St. Charles Borromeo, Prague, Gastroenterology and Digestive Endoscopy; Karel Mareš, Na Homolce Hospital, Department of Gastroenterology; Vladan Burda, Hospital Rudolf and Stefanie Benešov, Endoscopy Centre; Roman Staněk, Hospital Šumperk, Department of Gastroenterology; Ladislav Douda, Hospital Tábor, Department of Internal Medicine 1 – Gastroenterology; Martin Liberda, Hospital Valašské Meziříčí, Department of Gastroenterology; Ivana Mikoviny Kajzrlíková, Hospital Frýdek-Místek, Department of Internal Medicine, Beskydy Gastro Centre; Ján Ondrášik, Hospital Znojmo, Department of Internal Medicine; Zdeněk Papík, Regional Hospital Náchod; Petra Mertová, Regional Hospital Příbram, Department of Internal Medicine; Milan Široký, Pardubice Hospital, Internal Medicine Clinic; Luděk Hrdlička, ResTrial GastroEndo s.r.o., Gastroenterology Department, Prague; Jan Ulbrych, SurGal Clinic s.r.o.; Petra Mináriková, Central Military Hospital Prague, Department of Gastroenterology, Hepatology and Metabolism; Aleš Novotný, General University Hospital in Prague, Centre for the Diagnosis and Treatment of Inflammatory Bowel Diseases; Petr Jabandžiev, University Hospital Brno, Department of Paediatrics; Jan Melek, University Hospital Hradec Králové, Department of Paediatrics; Jiří Bronský, University Hospital Motol, Department of Paediatrics, 2nd Faculty of Medicine Charles University; Eva Karásková, University Hospital Olomouc, Department of Paediatrics; Astrida Šuláková, University Hospital Ostrava, Paediatric Clinic; Jan Schwarz, University Hospital Plzeň, Department of Paediatrics; Radim Vyhnánek, Thomayer University Hospital, Department of Paediatrics; Ivana Ženíšková, Hospital České Budějovice, Department of Paediatrics; Martin Zimen, Hospital Jihlava, Department of Paediatrics; Nabil El-Lababidi, General University Hospital in Prague, Clinic of Paediatrics and Adolescent Medicine.
) for English language editing. We also thank the Institute of Biostatistics and Analyses for operating the CREdIT registry. We thank all representatives of the centres participating in the CREdIT registry: Vladimír Zbořil, University Hospital Brno, Internal Department of Gastroenterology; Martin Pešta, University Hospital Bulovka, Department of Internal Medicine; Tomáš Douda, University Hospital Hradec Králové, 2nd Department of Gastroenterology; Premysl Falt, 2nd Department of Internal Medicine – Gastroenterology and Geriatrics, University Hospital and Faculty of Medicine, Palacký University, Olomouc, Czech Republic; Pavel Svoboda, Department of Internal Medicine and Cardiology, University Hospital Ostrava and Faculty of Medicine, University of Ostrava, Czech Republic; Jana Koželuhová, University Hospital Plzeň, 1st Department of Internal Medicine, Department of Gastroenterology; Luděk Bouchner, University Hospital Plzeň, Department of Internal Medicine; Jan Martínek, University Hospital St. Anne in Brno, 2nd Internal Medicine Clinic, Department of Gastroenterology; Pavel Kohout, Thomayer University Hospital, Department of Internal Medicine; Jan Hajer, University Hospital Královské Vinohrady, 2nd Department of Internal Medicine, Gastroenterology Outpatient Clinic; Václav Leksa, Gastromedic s.r.o., Gastroenterology Department, Pardubice; Tomáš Vaňásek, Hepato – Gastroenterology Hradec Králové; Pavel Drastich, IKEM, Clinic of Hepatogastroenterology; Milan Lukáš, ISCARE a.s., Clinical and Research Centre for Inflammatory Bowel Diseases; Lenka Nedbalová, Regional Hospital Liberec, Department of Gastroenterology; Vladimír Kojecký, Regional Hospital T. Baťa, Internal Medicine Clinic; Jan Valkovský, MAPO Gastro s.r.o., Prague; Jiří Stehlík, Masaryk Hospital Ústí and Labem, Department of Gastroenterology; Radka Košková, Municipal Hospital Ostrava, Gastroenterology Outpatient Clinic; Barbora Pipek, AGEL Hospital Ostrava-Vítkovice, Centre for Digestive Tract Care; Petr Konvička, AGEL Hospital Prostějov, Department of Gastroenterology; Jan Trna, Hospital Boskovice, Gastroenterology Outpatient Clinic; Martin Bortlík, Hospital České Budějovice, Department of Gastroenterology; Michal Voška, Hospital Hořovice, Department of Surgery; Marie Tomanová, Hospital Jihlava, Department of Internal Medicine; Štěpán Peterka, Hospital Jindřichův Hradec, Gastroenterology Outpatient Clinic; Blanka Zbořilová Pazdiorová, Hospital Karlovy Vary, Department of Internal Medicine – Gastroenterology; Magdalena Otrubová, Hospital of the Brothers of Mercy, Brno, Endoscopy Centre; Robert Mudr, Hospital of the Sisters of Mercy of St. Charles Borromeo, Prague, Gastroenterology and Digestive Endoscopy; Karel Mareš, Na Homolce Hospital, Department of Gastroenterology; Vladan Burda, Hospital Rudolf and Stefanie Benešov, Endoscopy Centre; Roman Staněk, Hospital Šumperk, Department of Gastroenterology; Ladislav Douda, Hospital Tábor, Department of Internal Medicine 1 – Gastroenterology; Martin Liberda, Hospital Valašské Meziříčí, Department of Gastroenterology; Ivana Mikoviny Kajzrlíková, Hospital Frýdek-Místek, Department of Internal Medicine, Beskydy Gastro Centre; Ján Ondrášik, Hospital Znojmo, Department of Internal Medicine; Zdeněk Papík, Regional Hospital Náchod; Petra Mertová, Regional Hospital Příbram, Department of Internal Medicine; Milan Široký, Pardubice Hospital, Internal Medicine Clinic; Luděk Hrdlička, ResTrial GastroEndo s.r.o., Gastroenterology Department, Prague; Jan Ulbrych, SurGal Clinic s.r.o.; Petra Mináriková, Central Military Hospital Prague, Department of Gastroenterology, Hepatology and Metabolism; Aleš Novotný, General University Hospital in Prague, Centre for the Diagnosis and Treatment of Inflammatory Bowel Diseases; Petr Jabandžiev, University Hospital Brno, Department of Paediatrics; Jan Melek, University Hospital Hradec Králové, Department of Paediatrics; Jiří Bronský, University Hospital Motol, Department of Paediatrics, 2nd Faculty of Medicine Charles University; Eva Karásková, University Hospital Olomouc, Department of Paediatrics; Astrida Šuláková, University Hospital Ostrava, Paediatric Clinic; Jan Schwarz, University Hospital Plzeň, Department of Paediatrics; Radim Vyhnánek, Thomayer University Hospital, Department of Paediatrics; Ivana Ženíšková, Hospital České Budějovice, Department of Paediatrics; Martin Zimen, Hospital Jihlava, Department of Paediatrics; Nabil El-Lababidi, General University Hospital in Prague, Clinic of Paediatrics and Adolescent Medicine.
Declarations
Use of artificial intelligence
Artificial intelligence tools were used to assist with language editing, translation and code development. Specifically, DeepL was used for translation, and ChatGPT (OpenAI) was used to support programming tasks and to assist with text shortening and stylistic refinement. All AI-assisted content was carefully reviewed, verified and edited by the authors. The authors take full responsibility for the accuracy, integrity and originality of the final manuscript.
Supplemental material
Supplemental material for this article is available online.
