Abstract
Background:
Acute kidney injury (AKI) in cirrhotic patients is associated with high morbidity and mortality. Serum creatinine (sCr) has limited utility for early detection and etiological differentiation. Novel biomarkers and predictive models offer potential to address these limitations.
Objectives:
To comprehensively evaluate the diagnostic performance of novel biomarkers and predictive models for the early detection and etiological differentiation of AKI in patients with liver cirrhosis.
Design:
Systematic review conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Data sources and methods:
A comprehensive literature search was performed in PubMed/Medline, EMBASE, and the Cochrane Library from inception to August 31, 2025. Studies evaluating biomarkers or models for AKI prediction or phenotyping in adult cirrhotic patients were included. Study selection, data extraction, and quality assessment (using Quality Assessment of Diagnostic Accuracy Studies-2, QUADAS-2) were performed independently by reviewers.
Results:
A total of 33 studies were included. For the early prediction of AKI, serum cystatin C (Cys C) demonstrated superior performance to sCr, with an area under the receiver operating characteristic curve (AUROC) of up to 0.85. Urinary neutrophil gelatinase-associated lipocalin (NGAL) exhibited strong predictive capability and was most reliable in differentiating acute tubular necrosis from hepatorenal syndrome (HRS), achieving an AUROC up to 0.87. In contrast, kidney injury molecule-1, interleukin-18, and liver-type fatty acid binding protein, showed only moderate or inconsistent performance across most studies. Seven studies developed predictive models by integrating clinical variables with biomarkers, some of which employed machine learning techniques. However, the clinical applicability of these models is currently constrained by significant heterogeneity and limited external validation.
Conclusion:
Serum Cys C and urinary NGAL offer distinct advantages over sCr in the early diagnosis and phenotypic differentiation of AKI. Although prediction models show promise, their routine clinical application requires further standardization and extensive external validation.
Trial registration:
PROSPERO (CRD420251126410).
Introduction
Cirrhosis is the advanced stage of chronic liver disease, characterized by fibrosis of the liver parenchyma. Patients with cirrhosis are at high risk for acute kidney injury (AKI). It has been reported that the incidence of AKI in hospitalized patients with cirrhosis ranges from 20% to 50%. 1 AKI imposes substantial healthcare costs and clinical burdens, with persistent AKI, manifested as hepatorenal syndrome (HRS) and acute tubular necrosis (ATN), constituting a major therapeutic challenge. Current diagnostic criteria define persistent AKI as failure to reduce serum creatinine (sCr) levels by 50% from peak values or return to baseline levels. 2 However, reliance on relative sCr changes for AKI diagnosis inadequately addresses etiologic distinctions or differentiates between functional impairments (e.g., prerenal azotemia (PRA) and HRS) and structural damage (e.g., ATN). 3 Accurate etiologic classification of AKI necessitates comprehensive diagnostic evaluation, including identification of underlying causes and implementation of targeted interventions. While prerenal AKI, characterized by reversible functional impairment, may be resolved with appropriate management, 4 structural injuries often lead to irreversible renal dysfunction, progression to chronic kidney disease, and potential requirement for long-term dialysis. This underscores the critical importance of precise etiologic determination in guiding AKI management.
Although histopathology remains the gold standard for AKI classification, clinical practice lacks reliable non-invasive methods for early diagnosis. Current AKI diagnostic guidelines based on dynamic sCr monitoring are limited by well-documented shortcomings of sCr as a biomarker. 5 Many investigations have identified urinary and plasma biomarkers in cirrhotic patients with AKI, 6 yet differentiation between HRS and ATN remains a persistent diagnostic dilemma. Currently, numerous studies have identified urinary and plasma biomarkers indicative of acute renal injury in patients with cirrhosis. These biomarkers demonstrate potential utility in differentiating HRS from ATN. In recent years, several systematic evaluations have investigated the diagnostic performance of emerging biomarkers, including neutrophil gelatinase-associated lipocalin (NGAL) and interleukin-18 (IL-18), specifically in the context of HRS among cirrhotic patients.7,8 These studies have consistently demonstrated moderate to high diagnostic accuracy for urinary and serum NGAL, with pooled area under the receiver operating characteristic curve (AUROCs) ranging from 0.82 to 0.91.8,9 However, substantial heterogeneity across studies, variability in assay methodologies, and inconsistent cutoff values limit clinical applicability. Moreover, most existing meta-analyses focus on individual biomarkers and lack comprehensive comparative assessments across multiple markers.
This review focuses on the early prediction of AKI and the differentiation of its etiological subtypes in patients with liver cirrhosis. Our analysis incorporated the most recent studies available up to 2025, extending beyond the previous literature cutoff of 2023. We comprehensively evaluated the clinical value of novel biomarkers, particularly increasing the evaluation of multi-parameter combined strategies and artificial intelligence models in the early prediction of AKI. By integrating the most recent evidence, this study not only clarifies the comparative performance of emerging biomarkers but also provides an evidence-based foundation for biomarker selection in clinical practice and trial design, thereby addressing key gaps in the current knowledge framework.
Methods
Data sources and search strategy
This systematic review was rigorously conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 10 The study protocol was prospectively registered with PROSPERO (registration number: CRD420251126410). A comprehensive literature search was performed across three major biomedical databases: PubMed/Medline, EMBASE, and the Cochrane Library, encompassing all available records from each database’s inception through August 31, 2025. Our search strategy employed a combination of controlled vocabulary (MeSH/Emtree terms) and free-text keywords, focusing on the following key concepts: (1) AKI (“acute kidney injury” OR “acute renal failure” OR “hepatorenal syndrome” OR “acute tubular necrosis”), (2) liver cirrhosis (“cirrhosis”), and (3) diagnostic markers (“biomarker” OR “predictor” OR “prediction” OR “diagnosis”). The search was limited to English-language publications, including original research articles, clinical trials, and systematic reviews.
Inclusion and exclusion criteria
Inclusion was restricted to studies that met all the following criteria: (1) enrollment of adult patients (age ⩾18 years); (2) a focus on individuals with liver cirrhosis who were either at risk of or diagnosed with AKI, including specific subtypes such as HRS and ATN. Studies were excluded if they were: (1) animal or non-human studies; (2) non-research publications (e.g., meeting abstracts, letters, editorials, case reports) that did not provide original data; or (3) investigations of AKI primarily attributable to non-cirrhotic causes, such as chronic kidney disease, contrast-induced nephropathy, or post-surgical AKI.
Data extraction and quality assessment
Reference management and duplicate removal were performed using Rayyan, a web-based systematic review platform. 11 The study selection was conducted independently by two researchers in two stages: an initial screening of titles and abstracts, followed by a full-text review to determine final eligibility, as detailed in the PRISMA flowchart (Figure 1). Articles selected by both reviewers were included; any discrepancies were adjudicated by a third reviewer to reach a consensus. For each included study, two independent reviewers (B.C. and L.Z.) extracted the relevant data, which was subsequently verified by two additional reviewers (J.W. and X.H.). In studies that evaluated multiple biomarkers, each biomarker was analyzed separately. Studies with a subgroup sample size of less than 20 were excluded. The methodological quality of the studies was appraised using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool, 12 which evaluates four key domains: patient selection, index test, reference standard, and flow and timing. Any disagreements arising during the quality assessment were resolved through consensus discussion.
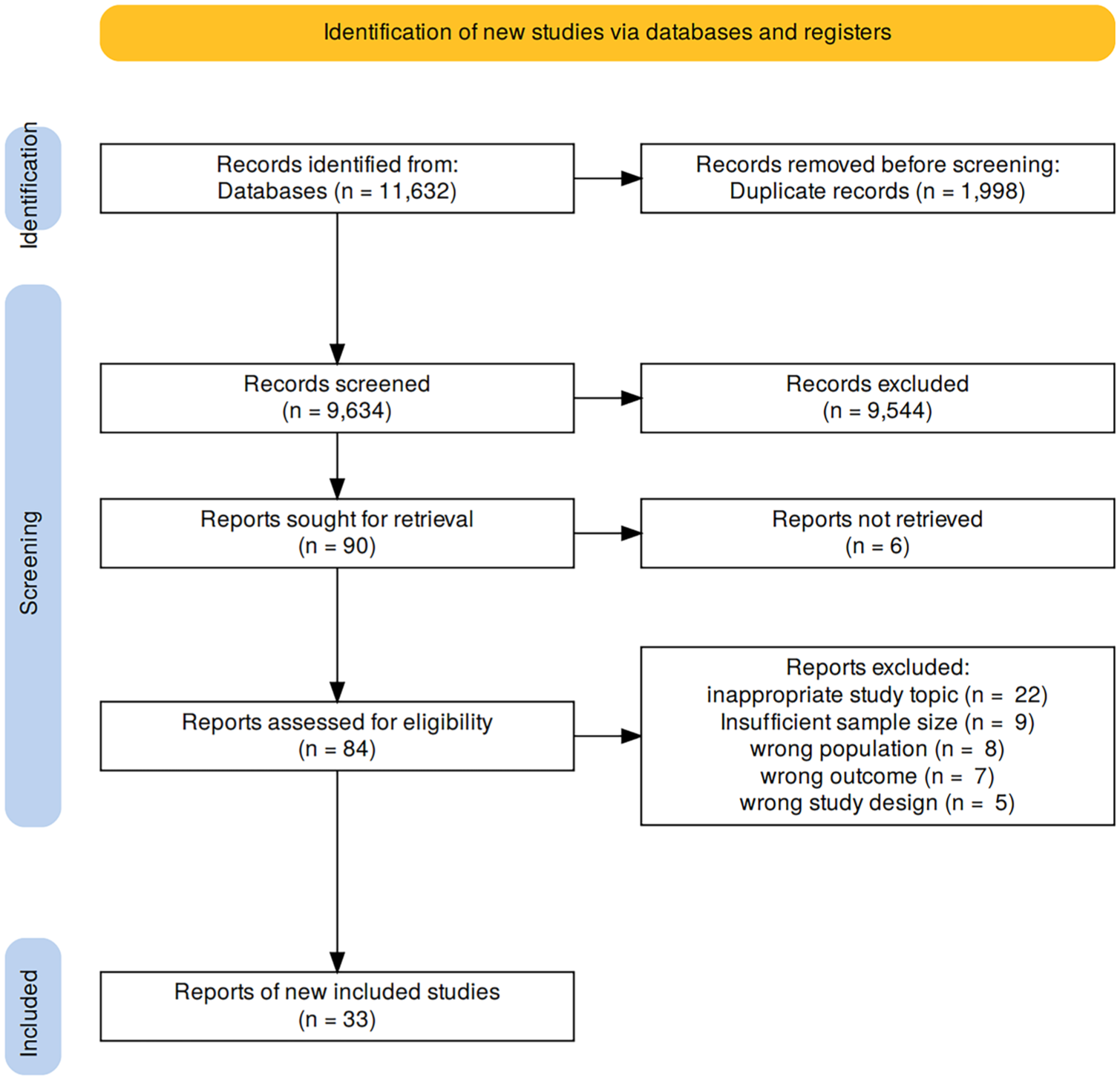
PRISMA flow flowchart.
Results
Search results and study characteristics
The initial search identified 9634 unique records. After applying the exclusion criteria, 9544 articles were removed. The remaining 90 articles underwent full-text screening, of which 33 studies met all inclusion criteria and were included in the final analysis. These studies predominantly employed prospective or retrospective cohort designs, with three case-control studies and one observational study. The study populations mainly consisted of hospitalized cirrhotic patients, with or without AKI. Only three studies reported performing a formal sample size calculation. The diagnosis of AKI in most studies was based on standardized criteria, including Kidney Disease: Improving Global Outcomes (KDIGO), International Club of Ascites (ICA), or Acute Kidney Injury Network. Only one study used renal biopsy histology as the diagnostic gold standard. Detailed characteristics of the included studies are summarized in the Supplemental Table.
Quality of the enrolled studies
QUADAS-2 appraisal showed variable quality among the 33 included studies. There was a low and/or unclear risk in each study in most domains of bias evaluation. The risk of bias was low for patient selection in 23 studies (69.7%); index test in 26 studies (78.8%); reference standard in 29 studies (87.9%); and flow and timing in 22 studies (66.7%). The applicability concerns were low for patient selection in 24 studies (72.7%); index test in 26 studies (78.8%); and reference standard in 29 studies (87.9%). Therefore, according to the criteria of overall quality, 17 studies (51.5%) were rated as low risk, 12 studies (36.4%) as unclear risk, and 4 studies (12.1%) as high risk. The results are shown in Figure 2 and the Supplemental Figure.

Risk-of-bias and applicability concerns summary generated with the QUADAS-2 tool for the full literature-based dataset.
Diagnostic criteria of AKI
In the studies reviewed, the diagnosis of AKI and its subtypes was primarily based on the KDIGO or ICA criteria, with only one study using renal histopathology as the gold standard. Given that patients with liver cirrhosis often present with coagulopathy, renal biopsy is associated with a high risk of bleeding and is therefore rarely performed in clinical practice, making non-invasive diagnostic criteria the mainstream approach.
The KDIGO criteria, 13 which rely on dynamic changes in sCr and urine output, demonstrate good sensitivity and broad applicability across populations. However, they exhibit relatively low specificity in patients with hemodynamically unstable liver disease. Studies have shown that among patients with decompensated cirrhosis, the incidence of AKI as defined by KDIGO is approximately 25%, yet nearly 40% of these cases are subsequently confirmed as reversible renal impairment after reassessment of volume status.14,15 In contrast, the ICA 2015 criteria take full account of the unique pathophysiological mechanisms of liver disease. 16 They stipulate that, after diuretic withdrawal and unsuccessful volume expansion, a sCr increase of ⩾50% and exceeding 1.5 mg/dL, in the absence of structural kidney disease, allows for better differentiation between functional renal injury (HRS) and intrinsic renal failure (ATN). By incorporating etiological assessment, this criterion significantly improves diagnostic specificity.
Biomarkers for early diagnosis of AKI in cirrhosis
Serum cystatin C (Cys C) serves as an excellent indicator for early decline in glomerular filtration rate (GFR). Unlike sCr, which is influenced by factors such as muscle mass, age, gender, and inflammatory status, Cys C exhibits minimal extrarenal variation. Clinical evidence indicates that serum Cys C levels rise earlier than sCr during the development of AKI, a characteristic particularly evident in high-risk populations such as patients with liver cirrhosis. Prospective cohort studies have demonstrated that the AUROC for predicting AKI using Cys C exceeds 0.8, with an optimal cutoff value ranging between 1.0 and 1.4 mg/L.17–19 Compared to sCr, Cys C consistently shows superior sensitivity and specificity. Furthermore, Cys C also facilitates early risk stratification, especially within the first 24–72 h of hospitalization, when interventions are likely to yield the most significant outcomes.17,20–22
Several studies have focused on the predictive capacity of NGAL for AKI development in hospitalized cirrhotic patients.18,20–28 In one prospective investigation, urinary NGAL achieved an AUROC of 0.966 for AKI prediction, with sensitivity and specificity of 86.92% and 100%, respectively. 27 Moreover, two studies reported that the interval to AKI onset was only 2 days.22,29 These findings suggest that urinary NGAL may detect early evidence of tubular injury before sCr reaches the diagnostic threshold for AKI.
Kidney injury molecule-1 (KIM-1) is specifically expressed in damaged proximal tubular epithelial cells and becomes detectable in urine within 6–12 h after injury. In a small prospective study, the AUROC of urinary KIM-1 for predicting AKI in decompensated cirrhosis was 0.55, 18 whereas another case-control study reported a value of 0.84. 30 Although KIM-1 exhibits high specificity due to its scarce extrarenal expression, its predictive performance for AKI in cirrhotic patients remains moderate.
N-acetyl-β-D-glucosaminidase (NAG), a lysosomal enzyme released during proximal tubular injury, increases within 12 h of renal damage and has been validated for early AKI detection. Its primary utility lies in identifying subclinical tubular dysfunction in high-risk cirrhotic patients before symptom onset. In a small retrospective study, Kim et al.19,21 reported that NAG excelled in predicting AKI in cirrhosis (AUROC = 0.833), while a larger prospective study found only moderate performance (AUROC = 0.625).
Research on liver-type fatty acid binding protein (L-FABP) and IL-18 remains relatively limited, with these markers more commonly applied to the progression and subtyping of AKI. A prospective study on the urinary cell cycle arrest biomarkers [TIMP2].[IGFBP-7] indicated modest performance in predicting AKI among cirrhotic patients. 20 Other emerging biomarkers, such as plasma uromodulin, microvesicles, and the neutrophil percentage-to-albumin ratio, have also shown potential in predicting AKI in cirrhotic patients.30–33 These novel markers may enhance diagnostic accuracy and provide deeper pathophysiological insights, holding promise for future clinical applications. Studies of biomarkers for early diagnosis of AKI are provided in Table 1.
Studies of biomarkers for early diagnosis of AKI.

AKI, acute kidney injury; AUROC, area under the receiver operating characteristic curve; Cr, creatinine; Cys C, cystatin C; KIM-1, kidney injury molecule-1; NAG, N-acetyl-β-D-glucosaminidase; NGAL, neutrophil gelatinase-associated lipocalin; [TIMP2].[IGFBP-7], tissue inhibitor of metalloproteinases-2 × insulin-like growth factor binding protein-7.
Biomarkers for differentiating the phenotypes of AKI
Urinary NGAL, a sensitive indicator of renal tubular injury, inflammation, and altered glomerular filtration function, has emerged as a key biomarker for identifying AKI phenotypes. Several studies have demonstrated that urinary NGAL exhibits strong performance in distinguishing ATN from non-ATN causes of kidney injury.18,20–30,35–38 In a prospective cohort study conducted by Huelin et al., 26 urinary NGAL was confirmed to possess high diagnostic accuracy in identifying ATN among various phenotypes of AKI in patients with cirrhosis. The study further revealed that in patients with AKI stage ⩾1B, the diagnostic performance of urinary NGAL was optimal when assessed within 2 days following AKI diagnosis and after albumin infusion. These findings strongly support the integration of urinary NGAL into clinical diagnostic algorithms for AKI in cirrhotic patients. Notably, urinary NGAL has also shown efficacy in differentiating between PRA and HRS. 28
In contrast, other biomarkers performed slightly less well. Urinary IL-18 can reflect renal tubular ischemic injury and inflammatory response, but its discriminatory ability is significantly lower than NGAL in large-scale prospective studies of decompensated liver cirrhosis (AUROC: 0.66 vs 0.87), possibly due to its insufficient specificity or weak signal strength in the context of liver cirrhosis.23,24,39 KIM-1 is expressed higher in ATN, suggesting its association with structural renal tubular damage, but existing studies show that its AUROC ranges from 0.54 to 0.78, with limited diagnostic accuracy, and is not yet sufficient for independent use in phenotype identification.23,24 L-FABP, as a marker of proximal renal tubular oxidative stress, is higher in ATN patients than in HRS or PRA patients, but there is currently insufficient validation data to establish its superiority over NGAL in diagnosis.
Serum Cys C is more accurate than sCr in estimating glomerular filtration rate (eGFR) and is not affected by muscle mass or nutritional status, but it mainly reflects changes in eGFR and cannot directly distinguish the causes of AKI. However, studies have found that the level of Cys C in the urine of ATN patients is significantly elevated, 40 suggesting its potential diagnostic significance.
Functional parameters such as fractional excretion of sodium (FENa) and fractional excretion of urea (FEUrea) are helpful in differentiating renal (especially ATN) and non-renal AKI, especially in the absence of oliguria.23,24,28,41 However, FENa is easily affected and reduces specificity in the presence of diuretics. Prospective studies have shown that at admission, FENa is a simple and effective clinical tool for differentiating ATN, PRA, and HRS in patients with liver cirrhosis, and its diagnostic efficacy is superior to FEUrea.41,42
Several other biomarkers have been explored but demonstrate limited clinical utility compared to those previously discussed. Urinary albumin, β2-microglobulin, and osteopontin, for instance, exhibit inferior diagnostic performance. 24 Meanwhile, emerging biomarkers such as microRNAs, urinary exosomes, and thromboxane-related markers require further validation to establish their diagnostic value and clinical applicability30,43,44 (Table 2).
Studies of biomarkers for differentiating the phenotypes of AKI.

AKI, acute kidney injury; ATN, acute tubular necrosis; AUROC, area under the receiver operating characteristic curve; FENa, fractional excretion of sodium; FEUrea, fractional excretion of urea; HRS, hepatorenal syndrome; IL-18, interleukin-18; KIM-1, kidney injury molecule-1; L-FABP, liver-type fatty acid binding protein; NGAL, neutrophil gelatinase-associated lipocalin; PRA, prerenal azotemia.
To synthesize these findings and provide a clear clinical framework, we have developed a schematic diagram that correlates key biomarkers with the pathophysiological process of AKI in patients with liver cirrhosis (Figure 3).

This diagram illustrates the various biomarkers of AKI in the setting of cirrhosis. In decompensated cirrhosis, portal hypertension causes vasodilation and reduced effective arterial volume, activating RAAS, SNS, and AVP, leading to renal vasoconstriction and sodium retention. Systemic inflammation from bacterial translocation promotes HRS and ATN. PRA influenced by diuretics and fluid imbalances may progress to HRS. ATN is manifested as tubular dysfunction (markers such as KIM-1, L-FABP, NAG, NGAL, Osteopontin). Biomarkers of glomerular filtration function include sCr, cystatin C, and so on, reflecting the degree of kidney function impairment.
Models for predicting AKI in liver cirrhosis
A total of seven studies on predictive models for AKI in patients with liver cirrhosis were included. These studies exhibited heterogeneity in design, patient populations, AKI criteria, and AKI incidence. Among them, five were retrospective, one was prospective, and one employed a cross-sectional design. In terms of clinical settings, four studies focused on general inpatients with cirrhosis, while three involved cirrhotic patients in the intensive care unit (ICU). The definition of AKI is mainly based on the KDIGO criteria. Only one study adopted the guidelines of the American Gastroenterological Association, and two cases were determined based on ICD-9 and ICD-10 codes. Considerable variation was observed across studies in sample size and the incidence of AKI events. See Supplemental Table.
Regarding modeling approaches, five studies utilized traditional regression analysis, and the remaining two incorporated machine learning algorithms. The number of predictor variables ranged from 3 to 11, encompassing demographic characteristics, clinical parameters, and novel biomarkers. Although all models underwent internal validation, only four were externally validated. The generalizability of these models, therefore, requires further assessment.
In summary, existing AKI prediction models in cirrhosis vary widely in terms of discriminative ability, calibration performance, and validation strategies. The absence of a unified modeling framework continues to limit its broad application in clinical practice. Studies of models for the prediction of AKI development are provided in Table 3.
Studies of models for the prediction of AKI development.

AKI, acute kidney injury; LASSO, Least Absolute Shrinkage and Selection Operator; ML, machine learning.
Discussion
This systematic review comprehensively synthesizes the current evidence on the use of novel biomarkers and predictive models for AKI in patients with liver cirrhosis. Our analysis confirms that several biomarkers, particularly NGAL and Cys C, demonstrate significant potential to surpass traditional sCr-based criteria by enabling earlier AKI detection and facilitating critical etiologic differentiation, especially between HRS and ATN. Furthermore, the emergence of prediction models, some incorporating machine learning, represents a promising shift in risk stratification from “post-event judgment” to “pre-event intervention,” thereby opening up valuable intervention windows for clinical practice. However, substantial heterogeneity in study designs, biomarker performance, and validation frameworks currently hinders their widespread clinical adoption.
Novel biomarkers offer significant advantages in detecting subclinical kidney injury, addressing the limited early sensitivity of conventional markers such as sCr. Among these, serum Cys C and urine NGAL have been the most extensively investigated. Substantial evidence supports the exceptional performance of Cys C in the early prediction of AKI. A large-scale cohort study by Maiwall et al. 17 reported that Cys C for diagnosing AKI achieved an AUROC of 0.75, with the optimal cut-off value approximately 1.4 mg/L, and its predictive efficacy was significantly superior to sCr; the AKI risk scoring model constructed by introducing Cys C further increased the AUROC to 0.85. Similarly, in a study of hospitalized patients with liver cirrhosis, Jaques et al. 18 confirmed that serum Cys C at admission predicted AKI with an AUROC of 0.85, markedly higher than that of NGAL (AUROC = 0.66), underscoring its superior discriminative capacity in this population. In contrast, urine NGAL demonstrates greater specificity for etiological differentiation. A systematic review and meta-analysis comprising 1219 hospitalized cirrhotic patients demonstrated that urine NGAL effectively distinguishes ATN from other phenotypes of AKI, yielding a summary AUROC of 0.88 at an optimal cut-off value approximating 220 ng/ml or 220 mg/g Cr. 9 This diagnostic utility stems from the role of NGAL as a direct marker of renal tubular epithelial injury, capturing early structural damage and thereby providing a mechanistically grounded tool for AKI phenotyping.37,51 To synthesize these complementary strengths into a practical clinical strategy, we propose an algorithm for the early diagnosis and management of AKI in cirrhosis (Figure 4).

Algorithm combining Cys C and NGAL for early diagnosis and management of AKI in cirrhosis.
Despite the growing number of emerging biomarkers for AKI, their validation in cirrhotic populations remains limited. While IL-18, KIM-1, and [TIMP2].[IGFBP7] show diagnostic promise in this setting, their reported performance varies markedly and lacks consistency. 52 This variability largely stems from the complex pathophysiology of cirrhosis. Systemic inflammation, bacterial translocation, and altered drug metabolism can independently influence biomarker levels, thereby reducing their specificity for kidney injury.53,54 Furthermore, differences in assay methods, variations in sample collection timing, and inconsistent AKI definitions across studies further limit the clinical translation of these biomarkers. 55
The development of predictive models represents an evolutionary step beyond single-marker diagnostics. By integrating demographic data, conventional clinical parameters, and novel biomarkers, these models aim to provide a holistic risk assessment. The inclusion of machine learning algorithms in some studies is particularly encouraging, as these techniques can uncover complex, non-linear interactions between variables that traditional regression might miss. However, as our review indicates, most existing models are hampered by their retrospective nature, limited sample sizes, and, most critically, a lack of robust external validation. Without external validation in distinct, independent cohorts, the generalizability and true clinical utility of these models remain unproven. The predominance of models built on ICU populations also limits their applicability to the broader inpatient or outpatient cirrhotic population.
Limitations and future research
The significant heterogeneity observed across studies, encompassing patient populations, research design, AKI criteria, and outcome indicators, precludes the feasibility of a formal meta-analysis. Moreover, differences in detection platforms, sample processing procedures, and reporting methods (such as the use of absolute concentrations or creatinine-adjusted values for urine biomarkers) further increase the difficulty of integrating the results. For instance, reported cutoff values for urinary NGAL vary widely, from 51 µg/g Cr to 365 ng/ml (see Tables 1 and 2), complicating direct comparison and clinical interpretation. Such inconsistencies undermine the reliability, reproducibility, and generalizability of findings across clinical settings. Therefore, establishing standardized analytical protocols and validated cirrhosis-specific reference intervals for key biomarkers like serum Cys C and urinary NGAL should be a research priority.
In the process of advancing precision nephrology for liver cirrhosis, a crucial step lies in developing and validating multivariate biomarker models. The pathophysiological mechanism of AKI involves multiple pathways, such as hemodynamic disorders, inflammatory responses, and renal tubular damage. A single biomarker is insufficient to comprehensively reflect its complexity. Integrating biomarkers with complementary mechanisms and combining them with clinical parameters to construct multi-parameter prediction models is expected to significantly improve the early identification and phenotyping of AKI. Such models can be further optimized through machine learning methods to enhance their discrimination and prognostic performance. However, the robustness and wide applicability of the models depend on rigorous external validation in independent cohorts with different geographical, racial, and socio-demographic characteristics. Establishing an international multicenter collaboration network will provide the necessary foundation for the validation and promotion of these multimodal tools, thereby shifting the management model for renal complications in patients with liver cirrhosis from passive response to active and individualized intervention, ultimately improving patient prognosis.
Although studies have explored the application of multiple biomarker combinations in the prediction and classification of AKI, 56 the cost–benefit assessment of these approaches remains severely inadequate, hindering their translation into clinical practice. Future research should systematically integrate health economics analysis and implementation science methods to evaluate whether the early detection and stratification strategies based on biomarkers can bring substantial clinical benefits, such as shorter hospital stays, reduced dialysis requirements, improved survival rates, and cost savings in overall medical expenses. The large-scale clinical application of biomarkers like urinary NGAL and serum Cys C not only depends on their superior diagnostic performance compared to sCr, but also on their economic feasibility and the degree of integration with existing clinical pathways.
Conclusion
In conclusion, the diagnostic and prognostic landscape of AKI in liver cirrhosis is being significantly transformed by novel biomarkers and predictive models. Serum Cys C and urine NGAL have robustly demonstrated their superiority over sCr for early detection and phenotypic differentiation. The ongoing development of multi-parameter models and AI-driven tools further augurs a future of personalized, pre-emptive medicine. However, for this promise to be realized, the field must collectively address the current challenges of standardization, rigorous validation, and demonstration of clinical utility. Until then, these novel tools should be viewed as powerful complements to, rather than replacements for, comprehensive clinical assessment.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251413123 – Supplemental material for Clinical diagnosis and biomarkers of acute kidney injury in liver cirrhosis: a systematic review
Supplemental material, sj-docx-1-tag-10.1177_17562848251413123 for Clinical diagnosis and biomarkers of acute kidney injury in liver cirrhosis: a systematic review by Baode Chen, Luyan Zheng, Xuelin He, Jie Wu and Min Zheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251413123 – Supplemental material for Clinical diagnosis and biomarkers of acute kidney injury in liver cirrhosis: a systematic review
Supplemental material, sj-docx-2-tag-10.1177_17562848251413123 for Clinical diagnosis and biomarkers of acute kidney injury in liver cirrhosis: a systematic review by Baode Chen, Luyan Zheng, Xuelin He, Jie Wu and Min Zheng in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
