Abstract
Background:
Parental psychological and behavioral factors may influence outcomes in youths with functional abdominal pain disorders (FAPDs), yet limited research has explored these associations in families referred for hospital-based psychological treatment.
Objectives:
To describe parental emotional and behavioral factors in families referred to internet-based cognitive behavioral therapy (ICBT) for FAPDs, and examine their associations with youth-reported outcomes.
Design:
Cross-sectional exploratory study.
Methods:
Eighty-seven parent-youth dyads (youth aged 8–17 years) enrolled in an ICBT intervention study at a Danish hospital participated. Parental factors included emotional distress (SCL-8), health anxiety by proxy (HAPYs), and behavioral responses (Adult Responses to Child Symptoms Monitor and Protect subscales). Youth-reported outcomes included gastrointestinal symptoms (Pediatric Quality of Life Gastrointestinal Symptom Scale (PedsQL-Gastro)), pain intensity (Faces Pain Rating Scale-Revised), quality of life (PedsQL), gastrointestinal-anxiety (Visceral Sensitivity Index-Child adapted short scale), and avoidance/control behaviors (BRQ-C). Associations were examined using Spearman’s correlations and hierarchical linear regression models, adjusting for all parental factors and youth sex.
Results:
Most parents were mothers, highly educated, employed, cohabiting, with a middle to high household income. They generally reported low emotional distress, moderate health anxiety by proxy (HAPY) and monitoring behavior, and low protective behavior. Parental emotional distress and HAPY were significantly correlated with lower youth-reported quality of life and higher gastrointestinal-specific anxiety, particularly among adolescents. Monitoring behavior correlated with greater pain intensity, especially in children. In adjusted analyses, higher emotional distress remained significantly associated with lower youth-reported quality of life (β = –0.95, p = 0.02) and higher gastrointestinal-specific anxiety (β = 0.63, p = 0.008). Monitoring behavior remained significantly associated with greater youth-reported pain intensity (β = 0.22, p = 0.01). HAPY and protective behavior showed no significant independent associations with adverse youth outcomes. However, in age-stratified analyses, protective behavior was associated with fewer gastrointestinal symptoms in adolescents (β = 2.84, p = 0.02). No notable significant sex differences were observed.
Conclusion:
Parental emotional distress and monitoring behavior were the most consistent parental factors associated with adverse youth outcomes and may represent key targets in intervention for pediatric FAPDs. The potential protective role of certain parental behaviors in adolescents warrants further exploration. Findings should be replicated in larger, more diverse samples.
Keywords
Introduction
Functional abdominal pain disorders (FAPDs) are a group of conditions where individuals experience recurring or chronic abdominal pain without any identifiable structural or biochemical cause. 1 According to the pediatric Rome IV criteria, diagnoses include Irritable Bowel Syndrome (IBS), functional dyspepsia, abdominal migraine, and Functional Abdominal Pain Not Otherwise Specified (FAP-NOS). 2 They are among the most common chronic pain conditions in children and adolescents, affecting approximately 10%–15% of this population, with higher prevalence in girls than boys, but no difference between age groups.3,4 FAPDs are classified as disorders of gut-brain interaction, suggesting that disrupted communication between the gut and brain causes normal digestive sensations—such as gas, movement, or stretching—to be perceived as painful or uncomfortable. 5 The diagnoses are based on specific symptom patterns centered around abdominal pain, with or without additional gastrointestinal symptoms such as diarrhea or constipation.
FAPDs are frequently associated with comorbid anxiety and depression, school absenteeism, 6 and reduced health-related quality of life, 7 placing a substantial burden not only on the affected child but also on their family. Parental emotional distress, including anxiety and depression, is commonly observed among parents of children with chronic pain, including FAPDs, with higher prevalences among parents of younger children versus older. 8 Such distress has been linked to poorer child outcomes, including higher pain severity and reduced functional ability. 8 Donnelly et al. 9 further reported that parental pain catastrophizing, that is, the tendency to overestimate the severity or threat of the child’s pain, is linked to increased child disability and depression, although its association with pain intensity was less robust. Conversely, decreased parental catastrophizing has been shown to mediate improvements in gastrointestinal symptoms in children undergoing internet-delivered cognitive behavioral therapy (ICBT) for FAPDs. 10 A concept related to catastrophizing, and recently introduced in the literature, is health anxiety by proxy (HAPY)—where parents experience intense and distressing health-related worries on behalf of their child.11,12 This concept, which can be measured by the Health Anxiety by Proxy Scale (HAPYs), 13 suggests that parents who catastrophize about their child’s symptoms may also struggle with more generalized anxiety about their child’s overall health, even if the child is otherwise healthy. However, HAPY has not yet been studied in parents of children with FAPDs, and its association with child symptoms and functioning remains unknown.
In addition to such emotional distress, specific parental behaviors—such as being overly protective or highly monitoring the child’s symptoms—have also been associated with increased child disability and school impairment, as demonstrated in the meta-analyses by Harrison et al. 14 and Donnelly et al. 9
Thus, a deeper understanding of the impact of parental factors could be essential to determine whether they should be systematically evaluated as part of the clinical assessment before initializing treatment of children and adolescents with FAPDs, and whether existing interventions should be adapted to further support parental mental health and illness behavior alongside child-focused care. 15
Against this background, this study aimed to examine various parental psychological and behavioral factors, that is, emotional distress, health-related worries about the child, and monitoring or protective responses to child symptoms at the time of their child being referred to specialized ICBT for FAPDs in a Danish hospital setting. Furthermore, the study examined associations between these parental factors and youth-reported outcomes, including symptom severity, pain intensity, quality of life, gastrointestinal anxiety, avoidance, and control behaviors. Given the limited existing research on parental psychological and behavioral factors in families referred to ICBT for pediatric FAPDs—particularly concerning HAPY—this study was designed as exploratory. From a developmental perspective, analyses were performed both for the full sample and separately for children and adolescents. 16
Methods
This study is part of “The Danish FGID Treatment Study,” 17 and the reporting follows the principles of the Equator Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline for cross-sectional studies. 18
Study design and setting
This study used cross-sectional data collected as part of a single-case design and pilot study involving parent-youth dyads enrolled in ICBT for FAPDs in a clinical setting at the Department of Child and Adolescent Psychiatry, Aarhus University Hospital, Denmark.17,19 Enrollment took place between June 2022 and January 2025.
Participants and procedures
Participants were referred from pediatric departments or pediatricians in private practice in the Central Denmark Region, North Denmark Region, and the Region of Southern Denmark following medical evaluation. Inclusion criteria in the original studies were: age 8–12 years (children) and 13–17 years (adolescents); a diagnosis of IBS or FAP-NOS according to the pediatric ROME-IV Criteria 2 ; stable medication use for the past month; and normal paraclinical test results. Required paraclinical tests included fecal calprotectin and blood samples (TSH, total IgA, IgA-tissue transglutaminase, complete blood count, C-reactive protein analysis, and liver enzymes). Exclusion criteria were severe psychiatric conditions requiring other intervention (e.g., psychotic symptoms, severe anxiety, high suicidal ideation, etc.), significant social issues (e.g., school absence >40% during the past month, custody disputes, child abuse, or substance use), ongoing psychological therapy or other treatment for FAPDs, and insufficient computer, internet, or language skills.
Participants were recruited through two procedures. In the first (June 2022–December 2022), eligibility was assessed during an in-person visit with a child and adolescent psychiatry trainee (E.S.N.), and questionnaires were completed online during the visit, that is, after informed consent but before the assessment. In the second (December 2023–January 2025), informed consent was obtained during a video consultation by a medical student, followed by an in-person assessment by either a child and adolescent psychiatrist (A.S.H.) or the trainee (E.S.N.). In this case, questionnaires were completed online before the assessment.
At inclusion, parents decided on one parent being responsible for study participation, here named the primary parent. Demographic data and questionnaires were managed and stored online through REDCap.20,21
Variables
Parental factors
Emotional distress was assessed by the Symptom Checklist-8 (SCL-8), a subscale from the Symptom Checklist-90-Revised. 22 It contains eight items covering anxiety and depression symptoms during the last 4 weeks, with items such as “feeling blue” or “worrying too much about things.” Items are scored on a 5-point scale (0 “not at all” to 4 “a whole lot”), with a sum score range from 0 to 32. It has shown satisfactory sensitivity and specificity as a screening tool to detect emotional mental disorders, with a cut-off score of 3, in Danish primary care patients.23,24 The Cronbach’s alpha coefficient in this study population was 0.87.
HAPY was assessed by The HAPYs.13,25 It covers 26 items regarding parental illness worries about their child’s health, covering thoughts, feelings, and behavior with items such as “I keep having thoughts about my child’s health,” “I am worried that my child has a serious illness,” and “I spend a lot of time seeking information about symptoms and illnesses.” Items are scored on a 5-point scale (0 “not at all” to 4 “a whole lot/most of the time”) with a sum score range from 0 to 104, a higher score indicating a higher level of HAPY. HAPYs has been validated in Danish parents with good known-groups validity and internal reliability. 25 The original validation of HAPYs was conducted across four parent groups representing a continuum of risk for HAPY: parents with health anxiety (highest risk), followed by those with other anxiety disorders, parents with functional somatic disorders, and healthy controls (lowest risk). 13 The Cronbach’s alpha coefficient in this study population was 0.89.
Parental responses to child symptoms were measured with the Adult Responses to Child Symptoms (ARCS). 26 The 15 included items covered the Monitor and Protect subscales based on the version proposed for children by Noel et al., 27 with items such as “Ask your child questions about how he/she feels” (monitor behavior) and “Let your child stay home from school” (protective behavior). Items were scored on a 5-point scale (0 “never” to 4 “always”). For age group sub-analyses, the child version with 15 items (four monitor items (sum score range 0–16), and 11 protect items (sum score range 0–44)) was used for younger children, while the adolescent version of the ARCS with 10 items (four monitor items (sum score range 0–16), 6 protect items (sum score range 0–24)) was used for the adolescent group. Higher scores indicate more frequent monitoring and protective parental behaviors, respectively. 28 In this study sample, Cronbach’s alpha was 0.63 for the monitor subscale and 0.72 for the protect subscale.
Youth outcomes
Gastrointestinal symptoms were assessed by the short Pediatric Quality of Life Gastrointestinal Symptom Scale (PedsQL-Gastro) based on nine items assessing the frequency of common FAPDs symptoms (abdominal pain, diarrhea, constipation, nausea, vomiting, abdominal discomfort, passing gas, nausea, and bloating) during the last month. Items were rated on a 5-point scale (0 “never” to 4 “almost always”), and scores were subsequently reversed and transformed to a sum score between 0 and 100. Higher scores indicated fewer symptoms. The PedsQL-Gastro was developed for children and adolescents with FAPDs and tested in American children with Cronbach’s alpha coefficient of 0.77, 29 and used in studies of ICBT in Swedish children with Cronbach’s alpha coefficient of 0.62. 30 For this study, it was translated into Danish by the author group following the World Health Organization (WHO) guideline for adaptation and translation of instruments. 31 The Cronbach’s alpha coefficient was 0.65 in the current sample.
Pain intensity was assessed by The Faces Pain Rating Scale-Revised (FPS-R). It assessed pain intensity on a 6-step scale with pictures indicating different pain intensities from 0 “no pain” to 10 “worst pain,” with a higher score indicating higher pain intensity. The FPS-R is validated in children and adolescents with good test–retest reliability. 32
Health-related quality of life was assessed by The Pediatric Quality of Life Inventory (PedsQL), which included 23 items that assessed physical and psychosocial health during the last month. Including items such as “hard to run” “feel afraid or scared,” “trouble keeping up with school work” and is rated on a 5-point scale (0 “never” to 4 “almost always”). Scores were reversed and transformed to a sum score between 0 and 100, with higher scores indicating better quality of life. The PedsQL has been developed for and validated in healthy and chronically ill children and adolescents with excellent internal reliability (Cronbach’s alpha 0.88). 33 Cronbach’s alpha coefficient was 0.90 in this sample.
Gastrointestinal anxiety was assessed by The Visceral Sensitivity Index-Child adapted short scale (VSI-C). With seven items such as “I often worry about problems in my belly” and “When I feel discomfort in my belly, it frightens me” being rated on a 6-point scale (0 “strongly disagree” to 5 “strongly agree,” sum score range 0–35, higher scores indicate more gastrointestinal anxiety). The VSI-C has been validated in Swedish children with FAPDs included for ICBT with good internal reliability (Cronbach’s alpha coefficient = 0.80). 30 For this study, it was translated into Danish by the author group following the WHO guideline, 31 and the Cronbach’s alpha coefficient was found to be excellent (0.90).
Control and avoidance behavior related to FAPDs was assessed by The IBS Behavioral Response Questionnaire Child adapted Short Scale (BRQ-C). With 11 items assessing avoidance and bowel control, with items such as “I avoid certain social situations (e.g., going to the cinema, eating at a friend’s house) because of my abdominal problems” and “I spend more time on the toilet than I ideally would like” rated on a 6-point scale (1 “never” to 7 “always”), with a sum score range from 11 to 77, where higher scores indicate more avoidance and control behavior. The BRQ-C has been validated in Swedish children with FAPDs with good internal reliability (Cronbach’s alpha coefficient = 0.84). 30 For this study, it was translated into Danish by the author group following the WHO guideline and with a similar Cronbach’s alpha coefficient of 0.85.
Additional variables
Demographic information was obtained at assessment and included information about sex, education level, employment status, and yearly household income of the primary parent, as well as marital status and medical history of both/all parents. Youth information included sex, age, FADPs diagnosis, duration of symptoms, and other health issues, including psychiatric comorbidity.
Study size
For this explorative study, no a priori power calculations were made. The sample size is the largest possible within the study setting. 17
Analyses
Descriptive statistics were used to characterize the total sample and stratified by age group. Demographic variables and questionnaire scores were summarized using frequencies and percentages, means with standard deviations, or medians with interquartile ranges (IQR), as appropriate. Between age group differences were examined using either independent samples t—, Chi-square, or Wilcoxon rank-sum—tests, as appropriate.
To examine simple correlations between parent-, youth-, and parent–youth variables, bivariate correlations were computed using Spearman’s rank correlation coefficients. To further explore potential associations between parental factors and child outcomes, hierarchical multiple linear regression analyses were conducted for the total sample and stratified by age group. Each regression model included one of the following youth-reported outcomes as the dependent variable: gastrointestinal symptoms (PedsQL-Gastro), pain intensity (FPS-R), health-related quality of life (PedsQL), gastrointestinal-specific anxiety (VSI-S), and avoidance and control behaviors (BRQ-C). In Step 1, the parent-reported measures were entered simultaneously as the independent variables: Emotional distress (SCL-8), HAPYs, and symptom response (ARCS-Monitor and ARCS-Protect). In Step 2, youth sex was added as an additional covariate.
Statistical significance was defined as p < 0.05. Given the study’s exploratory nature, no adjustments for multiple comparisons were applied. All statistical analyses were performed using Stata version 18.0. 34
Results
Participants characteristics
The final sample comprised 87 youth-parent dyads, including 54 children and 33 adolescents and their primary parent, respectively (for full study flowchart see Figure 1).

Study flowchart.
Missing data comprised one PedsQL-Gastro item for one participant and were handled according to the questionnaire-specific procedure described by Varni et al. 29 Data on medication use or school absence were missing for three participants, so these cases were excluded from the respective descriptive analyses.
Characteristics of included parents and youths are presented in Table 1. Most parents were employed (85%) mothers (86%), with higher education (58%), and living with the other parent (75%). Youths were predominantly female (75%); 39% were diagnosed with IBS and 62% with FAP-NOS. Median symptom duration was 2.5 years.
Study sample characteristics.
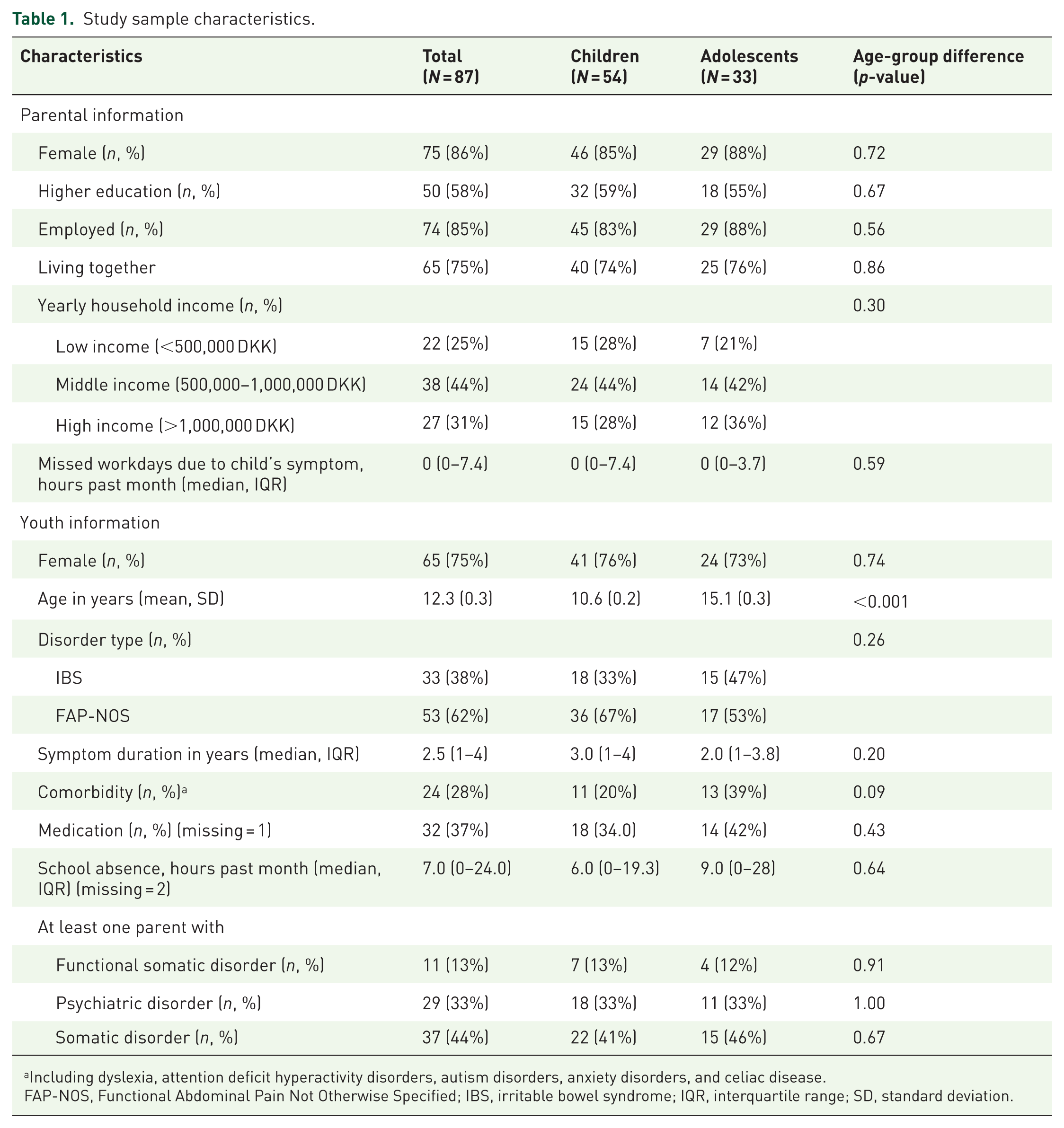
Including dyslexia, attention deficit hyperactivity disorders, autism disorders, anxiety disorders, and celiac disease.
FAP-NOS, Functional Abdominal Pain Not Otherwise Specified; IBS, irritable bowel syndrome; IQR, interquartile range; SD, standard deviation.
Summary statistics of parent factors and child outcomes, for the total sample and by age group, are presented in Table 2.
Parent factors and youth outcome scores for the total study sample and age groups separately.

↓, a lower score indicates the most positive value, and ↑, a higher score indicates the most positive value.
Median and IQR.
Child scale 0–44, adolescents scale 0–24.
Bold indicate statistically significant differences.
ARCS Monitor, Adult Responses to Child Symptoms—Monitor subscale; ARCS Protect, Adult Responses to Child Symptoms—Protect subscale; BRQ-C, IBS Behavioral Response Questionnaire—Child version; FPS-R, Faces Pain Scale—Revised; HAPYs, The Health Anxiety by Proxy Scale; PedsQL, Pediatric Quality of Life Inventory; PedsQL-Gastro, Pediatric Quality of Life Inventory—Gastrointestinal Symptoms; SCL-8, Symptom Checklist-8—Emotional Distress; VSI-C, Visceral Sensitivity Index—Child version (short scale).
Parents-reported factors
For the total sample, parents generally reported low emotional distress (median 2.0), moderate HAPY (mean 19.3) and monitoring behavior (mean 10.7) and low protective behavior (mean 7.9). No statistically significant differences in parental factors were observed between child and adolescent groups (see Table 2).
Youth-reported outcomes
For the total sample, youth reported high levels of gastrointestinal symptoms (mean 57.36), high pain intensity (mean 6.23), lowered quality of life (mean 68.11), moderate gastrointestinal anxiety (median 9), and avoidance and control behavior (median 27). Adolescents reported significantly higher levels of gastrointestinal symptoms, more gastrointestinal anxiety, and a lower quality of life than children (see Table 2).
Unadjusted correlations between parental factors and youth outcomes
Statistically significant correlations were observed among all youth-reported outcomes and all parent-reported factors (see correlation matrices in Supplemental Table S1(A) (youth reports) and (B) (parent reports)). Correlations between parental factors and youth outcomes are presented in Table 3.
Parent–child Spearman’s correlations total sample (n = 87), children (n = 54), and adolescents (n = 33).

Bold indicate statistically significant correlations.
ARCS Monitor, Adult Responses to Child Symptoms—Monitor subscale; ARCS Protect, Adult Responses to Child Symptoms—Protect subscale; BRQ-C, IBS Behavioral Response Questionnaire—Child version; FPS-R, Faces Pain Scale—Revised; HAPYs, The Health Anxiety by Proxy Scale; PedsQL, Pediatric Quality of Life Inventory; PedsQL-Gastro, Pediatric Quality of Life Inventory—Gastrointestinal Symptoms; SCL-8, Symptom Checklist-8—Emotional Distress; VSI-C, Visceral Sensitivity Index—Child version (short scale).
In the total sample, parental emotional distress and HAPY were significantly correlated with lower quality of life (r = –0.30, p = 0.004; r = −0.22, p = 0.036) and higher gastrointestinal anxiety (r = 0.38, p < 0.001; r = 0.22, p = 0.044). Parental monitoring correlated with greater pain intensity (r = 0.28, p = 0.009).
When stratified by age group, the correlations for emotional distress and HAPY were not statistically significant in children but remained so in adolescents, emotional distress was significantly correlated to pain intensity in children, and parental monitoring remained significantly correlated to pain intensity in children, but not adolescents.
Adjusted association between parental factors and youth outcomes
After mutual adjustment for all parental factors (Tables 4–6), the unique contribution of each factor showed that higher emotional distress was significantly associated with lower youth-reported quality of life and gastrointestinal anxiety (β = −0.93, p = 0.02; β = 0.63, p = 0.008). When stratified by age, the association for quality of life remained statistically significant in children (β = −1.00, p = 0.02) not adolescents (β = −0.56, p = 0.51). For gastrointestinal anxiety, the reverse pattern was seen, that is, the association was significant in adolescents (β = 0.94, p = 0.02) not children (β = 0.40, p = 0.16).
Hierarchical multiple linear regression analyses, total sample (n = 87).

Bold indicate statistically significant associations.
ARCS Monitor, Adult Responses to Child Symptoms—Monitor subscale; ARCS Protect, Adult Responses to Child Symptoms—Protect subscale; BRQ-C, IBS Behavioral Response Questionnaire—Child version; FPS-R: Faces Pain Scale—Revised; HAPYs, The Health Anxiety by Proxy Scale; PedsQL, Pediatric Quality of Life Inventory; PedsQL-Gastro, Pediatric Quality of Life Inventory—Gastrointestinal Symptoms; SCL-8, Symptom Checklist-8—Emotional Distress; VSI-C, Visceral Sensitivity Index—Child version (short scale).
Hierarchical multiple linear regression analyses, children (n = 54).
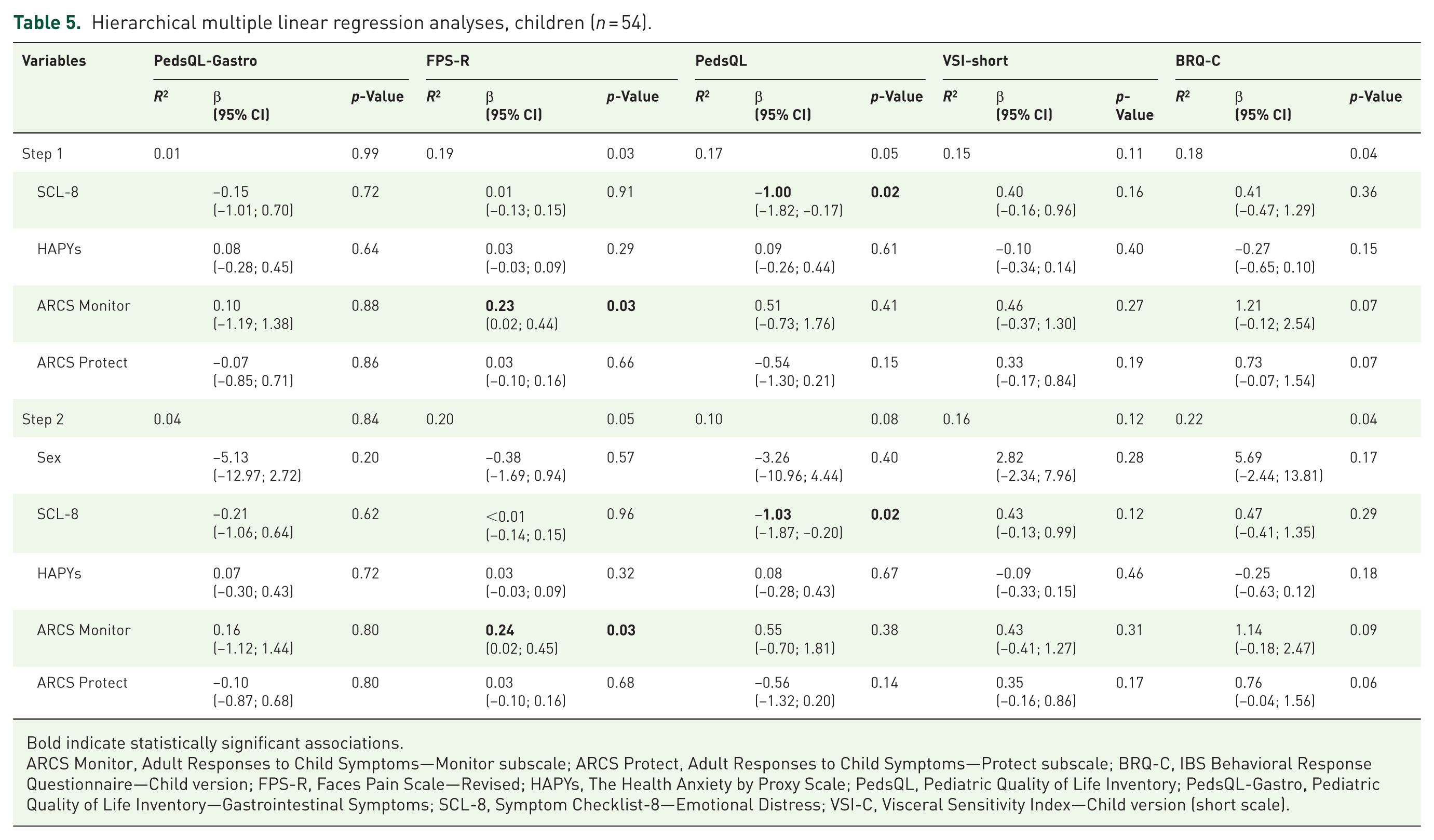
Bold indicate statistically significant associations.
ARCS Monitor, Adult Responses to Child Symptoms—Monitor subscale; ARCS Protect, Adult Responses to Child Symptoms—Protect subscale; BRQ-C, IBS Behavioral Response Questionnaire—Child version; FPS-R, Faces Pain Scale—Revised; HAPYs, The Health Anxiety by Proxy Scale; PedsQL, Pediatric Quality of Life Inventory; PedsQL-Gastro, Pediatric Quality of Life Inventory—Gastrointestinal Symptoms; SCL-8, Symptom Checklist-8—Emotional Distress; VSI-C, Visceral Sensitivity Index—Child version (short scale).
Hierarchical multiple linear regression analyses adolescents (n = 33).

Bold indicate statistically significant associations.
ARCS Monitor, Adult Responses to Child Symptoms—Monitor subscale; ARCS Protect, Adult Responses to Child Symptoms—Protect subscale; BRQ-C, IBS Behavioral Response Questionnaire—Child version; FPS-R, Faces Pain Scale—Revised; HAPYs, The Health Anxiety by Proxy Scale; PedsQL, Pediatric Quality of Life Inventory; PedsQL-Gastro, Pediatric Quality of Life Inventory—Gastrointestinal Symptoms; SCL-8, Symptom Checklist-8—Emotional Distress; VSI-C, Visceral Sensitivity Index—Child version (short scale).
Higher parental monitoring behavior was significantly associated with greater youth-reported pain intensity in the total sample (β = 0.22, p = 0.01) and in the child subgroup (β = 0.23, p = 0.03), but not in adolescents (β = 0.15, p = 0.24).
Parental protective behaviors were not significantly associated with any youth outcomes in the total sample. However, in the adolescent subgroup, higher protective behavior was significantly associated with fewer gastrointestinal symptoms (PedsQL-Gastro; β = 2.84, p = 0.02).
Including youth sex as a covariate did not change the overall pattern of findings or the direction of associations between specific parent factors and youth outcomes. However, among adolescents, ARCS Monitor was statistically significantly associated with more gastrointestinal symptoms only in the model adjusted for sex (adjusted: β = −2.09, p = 0.03 vs not-adjusted −1.79, p = 0.05).
Discussion
This study is the first to examine parental psychological and behavioral characteristics concerning symptom-related outcomes in youth with FAPDs referred to specialized ICBT in a Danish hospital setting. The findings suggest that even among relatively high-functioning parents, emotional distress and monitoring behaviors are linked to children’s symptom burden and well-being, highlighting their potential as possible intervention targets. However, further research with larger, more diverse samples is needed to confirm these findings and guide clinical practice.
Interpretation
The demographic profile of participating families offers important context for the interpretation of the current findings. Parents were relatively socioeconomically advantaged, with higher employment rates and lower divorce rates than national averages,35,36 while educational attainment and household income were high, they were comparable to national means.35,37 The parents reported low levels of emotional distress (SCL-8), consistent with the findings by Kalomiris et al. 38 showing non-elevated anxiety among parents of children with FAPDs undergoing CBT. This may contrast with Beveridge et al. 8 ’s meta-analysis, which reported high rates of anxiety (28.8%) and depression (20.0%) among parents of youth with chronic pain. The current study also introduced the first assessment of HAPY in this population using the HAPYs, 25 with a median score of 17 (IQR: 12–26), indicating moderate levels—similar to those in parents with functional somatic disorders. 13 Parental monitoring (ARCS Monitor) was comparable to a Swedish ICBT sample of parents of children with FAPDs 10 but lower than in North American samples of parents of children with chronic pain or IBD. 28 Finally, protective behavior (ARCS Protect), was slightly lower than in the Swedish sample and markedly lower than in the North American counterparts.10,28 Overall, these findings suggest a relatively high-functioning and possibly self-selected group of parents, aligned with Haine-Schlagel et al.’s 39 review, which identified, high education, employment, and income among parents engaged in child mental health care. This may reflect both the applied inclusion criteria and possible selective referral patterns by pediatricians in the current study.
Still, despite generally low to moderate levels of distress, HAPY, monitoring, and protective behaviors, these parental factors remained meaningfully associated with youth symptom severity and functioning, underscoring their clinically relevance. Thus, in the total sample, parental variables explained a modest proportion of the variance in child- and adolescent-reported outcomes. With regard to the individual factors, higher parental emotional distress was correlated with lower youth quality of life (PedsQL) and higher gastrointestinal-specific anxiety (VSI-C) in unadjusted correlation analyses. These associations remained significant after adjusting for other parental variables, suggesting that emotional distress is independently linked to youth well-being consistent with prior research, including Beveridge et al., 8 who found parental psychological distress to be associated with poorer child functioning.
Another important parental factor was monitoring behavior (ARCS Monitor), as higher parental monitoring was significantly associated with increased youth-reported pain intensity in the correlation analyses as well as in the adjusted regression analyses. This is consistent with previous research suggesting that excessive parental monitoring may inadvertently reinforce symptoms in youth with chronic pain.16,40
In contrast, parental protective behavior was not significantly correlated with any youth-rated outcomes in the total sample, neither in the unadjusted correlation analyses nor the regression model controlling for other parental variables. These findings are consistent with Van der Veek et al. 41 who reported no significant associations between parental solicitous behavior and youth outcomes in FAPDs in Dutch families included for CBT, suggesting that such behaviors may not be inherently maladaptive. Similarly, Stone et al. 42 found no links between parental solicitous responses and FAPD-related outcomes in adolescents with chronic pain. Taken together, these findings suggest that protective parental behaviors at the level in this sample may not be strongly associated with poorer child outcomes.
Although the new concept of HAPY was also correlated with poorer youth outcomes in unadjusted analyses, these associations did not remain significant when adjusting for other parental factors. This may indicate that HAPY shares substantial variance with more general parental emotional distress and/or monitoring behaviors, which may serve as more immediate drivers of child functioning in this context. In other words, once emotional distress and behavioral responses are considered, HAPY may not add unique explanatory value. Alternatively, the lack of significant adjusted effects could reflect limited variability in HAPYs scores within this relatively homogeneous, treatment-seeking sample, reducing statistical power to detect independent associations.
Age-stratified analyses revealed developmental differences in how parental distress and behavior impact youth. In children, higher parental emotional distress was significantly associated with lower quality of life, while in adolescents, it was linked to greater gastrointestinal-specific anxiety suggesting a shift from general to more symptom-specific effects with age. Although parental distress initially correlated with child-reported pain intensity in unadjusted analyses, this did not hold after adjustment—consistent with prior studies showing mixed evidence for this association.8,9,43
Parental monitoring was tied to higher pain intensity in children and to increased GI-symptoms in adolescents, highligting potential developmental shifts in how symptoms are expressed.
Protective behavior showed no effect in children, but in adolescents, it was linked to fewer gastrointestinal symptoms—possibly reflecting a developmental shift where tailored support becomes beneficial. Small subsample sizes limit conclusions, but the findings suggest that
Adjusting for youth sex did not substantially change the associations between parental factors and youth outcomes, suggesting that the influence of parental distress and behavior may operate similarly across sexes in this context. This aligns with the meta-analysis by Donnelly et al. 9 on parental cognitive, behavioral, and affective factors and associations to child chronic pain, which did not report sex differences. Similarly, Beveridge et al. 8 found no moderating effect of child sex on the associations between parental mental health and child outcomes in chronic pain population. 8 However, this area is still relatively under-investigated, and the number of boys in the present sample was relatively small, which limits the ability to draw firm conclusions. These findings should therefore be interpreted with caution and validated in larger, more sex-balanced samples.
Strengths and limitations
Several limitations of this study should be acknowledged. First, the exploratory nature of the analyses involved multiple testing, increasing the risk of Type I errors. Although no formal hypotheses were specified, analyses were informed by prior research and clinical knowledge to enhance relevance. Nonetheless, the possibility of chance associations inherent in exploratory designs should be acknowledged. Second, the internal consistency of the PedsQL-Gastro was suboptimal in this sample. This may have introduced measurement error, potentially affecting associations between gastrointestinal symptoms and other variables. However, the PedsQL-Gastro includes distinct items concerning various symptoms (e.g., bloating, pain), and lower internal consistency may be expected in heterogeneous populations such as children and adolescents with FAPDs, where symptom profiles vary. Third, not all questionnaires used in the study had been formally validated in their Danish translations (ARCS, PedsQL-Gastro, VSI-C, and BRQ-C). Although rigorous translation procedures were followed, the absence of formal validation may limit measurement reliability and validity, potentially introducing biased results. Fourth, for the ARCS questionnaire, the factor structure proposed by Noel et al. 27 was applied in the age group-specific analyses. However, for the analysis of the total sample, the factor structure validated in children was used across all participants, which may have affected validity and biased potential associations. Fifth, the cross-sectional design precludes any determination of causal direction between parental and youth variables, which may likely be bidirectional as well as dynamic over time and should therefore be considered when interpreting the findings. Sixth, the low levels of most parental factors observed in the sample may impact the ability to detect associations. Finally, the limited statistical power due to small sample sizes further constrains the strength of conclusions that can be drawn.
Generalizability
This study represents parents who actively consented to psychological treatment for their child’s FAPDs, which may indicate higher levels of psychological insight or engagement compared to the broader population of parents of youth with FAPDs. As such, direct comparisons with the general population of this pediatric patient group should be made with care. Additionally, the lack of data on gender identity, sexual orientation, and ethnic or cultural background limits the ability to assess the representativeness and inclusivity of the sample—factors that are increasingly recognized as important in pediatric pain research.45,46
Conclusion
This study offers a comprehensive characterization of parental psychological and behavioral factors in families of children and adolescents with FAPDs referred to ICBT in a Danish hospital setting. While overall levels of parental emotional distress were low, this factor was significantly associated with poorer youth outcomes, including reduced health-related quality of life and increased gastrointestinal-specific anxiety. Monitoring behavior also emerged as a relevant correlate of youth symptom severity. In age-stratified analyses, parental emotional distress was linked to lower quality of life in children and to greater gastrointestinal-specific anxiety in adolescents, suggesting age-specific effects on child functioning. Additionally, parental monitoring behavior was associated with higher pain intensity in children but more gastrointestinal symptoms in adolescents. Notably, protective behavior appeared beneficial for adolescents. However, replication in larger and more diverse samples is needed to confirm these findings.
Future research should replicate these findings in larger and more diverse samples and apply longitudinal designs to clarify causal links between parental factors and youth outcomes. It will also be important to examine developmental differences more systematically, as parental distress and behaviors appeared to influence children and adolescents in distinct ways. In addition, the specific role of the newer concept of HAPY warrants further studies to determine its potential impact on youth functioning. Finally, more research should explore whether parental factors act as mediators or moderators of treatment effects, which could guide the development of more refined and targeted parent-focused components in future interventions for FAPDs in youths.
Clinical implications
These findings underscore the importance of systematically evaluating parental factors at the outset of psychological treatment for children and adolescents with FAPDs. Parental emotional distress was significantly associated with impaired youth functioning, highlighting the clinical relevance of routine screening—potentially using brief tools like the SCL-8—to identify families in need of additional or tailored support. Although HAPY was not independently associated with youth outcomes in the adjusted cross-sectional analyses, parents reported moderately elevated levels, which may contribute to overall parental strain and merit further attention, particularly whether it becomes more relevant in shaping change processes during interventions targeting symptom-related parent–youth interactions.
Moreover, the differential associations of parental protective behaviors across age-groups, might point to the need for developmentally sensitive parental guidance in finding an appropriate balance between supervision and fostering autonomy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251383414 – Supplemental material for Parental distress and symptom severity in referred youths with functional abdominal pain disorders: a cross-sectional study prior to internet-based cognitive behavioral therapy
Supplemental material, sj-docx-1-tag-10.1177_17562848251383414 for Parental distress and symptom severity in referred youths with functional abdominal pain disorders: a cross-sectional study prior to internet-based cognitive behavioral therapy by Eva Skovslund Nielsen, Karen Hansen Kallesøe, Anne Sofie Hansen, Lisbeth Frostholm, Maria Lalouni, Marianne Bonnert and Charlotte Ulrikka Rask in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251383414 – Supplemental material for Parental distress and symptom severity in referred youths with functional abdominal pain disorders: a cross-sectional study prior to internet-based cognitive behavioral therapy
Supplemental material, sj-pdf-1-tag-10.1177_17562848251383414 for Parental distress and symptom severity in referred youths with functional abdominal pain disorders: a cross-sectional study prior to internet-based cognitive behavioral therapy by Eva Skovslund Nielsen, Karen Hansen Kallesøe, Anne Sofie Hansen, Lisbeth Frostholm, Maria Lalouni, Marianne Bonnert and Charlotte Ulrikka Rask in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We gratefully acknowledge all participants for their time and effort in completing the extensive questionnaires. We extend special thanks to statistician Anders Helles Carlesen for statistical support, medical students Helene Søgaard Singh and Laura Amalie Poulsen Dam for their valuable contributions to data collection and participant coordination. We also thank the pediatric departments and pediatricians in private practice pediatricians for referring participants, and MD, PhD Iben Møller Jönsson and MD Klaus Birkelund Johansen for their expert input in pediatric gastroenterology.
Declarations
ORCID iDs
Declaration of generative AI in the writing process
During the preparation of this work, the authors used Grammarly and ChatGPT 4.0 to edit language. After using the tools, the authors reviewed and edited the content as needed and took full responsibility for the publication’s content.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
