Abstract
Background:
Crohn’s disease (CD) remains a challenging condition, especially in patients with moderate-to-severe disease. Risankizumab (RZB), an anti-IL-23p19 monoclonal antibody, has shown efficacy in clinical trials. However, real-world data (RWD) in Asian populations are limited.
Objectives:
To assess the effectiveness and safety of Risankizumab in Asian patients with CD.
Design:
Multicenter cohort study.
Methods:
This study enrolled adult patients with moderate-to-severe CD who received Risankizumab between September 2024 and May 2025 in Taiwan. Efficacy was assessed at weeks 4, 8, and 12 using CD Activity Index (CDAI), patient-reported outcomes-2 (PRO2), and inflammatory bowel disease (IBD)-disk scores. Safety outcomes and treatment response by prior biologic exposure, including Ustekinumab (UST), were evaluated.
Results:
Forty-nine patients (mean age 41.5 years, 69.4% male) were included. Clinical response rates were 53.1%, 75.5%, and 91.8% at weeks 4, 8, and 12, respectively; clinical remission was achieved in 12.2%, 22.4%, and 42.9%. PRO2 remission reached 53.5% by week 12. Both CDAI and IBD-Disk scores improved at weeks 4, 8, and 12 with statistical significance (p < 0.0001). Transmural healing was observed in 16.3% of patients at week 12. Clinical remission at week 12 was consistent regardless of prior Ustekinumab exposure (exposed 36.36% vs naïve 44.74%, p = 0.630) or biologic-naïve status (exposed 36.84% vs naïve 63.63%, p = 0.119). No severe adverse events were reported, but mild events included headache and transient liver enzyme elevation (each 2.04%).
Conclusion:
Risankizumab may demonstrate significant short-term efficacy and favorable safety in real-world treatment of moderate-to-severe CD in an Asian cohort. Long-term data are needed to confirm sustained outcomes and guide their optimal use across diverse CD populations.
Plain language summary
Introduction
Inflammatory bowel disease (IBD) refers to a group of chronic inflammatory conditions affecting the gastrointestinal tract, primarily including Crohn’s disease (CD) and ulcerative colitis (UC), which are characterized by persistent inflammation of the intestines, leading to a variety of symptoms and complications. 1 IBD can profoundly affect various dimensions of quality of life (QoL), leading to not only physical limitations, but also psychological distress and social challenges. 2 The exact causes are not fully understood but are believed to be linked to a combination of genetic predisposition, abnormal immune responses, alterations in gut microbiota, and environmental factors.1,3 There has been a noticeable global increase in the incidence and prevalence of IBD over the past several decades, particularly in industrialized Western regions. 4 In Asian countries, recent studies indicated that the disease burden is at the stages of its emergence and acceleration in incidence.4–6 This shift highlights the need for improved understanding, diagnosis, and management of IBD in Asian countries, alongside ongoing research into its genetic, environmental, and clinical aspects.
Early diagnosis, risk stratification, setting treat-to-targets, and monitoring treatment response with tight control of disease are crucial strategies for the management of IBD.7,8 Emerging evidence revealed the favorable outcomes with early use of advanced therapies rather than gradual step-up care in IBD management.9–11 Therefore, with the early and effective control of inflammation in the window of opportunity, the risk of long-term complications and bowel disability could be ameliorated. Advanced therapies that target the complex inflammatory pathway in the development of IBD have evolved significantly in the past two decades.12,13 Among them, interleukin-23 (IL-23) plays an important role in several immune-mediated inflammatory diseases (IMIDs), including rheumatoid arthritis, psoriatic arthritis, axial spondyloarthritis, and IBD.14–17 Clinical trials of Risankizumab (RZB) which is a monoclonal antibody targeting p19 subunit of IL-23 demonstrated higher efficacy than placebo in terms of several endpoints for moderate-to-severe CD.18,19 Therefore, advanced therapy using anti-p19 antibodies to block IL-23 inflammatory pathway is promising for the management of CD and further research on the real-world data (RWD) are warranted. In this study, we aimed to evaluate the efficacy and safety of Risankizumab (Skyrizi) in Taiwan for the Assessment of RWD in Crohn’s Disease (STAR Trial) in a cohort of patients with moderate-to-severe disease status.
Methods
Study designs and subject enrollment
It was a multicenter retrospective cohort study conducted between September 2024 and March 2025 at seven (FEMH, VGHTC, CGMH, CMUH, TSGH, KMUH, MMH) tertiary centers in Taiwan. Patients with confirmed CD aged over 18 years old with disease activity of moderate-to-severe status, which was defined as Crohn’s Disease Activity Index (CDAI) of 220–450 points at baseline, were consecutively enrolled (Figure 1). Patients who received RZB treatment as first-line top-down therapy or advanced therapy after inadequate responses to step-up conventional care or other advanced therapies were enrolled. Clinical, laboratory, and endoscopic data were reviewed for analysis. The study followed the guidelines established by the Committee on Publication Ethics and the Helsinki Declaration. It was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 20 The Research Ethics Review Committees of the participating institutions approved the study.

Study flow diagram.
Definition of endpoints
Clinical response was defined as a decrease in baseline CDAI ⩾ 100 points. Clinical remission was defined as CDAI < 150 points or Harvey-Bradshaw Index (HBI) < 5, and patient-reported outcome-2 (PRO2) clinical remission was defined as 7-day average of very soft or liquid stool frequency ⩽2.8 and 7-day average of abdominal pain score ⩽1 (0, no pain; 1, mild pain; 2, moderate pain; and 3, severe pain) as well as not worse than baseline. Endoscopic response was defined as a decrease in SES-CD > 50% from baseline, while endoscopic remission was defined as SES-CD < 4 points. Mucosal healing without endoscopically visible ulcerations was considered ulcer-free endoscopic remission. Bowel wall thickness less than 3 mm by intestinal ultrasound (IUS), or absence of transmural inflammation by magnetic resonance enterography or computed tomography enterography was considered transmural healing (TH). Bowel wall thickness was measured by IUS using the methods recommended by the International Bowel Ultrasound Group. 21 Corticosteroid-free clinical remission (CFCR) was defined as achieving clinical remission without requiring any treatment with corticosteroids for more than 14 consecutive days after day 90 post-index. Adverse events (AEs) were recorder as grade 1 (mild, no limitation in activity; no medical intervention/therapy required), grade 2 (moderate, some assistance might be needed; no or minimal medical intervention/therapy required), grade 3 (severe, some assistance usually required; medical intervention/therapy required, hospitalization possible), and grade 4 (potential life-threatening, significant assistance required; significant medical intervention/therapy, hospitalization, or hospice care very likely). IBD-related hospitalization, emergent visits, and surgeries were recorded.
Statistical analysis
The patients’ basic characteristics were assessed using descriptive statistics. Discrete data are presented as counts and percentages, while continuous variables are represented as mean values ± standard deviation (SD), along with the maximum and minimum values. As this study was based on a consecutively enrolled cohort, the sample size was not determined by a priori calculation. Instead, all eligible patients who initiated Risankizumab and met the inclusion criteria during the study period were included. For the primary week-12 analysis, binary endpoints were analyzed using non-responder imputation: patients lacking an evaluable assessment within the week-12 window (±4 weeks) were imputed as non-responders. The chi-squared test was used to evaluate categorical variables, and a two-tailed p-value of less than 0.05 was deemed statistically significant. All statistical analyses were performed using STATA software (version 14.0; Stata Corp, College Station, TX, USA).
Results
A total of 49 patients were enrolled in the study, with a mean (±SD) age of 41.49 (±17.39) years. Among them, 34 (69.39%) were males and 15 (30.61%) were females (Table 1). The mean duration of disease was 77.88 (±71.89) months. Baseline CDAI and HBI scores averaged 353.92 (±65.94) and 7.98 (±2.24), respectively. Regarding disease location, 15 patients (30.61%) had isolated ileal involvement, 10 (20.41%) had colonic disease, 22 (44.90%) had ileocolonic involvement, and 2 (4.08%) had isolated upper gastrointestinal tract disease. Based on the Montreal Classification for disease behavior, 26 patients (53.06%) had B1 (inflammatory, non-stricturing, non-penetrating) disease, 17 (34.69%) had B2 (stricturing), and 6 (12.25%) had B3 (penetrating) disease. In addition, 10 patients (20.41%) presented with perianal disease, 11 (22.45%) had a history of intestinal resection, and 14 (28.57%) exhibited extraintestinal manifestations, including articular involvement in 6 (12.24%), cutaneous in 3 (6.12%), and ocular involvement in 5 (10.20%). Of the enrolled patients, 11 (22.45%) were biologics-naïve. Among those previously treated with biologics, 25 (51.02%) had been exposed to one, 7 (18.37%) to two, and 6 (8.16%) to more than three biologics with varying mechanisms of action.
Demographic data of enrolled subjects.

BMI, body mass index; CDAI, Crohn’s Disease Activity Index; HBI, Harvey-Bradshaw Index.
The clinical response rates following RZB treatment were 53.1% at week 4, 75.5% at week 8, and 91.8% at week 12 (Figure 2). Clinical remission was achieved in 12.2% of patients at week 4, increasing to 22.4% at week 8 and 42.9% at week 12. PRO2-defined clinical remission rates were 34.8%, 44.2%, and 53.5% at weeks 4, 8, and 12, respectively. A total of 15 patients (30.61%) had CFCR at week 12. The mean CDAI scores at weeks 4, 8, and 12 were 231.82 (±64.56), 207.73 (±64.90), and 165.60 (±59.47), respectively, all demonstrating statistically significant improvements from baseline (p < 0.0001; Figure 3(a)). Similarly, the mean IBD-Disk scores at weeks 4, 8, and 12 were 38.53 (±18.58), 21.32 (±17.61), and 25.84 (±18.51), respectively, also showing significant reductions compared to baseline (Figure 3(b)). IUS and ileocolonoscopic evaluation were performed in 21 (42.86%) and 15 (30.61%) patients, respectively, while the remaining 28 and 34 patients without IUS and endoscopic assessment were conservatively imputed as non-responders. At week 12, 8 (16.33%), 14 (28.57%), and 9 (18.37%) patients achieved endoscopic response, endoscopic remission, and TH, respectively. Ulcer-free endoscopic remission was found in 8 (16.33%) patients. Among patients with isolated ileal involvement, clinical response, clinical remission, and PRO2 remission were achieved in 9 (60.00%), 4 (26.67%), and 5 (33.33%) patients at week 8, and in 13 (86.67%), 9 (60.00%), and 11 (73.33%) patients at week 12, respectively.
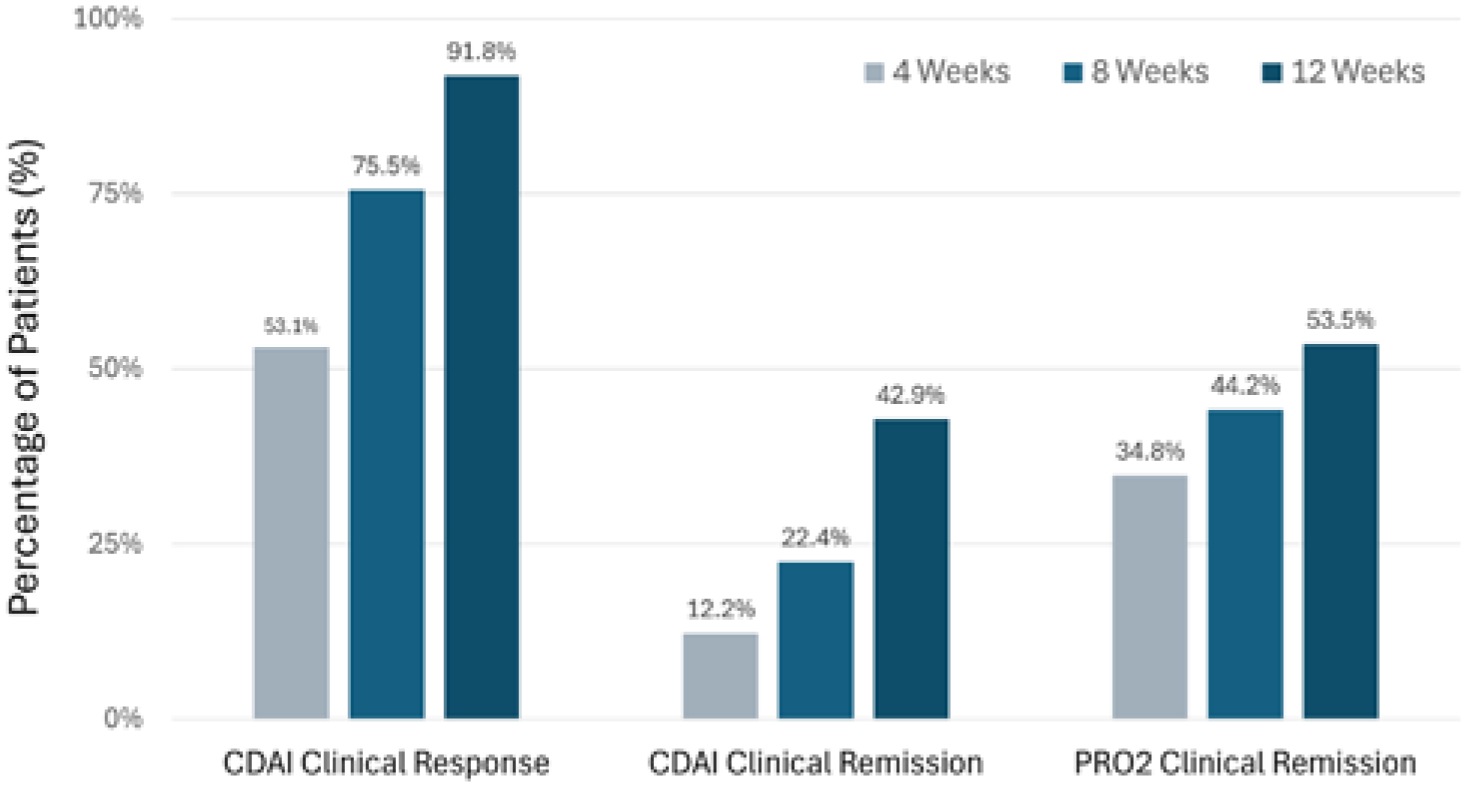
Short-term efficacy of Risankizumab for moderate-to-severe CD (clinical response defined as a decrease in baseline CDAI ⩾ 100 points; clinical remission defined as CDAI score ⩽150 or HBI <5; PRO2 clinical remission defined as stool frequency ⩽2.8 and abdominal pain score ⩽1 as well as not worse than baseline) at weeks 4, 8, and 12 of the treatment phase.
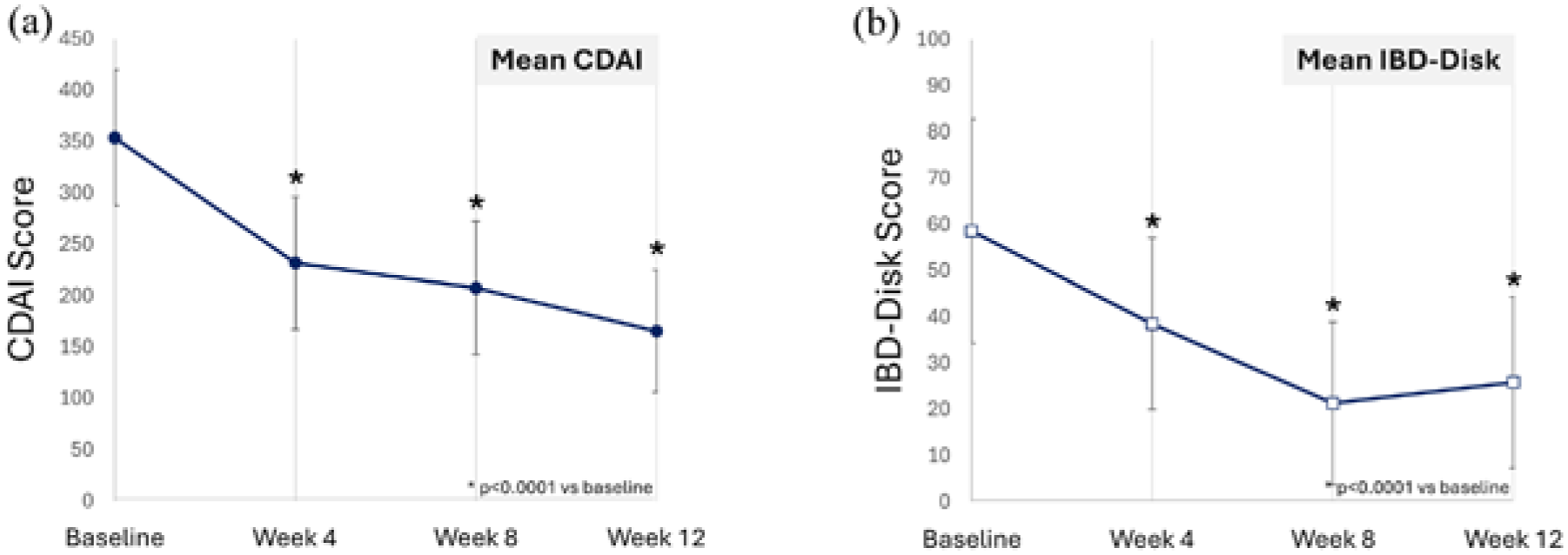
The CDAI (a) and IBD-disk scores (b) over time after Risankizumab treatment.
At week 8, clinical remission and PRO2 remission rates were 36.36% versus 18.42% (p = 0.217) and 54.55% versus 26.32% (p = 0.082), respectively, for biologic-naïve versus biologic-exposed patients. By week 12, the rates were 63.64% versus 36.84% (p = 0.119) for clinical remission, and 54.55% versus 52.63% (p = 0.913) for PRO2 remission (Table 2 and Figure 4). Regarding Ustekinumab (UST), an anti-IL12/23 agent, patients previously exposed to UST had clinical remission rates of 18.18% at week 8 and 36.36% at week 12, compared to 10.53% (p = 0.505) and 44.74% (p = 0.630), respectively, in UST-naïve patients. For PRO2-defined remission, UST-exposed patients achieved rates of 36.36% and 45.45% at weeks 8 and 12, respectively, which were not significantly different from those in UST-naïve patients (p = 0.772 and 0.575, respectively).
Short-term efficacy of RZB for moderate-to-severe Crohn’s disease stratified by biologics exposure history.

Clinical response: At week 8, p = 0.397 for biologics-naïve versus biologics-exposed, p = 0.069 for Ustekinumab-non-exposed versus Ustekinumab-exposed; at week 12, p = 0.175 for biologics-naïve versus biologics-exposed, p = 0.901 for Ustekinumab-non-exposed versus Ustekinumab-exposed. Clinical remission: At week 8, p = 0.217 for biologics-naïve versus biologics-exposed, p = 0.505 for Ustekinumab-non-exposed versus Ustekinumab-exposed; at week 12, p = 0.119 for biologics-naïve versus biologics-exposed, p = 0.630 for Ustekinumab-non-exposed versus Ustekinumab-exposed. PRO2 clinical remission: At week 8, p = 0.082 for biologics-naïve versus biologics-exposed, p = 0.772 for Ustekinumab-non-exposed versus Ustekinumab-exposed; at week 12, p = 0.913 for biologics-naïve versus biologics-exposed, p = 0.575 for Ustekinumab-non-exposed versus Ustekinumab-exposed.
PRO2, patient-reported outcome-2; RZB, Risankizumab; UST, Ustekinumab.

(a) Efficacy outcomes at weeks 8 and 12 by biologic treatment history. (b) Treatment response at weeks 8 and 12 by prior Ustekinumab use.
Four (8.16%) patients had been hospitalized, including one (2.04%) for endoscopic balloon dilatation of ileal stricture, one (2.04%) for appendicitis, one (2.04%) for cellulitis, and one (2.04%) for perianal abscess and fistula, who underwent surgery. No severe AEs or severe infections were reported. However, one patient (2.04%) experienced a headache, and another one (2.04%) had an abnormal liver function test, both classified as grade 1 events.
Discussion
To our knowledge, this is the first RWD evaluating RZB’s efficacy and safety for moderate-to-severe CD in an Asian population. Results aligned with clinical trials: clinical remission rates were 12.2% at week 4, 22.4% at week 8, and 42.9% at week 12; PRO2-defined remission reached 34.8%, 44.2%, and 53.5%, respectively. By week 12, about one-third of patients had discontinued corticosteroids. Significant improvements were seen in mean CDAI scores and QoL. The rate of endoscopic response, endoscopic remission, and TH at week 12 was 28.57%, 18.37%, and 16.33%, respectively. RZB was similarly effective in patients with isolated ileal disease and showed no significant difference in response between UST-experienced and UST-naïve patients. No serious AEs or infections occurred; only mild side effects such as headache (2.04%) and minor liver test abnormalities (2.04%) were noted.
Advanced therapies, including biologics and small molecules, have significantly improved IBD treatment outcomes and reduced surgical needs.22–25 The first biologic for CD was the anti-TNF-α monoclonal antibody cA2. 23 With advances in cytokine-targeting agents, therapies now include lymphocyte trafficking blockers, intracellular signaling inhibitors, and cytokine blockers. 22 RZB, a monoclonal antibody targeting the IL-23 p19 subunit, was approved for CD in June 2022 and UC in June 2024.18,19 In phase III trials, RZB showed strong efficacy in moderate-to-severe CD, including in patients with prior biologic failure (60% in ADVANCE, 100% in MOTIVATE). 18 At week 12, clinical remission and endoscopic response were significantly higher with RZB 600 mg versus placebo (ADVANCE: 43% vs 22%, p < 0.0001; 40% vs 12%, p < 0.0001; MOTIVATE: 35% vs 19%, p = 0.0007; 29% vs 11%, p < 0.0001). 18 In the FORTIFY maintenance study, week-52 remission and response rates were also superior with RZB (52% vs 40%, p < 0.01; 47% vs 22%, p < 0.0001). 19 Ulcer-free remission ranged from 14% to 21% during induction and reached 31% in maintenance.18,19 Among patients with prior biologic failure, remission and response rates remained significantly better than placebo during both induction (37.6%, 48.0%) and maintenance (30.8%, 44.1%) phases, while no major safety concerns or treatment-related AEs were identified.18,19 These findings confirm RZB as an effective and safe treatment option for moderate-to-severe CD.
RWD plays a crucial role in confirming the treatment of CD, offering insights and holistic views that are often not captured in clinical trials of new therapies. Some Western studies about the RWD of RZB for the management of moderate-to-severe CD patients have been published in the literature. About 67%–100% of enrolled patients were inadequately responsive to more than two biologics, and the clinical remission rates at 12-week induction and 52-week maintenance therapy were 16%–78% and 25%–54%, respectively.26–33 For those who failed to respond to anti-IL-12/23 had a clinical remission rate of 64% and 49% at induction and maintenance phase.31,32 Even among patients with CD who are refractory to multiple biologics, RZB demonstrates a positive effect on clinical and biochemical disease activity, as well as on patient-reported outcomes, in UK RWD. 33 The overall CFCR rates (Ustekinumab failure) were 16%–75% (52%) and 25%–50% (42%) at induction and maintenance.26–32 In our study of the Asian population, almost 80% of enrolled subjects were biologics-experienced, and the clinical remission rate at week 12 was not statistically different from those with biologics-naïve (36.84% vs 63.64%, p = 0.119). The efficacy of RZB on patients with UST-exposed was not different from those without prior UST usage (clinical remission rate at week 12 of 36.36% vs 44.74%, p = 0.630); however, the subgroup analysis may be underpowered to detect such differences. In Taiwan, before advanced therapies can be reimbursed by the National Health Insurance, patients are required to undergo 6 months of conventional treatment, including corticosteroids. As a result, the majority of patients were still on corticosteroids at the time RZB was initiated. In addition, due to concerns about adrenal insufficiency, tapering of corticosteroid dosage often needs to be gradual. These factors likely contributed to the relatively low CFCR of 30.61% at week 12 of RZB treatment. Regarding the safety issues in the French and USA RWD, the rate of CD-related hospitalization or flare during treatment was 6.6%–9.8%, while the most common reported AEs were fatigue, headache, injection site reaction, arthralgia, nausea, upper respiratory infection, and otitis, with incidence rates all less than 5%.28,31 The treatment persistence was 67%–79% at 52 weeks, with only 3.4% of patients discontinued RZB due to AEs in the French GETAID study.28,31 In our study, no treatment-emergent AEs were identified. Only two patients (4.08%) required hospitalization due to IBD-related complications, including one for an ileal stricture and another for perianal disease. Mild AEs reported included headache and hepatitis.
This study has several limitations. First, its observational design and small, region-specific sample may limit generalizability, though the multicenter RWD offers useful insights for CD management in Asian patients. The relatively modest overall study population limits the ability to draw definitive conclusions for specific subgroups. Second, the short follow-up period restricted the evaluation of long-term efficacy, treatment durability, and safety outcomes, particularly in regard to TH and CFCR. The notably low CFCR rate at week 12 indicates that interpretations of remission and response rates should be qualified by the fact that a substantial proportion of patients were still receiving corticosteroids at the time of disease assessment. Third, while efforts were made to standardize data collection across multiple centers, potential inconsistencies in diagnostic modalities, treatment protocols, and follow-up intervals may have introduced variability. In addition, the lack of a control group or comparator arm limits the ability to draw definitive conclusions regarding the relative effectiveness of RZB.
Conclusion
In conclusion, this multicenter RWD demonstrates that RZB may be an effective and well-tolerated treatment option for patients with moderate-to-severe CD in an Asian population. The clinical outcomes, including remission and QoL improvements, were consistent with those observed in clinical trials. These findings support the use of RZB as a valuable therapeutic option, particularly in settings with a high prevalence of biologics-experienced conditions and isolated ileal diseases. While outcomes aligned with clinical trials, further long-term data are needed to confirm sustained effectiveness and safety across diverse CD populations.
