Abstract
Endoscopic ultrasound-guided pancreatic duct drainage (EUS-PDD) is becoming an alternative treatment in patients with difficult retrograde access to the pancreatic duct due to difficulty in cannulation or surgically altered anatomy. This is narrative review for technical tips and outcomes for EUS-PDD. EUS-PDD is performed via one of two approaches: EUS-guided rendezvous and EUS-guided transmural drainage and involves the insertion of plastic or metal stents. While the use of plastic has been extensively described, fully covered self-expandable metal stents were reported in only six studies, and with a small number of patients. A meta-analysis including 27 trials and 902 patients reported technical success, clinical success, and adverse event (AE) rates of 89%, 88%, and 17%, respectively. The most common AE for post-EUS-PDD was acute pancreatitis, which occurred at a rate of 3%. The cumulative rates of bleeding, perforation, pancreatic leak, and infection were 2%, 2%, 1%, and 1%, respectively. The re-intervention rate due to stent dislocation, occlusion, or other reasons was 19%. In cases of surgically altered anatomy, the EUS-guided approach is significantly superior to the enteroscopy-guided approach with regard to pancreatic duct opacification (87% vs 30%; p < 0.001), cannulation success (79% vs 26%; p < 0.001), stent placement (72% vs 20%; p < 0.001), and clinical outcomes (79% vs 19%; p < 0.001), suggesting that EUS-PDD is acceptable as the first option in cases of surgically altered anatomy. Most reports on EUS‑PDD, however, evaluated only a small number of patients, and there are only few long-term follow-up studies. Further prospective studies are needed to assess the efficacy of EUS‑PDD. The development of dedicated devices and standardization of EUS-PDD procedures are also necessary.
Keywords
Introduction
Endoscopic pancreatic duct drainage with stent insertion is a therapeutic procedure for the treatment of increased pancreatic ductal pressure, which can be caused by strictures or stones associated with chronic pancreatitis (CP) or postoperative pancreatic duct–gastrointestinal anastomotic strictures. Algorithm for management of endoscopic pancreatic duct drainage is shown in Figure 1. Endoscopic retrograde cholangiopancreatography (ERCP), the technique conventionally used for pancreatic duct drainage, is often complicated by severe ductal tortuosity or tight strictures, which can make stent placement difficult. Reportedly, the treatment of pancreatic duct obstruction or stenosis with ERCP has a failure rate of 3%–10%. 1 Postoperative strictures can be treated by enteroscopy; however, difficulties during enteroscope insertion or failure to identify the anastomotic site for pancreatic duct cannulation often limit the success of this procedure. In such cases, endoscopic ultrasound (EUS)-guided pancreatic duct drainage (EUS-PDD) is a potential alternative. EUS-PDD was first reported by François et al. 2 in 2002, and has been effectively used to treat symptoms related to pancreatic duct hypertension, although, currently, EUS-guided biliary drainage (EUS-BD) is considered the alternative treatment of choice in cases in which drainage with ERCP is difficult. 3 EUS-PDD has certain limitations compared with EUS-BD; although both procedures are effective for EUS-guided drainage, they differ in two aspects. First, the diameter of the pancreatic duct, which is the target for puncture in EUS-PDD, is narrower than that of the bile duct. In addition, CP and recurring inflammation cause hardening of the pancreatic parenchyma and ducts, which make puncture, dilation, and stent placement difficult. This leads to a lower success rate and a greater incidence of complications with EUS-PDD than EUS-BD. Second, in EUS-BD, a failed procedure can be corrected using alternative methods such as percutaneous drainage or gallbladder drainage. With EUS-PDD, however, a failed procedure requires surgery. Therefore, this technique is dependent on the operator’s experience, such as the learning curve and level of expertise. Thus, EUS-PDD should be performed after gaining sufficient experience with EUS-BD.

Algorithm for management of endoscopic pancreatic duct drainage.
Comparing the results of EUS-PDD versus surgery in patients with CP, the early surgery group reportedly experienced significantly superior results in terms of improvement in the inflammation and relief of abdominal pain as compared to endoscopic treatment. A larger number of patients who underwent early surgery treatment also were “very satisfied” with the results in comparison with those who underwent endoscopic treatment. 4 The total number of interventions was also lower in the early surgery group. 5 However, while surgical treatment can provide long-term symptom relief for CP, it is not universally applicable in high-risk individuals such as the elderly, those with poor nutritional status, or those with severe systemic diseases. Additionally, some patients may have distorted and deformed pancreatic ducts due to chronic inflammation, making conventional ERCP procedures unsuccessful. EUS-PDD offers a minimally invasive and safe decompression treatment for these patients. Although EUS-PDD cannot bridge the gap with surgical treatment, it provides a feasible alternative for patients who cannot tolerate surgery.
Methods
We performed PubMed search to identify relevant English-language articles using the following keywords: “EUS guided pancreatic drainage,” or “EUS-guided pancreatic duct drainage.” Screening of their titles identified 387 articles that were deemed relevant to this review. We present a narrative review of these articles, describing technical tips of EUS-PDD and possible areas of future research.
Indications and contraindications of EUS-PDD
The indications and contraindications of EUS-PDD are summarized in Table 1. EUS-PDD is used to alleviate the abdominal and back pain associated with elevated pancreatic duct pressure in diseases such as CP, anastomotic stricture after pancreaticojejunostomy, pancreatic fistula after pancreatectomy, and pancreatic duct disruption due to severe pancreatitis, trauma, or tumors. In patients with normal anatomy, EUS-PDD should only be attempted in those with failure of ERCP.
Indications and contraindications of EUS-PDD.

EUS-PDD, endoscopic ultrasound-guided pancreatic duct drainage.
The contraindications for EUS-PDD include poor visualization of the pancreatic duct on EUS, intervening vessels in the puncture route, severe coagulopathy, and unstable conditions unfit for interventional EUS. In cases of massive ascites, EUS-PDD should be performed with caution because of associated risks such as pancreatic fistula, stent migration, and peritonitis. Although there are no reports of EUS-PDD in patients with massive ascites, there is one report of EUS-guided hepaticogastrostomy (EUS-HGS) in patients with ascites. 6 In that report, initiating continuous ascites drainage before EUS-HGS was important for technical success and preventing adverse events (AEs). This suggests that percutaneous insertion of a drainage tube and continuous ascites drainage prior to, during, and after the procedure may prevent complications such as peritonitis and stent migration. Additionally, in patients with ascites, bile leakage has been observed more frequently in patients receiving plastic stents (10%) than in those receiving covered self-expandable metal stent (SEMS; 4%) 6 ; therefore, a covered SEMS is recommended for patients with ascites. Hence, when performing in EUS-PDD in cases with ascites, a percutaneous drainage tube should be inserted before the procedure and drainage should be performed with a covered SEMS.
EUS-PDD techniques
EUS-PDD can be performed using one of two techniques: transpapillary or transanastomotic EUS-guided rendezvous (EUS-RV; Figures 2 and 3) and EUS-guided transmural drainage (EUS-TMD) including EUS-guided pancreatico-enterostomy (Figure 4) and transenteric antegrade stenting.

Retrograde stent placement (rendezvous technique) in patients with normal anatomy. The pancreatic duct is punctured with a 19G needle under EUS guidance (a). A guidewire is placed in the duodenum beyond the stenosis and papilla up to a sufficient length (arrow) (b). The duodenoscope is replaced, and the location of the guidewire protruding from the papilla (arrow) was confirmed (c). The guidewire is grasped, and cannulation is performed through the guidewire (d). The stent is placed using the rendezvous technique (e).
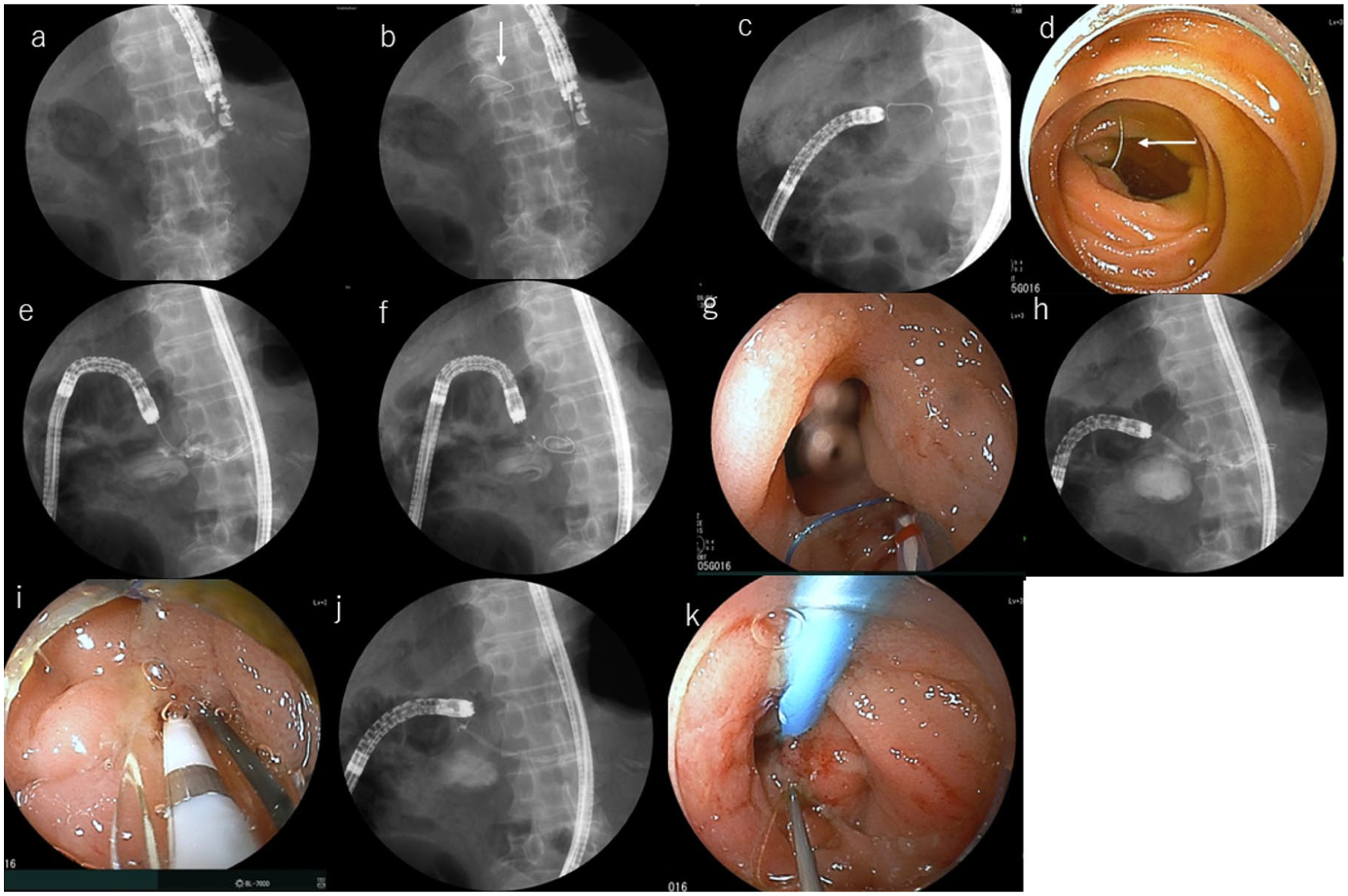
Rendezvous technique in cases with surgically altered anatomy. Under EUS guidance, the pancreatic duct is punctured with a 22G needle due to the slight dilation of the pancreatic duct (a). Next, after injection of contrast agent, the guidewire is inserted and placed beyond the anastomotic stenosis (arrow) and left in the digestive tract (b). The EUS scope is changed to an enteroscope, which is inserted up to the anastomosis (c) and the guidewire (arrow) was confirmed (d). After grasping the guidewire, the guidewire is pulled into the channel using forceps (e). The catheter is inserted through the guidewire (f/g). After dilating the anastomotic stenosis with a 4 mm balloon (h/i), the stent is placed into the pancreatic duct (j/k).

EUS-guided transenteric antegrade stenting. The pancreatic duct is punctured with a 19G needle under EUS guidance (a) and contrast agent is injected (b). It is difficult to insert the guidewire beyond pancreatic duct calculi (arrow) and the guidewire is placed into the pancreatic duct (c). A stent is inserted into the pancreatic duct (d) and the antegrade stent is placed as the pancreatico-enterostomy (e/f).
EUS-RV is performed when the major or minor papilla and anastomotic site are accessible. The procedure is performed as follows: the pancreatic duct is first punctured, and the contrast agents are injected; a guidewire is then inserted into the intestine beyond the stenotic area and the papilla/anastomotic site in an antegrade manner. The EUS scope is removed, leaving the guidewire in place. Then, the duodenoscope or balloon enteroscope is inserted up to the papilla/anastomotic site, the guidewire protruding from the papilla/anastomotic site is pulled into the scope, the cannula is inserted into the pancreatic duct along the guidewire, and the stent is placed across the stenosis.
EUS-TMD is performed when it is difficult to insert a guidewire beyond the stenotic area in the pancreatic duct or the papilla and the anastomotic site. EUS-TMD is performed as follows: the pancreatic duct is punctured, and contrast agent is injected, following which a guidewire is inserted into the pancreatic duct. In cases in which inserting the guidewire beyond the stenotic area and the papilla/anastomotic site is difficult, a stent is inserted between the pancreatic duct and gastrointestinal lumen (EUS-guided pancreatico-enterostomy). After inserting the guidewire beyond the stenotic area and the papilla/anastomotic site, the stent is inserted into the duodenum/intestine through the papilla/anastomotic site (transenteric antegrade stenting).
Role of fluoroscopy in EUS-PDD
Fluoroscopy plays important roles in EUS-PDD. First, contrast agent injection into the pancreatic ducts enables fluoroscopic visualization of the ductal system in detail, identifying any blockages, narrowing, or other abnormalities requiring treatment. Fluoroscopy enables endoscopists to visually guide catheters, guidewires, and stents through the intricate anatomy of the pancreatic ducts and into the stomach or duodenum. This is particularly important in cases involving anatomical variations or difficult strictures. When the endoscopic approach is difficult due to tight or tortuous strictures, acute angulation, or other technical issues, fluoroscopic guidance can facilitate successful instrument placement. Second, fluoroscopy enables confirmation of the correct position of implanted stents or drainage tubes in the targeted location, ensuring proper pancreatic duct decompression. Third, postprocedure fluoroscopy helps to verify the integrity of the new connection between the pancreas and the stomach or duodenal bulb by checking for leaks and the successful closure of any fistulous tracts.
On the other hand, fluoroscopy carries the risk of radiation exposure during the examination. Also, the fluoroscopic image does not allow detection of the distance between the pancreas and stomach or duodenum. Therefore, it is important to confirm the distance using ultrasound imaging.
Technical tips for EUS-PDD
EUS scope
There are two types of EUS scopes (forward-viewing and oblique-viewing EUS scope) for performing biliary drainage. Reportedly, determination of the optimal access site is more time consuming with the use of the forward-viewing EUS scope, which may be explained by the smaller scanning range (90° vs 180°) and the unusual orientation of the scanning plane. Time from puncture to stent placement, however, was seen to be short when the forward-viewing echoendoscope was used. Additionally, the forward-viewing EUS was easier to insert than the oblique-viewing EUS. 7 This suggests that the forward-viewing EUS may be preferable when the puncture site can be easily identified.
Puncture needle
A needle with excellent puncture capability and minimal risk of guidewire damage is recommended for EUS-PDD. Typically, a 19G needle I used with a 0.025-inch guidewire. In difficult cases, however, a 22G puncture needle is used along with a 0.018-inch guide wire. In cases with a longer distance between the EUS and the pancreatic duct or when a more angled puncture is necessary, the EZ shot 3 Plus (Olympus Medical Systems, Tokyo, Japan) is a good choice because it has a smaller curvature during puncture and has the largest range of deflection angles using an elevator device when used along with an elevator device due to needle made by coil sheath. 8
Use of a 22G needle and 0.018-inch guidewire is recommended for small main pancreatic duct dilations or when there is increased stiffness for puncture. When the 0.018-inch guidewire is used, it is replaced by a different guidewire with a lager caliber and greater stiffness for subsequent intervention.
In cases in which the pancreatic duct wall cannot be penetrated or pancreatic duct is not dilated, duct puncture is attempted by increasing the puncture speed. The needle is used to puncture the area beyond the pancreatic duct once and is then slowly pulled back to the pancreatic duct using the Seldinger technique. The contrast agent is then injected into the pancreatic duct. During manipulation of the guidewire through the needle, torque transmission is poorer than with an ERCP catheter, and guidewire damage can occur at the needle tip during advancement or withdrawal. Therefore, the needle needs to be pulled back slightly toward the pancreatic parenchyma to avoid damage to the guidewire (Figure 5).

Tips for overcoming difficulties with puncture. The puncture is performed by increasing the speed of needle insertion (arrow) (a). A needle is used to puncture beyond the pancreatic duct once (arrow) (b) and then the needle is slowly pulled back into the pancreatic duct using the Seldinger technique (arrow) (c, d). The needle (arrow) needs to be pulled back slightly toward the pancreatic parenchyma to avoid damage to the guidewire (arrowhead) (e).
Puncture site
Selection of the appropriate puncture site for EUS-PDD is also important. Typically, imaging modalities such as contrast-enhanced computed tomography and magnetic resonance imaging/magnetic retrograde cholangiopancreatography are used to map the area before EUS-PDD. These modalities enable evaluation of the size and tortuosity of the pancreatic duct, and the presence or absence of pancreatic duct stenosis and pancreatic stones, ascites, and the intervening vessels. The distance to the stricture, the angle of the needle, and the shape of the pancreatic duct at the puncture site influence the difficulty in guidewire manipulation. A gentle puncture angle facilitates later tract dilation. A perpendicular angle between the needle and pancreatic duct makes the guidewire manipulation and subsequently guidewire insertion. Therefore, before puncturing the pancreatic duct, it is important to evaluate the angle between the scope and the needle using fluoroscopy. In patients who have previously undergone partial gastrectomy, the puncture site is limited by the size of the remaining stomach. In patients who have undergone pancreaticoduodenectomy, the distal pancreatic duct should be punctured because the distance from the anastomosis is smaller. If the pancreatic duct is punctured at a site in which the stomach and pancreas are far apart, subsequent device insertion can be extremely difficult, and there is a risk of guidewire loss. Therefore, the puncture location should be at a short distance between the pancreatic duct and the EUS transducer. To ensure adequate guidewire placement, the pancreatic duct should be punctured as far as possible from the stenotic area, which provides stability for subsequent procedures such as guidewire and stent placement. However, puncturing upstream of the pancreatic duct may result in a vertical puncture angle or introduction of the guidewire at the tail side. Additionally, puncturing in areas with severe tortuosity should be avoided because it can increase the difficulty of subsequent procedures. These factors underscore the importance of selecting the appropriate puncture site.
Dilation
In EUS-TMD, dilation of the gastric wall, pancreatic parenchyma, and pancreatic duct are important for technical success. The guidewire should be inserted deep enough into the pancreatic duct to ensure the safety of subsequent procedures. However, insertion of a dilator can be difficult in cases of fibrotic and hardened pancreas, such as due to CP. Therefore, it is important to ensure that the guidewire is visible in the longitudinal direction on EUS imaging during the procedure. There are two types of endoscopic dilation devices: nonelectrocautery and electrocautery devices. Nonelectrocautery devices include mechanical dilators and balloon dilators, while electrocautery devices are classified into noncoaxial and coaxial types. When using electrocautery devices, however, only coaxial cautery dilators should be used. Further, electrocautery dilators have their own disadvantages, such as the risk of bleeding and pancreatitis. 9 Hence, mechanical dilators are more suitable for interventional EUS because they have a low risk of AEs such as bleeding, compared with cautery dilators. 10 Cautery-assisted devices are effective when dilation is difficult due to severe fibrosis of the pancreatic parenchyma, and should only be used when dilation with mechanical devices fails. A third dilation device that was recently developed is the drill dilator, which provides easy and safe tract dilation. The drill dilator can be inserted without the application of force because it uses rotatory action to move forward. This device facilitates safe performance of the EUS-BD procedure even by nonexperts. 11 In EUS-BD, the drill dilator is maintained at a constant distance between the gastric wall and the liver parenchyma, preventing its movement due to the pushing force. In EUS-PDD, the stiffness of the pancreatic parenchyma and pancreatic duct can increase the distance between the stomach and pancreas during insertion of the dilator. In such cases, the drill dilator may be useful as it can be inserted even through a narrow stricture. In fact, Ogura et al. 12 reported that a drill dilator was more useful than a mechanical dilator in EUS-PDD.
Stent placement
There are two types of stents: plastic stent and SEMS. Most stents that are placed transmurally in EUS-PDD are plastic stents. Single and double pigtail 5–7 Fr stents are frequently used. When selecting a plastic stent for EUS-PDD, an “all-in-one stent” is recommended to avoid insertion failure. The “all-in-one stent” has the advantage that it can return to its prerelease state if the stent length is inappropriate or if stent advancement is not possible. This is due to the presence of a string between the stent and the delivery system. This enables alternative stent placement or additional pathway expansion in cases in which stent insertion is difficult.
To prevent stent migration, covered SEMS with anchoring flaps or flared end for antimigration properties have been designed. To prevent stent migration, plastic stents with pigtail anchor or flanges have designed.
Use of SEMS in EUS-PDD is associated with both advantages and disadvantages. A covered SEMS can compress the fistula tract and prevent further pancreatic fluid leakage. Thus, EUS-PDD using a covered SEMS can be used as a rescue or primary therapy alternative to EUS-PDD using a plastic stent. Major issues of the EUS-PDD with a covered SEMS, on the other hand, include stent migration, pancreatic duct obstruction by the cross-stream blockage of the pancreatic duct with the covering membrane of the SEMS, and difficulty in stent insertion due to the width of the delivery system. Moreover, the high cost of using SEMS in EUS-PDD, which is often used for benign diseases, is another disadvantage.
In summary, many institutions still use plastic stents, and considering the prevalence of benign diseases, medical costs, and the lack of development of SEMS dedicated to EUS-PDD, drainage using pigtail plastic stents is currently being considered. Further, comparative studies evaluating the safety and feasibility of covered SEMS with plastic stent is thus necessary.
Clinical outcomes
Although there have been many studies on EUS-PDD, the most of them have described only a smaller number of cases. Table 2 presents reports involving a larger number of cases of EUS-PDD (>10).13–38 A meta-analysis of three of reports that included 27 trials involving 902 patients who underwent EUS-PDD reported a cumulative technical success rate of 89% (95% confidence interval (CI): 85–93, I2 = 77.7%), and a clinical success rate of 88% (95% CI: 83–93, I2 = 81.2%), whereas the rate of AEs was 17% (95% CI: 14–20, I2 = 28.9%). The most common AE in EUS-PDD was acute pancreatitis, which occurred at a rate of 3% (95% CI: 2–4, I2 = 0%). In addition, the cumulative rates of bleeding, perforation, pancreatic leakage, and infection were 2% (95% CI: 1–3, I2 = 0%), 2% (95% CI: 1–2, I2 = 0%), 1% (95% CI: 1–2, I2 = 0%), and 1% (95% CI: 1–2, I2 = 0%), respectively. Postoperative stent-related AEs occurred at a rate of 24% (95% CI: 12–35, I2 = 92.8%). The rate of reintervention due to stent dislocation, occlusion, and other causes was 19% (95% CI: 9–29, I2 = 80%). 39 In summary of previous meta-analyses and articles, the technical success and AE rates of EUS-PDD ranged from 48% to 100% and 12% to 42.9%, respectively.40–44 Hence, EUS-PDD demonstrates highly favorable technical and clinical success rates, along with acceptable AE rates, making it a highly effective procedure.
Previous reports of EUS-PDD.

EUS-PDD, endoscopic ultrasound-guided pancreatic duct drainage; LAMS, lumen-apposing metal stent; PS, plastic stent; RV, rendezvous technique; SEMS, self-expandable metal stent; TMD, transmural drainage.
Regarding the approach for EUS-PDD, the technical success rate for the rendezvous technique was 55.6%, which reflects the difficulty of the procedure. The success rate of TMD, including antegrade stenting, on the other hand, was 93.8%. 16 Prompt switching from rendezvous to antegrade stenting, especially when stricture traversal is prolonged, can prevent complications such as pancreatic fluid leakage. Therefore, in cases in which the rendezvous procedure is difficult, antegrade stenting should be considered.
A systematic review compared endoscopic retrograde pancreatography (ERP)-guided techniques, as a conventional drainage procedure, and EUS-guided techniques for pancreatic duct cannulation in patients with pancreaticojejunostomy stenosis. 45 The article analyzed 13 studies, including 77 patients with ERP-guided drainage, 145 patients with EUS-guided drainage, and 12 patients with both drainage modalities. The results showed that the EUS-PDD approach was significantly superior to the ERP-guided approach with regard to pancreatic duct opacification (87% vs 30%; p < 0.001), cannulation success (79% vs 26%; p < 0.001), and stent placement (72% vs 20%; p < 0.001). The EUS-PDD approach was also superior to the ERP-guided approach with regard to clinical outcomes such pain resolution (79% vs 19%; p < 0.001). In that analysis, however, the AEs rate was not compared between the two treatment modalities due to insufficient data. 45 Despite this, however, the results indicating the superiority of EUS-PDD suggest that it should be used as the first option. However, the use of EUS-PDD is limited by the lack of specialized devices.
Future research
Patients with pancreaticogastrostomies experience mixing of pancreatic and gastric juices. Gastric pH exerts significant effects on pancreatic enzyme activity in EUS-PDD patients. When the pH is below 3.0, both p-type amylase and lipase remain inactivated. 46 The impact of this on stent placement and fistula healing needs to be investigated in the future.
Conclusion
This review described the outcomes, techniques, and practical tips for EUS-PDD. Although this procedure is increasingly performed worldwide, the technical success rates vary, and complication rates remain high. Therefore, close collaboration with surgery and radiology teams is essential to prevent and effectively manage complications. In addition, most previous reports on the use of EUS‑PDD were small retrospective studies, and few long-term, follow-up studies have been published. In the future, the development of dedicated devices, refinement of techniques, and strategies to manage complications of EUS-PDD required.
